Screening the Capacity of 34 Wetland Plant Species to Remove Heavy Metals from Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Experimental Setup
2.3. Analysis of Samples
2.4. Data Analysis
Vtx)/mfine root (DW))/t
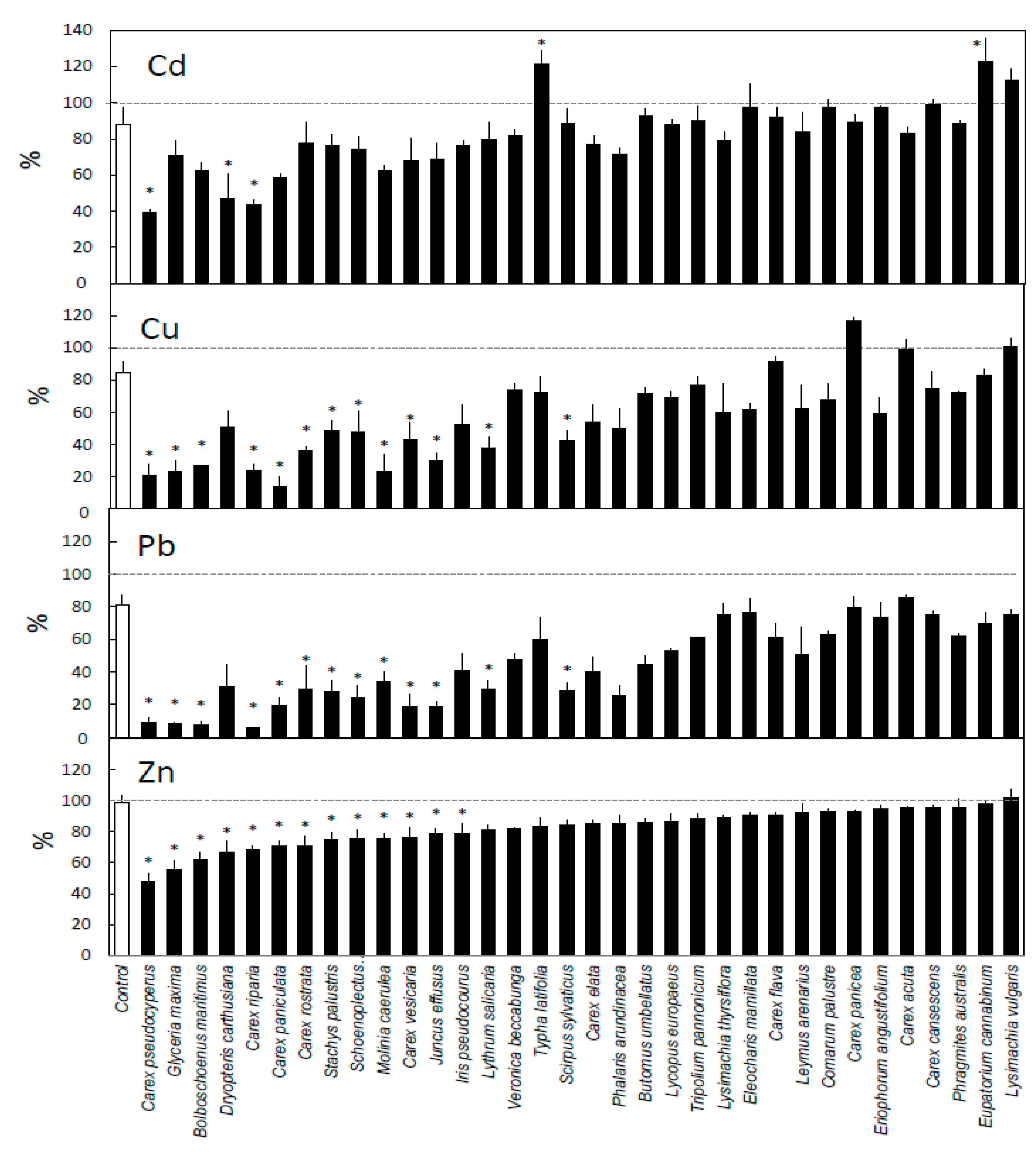
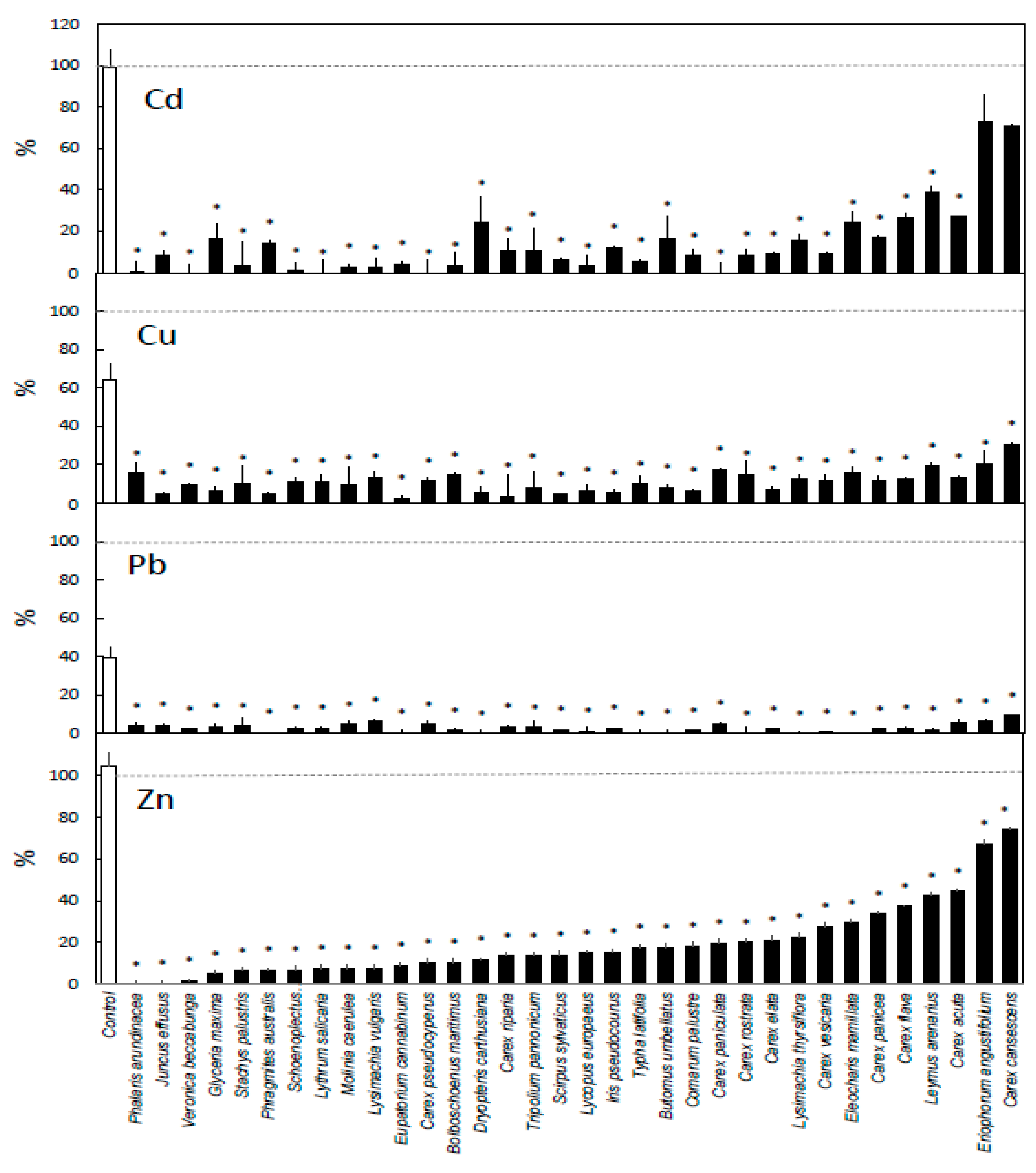
3. Results
4. Discussion
4.1. General Removal Patterns
4.2. Differences Between Metals
4.3. Differences Between Species
4.4. Differences Between Plant Groups
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, A.; Goonetilleke, A.; Egodawatta, P. Role of Rainfall and Catchment Characteristics on Urban Stormwater Quality; Springer Briefs in Water Science and Technology; Springer Singapore: Singapore, 2015; ISBN 978-981-287-458-0. [Google Scholar]
- Tanner, C.C.; Headley, T.R. Components of floating emergent macrophyte treatment wetlands influencing removal of stormwater pollutants. Ecol. Eng. 2011, 37, 474–486. [Google Scholar] [CrossRef]
- Revitt, D.; Morrison, G.M. Metal speciation variations within separate stormwater systems. Environ. Technol. Lett. 1987, 8, 373–380. [Google Scholar] [CrossRef]
- Billberger, M. Road Runoff—Advice and Recommendations for Choosing Environmental Measures (In Swedish). Borlänge, 2011. Available online: https://trafikverket.ineko.se/Files/sv-SE/11439/RelatedFiles/2011_112_vagdagvatten_rad_och_rekommendationer_for_val_av_miljoatgard.pdf (accessed on 25 June 2020).
- Kerr-Upal, M.; Seasons, M.; Mulamoottil, G. Retrofitting a stormwater management facility with a wetland component. J. Environ. Sci. Health Part A 2000, 35, 1289–1307. [Google Scholar] [CrossRef]
- Borne, K.E.; Fassman-Beck, E.; Tanner, C.C. Floating treatment wetland retrofit to improve stormwater pond performance for suspended solids, copper and zinc. Ecol. Eng. 2013, 54, 173–182. [Google Scholar] [CrossRef]
- Headley, T.R.; Tanner, C.C. Constructed Wetlands with Floating Emergent Macrophytes: An Innovative Stormwater Treatment Technology. Crit. Rev. Environ. Sci. Technol. 2012, 42, 2261–2310. [Google Scholar] [CrossRef]
- Revitt, D.; Shutes, R.; Llewellyn, N.; Worrall, P. Experimental reedbed systems for the treatment of airport runoff. Water Sci. Technol. 1997, 36, 385–390. [Google Scholar] [CrossRef]
- Shahid, M.J.; Tahseen, R.; Siddique, M.; Ali, S.; Iqbal, S.; Afzal, M. Remediation of polluted river water by floating treatment wetlands. Water Supply 2018, 19, 967–977. [Google Scholar] [CrossRef]
- Ning, D.; Huang, Y.; Pan, R.; Wang, F.; Wang, H. Effect of eco-remediation using planted floating bed system on nutrients and heavy metals in urban river water and sediment: A field study in China. Sci. Total. Environ. 2014, 485, 596–603. [Google Scholar] [CrossRef]
- Borne, K.E.; Fassman-Beck, E.; Tanner, C.C. Floating Treatment Wetland influences on the fate of metals in road runoff retention ponds. Water Res. 2014, 48, 430–442. [Google Scholar] [CrossRef]
- Ladislas, S.; Gerente, C.; Chazarenc, F.; Brisson, J.; Andres, Y. Floating treatment wetlands for heavy metal removal in highway stormwater ponds. Ecol. Eng. 2015, 80, 85–91. [Google Scholar] [CrossRef]
- Huang, X.; Zhao, F.; Yu, G.; Song, C.; Geng, Z.; Zhuang, P. Removal of Cu, Zn, Pb, and Cr from Yangtze Estuary Using thePhragmites australisArtificial Floating Wetlands. BioMed Res. Int. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Marschner, P., Ed.; Elsevier: London, UK, 2012; ISBN 9780123849052. [Google Scholar]
- Li, J.; Yu, H.; Luan, Y. Meta-Analysis of the Copper, Zinc, and Cadmium Absorption Capacities of Aquatic Plants in Heavy Metal-Polluted Water. Int. J. Environ. Res. Public Health 2015, 12, 14958–14973. [Google Scholar] [CrossRef] [PubMed]
- Weiss, J.D.; Hondzo, M.; Biesboer, D.; Semmens, M. Laboratory Study of Heavy Metal Phytoremediation by Three Wetland Macrophytes. Int. J. Phytoremediation 2006, 8, 245–259. [Google Scholar] [CrossRef] [PubMed]
- Rai, U.; Sinha, S.; Tripathi, R.; Chandra, P. Wastewater treatability potential of some aquatic macrophytes: Removal of heavy metals. Ecol. Eng. 1995, 5, 5–12. [Google Scholar] [CrossRef]
- Ladislas, S.; Gérente, C.; Chazarenc, F.; Brisson, J.; Andrès, Y. Performances of Two Macrophytes Species in Floating Treatment Wetlands for Cadmium, Nickel, and Zinc Removal from Urban Stormwater Runoff. Water Air Soil Pollut. 2013, 224, 224. [Google Scholar] [CrossRef]
- Deng, H.; Ye, Z.; Wong, M.H. Lead and zinc accumulation and tolerance in populations of six wetland plants. Environ. Pollut. 2006, 141, 69–80. [Google Scholar] [CrossRef]
- Sricoth, T.; Meeinkuirt, W.; Saengwilai, P.; Pichtel, J.; Taeprayoon, P. Aquatic plants for phytostabilization of cadmium and zinc in hydroponic experiments. Environ. Sci. Pollut. Res. 2018, 25, 14964–14976. [Google Scholar] [CrossRef]
- Landberg, T.; Greger, M. Differences in uptake and tolerance to heavy metals in Salix from unpolluted and polluted areas. Appl. Geochem. 1996, 11, 175–180. [Google Scholar] [CrossRef]
- Eliasson, L. Effects of Nutrients and Light on Growth and Root Formation in Pisum sativum Cuttings. Physiol. Plant. 1978, 43, 13–18. [Google Scholar] [CrossRef]
- Larm, T. Watershed-Based Design of Stormwater Treatment Facilities: Model Development and Applications. Available online: https://pdfs.semanticscholar.org/cc4a/95fbcb90c05ed6a6a186718eb430e42bf1e6.pdf (accessed on 11 May 2020).
- Keizer-Vlek, H.E.; Verdonschot, P.F.M.; Verdonschot, R.C.M.; Dekkers, D. The contribution of plant uptake to nutrient removal by floating treatment wetlands. Ecol. Eng. 2014, 73, 684–690. [Google Scholar] [CrossRef]
- Göthberg, A.; Greger, M.; Holm, K.; Bengtsson, B.-E. Influence of Nutrient Levels on Uptake and Effects of Mercury, Cadmium, and Lead in Water Spinach. J. Environ. Qual. 2004, 33, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Fritioff, Å.; Kautsky, L.; Greger, M. Influence of temperature and salinity on heavy metal uptake by submersed plants. Environ. Pollut. 2005, 133, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Rezania, S.; Taib, S.M.; Din, M.F.M.; Dahalan, F.A.; Kamyab, H. Comprehensive review on phytotechnology: Heavy metals removal by diverse aquatic plants species from wastewater. J. Hazard. Mater. 2016, 318, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Juang, K.-W.; Lai, H.-Y.; Chen, B.-C. Coupling bioaccumulation and phytotoxicity to predict copper removal by switchgrass grown hydroponically. Ecotoxicology 2011, 20, 827–835. [Google Scholar] [CrossRef]
- McGeer, J.C.; Brix, K.V.; Skeaff, J.M.; Deforest, D.K.; Brigham, S.I.; Adams, W.J.; Green, A. Inverse relationship between bioconcentration factor and exposure concentration for metals: Implications for hazard assessment of metals in the aquatic environment. Environ. Toxicol. Chem. 2003, 22, 1017–1037. [Google Scholar] [CrossRef]
- Christofilopoulos, S.; Syranidou, E.; Gkavrou, G.; Manousaki, E.; Kalogerakis, N. The role of halophyteJuncus acutusL. in the remediation of mixed contamination in a hydroponic greenhouse experiment. J. Chem. Technol. Biotechnol. 2016, 91, 1665–1674. [Google Scholar] [CrossRef]
- Weiss, P.; Westbrook, A.; Weiss, J.; Gulliver, J.; Biesboer, D. Effect of Water Velocity on Hydroponic Phytoremediation of Metals. Int. J. Phytoremediation 2013, 16, 203–217. [Google Scholar] [CrossRef]
- Ngoc, M.N.; Dultz, S.; Kasbohm, J. Simulation of retention and transport of copper, lead and zinc in a paddy soil of the Red River Delta, Vietnam. Agric. Ecosyst. Environ. 2009, 129, 8–16. [Google Scholar] [CrossRef]
- Nyquist, J.; Greger, M. Uptake of Zn, Cu, and Cd in metal loaded Elodea canadensis. Environ. Exp. Bot. 2007, 60, 219–226. [Google Scholar] [CrossRef]
- Moog, P.R. Planta Flooding tolerance of Carex species. I. Root structure. Planta 1998, 207, 189–198. [Google Scholar] [CrossRef]
- Chayapan, P.; Kruatrachue, M.; Meetam, M.; Pokethitiyook, P. Phytoremediation potential of Cd and Zn by wetland plants, Colocasia esculenta L. Schott., Cyperus malaccensis Lam. and Typha angustifolia L. grown in hydroponics. J. Environ. Biol. 2015, 36, 1179–1183. [Google Scholar] [PubMed]
| Type, Family, Species | Biomass Dry Weight (g) | Origin of Plant Material a | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fine Roots | Coarse Roots, Rhizomes | Leaves | Stalk, Inflorescence | Total | |||||||
| Fern, Dryopteridaceae | |||||||||||
| Dryopteris carthusiana | 0.71 | ±0.17 | 9.22 | ±0.81 | 3.13 | ±0.64 | - | - | 13.06 | ±0.19 | 5 |
| Monocot, Butomaceae | |||||||||||
| Butomus umbellatus | 0.08 | ±0.01 | 0.74 | ±0.21 | 1.16 | ±0.27 | - | - | 1.98 | ±0.15 | 1 |
| Cyperaceae | |||||||||||
| Bolboschoenus maritimus | 0.80 | ±0.32 | 1.47 | ±0.69 | 0.88 | ±0.26 | 0.74 | ±0.26 | 3.88 | ±0.97 | 2 |
| Carex acuta | 0.22 | ±0.05 | 0.21 | ±0.15 | 0.47 | ±0.15 | - | - | 0.89 | ±0.25 | 3 |
| Carex cansescens | 0.05 | ±0.01 | 0.02 | ±0.01 | 0.19 | ±0.05 | - | - | 0.26 | ±0.06 | 3 |
| Carex elata | 0.37 | ±0.04 | 0.14 | ±0.03 | 1.24 | ±0.19 | - | - | 1.74 | ±0.23 | 1 |
| Carex flava | 0.17 | ±0.03 | 0.10 | ±0.01 | 0.58 | ±0.07 | 0.14 | ±0.01 | 0.91 | ±0.05 | 4 |
| Carex panicea | 0.18 | ±0.02 | 0.13 | ±0.02 | 1.05 | ±0.15 | - | - | 1.36 | ±0.18 | 4 |
| Carex paniculata | 0.51 | ±0.06 | 0.58 | ±0.15 | 4.31 | ±0.99 | - | - | 5.41 | ±0.16 | 1 |
| Carex pseudocyperus | 1.16 | ±0.21 | 0.18 | ±0.08 | 3.36 | ±0.12 | - | - | 4.71 | ±1.32 | 5 |
| Carex riparia | 0.44 | ±0.03 | 0.29 | ±0.05 | 4.51 | ±0.15 | - | - | 5.23 | ±0.15 | 3 |
| Carex rostrata | 0.64 | ±0.23 | 0.40 | ±0.13 | 1.77 | ±0.66 | 0.26 | ±0.00 | 2.86 | ±0.57 | 5 |
| Carex vesicaria | 0.46 | ±0.11 | 0.23 | ±0.04 | 1.86 | ±0.12 | - | - | 2.55 | ±0.16 | 4 |
| Eleocharis mamillata | 0.15 | ±0.12 | 0.04 | ±0.02 | - | - | 0.67 | ±0.02 | 0.86 | ±0.84 | 5 |
| Eriophorum angustifolium | 0.10 | ±0.02 | 0.06 | ±0.03 | 0.18 | ±0.04 | - | - | 0.34 | ±0.04 | 6 |
| Schoenoplectus tabernaemontani | 0.40 | ±0.01 | 0.51 | ±0.18 | - | - | 2.05 | ±0.36 | 2.96 | ±0.16 | 2 |
| Scirpus sylvaticus | 0.43 | ±0.04 | 0.29 | ±0.05 | 1.51 | ±0.16 | - | - | 2.23 | ±0.22 | 5 |
| Iridaceae | |||||||||||
| Iris pseudocourus | 0.97 | ±0.09 | 2.41 | ±0.69 | 2.64 | ±0.19 | - | - | 6.03 | ±0.80 | 5 |
| Juncaceae | |||||||||||
| Juncus effusus | 0.58 | ±0.01 | 0.27 | ±0.02 | 2.42 | ±0.14 | - | - | 3.27 | ±0.16 | 3 |
| Poaceae | |||||||||||
| Glyceria maxima | 1.96 | ±0.31 | 1.08 | ±0.24 | 2.64 | ±0.70 | 2.80 | ±0.84 | 8.44 | ±1.67 | 5 |
| Leymus arenarius | 0.09 | ±0.02 | 0.10 | ±0.02 | 0.30 | ±0.00 | 0.27 | ±0.01 | 0.77 | ±0.01 | 2 |
| Molinia caerulea | 0.70 | ±0.04 | 0.23 | ±0.02 | 3.01 | ±0.15 | - | - | 3.93 | ±0.16 | 1 |
| Phalaris arundinacea | 0.40 | ±0.06 | 0.15 | ±0.01 | 0.72 | ±0.15 | 0.69 | ±0.09 | 1.80 | ±0.41 | 1 |
| Phragmites australis | 0.30 | ±0.06 | 0.59 | ±0.10 | 0.56 | ±0.06 | 1.05 | ±0.19 | 2.66 | ±0.21 | 1 |
| Typhaceae | |||||||||||
| Typha latifolia | 0.14 | ±0.02 | 0.86 | ±0.12 | 1.67 | ±0.11 | - | - | 2.67 | ±0.06 | 5 |
| Dicot, Asteraceae | |||||||||||
| Eupatorium cannabinum | 0.35 | ±0.03 | 0.37 | ±0.15 | 1.12 | ±0.28 | 1.68 | ±0.36 | 3.53 | ±0.08 | 1 |
| Tripolium pannonicum | 0.46 | ±0.08 | 0.50 | ±0.07 | 0.43 | ±0.12 | - | - | 1.39 | ±0.21 | 1 |
| Lamiaceae | |||||||||||
| Lycopus europaeus | 0.09 | ±0.02 | 0.08 | ±0.03 | 0.61 | ±0.11 | 0.27 | ±0.05 | 1.04 | ±0.15 | 5 |
| Stachys palustris | 0.30 | ±0.07 | 0.27 | ±0.06 | 0.85 | ±0.04 | 1.14 | ±0.09 | 2.47 | ±0.57 | 1 |
| Lythraceae | |||||||||||
| Lythrum salicaria | 0.25 | ±0.05 | 0.56 | ±0.12 | 0.39 | ±0.11 | 1.43 | ±0.12 | 3.24 | ±0.13 | 1 |
| Plantaginaceae | |||||||||||
| Veronica beccabunga | 0.14 | ±0.01 | 0.06 | ±0.01 | 0.86 | ±0.08 | 0.63 | ±0.05 | 1.70 | ±0.15 | 5 |
| Primulaceae | |||||||||||
| Lysimachia thyrsiflora | 0.08 | ±0.03 | 0.69 | ±0.10 | 0.95 | ±0.09 | 0.51 | ±0.11 | 1.68 | ±0.26 | 4 |
| Lysimachia vulgaris | 0.23 | ±0.02 | 0.43 | ±0.03 | 1.00 | ±0.11 | - | - | 1.62 | ±0.19 | 5 |
| Rosaceae | |||||||||||
| Comarum palustre | 0.14 | ±0.05 | 0.41 | ±0.22 | 0.37 | ±0.11 | 0.13 | ±0.04 | 1.06 | ±0.38 | 4 |
| Species | Removal Rate 0–0.5 h (ug [Me] g DW−1 h−1) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cd | Cu | Pb | Zn | |||||
| Bolboschoenus maritimus | 1.1 ± 0.2 | ab | 195 ± 4 | a | 46 ± 6 | ab | 1015 ± 17 | abc |
| Butomus umbellatus | −0.6 ± 0.5 | ab | 258 ± 28 | a | 87 ± 23 | ab | 1956 ± 107 | ab |
| Carex acuta | 0.7 ± 0.3 | ab | −57 ± 4 | a | −26 ± 4 | b | 459 ± 17 | bc |
| Carex cansescens | −4.6 ± 2.1 | b | 592 ± 161 | a | 145 ± 126 | ab | 1534 ± 466 | abc |
| Carex elata | 0.8 ± 0.4 | ab | 89 ± 61 | a | 118 ± 5 | ab | 384 ± 278 | bc |
| Carex flava | −0.5 ± 0.4 | ab | −29 ± 28 | a | 80 ± 27 | ab | 812 ± 73 | abc |
| Carex panicea | 0.3 ± 0.2 | ab | −146 ± 18 | a | 4 ± 6 | ab | 606 ± 180 | abc |
| Carex paniculata | 1.6 ± 0.7 | ab | 156 ± 15 | a | 55 ± 23 | ab | 312 ± 276 | bc |
| Carex pseudocyperus | 0.5 ± 1.8 | ab | 47 ± 22 | a | 32 ± 59 | ab | 155 ± 241 | bc |
| Carex riparia | 1.8 ± 1.8 | ab | 91 ± 431 | a | 69 ± 73 | ab | 282 ± 407 | bc |
| Carex rostrata | 0.8 ± 0.1 | ab | 73 ± 13 | a | 67 ± 12 | ab | 777 ± 55 | abc |
| Carex vesicaria | 0.5 ± 2 | ab | 109 ± 458 | a | 100 ± 71 | ab | 712 ± 218 | abc |
| Comarum palustre | −1.3 ± 0.1 | ab | 147 ± 17 | a | 61 ± 37 | ab | 1011 ± 160 | abc |
| Dryopteris carthusiana | 1.4 ± 0.9 | ab | 47 ± 101 | a | 6 ± 21 | ab | 611 ± 606 | abc |
| Eleocharis mamillata | 0 ± 1.5 | ab | 147 ± 396 | a | 60 ± 10 | ab | 947 ± 775 | abc |
| Eriophorum angustifolium | −3.5 ± 0.3 | b | 592 ± 18 | a | 1 ± 5 | ab | 724 ± 37 | abc |
| Eupatorium cannabinum | −4.9 ± 0.7 | b | −4 ± 70 | a | 71 ± 28 | ab | 121 ± 442 | bc |
| Glyceria maxima | 0.2 ± 0.4 | ab | 44 ± 14 | a | 45 ± 22 | ab | 317 ± 159 | bc |
| Iris pseudocourus | 0.4 ± 0.6 | ab | 37 ± 15 | a | 17 ± 2 | ab | 242 ± 20 | bc |
| Juncus effusus | 0.6 ± 1.1 | ab | 57 ± 28 | a | 60 ± 37 | ab | 148 ± 35 | bc |
| Leymus arenarius | 0.6 ± 0 | ab | 337 ± 4 | a | 215 ± 2 | ab | 736 ± 17 | abc |
| Lycopus europaeus | 0.3 ± 0.1 | ab | 245 ± 16 | a | 252 ± 4 | a | 1854 ± 83 | abc |
| Lysimachia thyrsiflora | −1.0 ± 0 | ab | −47 ± 38 | a | 5 ± 7 | ab | 4 ± 35 | c |
| Lysimachia vulgaris | 3.7 ± 0.8 | a | 467 ± 47 | a | 8 ± 18 | ab | 2440 ± 78 | a |
| Lythrum salicaria | 1.3 ± 0 | ab | 252 ± 13 | a | 106 ± 4 | ab | 1116 ± 51 | abc |
| Molinia caerulea | 0.5 ± 0.5 | ab | 125 ± 22 | a | 41 ± 41 | ab | 149 ± 157 | bc |
| Phalaris arundinacea | 1.2 ± 0.3 | ab | 100 ± 36 | a | 143 ± 21 | ab | 404 ± 145 | bc |
| Phragmites australis | 0.1 ± 0.3 | ab | 80 ± 28 | a | 115 ± 25 | ab | 118 ±25 | bc |
| Schoenoplectus tabernaemontani | 0.7 ± 0.5 | ab | 121 ± 36 | a | 115 ± 42 | ab | 818 ± 57 | abc |
| Scirpus sylvaticus | 0.1 ± 0.6 | ab | 124 ± 54 | a | 115 ± 22 | ab | 440 ± 677 | bc |
| Stachys palustris | 1.2 ± 1 | ab | 143 ± 65 | a | 186 ± 27 | ab | 817 ± 522 | abc |
| Tripolium pannonicum | 0.3 ± 2.8 | ab | 29 ± 19 | a | 32 ± 56 | ab | 249 ± 72 | bc |
| Typha latifolia | −4.9 ± 0.9 | b | 126 ± 40 | a | 29 ± 23 | ab | 1577 ± 73 | abc |
| Veronica beccabunga | 1.3 ± 0.5 | ab | 101 ± 20 | a | 200 ± 3 | ab | 1120 ± 63 | abc |
| Species | Removal Rate 0.5 h–5 days (ug [Me] g DW−1 h−1) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cd | Cu | Pb | Zn | |||||
| Bolboschoenus maritimus | 0 ± 0 | cd | −0.1 ± 0 | a | 0.3 ± 0 | d | 7 ± 0 | def |
| Butomus umbellatus | 0.1 ± 0 | ab | 3 ± 0.1 | a | 3.2 ± 0.1 | ab | 38 ± 0 | abc |
| Carex acuta | 0 ± 0 | bcd | 1.8 ± 0 | a | 1.1 ± 0 | abcd | 15 ± 0 | bcdef |
| Carex cansescens | 0.1 ± 0 | bcd | 2.3 ± 1.3 | a | 2.9 ± 0.9 | abc | 28 ± 7 | bcdef |
| Carex elata | 0 ± 0 | cd | 0.3 ± 0 | a | 0.1 ± 0.1 | d | 7 ± 2 | def |
| Carex flava | 0.1 ± 0 | bcd | 1.7 ± 0.3 | a | 0.8 ± 0.1 | bcd | 21 ± 3 | bcdef |
| Carex panicea | 0 ± 0 | bcd | 1.7 ± 0.7 | a | 0.9 ± 0.3 | bcd | 20 ± 2 | bcdef |
| Carex paniculata | 0 ± 0 | cd | −0.3 ± 0 | a | 0.2 ± 0 | d | 2 ± 2 | ef |
| Carex pseudocyperus | 0 ± 0 | d | 0 ± 0.4 | a | −0.1 ± 0.3 | d | 1 ± 2 | f |
| Carex riparia | 0 ± 0 | cd | 0 ± 0.7 | a | −0.1 ± 0.3 | d | 2 ± 2 | ef |
| Carex rostrata | 0 ± 0 | cd | 0 ± 0 | a | 0.1 ± 0 | d | 6 ± 0 | def |
| Carex vesicaria | 0 ± 0 | cd | 0.3 ± 0.8 | a | 0.1 ± 1.3 | d | 10 ± 5 | bcdef |
| Comarum palustre | 0.1 ± 0 | bcd | 2.2 ± 0.3 | a | 1.4 ± 0.1 | abcd | 40 ± 3 | ab |
| Dryopteris carthusiana | 0 ± 0 | cd | 0.2 ± 0.5 | a | 0.3 ± 0.1 | d | 5 ± 2 | def |
| Eleocharis mamillata | 0 ± 0 | bcd | 0.7 ± 1.8 | a | 1.6 ± 0.9 | abcd | 24 ± 17 | bcdef |
| Eriophorum angustifolium | 0 ± 0 | bcd | 1.1 ± 0.3 | a | 2.9 ± 0.2 | abc | 22 ± 5 | bcdef |
| Eupatorium cannabinum | 0.1 ± 0 | bcd | 1.6 ± 0.8 | a | 0.8 ± 0.4 | bcd | 22 ± 9 | bcdef |
| Glyceria maxima | 0 ± 0 | d | 0 ± 0.2 | a | −0.1 ± 0.2 | d | 2 ± 3 | ef |
| Iris pseudocourus | 0 ± 0 | cd | 0.2 ± 0.1 | a | 0.2 ± 0.1 | d | 4 ± 2 | ef |
| Juncus effusus | 0 ± 0 | cd | 0 ± 0.4 | a | 0 ± 0.3 | d | 2 ± 2 | ef |
| Leymus arenarius | 0.1 ± 0 | bcd | 2.3 ± 0 | a | 1.7 ± 0 | abcd | 31 ± 0 | bcde |
| Lycopus europaeus | 0.1 ± 0 | ab | 2.7 ± 0.2 | a | 2.1 ± 0.2 | abcd | 35 ± 2 | abcd |
| Lysimachia thyrsiflora | 0.2 ± 0 | bcd | 1.9 ± 0.1 | a | 3.6 ± 0 | cd | 63 ± 0 | def |
| Lysimachia vulgaris | 0 ± 0 | a | 1 ± 0.1 | a | 0.5 ± 0.1 | a | 8 ± 1 | a |
| Lythrum salicaria | 0 ± 0 | bcd | 0.1 ± 0.1 | a | 0.5 ± 0 | cd | 20 ± 1 | bcdef |
| Molinia caerulea | 0 ± 0 | cd | −0.1 ± 0.1 | a | 0 ± 0.1 | d | 2 ± 1 | ef |
| Phalaris arundinacea | 0 ± 0 | cd | 0.1 ± 0.1 | a | −0.1 ± 0.1 | d | cdef | |
| Phragmites australis | 0 ± 0 | bcd | 1.1 ± 0.2 | a | 0.5 ± 0.2 | cd | 14 ± 1 | bcdef |
| Schoenoplectus tabernaemontani | 0 ± 0 | cd | 0.4 ± 0.1 | a | 0.2 ± 0 | d | 12 ± 1 | bcdef |
| Scirpus sylvaticus | 0 ± 0 | cd | 0.2 ± 0.6 | a | 0.1 ± 0.6 | d | 8 ± 4 | def |
| Stachys palustris | 0 ± 0 | bcd | 0.3 ± 0.3 | a | 0 ± 0.5 | d | 10 ± 4 | cdef |
| Tripolium pannonicum | 0 ± 0 | cd | 0.8 ± 0.7 | a | 0.4 ± 0.3 | cd | 8 ± 9 | def |
| Typha latifolia | 0.1 ± 0 | abc | 1.4 ± 0.1 | a | 1.6 ± 0 | abcd | 26 ± 2 | bcdef |
| Veronica beccabunga | 0.1 ± 0 | bcd | 1.3 ± 0.2 | a | 0.5 ± 0.1 | cd | 21 ± 2 | bcdef |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schück, M.; Greger, M. Screening the Capacity of 34 Wetland Plant Species to Remove Heavy Metals from Water. Int. J. Environ. Res. Public Health 2020, 17, 4623. https://doi.org/10.3390/ijerph17134623
Schück M, Greger M. Screening the Capacity of 34 Wetland Plant Species to Remove Heavy Metals from Water. International Journal of Environmental Research and Public Health. 2020; 17(13):4623. https://doi.org/10.3390/ijerph17134623
Chicago/Turabian StyleSchück, Maria, and Maria Greger. 2020. "Screening the Capacity of 34 Wetland Plant Species to Remove Heavy Metals from Water" International Journal of Environmental Research and Public Health 17, no. 13: 4623. https://doi.org/10.3390/ijerph17134623
APA StyleSchück, M., & Greger, M. (2020). Screening the Capacity of 34 Wetland Plant Species to Remove Heavy Metals from Water. International Journal of Environmental Research and Public Health, 17(13), 4623. https://doi.org/10.3390/ijerph17134623





