Effects of Manual Therapy on Fatigue, Pain, and Psychological Aspects in Women with Fibromyalgia
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Declarations: Ethics Approval, Consent to Participate, and Consent for Publication
2.4. Eligibility Criteria
2.5. Study Intervention
2.6. Outcome Measurements
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chinn, S.; Caldwell, W.; Gritsenko, K. Fibromyalgia Pathogenesis and Treatment Options Update. Curr. Pain Headache Rep. 2016, 20, 25. [Google Scholar] [CrossRef]
- Häuser, W.; Brähler, E.; Ablin, J.; Wolfe, F. 2016 modified American College of Rheumatology fibromyalgia criteria, ACTTION-APS Pain Taxonomy criteria and the prevalence of fibromyalgia. Arthritis Care Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.A.; Goldenberg, D.L.; Häuser, W.; Katz, R.L.; Mease, P.J.; Russell, A.S.; Russell, I.J.; Walitt, B. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin. Arthritis Rheum. 2016, 46, 319–329. [Google Scholar] [CrossRef]
- Jones, G.T.; Atzeni, F.; Beasley, M.; Flüß, E.; Sarzi-Puttini, P.; Macfarlane, G.J. The prevalence of fibromyalgia in the general population: A comparison of the American College of Rheumatology 1990, 2010, and modified 2010 classification criteria. Arthritis Rheumatol. 2015, 67, 568–575. [Google Scholar] [CrossRef] [PubMed]
- White, H.D.; Robinson, T.D. A novel use for testosterone to treat central sensitization of chronic pain in fibromyalgia patients. Int. Immunopharmacol. 2015, 27, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Okifuji, A.; Bradshaw, D.H.; Donaldson, G.W.; Turk, D.C. Sequential analyses of daily symptoms in women with fibromyalgia syndrome. J. Pain 2011, 12, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.F.; Tomassi, S.; Russell, A.; Mondelli, V.; Pariante, C.M. Clinical Challenges in the Biopsychosocial Interface. Adv. Psychosom. Med. 2015, 34, 61–77. [Google Scholar] [PubMed]
- Dailey, D.L.; Keffala, V.J.; Sluka, K.A. Do cognitive and physical fatigue tasks enhance pain, cognitive fatigue, and physical fatigue in people with fibromyalgia? Arthritis Care Res. 2015, 67, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Dailey, D.L.; A Frey-Law, L.; Vance, C.; Rakel, B.A.; Merriwether, E.N.; Darghosian, L.; Golchha, M.; Geasland, K.M.; Spitz, R.; Crofford, L.J.; et al. Perceived function and physical performance are associated with pain and fatigue in women with fibromyalgia. Arthritis Res. Ther. 2016, 18, 68. [Google Scholar] [CrossRef]
- Bennett, R.M.; Jones, J.; Turk, D.C.; Russell, I.J.; Matallana, L. An internet survey of 2,596 people with fibromyalgia. BMC Musculoskelet. Disord. 2007, 8, 27. [Google Scholar] [CrossRef]
- Wu, Y.L.; Chang, L.Y.; Lee, H.C.; Fang, S.C.; Tsai, P.S. Sleep disturbances in fibromyalgiaA meta-analysis of case-control studies. J. Psychosom. Res. 2017, 96, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, 2–15. [Google Scholar] [CrossRef]
- Góes, S.M.; Leite, N.; Shay, B.L.; Homann, D.; Stefanello, J.M.F.; Rodacki, A.L.F. Functional capacity, muscle strength and falls in women with fibromyalgia. Clin. Biomech. 2012, 27, 578–583. [Google Scholar] [CrossRef]
- Écija, C.; Luque-Reca, O.; Suso-Ribera, C.; Catala, P.; Peñacoba, C. Associations of Cognitive Fusion and Pain Catastrophizing with Fibromyalgia Impact through Fatigue, Pain Severity, and Depression: An Exploratory Study Using Structural Equation Modeling. J. Clin. Med. 2020, 9, 1763. [Google Scholar] [CrossRef]
- Choy, E.H.S. The role of sleep in pain and fibromyalgia. Nat. Rev. Rheumatol. 2015, 11, 513–520. [Google Scholar] [CrossRef]
- Mork, P.J.; Nilsen, T.I.L. Sleep problems and risk of fibromyalgia: Longitudinal data on an adult female population in Norway. Arthritis Rheum. 2012, 64, 281–284. [Google Scholar] [CrossRef]
- Spaeth, M.; Rizzi, M.; Sarzi-Puttini, P. Fibromyalgia and sleep. Best Practice and Research: Clin Rheumatol. 2011, 25, 227–239. [Google Scholar] [CrossRef]
- Choy, E.H. Current treatments to counter sleep dysfunction as a pathogenic stimulus of fibromyalgia. Pain Manag. 2016, 6, 339–346. [Google Scholar] [CrossRef]
- Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Carbonell-Baeza, A.; Aparicio, V.A.; Ortega, F.B.; Casimiro, A.J.; Delgado-Fernández, M. Fibromyalgia has a larger impact on physical health than on psychological health, yet both are markedly affected: The al-Ándalus project. Semin. Arthritis Rheum. 2015, 44, 563–570. [Google Scholar] [CrossRef]
- Peñacoba Puente, C.; Velasco Furlong, L.; Écija Gallardo, C.; Cigarán Méndez, M.; McKenney, K. Anxiety, Depression and Alexithymia in Fibromyalgia: Are There Any Differences According to Age? J. Women Aging 2013, 25, 305–320. [Google Scholar] [CrossRef]
- Mercieca, C.; Borg, A.A. EULAR recommendations underplay importance of severe anxiety and depression in fibromyalgia treatment. Ann. Rheum. Dis. 2017, 76, 53. [Google Scholar] [CrossRef]
- Córdoba-Torrecilla, S.; Aparicio, V.A.; Soriano-Maldonado, A.; Estévez-López, F.; Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Femia-Marzo, P.; Delgado-Fernández, M. Physical fitness is associated with anxiety levels in women with fibromyalgia: The al-Ándalus project. Qual. Life Res. 2016, 25, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Kilic, O.; Sar, V.; Taycan, O.; Poyraz, C.A.; Erol, T.C.; Tecer, Ö.; Emul, M.H.; Özmen, M. Dissociative Depression among Women with Fibromyalgia or Rheumatoid Arthritis. J. Trauma Dissociation 2014, 15, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Pulido-Martos, M.; Luque-Reca, O.; Segura-Jiménez, V.; Álvarez-Gallardo, I.C.; Soriano-Maldonado, A.; Acosta-Manzano, P.; Gavilán-Carrera, B.; McVeigh, J.G.; Geenen, R.; Delgado-Fernández, M.; et al. Physical and psychological paths toward less severe fibromyalgia: A estructural equation model. Ann. Phys. Rehabil. Med. 2020, 63, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Ángel García, D.; Martínez Nicolás, I.; Saturno Hernández, P.J. Abordaje clínico de la fibromialgia: Síntesis de recomendaciones basadas en la evidencia, una revisión sistemática. Reumatol. Clin. 2016, 12, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Valentini, E.; Fetter, E.; Orbell, S. Treatment preferences in fibromyalgia patients: A cross-sectional web-based survey. Eur. J. Pain 2020. [Google Scholar] [CrossRef]
- Diaz-Piedra, C.; Catena, A.; Sánchez, A.I.; Miró, E.; Pilar Martínez, M.; Buela-Casal, G. Sleep disturbances in fibromyalgia syndrome: The role of clinical and polysomnographic variables explaining poor sleep quality in patients. Sleep Med. 2015, 16, 917–925. [Google Scholar] [CrossRef]
- Kalichman, L. Massage therapy for fibromyalgia symptoms. Rheumatol. Int. 2010, 30, 1151–1157. [Google Scholar] [CrossRef]
- Arnold, L.M.; Crofford, L.J.; Mease, P.J.; Burgess, S.M.; Palmer, S.C.; Abetz, L.; Martin, S.A. Patient perspectives on the impact of fibromyalgia. Patient Educ. Couns. 2008, 73, 114–120. [Google Scholar] [CrossRef]
- Bervoets, D.C.; Luijsterburg, P.A.J.; Alessie, J.J.N.; Buijs, M.J.; Verhagen, A.P. Massage therapy has short-term benefits for people with common musculoskeletal disorders compared to no treatment: A systematic review. J. Physiother. 2015, 61, 106–116. [Google Scholar] [CrossRef]
- Roberts, M.A. CMTL Effects of Patterns of Pressure Application on Resting Electromyography during Massage. Int. J. Ther. Massag. Bodyw. 2011, 30, 4. [Google Scholar]
- Cimmino, M.A.; Grassi, W.; Cutolo, M. Best practice & research clinical rheumatology. Imaging and musculoskeletal conditions. Preface. Best Pract. Res. Clin. Rheumatol. 2008, 22, 949. [Google Scholar] [PubMed]
- Yuan, S.L.K.; Matsutani, L.A.; Marques, A.P. Effectiveness of different styles of massage therapy in fibromyalgia: A systematic review and meta-analysis. Man. Ther. 2015, 20, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Brattberg, G. Connective tissue massage in the treatment of fibromyalgia. Eur. J. Pain 1999, 3, 235–244. [Google Scholar] [CrossRef]
- Ekici, G.; Bakar, Y.; Akbayrak, T.; Yuksel, I. Comparison of Manual Lymph Drainage Therapy and Connective Tissue Massage in Women with Fibromyalgia: A Randomized Controlled Trial. J. Manip. Physiol. Ther. 2009, 32, 127–133. [Google Scholar] [CrossRef]
- Saghaei, M. An overview of randomization and minimization programs for randomized clinical trials. J. Med. Signals Sens. 2011, 1, 55–61. [Google Scholar] [CrossRef]
- Diego, M.A.; Field, T. Moderate pressure massage elicits a parasympathetic nervous system response. Int. J. Neurosci. 2009, 119, 630–638. [Google Scholar] [CrossRef]
- Krupp, L.B.; Larocca, N.G.; Muir Nash, J.; Steinberg, A.D. The fatigue severity scale: Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch. Neurol. 1989, 46, 1121–1123. [Google Scholar] [CrossRef]
- Takasaki, H.; Treleaven, J. Construct Validity and Test-Retest Reliability of the Fatigue Severity Scale in People with Chronic Neck Pain. Arch. Phys. Med. Rehabil. 2013, 94, 1328–1362. [Google Scholar] [CrossRef]
- Labronici, P.J.; Dos Santos-Viana, A.M.; Dos Santos-Filho, F.C.; Santos-Pires, R.E.; Labronici, G.J.; Penteado-da Silva, L.H. Evaluation of the pain in older people. Acta Ortop. Mex. 2016, 30, 73–80. [Google Scholar]
- Marques, A.P.; Assumpção, A.; Matsutani, L.A.; Bragança Pereira, C.A.; Lage, L. Pain in fibromyalgia and discriminativen power of the instruments: Visual Analog Scale. Dolorimetry and the McGill Pain Questionnaire. Acta Reumatol. Port. 2008, 33, 345–351. [Google Scholar] [PubMed]
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale. Arthritis Care Res. 2011, 63, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, M.B.; Quaresma, M.R.; Aquino, L.R.L.; Atra, E.; Tugwell, P.; Goldsmith, C.H. Reliability of pain scales in the assessment of literate and illiterate patients with rheumatoid arthritis. J. Rheumatol. 1990, 17, 1022–1024. [Google Scholar]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Escobar-Córdoba, F.; Eslava-Schmalbach, J. Colombian validation of the Pittsburgh Sleep Quality Index. Rev. Neurol. 2005, 40, 150–155. [Google Scholar] [PubMed]
- Smyth, M.S.N.C. The Pittsburgh Sleep Quality Index (PSQI). Insight 2000, 25, 97–98. [Google Scholar]
- Lin, S.; Hsiao, Y.-Y.; Wang, M. Test Review: The Profile of Mood States 2nd Edition. J. Psychoeduc. Assess. 2014, 32, 273–277. [Google Scholar] [CrossRef]
- Andrade, E.; Arce, C.; De Francisco, C.; Torrado, J.; Garrido, J. Versión breve en español del cuestionario POMS para deportistas adultos y población general. Rev. Psicol. Deporte 2013, 22, 95–102. [Google Scholar]
- Profile of Mood States (POMS)-Statistics Solutions; Multi-Health Systems Inc.: Toronto, ON, Canada, 2003.
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Academic Press: Cambridge, MA, USA, 1977; p. 474. [Google Scholar]
- Bernik, M.; Sampaio, T.P.A.; Gandarela, L. Fibromyalgia comorbid with anxiety disorders and depression: Combined medical and psychological treatment. Curr. Pain Headache Rep. 2013, 17, 358. [Google Scholar] [CrossRef]
- Bosch Romero, E.; Sáenz Moya, N.; Valls Esteve, M.; Viñolas Valer, S. Study of quality of life of patients with fibromyalgia: Impact of a health education programme. Atención Primaria 2002, 30, 16–21. [Google Scholar] [CrossRef]
- Brummitt, J. The role of massage in sports performance and rehabilitation: Current evidence and future direction. N. Am. J. Sports Phys. Ther. 2008, 3, 7–21. [Google Scholar]
- Netchanok, S.; Wendy, M.; Marie, C.; Siobhan, O. The effectiveness of Swedish massage and traditional Thai massage in treating chronic low back pain: A review of the literature. Complement. Ther. Clin. Pr. 2012, 18, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Weerapong, P.; Hume, P.A.; Kolt, G.S. The mechanisms of massage and effects on performance, muscle recovery and injury prevention. Sports Med. 2005, 35, 235–256. [Google Scholar] [CrossRef] [PubMed]
- Field, T.; Diego, M.; Hernandez-Reif, M. Moderate pressure is essential for massage therapy effects. Int. J. Neurosci. 2010, 120, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Bourke, J. Fibromyalgia and chronic fatigue syndrome: Management issues. Adv. Psychosom. Med. 2015, 34, 78–91. [Google Scholar] [PubMed]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. Pilot Feasibility Study 2016, 2, 1–32. [Google Scholar] [CrossRef]
- Holdcraft, L.C.; Assefi, N.; Buchwald, D. Complementary and alternative medicine in fibromyalgia and related syndromes. Best Pr. Res. Clin. Rheumatol. 2003, 17, 667–683. [Google Scholar] [CrossRef]
- De Oliveira, F.R.; Gonçalves, L.C.V.; Borghi, F.; Da Silva, L.G.R.V.; Gomes, A.E.; Trevisan, G.; De Souza, A.L.; Grassi-Kassisse, D.M.; Crege, D.R.X.D.O. Massage therapy in cortisol circadian rhythm, pain intensity, perceived stress index and quality of life of fibromyalgia syndrome patients. Complement. Ther Clin. Pract. 2018, 30, 85–90. [Google Scholar] [CrossRef]
- Goldenberg, D.L.; Bradley, L.A.; Arnold, L.M.; Glass, J.M.; Clauw, D.J. Understanding fibromyalgia and its related disorders. Prim. Care Companion J. Clin. Psychiatry 2008, 10, 133–144. [Google Scholar]
- Elbinoune, I.; Amine, B.; Shyen, S.; Gueddari, S.; Abouqal, R.; Hajjaj-Hassouni, N. Chronic neck pain and anxiety-depression: Prevalence and associated risk factors. Pan Afr. Med. J. 2016, 24. [Google Scholar] [CrossRef]
- Juan, W.; Rui, L.; Wei-Wen, Z. Chronic neck pain and depression: The mediating role of sleep quality and exercise. Psychol. Health Med. 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Von Korff, M.; Dworkin, S.F.; Le Resche, L.; Kruger, A. An epidemiologic comparison of pain complaints. Pain 1988, 32, 73–83. [Google Scholar] [CrossRef]
- Linton, S.J. A review of psychological risk factors in back and neck pain. Depression and anxiety as major determinants of neck pain: A cross-sectional study in general practice. Spine 2000, 25, 1148–1156. [Google Scholar] [CrossRef] [PubMed]
- Blozik, E.; Laptinskaya, D.; Herrmann-Lingen, C.; Schaefer, H.; Kochen, M.M.; Himmel, W.; Scherer, M. Depression and anxiety as major determinants of neck pain: A cross-sectional study in general practice. BMC Musculoskelet. Disord. 2009, 10. [Google Scholar] [CrossRef] [PubMed]
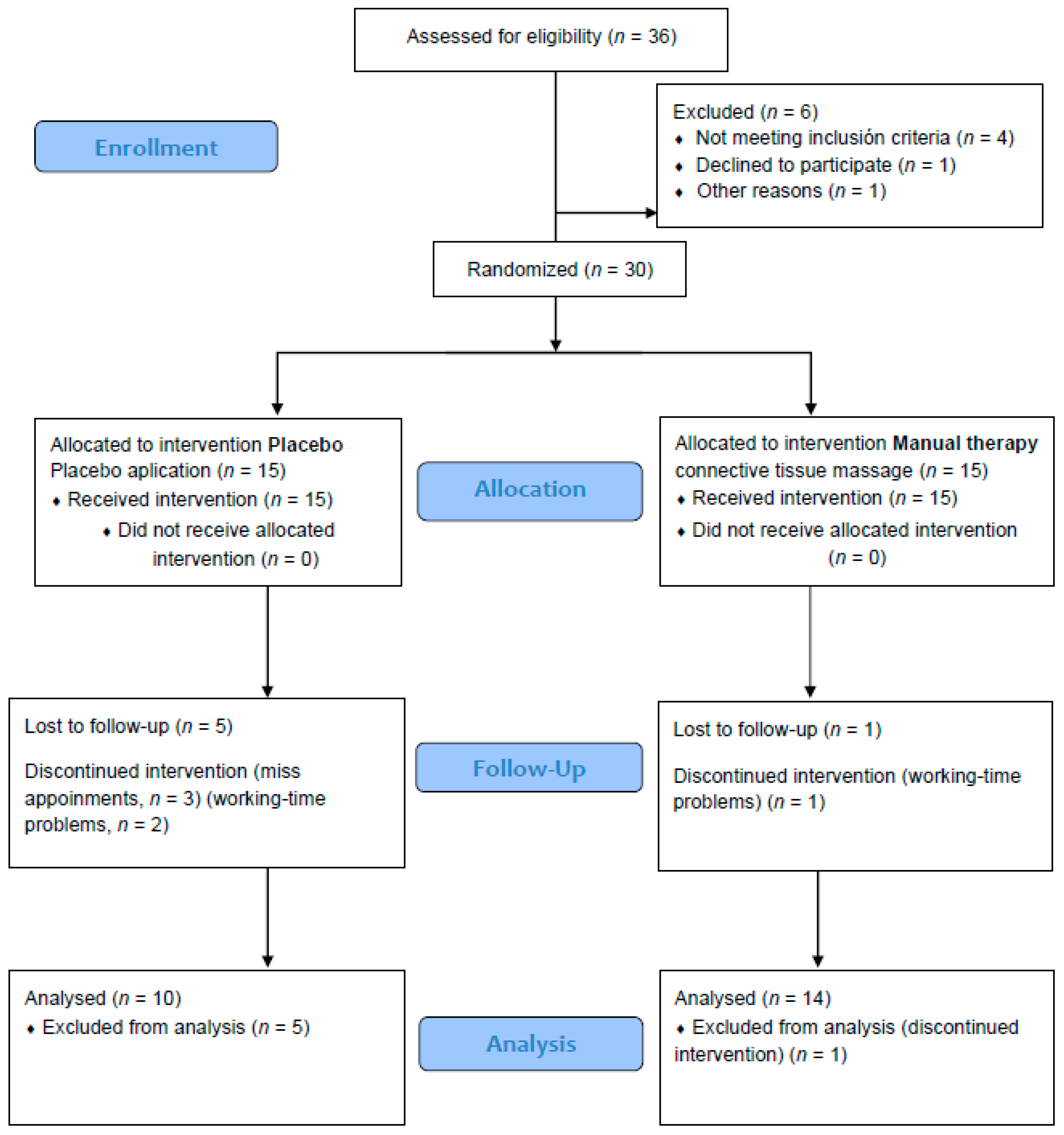
| Variables | Placebo (n = 10) | Manual Therapy (n = 14) | Basal Differences | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Basal | Post | Basal | Post | ||||||||
| Mean | SD 1 | Mean | SD | Mean | SD | Mean | SD | t | p | ES | |
| Height (m) | 1.6 | 0.1 | 1.5 | 0.1 | 0.768 | 0.451 | 0.318 | ||||
| Weight (kg) | 70.7 | 11.0 | 68.2 | 11.7 | 0.528 | 0.603 | 0.219 | ||||
| BMI (kg/m2) | 28.7 | 4.1 | 28.4 | 4.3 | 0.146 | 0.886 | 0.06 | ||||
| Fatigue (FSS) | 4.7 | 0.8 | 5.3 | 0.7 | 3.8 | 1.0 | 4.6 | 0.7 | 2.213 | 0.038 * | 0.916 |
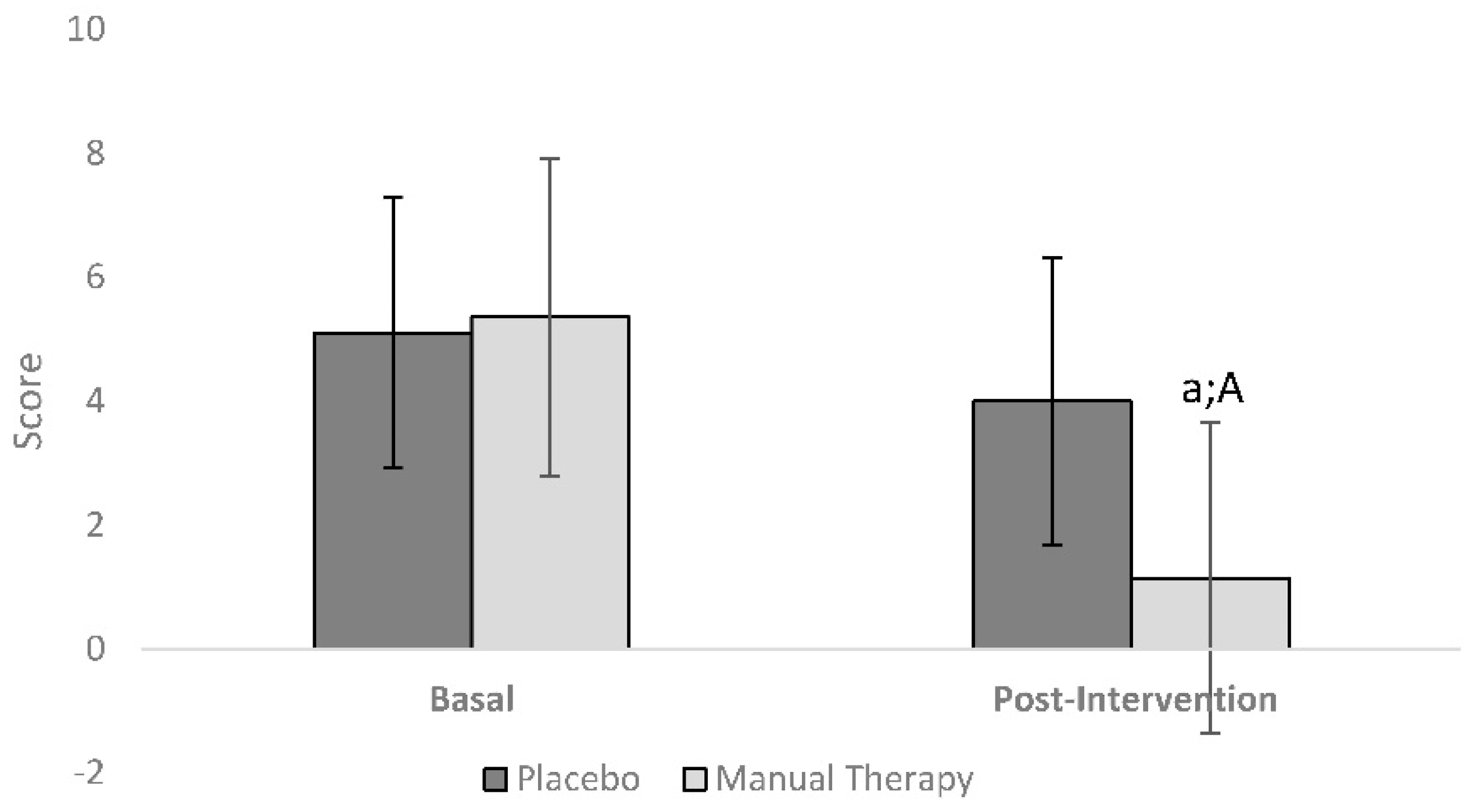
| Pain (VAS) | 5.1 | 2.2 | 4.0 | 2.3 | 5.4 | 2.6 | 1.1 | 2.5 | −0.257 | 0.799 | −0.107 |
| Sleep (Pittsburgh test) | 11.3 | 2.7 | 10.3 | 2.7 | 9.6 | 4.1 | 7.9 | 3.1 | 1.159 | 0.259 | 0.48 |
| POMS-29 total | 138.2 | 13.0 | 128.1 | 10.5 | 131.4 | 17.3 | 120.9 | 16.3 | 1.042 | 0.309 | 0.432 |
| Stress–Anxiety | 10.9 | 4.4 | 8.3 | 3.5 | 11.0 | 5.2 | 6.0 | 2.7 | −0.050 | 0.961 | −0.021 |
| Depression–Melancholy | 6.0 | 2.5 | 4.0 | 3.6 | 6.3 | 4.7 | 4.2 | 4.4 | −0.174 | 0.863 | −0.072 |
| Anger–Hostility | 14.1 | 5.5 | 11.3 | 3.7 | 10.9 | 5.4 | 7.1 | 3.9 | 1.060 | 0.146 | 0.624 |
| Vigor | 6.3 | 3.0 | 7.4 | 4.1 | 8.4 | 5.5 | 7.3 | 5.4 | −1.108 | 0.280 | −0.459 |
| Fatigue | 13.5 | 2.8 | 11.7 | 4.5 | 11.8 | 4.3 | 9.1 | 4.1 | 1.105 | 0.281 | 0.458 |
| Variable | Effect Time | Effect Time × Group | ||||
|---|---|---|---|---|---|---|
| F1 | p | η2 | F | p | η2 | |
| Fatigue (FSS) | 0.899 | 0.354 | 0.013 | 0.352 | 0.559 | 0.005 |
| Pain (VAS) | 0.035 | 0.854 | 0.000 | 23.635 | <0.001 * | 0.093 |
| Sleep (Pittsburgh test) | 0.479 | 0.496 | 0.004 | 0.288 | 0.597 | 0.003 |
| POMS total | 1.187 | 0.288 | 0.009 | 0.020 | 0.890 | 0.000 |
| Stress–Anxiety | 0.005 | 0.945 | 0.000 | 1.580 | 0.222 | 0.023 |
| Depression–Melancholy | 0.224 | 0.641 | 0.001 | 0.007 | 0.936 | 0.000 |
| Anger–Hostility | 0.585 | 0.453 | 0.008 | 0.380 | 0.544 | 0.002 |
| Vigor | 2.902 | 0.103 | 0.017 | 2.269 | 0.147 | 0.013 |
| Fatigue | 1.256 | 0.275 | 0.008 | 0.380 | 0.544 | 0.002 |
| Variables | Statistics | FSS 1 Fatigue | Pain | Sleep | POMS Total | Stress–Anxiety | Depression | Anger–Hostility | Vigor |
|---|---|---|---|---|---|---|---|---|---|
| Pain | Pearson (R) | −0.344 | |||||||
| p | 0.099 | ||||||||
| Sleep | Pearson (R) | 0.411 * | 0.323 | ||||||
| p | 0.046 | 0.124 | |||||||
| POMS Total | Pearson (R) | 0.073 | 0.253 | −0.019 | |||||
| p | 0.736 | 0.232 | 0.930 | ||||||
| Stress–Anxiety | Pearson (R) | 0.007 | 0.114 | −0.042 | 0.664 ** | ||||
| p | 0.992 | 0.597 | 0.845 | 0.000 | |||||
| Depression | Pearson (R) | 0.106 | −0.055 | −0.015 | 0.794 ** | 0.415 * | |||
| p | 0.623 | 0.799 | 0.945 | 0.000 | 0.044 | ||||
| Anger–Hostility | Pearson (R) | 0.091 | 0.436 * | 0.062 | 0.795 ** | 0.590 ** | 0.406 | ||
| p | 0.672 | 0.033 | 0.773 | 0.000 | 0.002 | 0.049 | |||
| Vigor | Pearson (R) | −0.065 | −0.157 | 0.122 | −0.656 ** | 0.010 | −0.585 ** | −0.376 | |
| p | 0.763 | 0.463 | 0.570 | 0.000 | 0.964 | 0.003 | 0.070 | ||
| Fatigue | Pearson (R) | −0.017 | 0.174 | 0.027 | 0.772 ** | 0.521 ** | 0.585 ** | 0.414 * | −0.427 * |
| p | 0.938 | 0.415 | 0.901 | 0.000 | 0.009 | 0.003 | 0.044 | 0.038 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nadal-Nicolás, Y.; Rubio-Arias, J.Á.; Martínez-Olcina, M.; Reche-García, C.; Hernández-García, M.; Martínez-Rodríguez, A. Effects of Manual Therapy on Fatigue, Pain, and Psychological Aspects in Women with Fibromyalgia. Int. J. Environ. Res. Public Health 2020, 17, 4611. https://doi.org/10.3390/ijerph17124611
Nadal-Nicolás Y, Rubio-Arias JÁ, Martínez-Olcina M, Reche-García C, Hernández-García M, Martínez-Rodríguez A. Effects of Manual Therapy on Fatigue, Pain, and Psychological Aspects in Women with Fibromyalgia. International Journal of Environmental Research and Public Health. 2020; 17(12):4611. https://doi.org/10.3390/ijerph17124611
Chicago/Turabian StyleNadal-Nicolás, Yolanda, Jacobo Ángel Rubio-Arias, María Martínez-Olcina, Cristina Reche-García, María Hernández-García, and Alejandro Martínez-Rodríguez. 2020. "Effects of Manual Therapy on Fatigue, Pain, and Psychological Aspects in Women with Fibromyalgia" International Journal of Environmental Research and Public Health 17, no. 12: 4611. https://doi.org/10.3390/ijerph17124611
APA StyleNadal-Nicolás, Y., Rubio-Arias, J. Á., Martínez-Olcina, M., Reche-García, C., Hernández-García, M., & Martínez-Rodríguez, A. (2020). Effects of Manual Therapy on Fatigue, Pain, and Psychological Aspects in Women with Fibromyalgia. International Journal of Environmental Research and Public Health, 17(12), 4611. https://doi.org/10.3390/ijerph17124611







