Identification of Time-Invariant Biomarkers for Non-Genotoxic Hepatocarcinogen Assessment
Abstract
1. Introduction
2. Materials and Methods
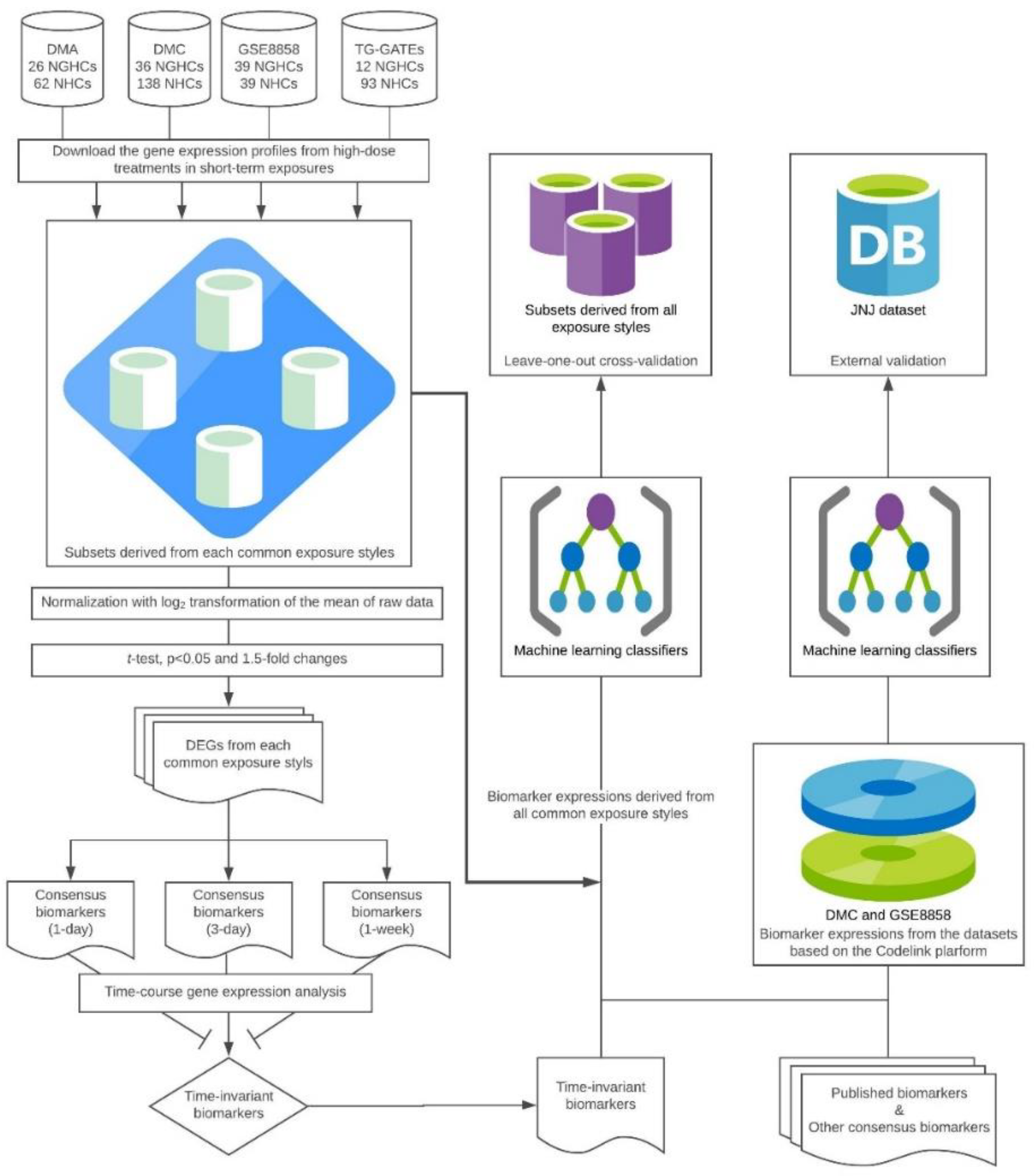
2.1. Chemical List and Microarray Datasets
2.2. Identification of the Time-Invariant Biomarker Sets
2.3. Model Development
2.4. External Validation
2.5. Enrichment Analysis
3. Results and Discussion
3.1. Time-Invariant Biomarkers and Machine Learning Classifiers
3.2. External Validation
3.3. Analysis of the Time-Invariant Biomarkers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Wogan, G.N. Impacts of chemicals on liver cancer risk. Semin. Cancer Biol. 2000, 10, 201–210. [Google Scholar] [CrossRef]
- Santos, N.P.; Colaco, A.A.; Oliveira, P.A. Animal models as a tool in hepatocellular carcinoma research: A Review. Tumour Biol. J. Int. Soc. Oncodevelopmental Biol. Med. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Dieter, S. What is the meaning of ‘A compound is carcinogenic’? Toxicol. Rep. 2018, 5, 504–511. [Google Scholar] [CrossRef]
- Butterworth, B.E. Consideration of both genotoxic and nongenotoxic mechanisms in predicting carcinogenic potential. Mutat. Res. 1990, 239, 117–132. [Google Scholar] [CrossRef]
- Plant, N. Can systems toxicology identify common biomarkers of non-genotoxic carcinogenesis? Toxicology 2008, 254, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.D.; Jackson, M.; Lea, I. Characterizing and predicting carcinogenicity and mode of action using conventional and toxicogenomics methods. Mutat. Res. 2010, 705, 184–200. [Google Scholar] [CrossRef]
- Schaap, M.M.; Wackers, P.F.; Zwart, E.P.; Huijskens, I.; Jonker, M.J.; Hendriks, G.; Breit, T.M.; van Steeg, H.; van de Water, B.; Luijten, M. A novel toxicogenomics-based approach to categorize (non-)genotoxic carcinogens. Arch. Toxicol. 2015, 89, 2413–2427. [Google Scholar] [CrossRef]
- Fielden, M.R.; Brennan, R.; Gollub, J. A gene expression biomarker provides early prediction and mechanistic assessment of hepatic tumor induction by nongenotoxic chemicals. Toxicol. Sci. Off. J. Soc. Toxicol. 2007, 99, 90–100. [Google Scholar] [CrossRef]
- Fielden, M.R.; Adai, A.; Dunn, R.T., II; Olaharski, A.; Searfoss, G.; Sina, J.; Aubrecht, J.; Boitier, E.; Nioi, P.; Auerbach, S.; et al. Development and evaluation of a genomic signature for the prediction and mechanistic assessment of nongenotoxic hepatocarcinogens in the rat. Toxicol. Sci. Off. J. Soc. Toxicol. 2011, 124, 54–74. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Kelly, R.; Fang, H.; Ding, D.; Tong, W. Comparative analysis of predictive models for nongenotoxic hepatocarcinogenicity using both toxicogenomics and quantitative structure-activity relationships. Chem. Res. Toxicol. 2011, 24, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Uehara, T.; Ono, A.; Maruyama, T.; Kato, I.; Yamada, H.; Ohno, Y.; Urushidani, T. The Japanese toxicogenomics project: Application of toxicogenomics. Mol. Nutr. Food Res. 2010, 54, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Eichner, J.; Wrzodek, C.; Romer, M.; Ellinger-Ziegelbauer, H.; Zell, A. Evaluation of toxicogenomics approaches for assessing the risk of nongenotoxic carcinogenicity in rat liver. PLoS ONE 2014, 9, e97678. [Google Scholar] [CrossRef] [PubMed]
- Uehara, T.; Minowa, Y.; Morikawa, Y.; Kondo, C.; Maruyama, T.; Kato, I.; Nakatsu, N.; Igarashi, Y.; Ono, A.; Hayashi, H.; et al. Prediction model of potential hepatocarcinogenicity of rat hepatocarcinogens using a large-scale toxicogenomics database. Toxicol. Appl. Pharmacol. 2011, 255, 297–306. [Google Scholar] [CrossRef]
- Liu, Y.F.; Zha, B.S.; Zhang, H.L.; Zhu, X.J.; Li, Y.H.; Zhu, J.; Guan, X.H.; Feng, Z.Q.; Zhang, J.P. Characteristic gene expression profiles in the progression from liver cirrhosis to carcinoma induced by diethylnitrosamine in a rat model. J. Exp. Clin. Cancer Res. CR 2009, 28, 107. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.H.; Tung, C.W. Identification of consensus biomarkers for predicting non-genotoxic hepatocarcinogens. Sci. Rep. 2017, 7, 41176. [Google Scholar] [CrossRef] [PubMed]
- Uehara, T.; Hirode, M.; Ono, A.; Kiyosawa, N.; Omura, K.; Shimizu, T.; Mizukawa, Y.; Miyagishima, T.; Nagao, T.; Urushidani, T. A toxicogenomics approach for early assessment of potential non-genotoxic hepatocarcinogenicity of chemicals in rats. Toxicology 2008, 250, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Nicolaidou, V.; Koufaris, C. Application of transcriptomic and microRNA profiling in the evaluation of potential liver carcinogens. Toxicol. Ind. Health 2020. [Google Scholar] [CrossRef]
- Tung, C.-W.; Jheng, J.-L. Interpretable prediction of non-genotoxic hepatocarcinogenic chemicals. Neurocomputing 2014, 145, 68–74. [Google Scholar] [CrossRef]
- Ganter, B.; Tugendreich, S.; Pearson, C.I.; Ayanoglu, E.; Baumhueter, S.; Bostian, K.A.; Brady, L.; Browne, L.J.; Calvin, J.T.; Day, G.J.; et al. Development of a large-scale chemogenomics database to improve drug candidate selection and to understand mechanisms of chemical toxicity and action. J. Biotechnol. 2005, 119, 219–244. [Google Scholar] [CrossRef]
- Quinlan, J.R. C4.5: Programs for Machine Learning; Morgan Kaufmann Publishers Inc.: Burlington, MA, USA, 1993. [Google Scholar]
- Breiman, L. Bagging predictors. Mach. Learn. 1996, 24, 123–140. [Google Scholar] [CrossRef]
- Schapire Robert, E.; Freund, Y. Boosting: Foundations and Algorithms. Kybernetes 2013, 42, 164–166. [Google Scholar] [CrossRef]
- Keller, J.M.; Gray, M.R.; Givens, J.A. A fuzzy K-nearest neighbor algorithm. IEEE Trans. Syst. ManCybern. 1985, SMC-15, 580–585. [Google Scholar] [CrossRef]
- Rish, I. An empirical study of the naive Bayes classifier. In Proceedings of the IJCAI 2001 Workshop on Empirical Methods in Artificial Intelligence, Seattle, WA, USA, 4 August 2001; pp. 41–46. [Google Scholar]
- Noble, W.S. What is a support vector machine? Nat. Biotechnol. 2006, 24, 1565–1567. [Google Scholar] [CrossRef] [PubMed]
- Breiman, L.J.M.l. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Nie, A.Y.; McMillian, M.; Parker, J.B.; Leone, A.; Bryant, S.; Yieh, L.; Bittner, A.; Nelson, J.; Carmen, A.; Wan, J.; et al. Predictive toxicogenomics approaches reveal underlying molecular mechanisms of nongenotoxic carcinogenicity. Mol. Carcinog. 2006, 45, 914–933. [Google Scholar] [CrossRef] [PubMed]
- Lea, I.A.; Gong, H.; Paleja, A.; Rashid, A.; Fostel, J. CEBS: A comprehensive annotated database of toxicological data. Nucleic Acids Res. 2017, 45, D964–D971. [Google Scholar] [CrossRef]
- Davis, A.P.; Grondin, C.J.; Johnson, R.J.; Sciaky, D.; McMorran, R.; Wiegers, J.; Wiegers, T.C.; Mattingly, C.J. The Comparative Toxicogenomics Database: Update 2019. Nucleic Acids Res. 2019, 47, D948–D954. [Google Scholar] [CrossRef] [PubMed]
- Manica, G.C.; Ribeiro, C.F.; Oliveira, M.A.; Pereira, I.T.; Chequin, A.; Ramos, E.A.; Klassen, L.M.; Sebastião, A.P.; Alvarenga, L.M.; Zanata, S.M.; et al. Down regulation of ADAM33 as a Predictive Biomarker of Aggressive Breast Cancer. Sci. Rep. 2017, 7, 44414. [Google Scholar] [CrossRef] [PubMed]
- Ghasemkhani, N.; Shadvar, S.; Masoudi, Y.; Talaei, A.J.; Yahaghi, E.; Goudarzi, P.K.; Shakiba, E. Down-regulated MicroRNA 148b expression as predictive biomarker and its prognostic significance associated with clinicopathological features in non-small-cell lung cancer patients. Diagn. Pathol. 2015, 10, 164. [Google Scholar] [CrossRef]
- Kuhara, M.; Wang, J.; Flores, M.J.; Qiao, Z.; Koizumi, Y.; Koyota, S.; Taniguchi, N.; Sugiyama, T. Sexual dimorphism in LEC rat liver: Suppression of carbonic anhydrase III by copper accumulation during hepatocarcinogenesis. Biomed. Res. (Tokyo, Japan) 2011, 32, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Di Fiore, A.; Monti, D.M.; Scaloni, A.; De Simone, G.; Monti, S.M. Protective Role of Carbonic Anhydrases III and VII in Cellular Defense Mechanisms upon Redox Unbalance. Oxidative Med. Cell. Longev. 2018, 2018, 2018306. [Google Scholar] [CrossRef] [PubMed]
- Ellinger-Ziegelbauer, H.; Gmuender, H.; Bandenburg, A.; Ahr, H.J. Prediction of a carcinogenic potential of rat hepatocarcinogens using toxicogenomics analysis of short-term in vivo studies. Mutat. Res. 2008, 637, 23–39. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, K.; Kawano, Y.; Kawakami, Y.; Moriwaki, N.; Sekijima, M.; Otsuka, M.; Yakabe, Y.; Miyaura, H.; Saito, K.; Sumida, K.; et al. Differences in gene expression profiles in the liver between carcinogenic and non-carcinogenic isomers of compounds given to rats in a 28-day repeat-dose toxicity study. Toxicol. Appl. Pharmacol. 2006, 217, 299–307. [Google Scholar] [CrossRef]
- Romer, M.; Eichner, J.; Metzger, U.; Templin, M.F.; Plummer, S.; Ellinger-Ziegelbauer, H.; Zell, A. Cross-platform toxicogenomics for the prediction of non-genotoxic hepatocarcinogenesis in rat. PLoS ONE 2014, 9, e97640. [Google Scholar] [CrossRef]
- Albrethsen, J.; Miller, L.M.; Novikoff, P.M.; Angeletti, R.H. Gel-based proteomics of liver cancer progression in rat. Biochim. Biophys. Acta 2011, 1814, 1367–1376. [Google Scholar] [CrossRef]
- Hibuse, T.; Maeda, N.; Nagasawa, A.; Funahashi, T. Aquaporins and glycerol metabolism. Biochim. Biophys. Acta 2006, 1758, 1004–1011. [Google Scholar] [CrossRef]
- Aikman, B.; de Almeida, A.; Meier-Menches, S.M.; Casini, A. Aquaporins in cancer development: Opportunities for bioinorganic chemistry to contribute novel chemical probes and therapeutic agents. Met. Integr. Biometal Sci. 2018, 10, 696–712. [Google Scholar] [CrossRef]
- Beltran-Ramirez, O.; Sokol, S.; Le-Berre, V.; Francois, J.M.; Villa-Trevino, S. An approach to the study of gene expression in hepatocarcinogenesis initiation. Transl. Oncol. 2010, 3, 142–148. [Google Scholar] [CrossRef][Green Version]
- Ovando, B.J.; Vezina, C.M.; McGarrigle, B.P.; Olson, J.R. Hepatic gene downregulation following acute and subchronic exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Toxicol. Sci. Off. J. Soc. Toxicol. 2006, 94, 428–438. [Google Scholar] [CrossRef]
- Heise, T.; Schug, M.; Storm, D.; Ellinger-Ziegelbauer, H.; Ahr, H.J.; Hellwig, B.; Rahnenfuhrer, J.; Ghallab, A.; Guenther, G.; Sisnaiske, J.; et al. In vitro-in vivo correlation of gene expression alterations induced by liver carcinogens. Curr. Med. Chem. 2012, 19, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.S.; Wang, G.P.; Zhang, L.X.; Chang, C.F.; Zhi, J.; Hao, Y.P. Correlation between liver cancer occurrence and gene expression profiles in rat liver tissue. Genet. Mol. Res. GMR 2011, 10, 3480–3513. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M.; Barbacid, M. Cell cycle, CDKs and cancer: A changing paradigm. Nat. Rev.Cancer 2009, 9, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.X.; Wang, X.Q.; Chok, S.H.; Man, K.; Tsang, S.H.Y.; Chan, A.C.Y.; Ma, K.W.; Xia, W.; Cheung, T.T. Blocking CDK1/PDK1/β-Catenin signaling by CDK1 inhibitor RO3306 increased the efficacy of sorafenib treatment by targeting cancer stem cells in a preclinical model of hepatocellular carcinoma. Theranostics 2018, 8, 3737–3750. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Yin, J.; Gong, S.; Gu, J.; Xiao, J.; Shi, W.; Ding, W.; He, Y. Bioinformatics analysis of key genes and pathways for hepatocellular carcinoma transformed from cirrhosis. Medicine 2017, 96, e6938. [Google Scholar] [CrossRef]
- Xing, C.; Xie, H.; Zhou, L.; Zhou, W.; Zhang, W.; Ding, S.; Wei, B.; Yu, X.; Su, R.; Zheng, S. Cyclin-dependent kinase inhibitor 3 is overexpressed in hepatocellular carcinoma and promotes tumor cell proliferation. Biochem. Biophys. Res. Commun. 2012, 420, 29–35. [Google Scholar] [CrossRef]
- Drozdov, I.; Bornschein, J.; Wex, T.; Valeyev, N.V.; Tsoka, S.; Malfertheiner, P. Functional and topological properties in hepatocellular carcinoma transcriptome. PLoS ONE 2012, 7, e35510. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.H.; Yuan, X.; Li, J.F.; Xie, Y.F.; Zhang, A.Z.; Wang, X.L.; Yang, L.; Liu, C.X.; Liang, W.H.; Pang, L.J.; et al. Bioinformatics-based screening of key genes for transformation of liver cirrhosis to hepatocellular carcinoma. J. Transl. Med. 2020, 18, 40. [Google Scholar] [CrossRef]
- Torres-Mena, J.E.; Salazar-Villegas, K.N.; Sánchez-Rodríguez, R.; López-Gabiño, B.; Del Pozo-Yauner, L.; Arellanes-Robledo, J.; Villa-Treviño, S.; Gutiérrez-Nava, M.A.; Pérez-Carreón, J.I. Aldo-Keto Reductases as Early Biomarkers of Hepatocellular Carcinoma: A Comparison Between Animal Models and Human HCC. Dig. Dis. Sci. 2018, 63, 934–944. [Google Scholar] [CrossRef]
- Wu, M.; Liu, Z.; Li, X.; Zhang, A.; Lin, D.; Li, N. Analysis of potential key genes in very early hepatocellular carcinoma. World J. Surg. Oncol. 2019, 17, 77. [Google Scholar] [CrossRef]
- Tarao, K.; Nozaki, A.; Ikeda, T.; Sato, A.; Komatsu, H.; Komatsu, T.; Taguri, M.; Tanaka, K. Real impact of liver cirrhosis on the development of hepatocellular carcinoma in various liver diseases-meta-analytic assessment. Cancer Med. 2019, 8, 1054–1065. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, H.; Tatemichi, M.; Sawa, T. Chemical basis of inflammation-induced carcinogenesis. Arch. Biochem. Biophys. 2003, 417, 3–11. [Google Scholar] [CrossRef]
- Lu, H.; Ouyang, W.; Huang, C. Inflammation, a key event in cancer development. Mol. Cancer Res. MCR 2006, 4, 221–233. [Google Scholar] [CrossRef]
- Affo, S.; Yu, L.X.; Schwabe, R.F. The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer. Annu. Rev. Pathol. 2017, 12, 153–186. [Google Scholar] [CrossRef] [PubMed]
- Pérez, L.O.; González-José, R.; García, P.P. Prediction of Non-Genotoxic Carcinogenicity Based on Genetic Profiles of Short Term Exposure Assays. Toxicol. Res. 2016, 32, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Furihata, C.; Suzuki, T. Evaluation of 12 mouse marker genes in rat toxicogenomics public data, Open TG-GATEs: Discrimination of genotoxic from non-genotoxic hepatocarcinogens. Mutat. Res. Genet. Toxicol. Environ. Mutagenesis 2019, 838, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Kossler, N.; Matheis, K.A.; Ostenfeldt, N.; Bach Toft, D.; Dhalluin, S.; Deschl, U.; Kalkuhl, A. Identification of specific mRNA signatures as fingerprints for carcinogenesis in mice induced by genotoxic and nongenotoxic hepatocarcinogens. Toxicol. Sci. Off. J. Soc. Toxicol. 2015, 143, 277–295. [Google Scholar] [CrossRef] [PubMed]
- Felter, S.P.; Foreman, J.E.; Boobis, A.; Corton, J.C.; Doi, A.M.; Flowers, L.; Goodman, J.; Haber, L.T.; Jacobs, A.; Klaunig, J.E.; et al. Human relevance of rodent liver tumors: Key insights from a Toxicology Forum workshop on nongenotoxic modes of action. Regul. Toxicol. Pharmacol. RTP 2018, 92, 1–7. [Google Scholar] [CrossRef]
- Percival, B.C.; Gibson, M.; Wilson, P.B.; Platt, F.M.; Grootveld, M. Metabolomic Studies of Lipid Storage Disorders, with Special Reference to Niemann-Pick Type C Disease: A Critical Review with Future Perspectives. Int. J. Mol. Sci. 2020, 21, 2533. [Google Scholar] [CrossRef]
- Leenders, J.; Grootveld, M.; Percival, B.; Gibson, M.; Casanova, F.; Wilson, P.B. Benchtop Low-Frequency 60 MHz NMR Analysis of Urine: A Comparative Metabolomics Investigation. Metabolites 2020, 10, 155. [Google Scholar] [CrossRef]
| Biomarker Set | Gene Symbol | Affymetrix ID | Codelink ID | Modulation | Time-Invariant | Reference | ||
|---|---|---|---|---|---|---|---|---|
| 1D | 3D | 1W | ||||||
| Consensus Biomarkers (1-day) | A2m | 1367794_at | NM_012488 | − | − | − | Yes | Huang and Tung (2017) [16] |
| Ca3 | 1386977_at | NM_019292 | − | − | − | Yes | ||
| Cxcl1 | 1387316_at | NM_030845 | − | − | − | Yes | ||
| Cyp8b1 | 1368435_at | NM_031241 | − | − | +/− | No | ||
| Consensus Biomarkers (3-day) * | A2m | 1367794_at | NM_012488 | − | − | − | Yes | This study |
| Akr7a3 | 1368121_at | NM_013215 | + | + | + | Yes | ||
| Aqp7 | 1368317_at | NM_019157 | + | + | + | Yes | ||
| Ca3 | 1386977_at | NM_019292 | − | − | − | Yes | ||
| Cdc2a | 1367776_at | NM_019296 | + | + | + | Yes | ||
| Cdkn3 | 1372685_at | BE113362 | + | + | + | Yes | ||
| Cyp2c11 | 1387328_at | NM_019184 | − | − | − | Yes | ||
| Ntf3 | 1387267_at | NM_031073 | − | − | − | Yes | ||
| Sds | 1369864_a_at | NM_053962 | − | − | − | Yes | ||
| Consensus Biomarkers (1-week) | Akr7a3 | 1368121_at | NM_013215 | + | + | + | Yes | This study |
| Aqp7 | 1368317_at | NM_019157 | + | + | + | Yes | ||
| Atf3 | 1369268_at | NM_012912 | + | +/− | + | No | ||
| beta-sarcoglycan | 1374796_at | AI413058 | + | + | + | Yes | ||
| Ca3 | 1386977_at | NM_019292 | − | − | − | Yes | ||
| Cpt1b | 1367742_at | NM_013200 | + | + | + | Yes | ||
| Cyp2c11 | 1387328_at | NM_019184 | − | − | − | Yes | ||
| Cyp17a1 | 1387123_at | NM_012753 | − | +/− | + | No | ||
| Ntf3 | 1387267_at | NM_031073 | − | − | − | Yes | ||
| RGD1562428_predicted | 1376296_at | BF387347 | + | + | + | Yes | ||
| Snx10 | 1383585_at | AI043753 | + | + | + | Yes | ||
| E5 | Abcb4 | 1369161_at | NA | − | − | − | Yes | Eichner et al. (2014) [13] |
| Akr7a3 | 1368121_at | NM_013215 | + | + | + | Yes | ||
| Ccng1 | 1367764_at | NM_012923 | + | + | +/− | No | ||
| Cdkn1a | 1387391_at | NM_080782 | + | +/− | +/− | No | ||
| Phlda3 | 1375224_at | AW520812 | +/− | +/− | + | No | ||
| F19 | Akr7a3 | 1368121_at | NM_013215 | + | + | + | Yes | Fielden et al.(2011) [10] |
| Aldh1a1 | 1387022_at | CK222590 | + | + | + | Yes | ||
| Anxa2 | 1367584_at | AA956299 | +/− | + | + | No | ||
| Btg2 | 1386994_at | NM_017259 | − | +/− | +/− | No | ||
| Cdkn1a | 1387391_at | NM_080782 | + | +/− | +/− | No | ||
| Cited4 | 1390008_-at | NM_053699 | +/− | +/− | +/− | No | ||
| ESTs | NA | BM388029 | − | + | + | No | ||
| Gpr146 | 1373158_at | NA | − | − | − | Yes | ||
| Ica1 | 1367787_at | NM_030844 | + | + | + | Yes | ||
| LitaF | 1370928_at | U53184 | +/− | +/− | +/− | No | ||
| Mat1a | 1371031_at | X60822 | − | − | − | Yes | ||
| Mgmt | 1368311_at | NM_012861 | +/− | + | + | No | ||
| Mt1a | 1371237_at | CR458797 | − | − | − | Yes | ||
| Ppia | 1398850_at | BI303474 | +/− | +/− | +/− | No | ||
| Prodh2 | 1389645_at | AI058310 | − | − | − | Yes | ||
| Psmb9 | 1370186_at | NM_012708 | +/− | +/− | +/− | No | ||
| Tap1 | 1388149_at | X57523 | +/- | +/- | +/- | No | ||
| Trnt1 | 1383144_at | AI412002 | +/- | +/- | + | No | ||
| Usp2 | 1387703_at | NM_053774 | +/− | − | +/− | No | ||
| U9 | Abcb1a | 1370583_s_at | NA | + | + | + | Yes | Uehara et al. (2011) [12] |
| Acot9 | 1379262_at | NA | + | + | + | Yes | ||
| Cd276_1 | 1395737_at | BF398424 | +/− | +/− | + | No | ||
| Cd276_2 | 1374198_at | NA | +/− | + | + | No | ||
| Cdh13_1 | 1375719_s_at | NM_138889 | +/− | +/− | +/− | No | ||
| Cdh13_2 | 1373102_at | NA | +/− | +/− | +/− | No | ||
| Ica1 | 1367787_at | NM_030844 | + | + | + | Yes | ||
| Tes | 1383401_at | NM_173132 | + | +/− | +/− | No | ||
| Tmem184c | 1379419_at | NA | + | + | + | Yes | ||
| Algorithm | Performance (Median AUC from LOOCV) | Variance | ||
|---|---|---|---|---|
| IQR | C.V.d | C.V.e | ||
| Bagging Tree (BaT) | 0.809 | 0.035 | 5.33% | 6.17% |
| Boosting Tree (BoT) | 0.757 | 0.102 | 9.37% | 9.21% |
| Decision Tree (J48) | 0.598 | 0.197 | 22.98% | 24.35% |
| k-Nearest Neighbor (kNN) | 0.720 | 0.071 | 5.81% | 7.09% |
| Naive Bayes (NB) | 0.800 | 0.055 | 3.50% | 4.00% |
| Random Forest (RF) | 0.817 | 0.041 | 4.36% | 4.74% |
| Support Vector Machine (SVM) | 0.582 | 0.084 | 8.56% | 3.07% |
| Signature | Dataset (Exposure Style) | Performance (Median AUC from LOOCV) | Variance (All Exposure) | |||
|---|---|---|---|---|---|---|
| Short-Term 3 | All Exposure 4 | IQR | C.V.d | C.V.e | ||
| Consensus biomarkers (1-day) | Multiple datasets 1 (1 day) | 0.739 | 0.733 | 0.049 * | 4.89% | 4.02% |
| Time-invariant biomarkers /Consensus biomarkers (3-day) | Multiple datasets 1 (3 days) | 0.817 | 0.824 | 0.036 * | 4.34% | 4.72% |
| Consensus biomarker (1-week) | Multiple datasets 1 (5 or 7 days 2) | 0.780 | 0.810 | 0.111 | 7.41% | 7.25% |
| E5 | TG-GATEs (14 days) | 0.656 | 0.656 | 0.097 | 9.04% | 9.47% |
| F19 | DrugMatrix (5 days) | 0.796 | 0.809 | 0.085 | 6.47% | 3.88% |
| U9 | TG-GATEs (28 days) | 0.703 | 0.713 | 0.057 | 8.25% | 7.09% |
| Signature | Performance (AUC from the Training Datasets) | |
|---|---|---|
| DMC | GSE8858 | |
| Consensus biomarkers (1-day) | 0.753 | 0.852 |
| Time-invariant biomarkers /Consensus biomarkers (3-day) | 0.862 | 0.857 |
| Consensus biomarker (1-week) | 0.820 | 0.815 |
| E5 | 0.632 | 0.562 |
| F19 | 0.732 | 0.791 |
| U9 | 0.338 | 0.465 |
| Disease ID. | Disease Name | Involved Genes | Corrected p-Value * |
|---|---|---|---|
| MESH:D008106 | Liver Cirrhosis (Experimental) | A2m, Aqp7, Ca3, Cdc2a, Cdkn3, Sds | 2.97 × 10−7 |
| MESH:D008103 | Liver Cirrhosis | A2m, Aqp7, Ca3, Cdc2a, Cdkn3, Sds | 6.52 × 10−7 |
| MESH:D005355 | Fibrosis | A2m, Aqp7, Ca3, Cdc2a, Cdkn3, Sds | 9.82 × 10−7 |
| MESH:D008107 | Liver Diseases | A2m, Akr7a3, Aqp7, Ca3, Cdc2aCdkn3, Sds | 1.40 × 10−6 |
| MESH:D004066 | Digestive System Diseases | A2m, Akr7a3, Aqp7, Ca3, Cdc2aCdkn3, Sds | 1.61 × 10−5 |
| MESH:D006528 | Carcinoma, Hepatocellular | A2m Cdc2a, Cdkn3 | 0.015 |
| MESH:D008113 | Liver Neoplasms | A2m Cdc2a, Cdkn3 | 0.037 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, S.-H.; Lin, Y.-C.; Tung, C.-W. Identification of Time-Invariant Biomarkers for Non-Genotoxic Hepatocarcinogen Assessment. Int. J. Environ. Res. Public Health 2020, 17, 4298. https://doi.org/10.3390/ijerph17124298
Huang S-H, Lin Y-C, Tung C-W. Identification of Time-Invariant Biomarkers for Non-Genotoxic Hepatocarcinogen Assessment. International Journal of Environmental Research and Public Health. 2020; 17(12):4298. https://doi.org/10.3390/ijerph17124298
Chicago/Turabian StyleHuang, Shan-Han, Ying-Chi Lin, and Chun-Wei Tung. 2020. "Identification of Time-Invariant Biomarkers for Non-Genotoxic Hepatocarcinogen Assessment" International Journal of Environmental Research and Public Health 17, no. 12: 4298. https://doi.org/10.3390/ijerph17124298
APA StyleHuang, S.-H., Lin, Y.-C., & Tung, C.-W. (2020). Identification of Time-Invariant Biomarkers for Non-Genotoxic Hepatocarcinogen Assessment. International Journal of Environmental Research and Public Health, 17(12), 4298. https://doi.org/10.3390/ijerph17124298






