No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial
Abstract
1. Introduction
2. Materials and Methods
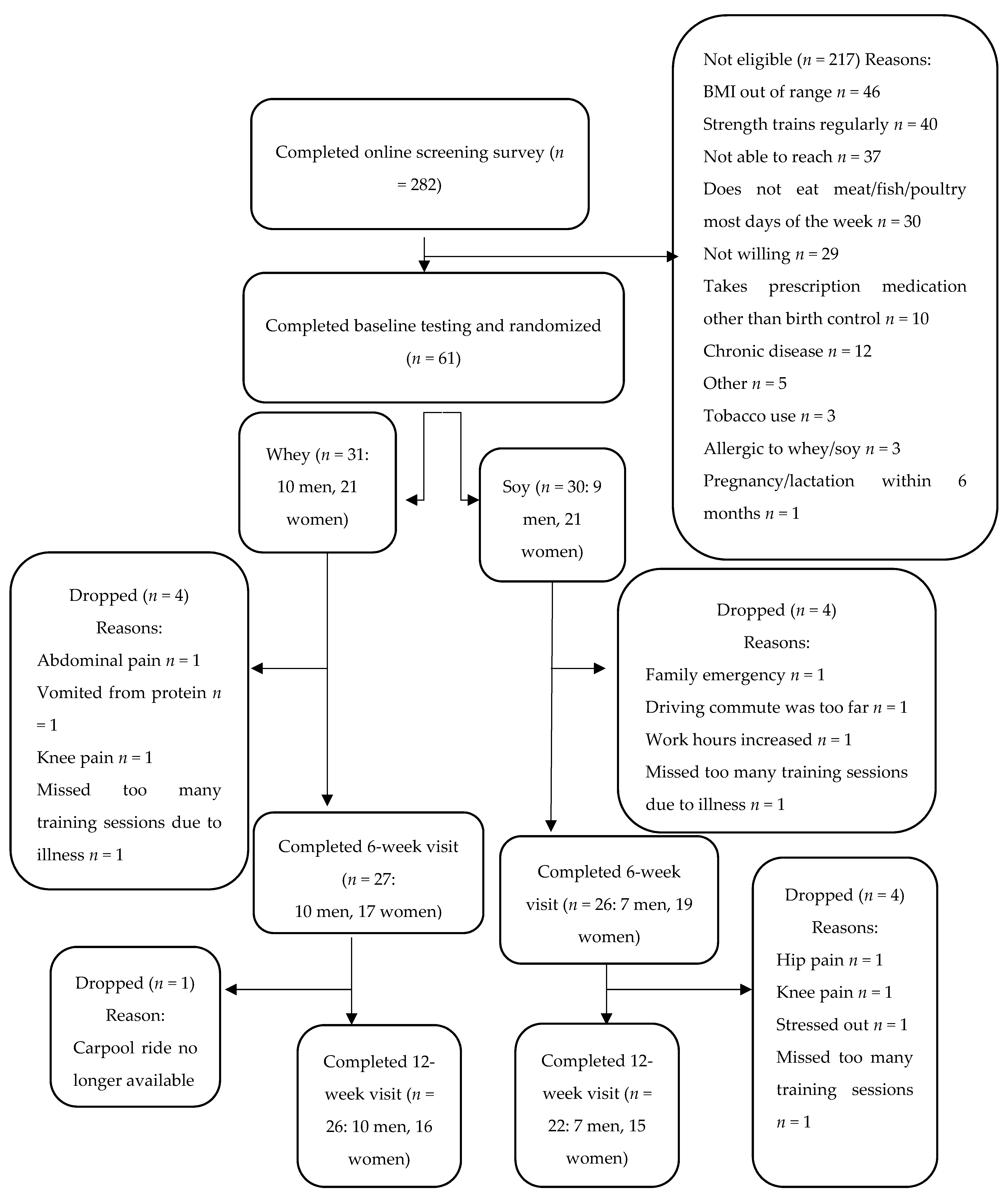
2.1. Participants
2.2. Protocol
2.3. Diet
2.4. Protein Supplement
2.5. Training Protocol
2.6. Outcomes
3. Results
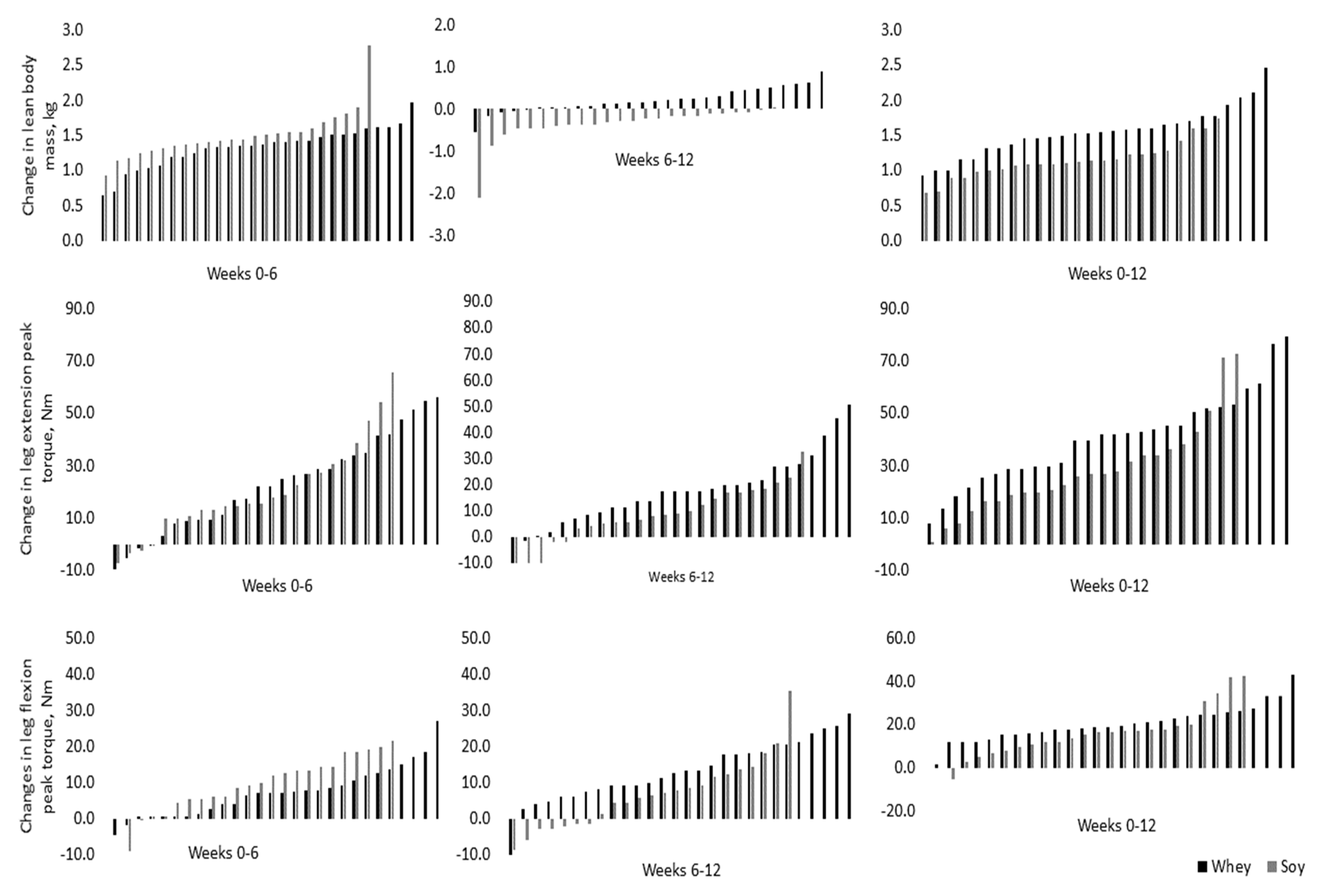
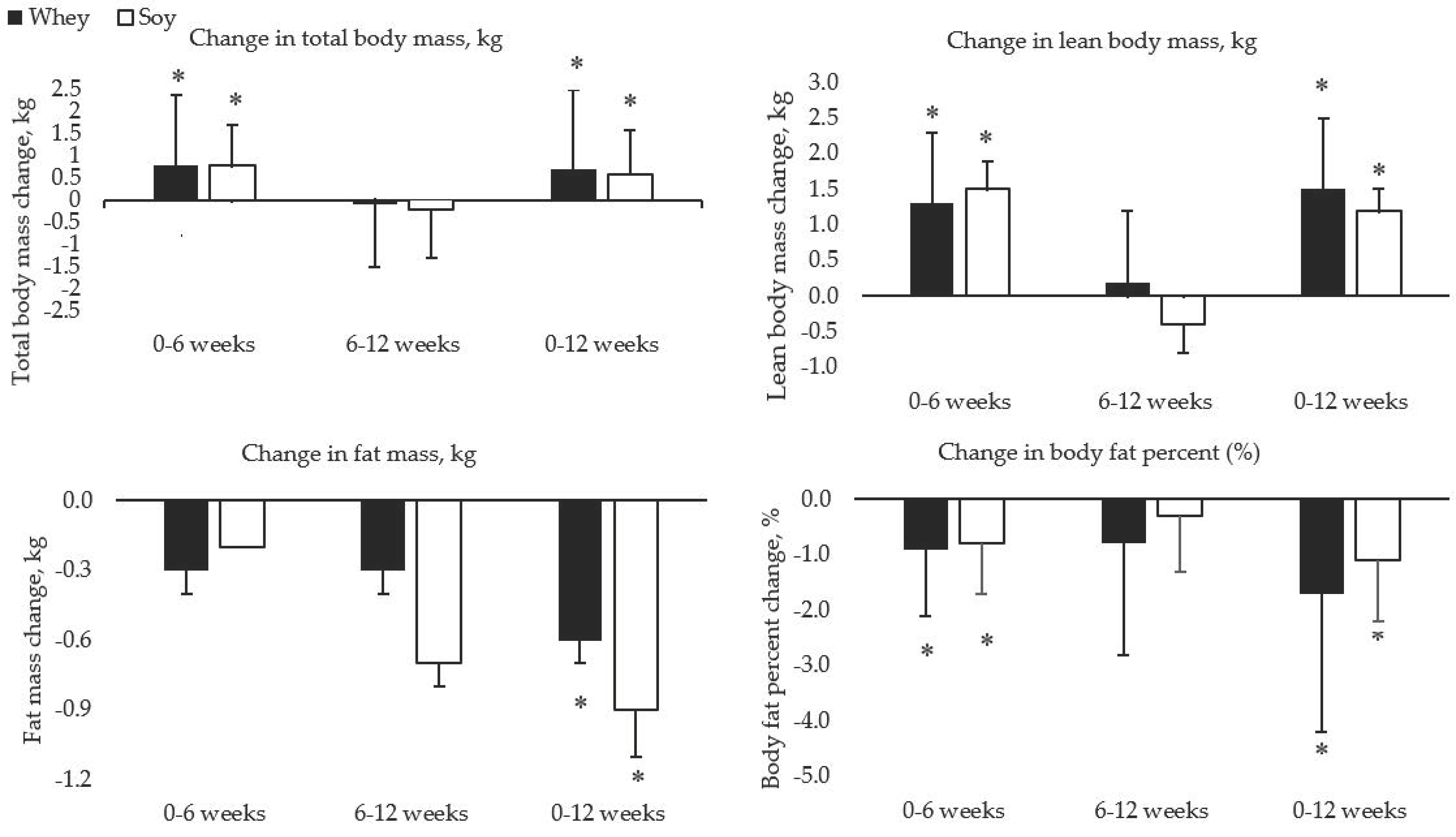
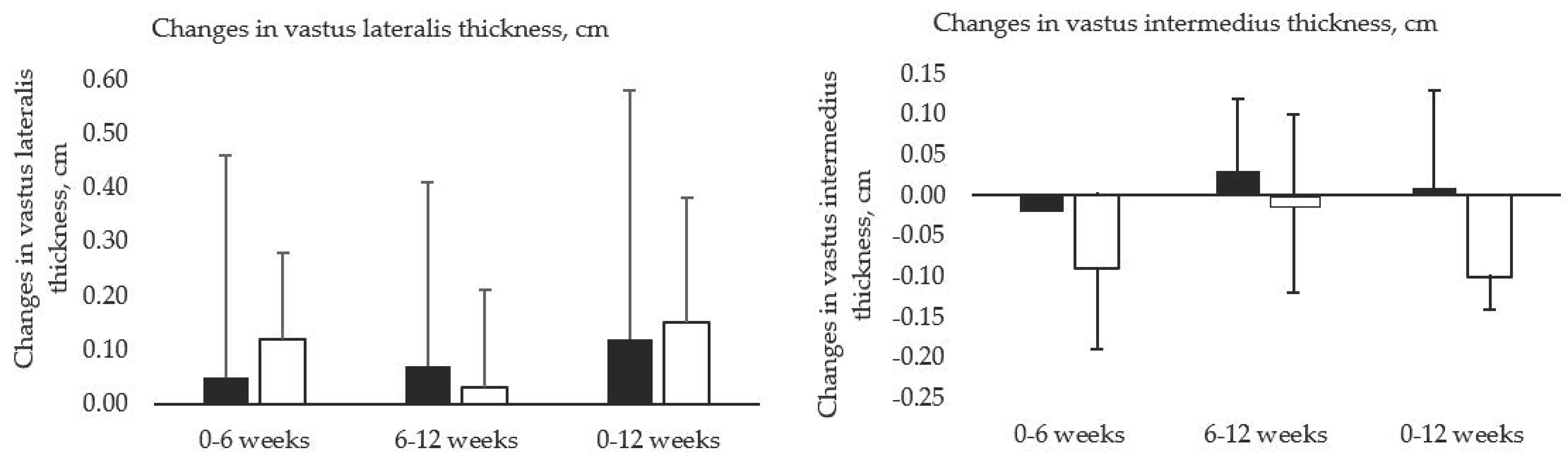
3.1. Anthropometric Changes and Strength Gains
3.2. Nutrient Intake
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mintel. Food and Drink Trends 2017; Mintel: London, UK, 2019; pp. 5–10. [Google Scholar]
- Mintel. Global Food & Drink Trends 2018; Zegler, J., Ed.; Available online: Mintel.com (accessed on 14 March 2020).
- Catsburg, C.; Kim, R.S.; Kirsh, V.A.; Soskolne, C.L.; Kreiger, N.; Rohan, T.E. Dietary patterns and breast cancer risk: A study in 2 cohorts. Am. J. Clin. Nutr. 2015, 101, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Tantamango-Bartley, Y.; Knutsen, S.F.; Knutsen, R.; Jacobsen, B.K.; Fan, J.; Beeson, W.L.; Sabate, J.; Hadley, D.; Jaceldo-Siegl, K.; Penniecook, J.; et al. Are strict vegetarians protected against prostate cancer? Am. J. Clin. Nutr. 2015, 103, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Orlich, M.; Singh, P.N.; Sabaté, J.; Fan, J.; Sveen, L.; Bennett, H.; Knutsen, S.F.; Beeson, W.L.; Jaceldo-Siegl, K.; Butler, T.L.; et al. Vegetarian dietary patterns and the risk of colorectal cancers. JAMA Intern. Med. 2015, 175, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Bae, Y.-J. Comparative Study of Serum Leptin and Insulin Resistance Levels Between Korean Postmenopausal Vegetarian and Non-vegetarian Women. Clin. Nutr. Res. 2015, 4, 175–181. [Google Scholar] [CrossRef]
- Kahleová, H.; Tura, A.; Hill, M.; Holubkov, R.; Barnard, N.D. A Plant-Based Dietary Intervention Improves Beta-Cell Function and Insulin Resistance in Overweight Adults: A 16-Week Randomized Clinical Trial. Nutrients 2018, 10, 189. [Google Scholar] [CrossRef]
- Kahleová, H.; Pelikanova, T. Vegetarian Diets in the Prevention and Treatment of Type 2 Diabetes. J. Am. Coll. Nutr. 2015, 34, 448–458. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishimura, K.; Barnard, N.D.; Takegami, M.; Watanabe, M.; Sekikawa, A.; Okamura, T.; Miyamoto, Y. Vegetarian Diets and Blood Pressure. JAMA Intern. Med. 2014, 174, 577. [Google Scholar] [CrossRef]
- Quiles-Guiñau, L.; Portolés, O.; Sorlí, J.V.; Corella, D. Short term effects on lipid profile and glycaemia of a low-fat vegetarian diet. Nutr. Hosp. 2015, 32, 156–164. [Google Scholar]
- Pimentel, D.; Pimentel, M. Sustainability of meat-based and plant-based diets and the environment. Am. J. Clin. Nutr. 2003, 78, 660S–663S. [Google Scholar] [CrossRef]
- Cordell, D.; Drangert, J.-O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Monsivais, P.; Scarborough, P.; Lloyd, T.; Mizdrak, A.; Luben, R.N.; Mulligan, A.A.; Wareham, N.J.; Woodcock, J. Greater accordance with the Dietary Approaches to Stop Hypertension dietary pattern is associated with lower diet-related greenhouse gas production but higher dietary costs in the United Kingdom. Am. J. Clin. Nutr. 2015, 102, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Masset, G.; Soler, L.-G.; Vieux, F.; Darmon, N. Identifying Sustainable Foods: The Relationship between Environmental Impact, Nutritional Quality, and Prices of Foods Representative of the French Diet. J. Acad. Nutr. Diet. 2014, 114, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Meyer, N.; Reguant-Closa, A. “Eat as If You Could Save the Planet and Win!” Sustainability Integration into Nutrition for Exercise and Sport. Nutrients 2017, 9, 412. [Google Scholar] [CrossRef]
- Mobley, C.B.; Haun, C.; Roberson, P.A.; Mumford, P.W.; Romero, M.A.; Kephart, W.C.; Anderson, R.G.; Vann, C.G.; Osburn, S.C.; Pledge, C.D.; et al. Effects of Whey, Soy or Leucine Supplementation with 12 Weeks of Resistance Training on Strength, Body Composition, and Skeletal Muscle and Adipose Tissue Histological Attributes in College-Aged Males. Nutrients 2017, 9, 972. [Google Scholar] [CrossRef] [PubMed]
- Volek, J.S.; Volk, B.M.; Gomez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.; et al. Whey Protein Supplementation During Resistance Training Augments Lean Body Mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Van Vliet, S.; Burd, N.A.; Van Loon, L.J.C. The Skeletal Muscle Anabolic Response to Plant- versus Animal-Based Protein Consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Volpi, E.; Kobayashi, H.; Sheffield-Moore, M.; Mittendorfer, B.; Wolfe, R.R. Essential amino acids are primarily responsible for the amino acid stimulation of muscle protein anabolism in healthy elderly adults. Am. J. Clin. Nutr. 2003, 78, 250–258. [Google Scholar] [CrossRef]
- Blomstrand, E.; Eliasson, J.; Karlsson, H.K.R.; Köhnke, R. Branched-Chain Amino Acids Activate Key Enzymes in Protein Synthesis after Physical Exercise. J. Nutr. 2006, 136, 269S–273S. [Google Scholar] [CrossRef]
- Dickinson, J.M.; Rasmussen, B.B. Essential amino acid sensing, signaling, and transport in the regulation of human muscle protein metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 83–88. [Google Scholar] [CrossRef]
- Dickinson, J.M.; Gundermann, D.M.; Walker, D.K.; Reidy, P.T.; Borack, M.S.; Drummond, M.J.; Arora, M.; Volpi, E.; Rasmussen, B.B. Leucine-enriched amino acid ingestion after resistance exercise prolongs myofibrillar protein synthesis and amino acid transporter expression in older men. J. Nutr. 2014, 144, 1694–1702. [Google Scholar] [CrossRef] [PubMed]
- Garlick, P.J. The Role of Leucine in the Regulation of Protein Metabolism. J. Nutr. 2005, 135, 1553S–1556S. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G. The Protein Digestibility–Corrected Amino Acid Score. J. Nutr. 2000, 130, 1865S–1867S. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.J.; Ryan, D.J.; Mukherjea, R.; Schasteen, C.S. Protein Digestibility-Corrected Amino Acid Scores (PDCAAS) for Soy Protein Isolates and Concentrate: Criteria for Evaluation. J. Agric. Food Chem. 2011, 59, 12707–12712. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations. Dietary Protein Quality Evaluation in Human Nutrition: Report of an FAO Expert Consultation, 31 March–2 April, 2011, Auckland, New Zealand; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013. [Google Scholar]
- Yang, Y.; Churchward-Venne, T.A.; Burd, N.A.; Breen, L.; Tarnopolsky, M.A.; Phillips, S.M. Myofibrillar protein synthesis following ingestion of soy protein isolate at rest and after resistance exercise in elderly men. Nutr. Metab. 2012, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.B.; Tarnopolsky, M.A.; Macdonald, M.J.; Macdonald, J.R.; Armstrong, D.; Phillips, S.M. Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. Am. J. Clin. Nutr. 2007, 85, 1031–1040. [Google Scholar] [CrossRef]
- Messina, M.; Lynch, H.; Dickinson, J.M.; Reed, K.E. No Difference Between the Effects of Supplementing With Soy Protein Versus Animal Protein on Gains in Muscle Mass and Strength in Response to Resistance Exercise. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 674–685. [Google Scholar] [CrossRef]
- Brown, E.C.; DiSilvestro, R.A.; Babaknia, A.; Devor, S.T. Soy versus whey protein bars: Effects on exercise training impact on lean body mass and antioxidant status. Nutr. J. 2004, 3, 22. [Google Scholar] [CrossRef]
- Glynn, E.L.; Fry, C.S.; Drummond, M.J.; Timmerman, K.L.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Excess leucine intake enhances muscle anabolic signaling but not net protein anabolism in young men and women. J. Nutr. 2010, 140, 1970–1976. [Google Scholar] [CrossRef]
- Mamerow, M.M.; Mettler, J.A.; English, K.L.; Casperson, S.L.; Arentson-Lantz, E.J.; Sheffield-Moore, M.; Layman, D.; Paddon-Jones, D. Dietary protein distribution positively influences 24-h muscle protein synthesis in healthy adults. J. Nutr. 2014, 144, 876–880. [Google Scholar] [CrossRef]
- Moore, D.R.; Areta, J.; Coffey, V.G.; Stellingwerff, T.; Phillips, S.M.; Burke, L.M.; Cleroux, M.; Godin, J.-P.; Hawley, J.A. Daytime pattern of post-exercise protein intake affects whole-body protein turnover in resistance-trained males. Nutr. Metab. 2012, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Borack, M.S.; Markofski, M.M.; Dickinson, J.M.; Deer, R.R.; Husaini, S.H.; Walker, D.K.; Igbinigie, S.; Robertson, S.M.; Cope, M.B.; et al. Protein Supplementation Has Minimal Effects on Muscle Adaptations during Resistance Exercise Training in Young Men: A Double-Blind Randomized Clinical Trial. J. Nutr. 2016, 146, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Rueden, C.T.; Hiner, M.C.; Eliceiri, K.W. The ImageJ ecosystem: An open platform for biomedical image analysis. Mol. Reprod. Dev. 2015, 82, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Feiring, D.C.; Ellenbecker, T.S.; Derscheid, G.L. Test-Retest Reliability of the Biodex Isokinetic Dynamometer. J. Orthop. Sports Phys. Ther. 1990, 11, 298–300. [Google Scholar] [CrossRef]
- Table, M. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; National Academy Press: Washington, DC, USA, 2005; Volume 5, pp. 589–768. [Google Scholar]
- Candow, D.G.; Burke, N.C.; Smith-Palmer, T.; Burke, D.G. Effect of whey and soy protein supplementation combined with resistance training in young adults. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 233–244. [Google Scholar] [CrossRef]
- Hartman, J.W.; Tang, J.E.; Wilkinson, S.B.; Tarnopolsky, M.A.; Lawrence, R.L.; Fullerton, A.V.; Phillips, S.M. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am. J. Clin. Nutr. 2007, 86, 373–381. [Google Scholar] [CrossRef]
- Haub, M.D.; Wells, A.M.; Tarnopolsky, M.A.; Campbell, W. Effect of protein source on resistive-training-induced changes in body composition and muscle size in older men. Am. J. Clin. Nutr. 2002, 76, 511–517. [Google Scholar] [CrossRef]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Mc Wilson, S.; Kalman, D.; E Dudeck, J.; Jäger, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12, 86. [Google Scholar] [CrossRef]
- Kalman, D.; Feldman, S.; Martinez, M.; Krieger, D.R.; Tallon, M.J. Effect of protein source and resistance training on body composition and sex hormones. J. Int. Soc. Sports Nutr. 2007, 4, 4. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Rasmussen, C.J.; Lancaster, S.L.; Magu, B.; Smith, P.; Melton, C.; Greenwood, M.; Almada, A.L.; Earnest, C.P.; Kreider, R.B. The effects of protein and amino acid supplementation on performance and training adaptations during ten weeks of resistance training. J. Strength Cond. Res. 2006, 20, 643. [Google Scholar]
- Reidy, P.T.; Rasmussen, B.B. Role of Ingested Amino Acids and Protein in the Promotion of Resistance Exercise–Induced Muscle Protein Anabolism. J. Nutr. 2016, 146, 155–183. [Google Scholar] [CrossRef] [PubMed]
- Glynn, E.L.; Fry, C.S.; Timmerman, K.L.; Drummond, M.J.; Volpi, E.; Rasmussen, B.B. Addition of carbohydrate or alanine to an essential amino acid mixture does not enhance human skeletal muscle protein anabolism. J. Nutr. 2013, 143, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Fry, C.S.; Borack, M.S.; Cope, M.B.; Mukherjea, R.; et al. Protein blend ingestion following resistance exercise promotes human muscle protein synthesis. J. Nutr. 2013, 143, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Churchward-Venne, T.A.; Pinckaers, P.; Smeets, J.S.J.; Peeters, W.M.; Zorenc, A.H.; Schierbeek, H.; Rollo, I.; Verdijk, L.B.; Van Loon, L.J.C. Myofibrillar and Mitochondrial Protein Synthesis Rates Do Not Differ in Young Men Following the Ingestion of Carbohydrate with Whey, Soy, or Leucine-Enriched Soy Protein after Concurrent Resistance- and Endurance-Type Exercise. J. Nutr. 2019, 149, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, B.; Bhammar, D.M.; Angadi, S.S.; Ryan, D.M.; Ryder, J.R.; Sussman, E.J.; Bertmann, F.; Gaesser, G.A. Predictors of Fat Mass Changes in Response to Aerobic Exercise Training in Women. J. Strength Cond. Res. 2015, 29, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Tesch, P.A. Skeletal muscle adaptations consequent to long-term heavy resistance exercise. Med. Sci. Sports Exerc. 1988, 20, S132–S134. [Google Scholar] [CrossRef]
- Ribeiro, A.; Dos Santos, L.; Silva, A.M.; Gobbo, L.A.; Schoenfeld, B.; Sardinha, L.B.; Cyrino, E.S.; Avelar, A. Hypertrophy-type Resistance Training Improves Phase Angle in Young Adult Men and Women. Int. J. Sports Med. 2016, 38, 35–40. [Google Scholar] [CrossRef]
- Bone, J.L.; Ross, M.L.; Tomcik, K.A.; Jeacocke, N.A.; Hopkins, W.G.; Burke, L.M. Manipulation of muscle creatine and glycogen changes DXA estimates of body composition. Med. Sci. Sports Exerc. 2017, 49, 1029–1035. [Google Scholar] [CrossRef]
- Nieman, D.; Shanely, R.A.; Zwetsloot, K.A.; Meaney, M.P.; Farris, G.E. Ultrasonic assessment of exercise-induced change in skeletal muscle glycogen content. BMC Sports Sci. Med. Rehabilitation 2015, 7, 9. [Google Scholar] [CrossRef]
- Baroni, B.M.; Geremia, J.M.; Rodrigues, R.; Franke, R.D.A.; Karamanidis, K.; Vaz, M.A. Muscle architecture adaptations to knee extensor eccentric training: Rectus femoris vs. vastus lateralis. Muscle Nerve 2013, 48, 498–506. [Google Scholar] [CrossRef]
- Brook, M.; Wilkinson, D.J.; Mitchell, W.K.; Lund, J.; Szewczyk, N.J.; Greenhaff, P.L.; Smith, K.; Atherton, P.J. Skeletal muscle hypertrophy adaptations predominate in the early stages of resistance exercise training, matching deuterium oxide-derived measures of muscle protein synthesis and mechanistic target of rapamycin complex 1 signaling. FASEB J. 2015, 29, 4485–4496. [Google Scholar] [CrossRef] [PubMed]
- Morton, R.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2017, 52, 376–384. [Google Scholar] [CrossRef] [PubMed]
| Nutrient | Whey Protein Isolate (21 g) | Soy Protein Isolate (29 g) |
|---|---|---|
| Protein (g) | 19 | 26 |
| Amino acid composition (mg) | ||
| Leucine | 1997 | 1967 |
| Isoleucine | 1243 | 1233 |
| Valine | 1067 | 1188 |
| Histidine | 315 | 668 |
| Lysine | 1930 | 1545 |
| Methionine | 405 | 328 |
| Phenylalanine | 596 | 1332 |
| Threonine | 1292 | 910 |
| Tryptophan | 374 | 324 |
| Arginine | 433 | 1934 |
| Glutamic acid | 3318 | 5061 |
| Cystine | 449 | 303 |
| Alanine | 995 | 1041 |
| Glycine | 319 | 1045 |
| Proline | 1151 | 1438 |
| Serine | 872 | 1332 |
| Tyrosine | 569 | 934 |
| Aspartic acid | 2052 | 2959 |
| Baseline (Week 0) | Week 6 | Week 12 | Change Week 0–6 | Change Week 6–12 | Change Week 0–12 | Effect Size for Change Weeks 0–12 (Cohen’s d) | |
|---|---|---|---|---|---|---|---|
| Weight (kilograms, kg) | |||||||
| Whey | 66.9 ± 10.1 | 67.7 ± 10.2 * | 67.6 ± 10.0 * | 0.8 ± 1.6 | −0.1 ± 1.4 | 0.7 ± 1.8 | 0.07 |
| Soy | 65.5 ± 13.3 | 66.4 ± 13.7 * | 66.2 ± 13.2 * | 0.8 ± 0.9 | −0.2 ± 1.1 | 0.6 ± 1.0 | 0.05 |
| Lean body mass (kg) | |||||||
| Whey | 44.5 ± 8.7 | 45.8 ± 8.9 * | 46.0 ± 8.9 * | 1.3 ± 0.3 | 0.2 ± 0.3 | 1.5 ± 0.3 | 0.17 |
| Soy | 44.1 ± 10.3 | 45.6 ± 10.5 * | 45.2 ± 10.3 * | 1.5 ± 0.4 | −0.4 ± 0.4 | 1.2 ± 0.3 | 0.11 |
| Appendicular lean body mass (kg) | |||||||
| Whey | 20.3 ± 5.1 | 21.3 ± 5.2 * | 21.3 ± 5.2 * | 1.0 ± 0.1 | 0.0 ± 0.1 | 0.9 ± 0.1 | 0.19 |
| Soy | 19.8 ± 5.4 | 20.5 ± 5.5 * | 20.7 ± 5.4 * | 0.7 ± 0.1 | 0.1 ± 0.1 | 0.8 ± 0.1 | 0.17 |
| Fat mass (kg) | |||||||
| Whey | 20.2 ± 6.3 | 19.9 ± 6.3 | 19.6 ± 6.2 * | −0.3 ± 0.1 | −0.3 ± 0.1 | −0.6 ± 0.1 | −0.10 |
| Soy | 19.7 ± 6.8 | 19.5 ± 6.8 | 18.8 ± 6.8 * | −0.2 ± 0.0 | −0.7 ± 0.1 | −0.9 ± 0.2 | −0.13 |
| Body fat percent (%) | |||||||
| Whey | 31.4 ± 8.2 | 30.5 ± 8.3 * | 29.7 ± 8.9 * | −0.9 ± 1.2 | −0.8 ± 2.0 | −1.7 ± 2.5 | −0.20 |
| Soy | 30.9 ± 8.2 | 30.1 ± 8.1 * | 29.8 ± 7.8 * | −0.8 ± 0.9 | −0.3 ± 1.0 | −1.1 ± 1.1 | −0.14 |
| Vastus lateralis thickness (centimeters, cm) | |||||||
| Whey | 2.3 ± 0.5 | 2.4 ± 0.4 | 2.5 ± 0.5 | 0.05 ± 0.41 | 0.07 ± 0.34 | 0.12 ± 0.46 | 0.40 |
| Soy | 2.2 ± 0.3 | 2.3 ± 0.2 | 2.3 ± 0.3 | 0.12 ± 0.16 | 0.03 ± 0.18 | 0.15 ± 0.23 | 0.33 |
| Vastus intermedius thickness (cm) | |||||||
| Whey | 1.6 ± 0.4 | 1.6 ± 0.4 | 1.6 ± 0.4 | −0.02 ± 0.12 | 0.03 ± 0.09 | 0.01 ± 0.12 | 0.00 |
| Soy | 1.6 ± 0.3 | 1.5 ± 0.4 | 1.5 ± 0.4 | −0.10 ± 0.10 | −0.01 ± 0.11 | −0.10 ± 0.98 | −0.28 |
| Peak torque extensions (Newton-meters, Nm) | |||||||
| Whey | 124.4 ± 39.9 | 142.3 ± 34.3 * | 164.6 ± 39.4 * | 18.0 ± 16.1 | 12.5 ± 12.9 | 30.5 ± 15.6 | 1.01 |
| Soy | 132.0 ± 44.9 | 152.2 ± 43.6 * | 160.4 ± 43.8 * | 12.1 ± 13.5 | 7.6 ± 8.9 | 19.7 ± 15.4 | 0.64 |
| Peak torque flexions (Nm) | |||||||
| Whey | 60.5 ± 15.9 | 67.7 ± 14.6 | 80.9 ± 18.0 * | 4.3 ± 6.5 | 9.9 ± 9.6 | 14.2 ± 8.7 | 1.20 |
| Soy | 64.3 ± 15.0 | 71.5 ± 16.7 | 80.6 ± 20.0 * | 7.7 ± 8.9 | 3.7 ± 10.7 | 11.4 ± 12.9 | 0.92 |
| Baseline (Week 0) | Week 6 | Week 12 | Change Week 0–6 | Change Week 6–12 | Change Week 0–12 | |
|---|---|---|---|---|---|---|
| Kilocalories (kcal) | ||||||
| Whey | 2225 ± 406 | 1858 ± 341 | 2173 ± 351 | −367 ± 221 | 314 ± 230 | −53 ± 368 |
| Soy | 1839 ± 247 | 2018 ± 344 | 2331 ± 357 | 180 ± 236 | 313 ± 174 | 492 ± 214 |
| Carbohydrate (g) | ||||||
| Whey | 265 ± 66 | 223 ± 54 | 259 ± 52 | −42 ± 36 | 36 ± 32 | −6 ± 51 |
| Soy | 232 ± 36 | 245 ± 45 | 271 ± 44 | 13 ± 28 | 26 ± 22 | 39 ± 27 |
| Carbohydrate (% kcal) | ||||||
| Whey | 48 ± 7 | 48 ± 7 | 48 ± 7 | 0 ± 5 | 0 ± 3 | 0 ± 4 |
| Soy | 51 ± 5 | 50 ± 6 | 46 ± 4 | −1 ± 4 | −2 ± 3 | −3 ± 4 |
| Fat (g) | ||||||
| Whey | 87 ± 19 | 68 ± 16 | 85 ± 19 * | −19 ± 11 | 18 ± 12 | −2 ± 19 |
| Soy | 68 ± 13 | 81 ± 16 | 100 ± 19 * | 12 ± 10 | 20 ± 8 | 32 ± 11 |
| Fat (% kcal) | ||||||
| Whey | 35 ± 5 | 33 ± 5 | 35 ± 6 | −2 ± 3 | 2 ± 3 | 0 ± 4 |
| Soy | 33 ± 4 | 36 ± 3 | 39 ± 3 | 2 ± 2 | 2 ± 2 | 4 ± 3 |
| Protein (g) | ||||||
| Whey | 94 ± 23 | 85 ± 22 | 91 ± 22 | −9 ± 11 | 6 ± 12 | −3 ± 16 |
| Soy | 75 ± 14 | 75 ± 23 | 90 ± 20 | 1 ± 21 | 14 ± 13 | 15 ± 13 |
| Protein (g/kg) | ||||||
| Whey | 1.2 ± 0.3 | 1.1 ± 0.3 | 1.3 ± 0.2 | −0.2 ± 0.2 | 0.1 ± 0.2 | −0.1 ± 0.2 |
| Soy | 1.4 ± 0.5 | 1.3 ± 0.5 | 1.4 ± 0.4 | 0.0 ± 0.3 | 0.2 ± 0.2 | 0.2 ± 0.2 |
| Protein (% kcal) | ||||||
| Whey | 17 ± 3 | 19 ± 4 | 17 ± 4 | 1 ± 2 | −1 ± 2 | 0 ± 2 |
| Soy | 16 ± 2 | 17 ± 4 | 16 ± 3 | −1 ± 2 | 1 ± 2 | 0 ± 2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lynch, H.M.; Buman, M.P.; Dickinson, J.M.; Ransdell, L.B.; Johnston, C.S.; Wharton, C.M. No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial. Int. J. Environ. Res. Public Health 2020, 17, 3871. https://doi.org/10.3390/ijerph17113871
Lynch HM, Buman MP, Dickinson JM, Ransdell LB, Johnston CS, Wharton CM. No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial. International Journal of Environmental Research and Public Health. 2020; 17(11):3871. https://doi.org/10.3390/ijerph17113871
Chicago/Turabian StyleLynch, Heidi M., Matthew P. Buman, Jared M. Dickinson, Lynda B. Ransdell, Carol S. Johnston, and Christopher M. Wharton. 2020. "No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial" International Journal of Environmental Research and Public Health 17, no. 11: 3871. https://doi.org/10.3390/ijerph17113871
APA StyleLynch, H. M., Buman, M. P., Dickinson, J. M., Ransdell, L. B., Johnston, C. S., & Wharton, C. M. (2020). No Significant Differences in Muscle Growth and Strength Development When Consuming Soy and Whey Protein Supplements Matched for Leucine Following a 12 Week Resistance Training Program in Men and Women: A Randomized Trial. International Journal of Environmental Research and Public Health, 17(11), 3871. https://doi.org/10.3390/ijerph17113871






