The Risk of Subsequent Deep Vein Thrombosis and Pulmonary Embolism in Patients with Nontyphoidal Salmonellosis: A Nationwide Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Study Design
2.2. Study Population
2.3. Outcome and Relevant Variables
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Risk of DVT and PE
3.3. Analysis Stratified by Sex, Age, and Co-Morbidities
3.4. Sensitivity Analysis
3.5. Time Trends for Risk of DVT and PE
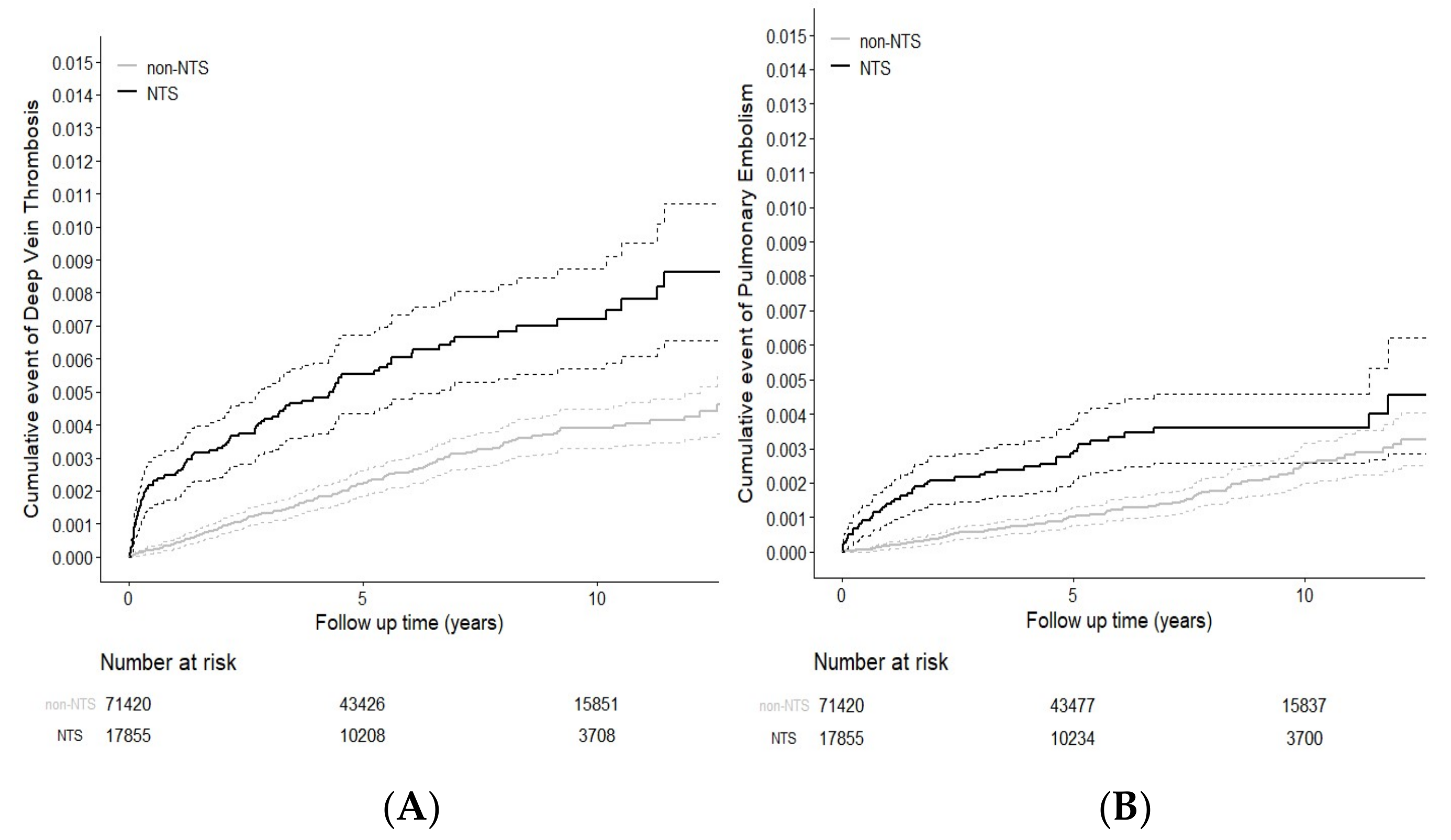
3.6. Cumulative Incidences Completing Risk Analysis of DVT and PE in NTS and non-NTS Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gromadzinski, L.; Targonski, R.; Januszko-Giergielewicz, B.; Ciurzynski, M.; Pruszczyk, P. The influence of acute pulmonary embolism on early and delayed prognosis for patients with chronic heart failure. Cardiol. J. 2012, 19, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Wolberg, A.S.; Rosendaal, F.R.; Weitz, J.I.; Jaffer, I.H.; Agnelli, G.; Baglin, T.; Mackman, N. Venous thrombosis. Nat. Rev. Dis. Primers 2015, 1, 15006. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Horvath-Puho, E.; Thomsen, R.W.; Smeeth, L.; Sorensen, H.T. Acute infections and venous thromboembolism. J. Intern. Med. 2012, 271, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Mejer, N.; Westh, H.; Schonheyder, H.C.; Jensen, A.G.; Larsen, A.R.; Skov, R.; Benfield, T.; Danish Staphylococcal Bacteraemia Study Group. Increased risk of venous thromboembolism within the first year after staphylococcus aureus bacteraemia: A nationwide observational matched cohort study. J. Intern. Med. 2014, 275, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Carey, J.; Buchstein, S.; Shah, S. Septic deep vein thrombosis due to salmonella johannesburg. J. Infect. 2001, 42, 79–80. [Google Scholar] [CrossRef] [PubMed]
- Salamon, S.A.; Prag, J. A case of superficial septic thrombophlebitis in a varicose vein caused by salmonella panama. Clin. Microbiol. Infect. 2001, 7, 34–36. [Google Scholar] [CrossRef]
- Schifferdecker, B.; Merchan, J.A.; Ahmar, C.; Worthington, M.; Griben, A.; Schainfeld, R.M.; Soukas, P.A. Endovascular treatment of septic thrombophlebitis: A case report of a rare complication and review of the literature. Vasc. Med. 2009, 14, 47–50. [Google Scholar] [CrossRef]
- Wright, A.E.; Knapp, H.H. A note on the causation and treatment of thrombosis occurring in connection with typhoid fever. Med. Chir. Trans. 1903, 86, 1–19. [Google Scholar]
- Hitchcock, J.R.; Cook, C.N.; Bobat, S.; Ross, E.A.; Flores-Langarica, A.; Lowe, K.L.; Khan, M.; Dominguez-Medina, C.C.; Lax, S.; Carvalho-Gaspar, M.; et al. Inflammation drives thrombosis after salmonella infection via clec-2 on platelets. J. Clin. Investig. 2015, 125, 4429–4446. [Google Scholar] [CrossRef]
- Stapels, D.A.C.; Hill, P.W.S.; Westermann, A.J.; Fisher, R.A.; Thurston, T.L.; Saliba, A.E.; Blommestein, I.; Vogel, J.; Helaine, S. Salmonella persisters undermine host immune defenses during antibiotic treatment. Science 2018, 362, 1156–1160. [Google Scholar] [CrossRef]
- Wu, H.H.; Chen, Y.T.; Shih, C.J.; Lee, Y.T.; Kuo, S.C.; Chen, T.L. Association between recent use of proton pump inhibitors and nontyphoid salmonellosis: A nested case-control study. Clin. Infect. Dis. 2014, 59, 1554–1558. [Google Scholar] [CrossRef] [PubMed]
- Surewaard, B.G.J.; Thanabalasuriar, A.; Zeng, Z.; Tkaczyk, C.; Cohen, T.S.; Bardoel, B.W.; Jorch, S.K.; Deppermann, C.; Bubeck Wardenburg, J.; Davis, R.P.; et al. Alpha-toxin induces platelet aggregation and liver injury during staphylococcus aureus sepsis. Cell Host Microbe 2018, 24, 271–284.e3. [Google Scholar] [CrossRef] [PubMed]
- Gardlund, B. Fatal pulmonary embolism in hospitalized non-surgical patients. Acta Med. Scand. 1985, 218, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Belch, J.J.; Lowe, G.D.; Ward, A.G.; Forbes, C.D.; Prentice, C.R. Prevention of deep vein thrombosis in medical patients by low-dose heparin. Scott. Med. J. 1981, 26, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Gardlund, B. Randomised, controlled trial of low-dose heparin for prevention of fatal pulmonary embolism in patients with infectious diseases. The heparin prophylaxis study group. Lancet 1996, 347, 1357–1361. [Google Scholar]
- Alikhan, R.; Cohen, A.T.; Combe, S.; Samama, M.M.; Desjardins, L.; Eldor, A.; Janbon, C.; Leizorovicz, A.; Olsson, C.G.; Turpie, A.G. Prevention of venous thromboembolism in medical patients with enoxaparin: A subgroup analysis of the medenox study. Blood Coagul. Fibrinolysis 2003, 14, 341–346. [Google Scholar] [CrossRef]
- Cohen, A.T.; Davidson, B.L.; Gallus, A.S.; Lassen, M.R.; Prins, M.H.; Tomkowski, W.; Turpie, A.G.; Egberts, J.F.; Lensing, A.W.; Investigators, A. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: Randomised placebo controlled trial. BMJ 2006, 332, 325–329. [Google Scholar] [CrossRef]
- Kaplan, D.; Casper, T.C.; Elliott, C.G.; Men, S.; Pendleton, R.C.; Kraiss, L.W.; Weyrich, A.S.; Grissom, C.K.; Zimmerman, G.A.; Rondina, M.T. Vte incidence and risk factors in patients with severe sepsis and septic shock. Chest 2015, 148, 1224–1230. [Google Scholar] [CrossRef]
- Ohl, M.E.; Miller, S.I. Salmonella: A model for bacterial pathogenesis. Annu. Rev. Med. 2001, 52, 259–274. [Google Scholar] [CrossRef]
- Van Sorge, N.M.; Zialcita, P.A.; Browne, S.H.; Quach, D.; Guiney, D.G.; Doran, K.S. Penetration and activation of brain endothelium by salmonella enterica serovar typhimurium. J. Infect. Dis. 2011, 203, 401–405. [Google Scholar] [CrossRef]
- Riegger, J.; Byrne, R.A.; Joner, M.; Chandraratne, S.; Gershlick, A.H.; Ten Berg, J.M.; Adriaenssens, T.; Guagliumi, G.; Godschalk, T.C.; Neumann, F.J.; et al. Histopathological evaluation of thrombus in patients presenting with stent thrombosis. A multicenter european study: A report of the prevention of late stent thrombosis by an interdisciplinary global european effort consortium. Eur. Heart J. 2016, 37, 1538–1549. [Google Scholar] [CrossRef] [PubMed]
- Engelmann, B.; Massberg, S. Thrombosis as an intravascular effector of innate immunity. Nat. Rev. Immunol. 2013, 13, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Von Bruhl, M.L.; Stark, K.; Steinhart, A.; Chandraratne, S.; Konrad, I.; Lorenz, M.; Khandoga, A.; Tirniceriu, A.; Coletti, R.; Kollnberger, M.; et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 2012, 209, 819–835. [Google Scholar] [CrossRef] [PubMed]
- Zarychanski, R.; Abou-Setta, A.M.; Kanji, S.; Turgeon, A.F.; Kumar, A.; Houston, D.S.; Rimmer, E.; Houston, B.L.; McIntyre, L.; Fox-Robichaud, A.E.; et al. The efficacy and safety of heparin in patients with sepsis: A systematic review and metaanalysis. Crit. Care Med. 2015, 43, 511–518. [Google Scholar] [CrossRef]
- Payne, H.; Ponomaryov, T.; Watson, S.P.; Brill, A. Mice with a deficiency in clec-2 are protected against deep vein thrombosis. Blood 2017, 129, 2013–2020. [Google Scholar] [CrossRef]
- Budnik, I.; Brill, A. Immune factors in deep vein thrombosis initiation. Trends Immunol. 2018, 39, 610–623. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Brinkmann, V.; Zychlinsky, A. Beneficial suicide: Why neutrophils die to make nets. Nat. Rev. Microbiol. 2007, 5, 577–582. [Google Scholar] [CrossRef]
- Averhoff, P.; Kolbe, M.; Zychlinsky, A.; Weinrauch, Y. Single residue determines the specificity of neutrophil elastase for shigella virulence factors. J. Mol. Biol. 2008, 377, 1053–1066. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Brill, A.; Wagner, D.D. Neutrophil extracellular trap (net) impact on deep vein thrombosis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1777–1783. [Google Scholar] [CrossRef]
- Yang, H.; Biermann, M.H.; Brauner, J.M.; Liu, Y.; Zhao, Y.; Herrmann, M. New insights into neutrophil extracellular traps: Mechanisms of formation and role in inflammation. Front. Immunol. 2016, 7, 302. [Google Scholar] [CrossRef] [PubMed]
- Kimball, A.S.; Obi, A.T.; Diaz, J.A.; Henke, P.K. The emerging role of nets in venous thrombosis and immunothrombosis. Front. Immunol. 2016, 7, 236. [Google Scholar] [CrossRef] [PubMed]
- Hazeldine, J.; Harris, P.; Chapple, I.L.; Grant, M.; Greenwood, H.; Livesey, A.; Sapey, E.; Lord, J.M. Impaired neutrophil extracellular trap formation: A novel defect in the innate immune system of aged individuals. Aging Cell 2014, 13, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Itagaki, K.; Kaczmarek, E.; Lee, Y.T.; Tang, I.T.; Isal, B.; Adibnia, Y.; Sandler, N.; Grimm, M.J.; Segal, B.H.; Otterbein, L.E.; et al. Mitochondrial DNA released by trauma induces neutrophil extracellular traps. PLoS ONE 2015, 10, e0120549. [Google Scholar] [CrossRef]
| Nontyphoidal Salmonellosis | p Value | ||
|---|---|---|---|
| No (N = 71,420) | Yes (N = 17,855) | ||
| n (%) | n (%) | ||
| Age, years | 1.00 | ||
| 18–39 | 18,900 (26.5) | 4725 (26.5) | |
| 40–64 | 26,048 (36.5) | 6512 (36.5) | |
| ≥65 | 26,472 (37.1) | 6618 (37.1) | |
| Mean (SD) * | 55.3 (19.7) | 55.4 (19.7) | 0.62 * |
| Gender | 1.00 | ||
| Female | 32,204 (0.45) | 8051 (0.45) | |
| Male | 39,216 (0.55) | 9804 (0.55) | |
| Comorbidities | 16,732 (23.4) | 7951 (44.5) | <0.001 |
| Hypertension | 9376 (0.13) | 4313 (0.24) | <0.001 |
| Diabetes | 4968 (0.07) | 2854 (0.16) | <0.001 |
| Hyperlipidemia | 2219 (0.03) | 1224 (0.07) | <0.001 |
| Coronary artery disease | 4465 (0.06) | 2117 (0.12) | <0.001 |
| Cerebrovascular accident | 4104 (0.06) | 1664 (0.09) | <0.001 |
| Chronic Kidney disease | 586 (0.01) | 687 (0.04) | <0.001 |
| Cancer | 2368 (0.03) | 1977 (0.11) | <0.001 |
| Chronic Obstructive Pulmonary Disease | 2359 (0.03) | 1453 (0.08) | <0.001 |
| Sleep apnea | 66 (0.001) | 41 (0.002) | <0.001 |
| Rheumatoid arthritis | 144 (0.002) | 190 (0.01) | <0.001 |
| Atrial fibrillation | 916 (0.01) | 537 (0.03) | <0.001 |
| Chronic liver disease | 553 (0.01) | 419 (0.02) | 0.75 |
| Pregnancy | 13 (0.0002) | 2 (0.0001) | <0.001 |
| Lower leg fracture or surgery | 2935 (0.04) | 1121 (0.06) | <0.001 |
| Mortality | 5560 (7.78) | 4290 (24.0) | <0.001 |
| Variables | Event | Crude HR ‡ | Adjusted HR § | SHR | |||||
|---|---|---|---|---|---|---|---|---|---|
| n | PY | Rate † | (95% CI) | (95% CI) | (95% CI) | ||||
| DVT | |||||||||
| Non-NTS | 215 | 450,270 | 4.77 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 109 | 88,183 | 12.36 | 2.54 | (2.02 to 3.20) *** | 2.35 | (1.86 to 2.98) *** | 1.83 | (1.44 to 2.31) *** |
| Gender | |||||||||
| Female | 154 | 248,049 | 6.21 | 1.00 | - | ||||
| Male | 170 | 290,404 | 5.85 | 0.94 | (0.75 to 1.17) | ||||
| Age | |||||||||
| 18–39 | 22 | 164,576 | 1.34 | 1.00 | - | 1.00 | - | 1.00 | - |
| 40–64 | 82 | 209,177 | 3.92 | 2.91 | (1.82 to 4.66) *** | 2.49 | (1.55 to 4.01) *** | 2.93 | (1.47 to 3.90) *** |
| ≥65 | 220 | 164,700 | 13.36 | 9.68 | (6.24 to 15.0) *** | 6.41 | (4.04 to 10.2) *** | 5.58 | (3.38 to 9.22) *** |
| Comorbidities | |||||||||
| No | 142 | 432,486 | 3.28 | 1.00 | - | 1.00 | - | 1.00 | - |
| Yes | 182 | 105,967 | 17.18 | 5.06 | (4.05 to 6.32) *** | 2.58 | (2.02 to 3.29) *** | 2.09 | (1.60 to 2.73) *** |
| PE | |||||||||
| Non-NTS | 109 | 450,270 | 2.42 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 53 | 88,183 | 6.01 | 2.46 | (1.77 to 3.41) *** | 2.36 | (1.69 to 3.30) *** | 1.84 | (1.30 to 2.60) *** |
| Gender | |||||||||
| Female | 76 | 248,049 | 3.06 | 1.00 | - | ||||
| Male | 86 | 290,404 | 2.96 | 0.96 | (0.71 to 1.31) | ||||
| Age | |||||||||
| 18–39 | 15 | 164,576 | 0.91 | 1.00 | - | 1.00 | - | 1.00 | - |
| 40–64 | 43 | 209,177 | 2.06 | 2.28 | (1.26 to 4.10) *** | 2.05 | (1.13 to 3.71) * | 1.96 | (1.08 to 3.56) * |
| ≥65 | 104 | 164,700 | 6.31 | 7.09 | (4.12 to 12.2) *** | 5.27 | (2.97 to 9.37) *** | 4.51 | (2.46 to 8.26) *** |
| Comorbidities | |||||||||
| No | 82 | 432,486 | 1.9 | 1.00 | - | 1.00 | - | 1.00 | - |
| Yes | 80 | 105,967 | 7.55 | 4.09 | (2.99 to 5.59) *** | 2.15 | (1.52 to 3.03) *** | 1.68 | (1.15 to 2.47) ** |
| Variables | Salmonellosis | Compared to Control | p for Interaction | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Yes | No | Crude HR ‡ | Adjusted HR § | ||||||||
| Events | Events | ||||||||||
| n | PY | Rate † | n | PY | Rate † | (95% CI) | (95% CI) | ||||
| DVT | |||||||||||
| Gender | 0.86 | ||||||||||
| Female | 54 | 42,315 | 12.8 | 100 | 205,734 | 4.86 | 2.61 | (1.87 to 3.63) *** | 2.34 | (1.67 to 3.28) *** | |
| Male | 55 | 45,868 | 12.0 | 115 | 244,536 | 4.70 | 2.48 | (1.80 to 3.42) *** | 2.26 | (1.63 to 3.14) *** | |
| Age, years | <0.001 | ||||||||||
| 18–39 | 16 | 30,710 | 5.21 | 6 | 133,866 | 0.45 | 11.4 | (4.44 to 29.0) *** | 5.95 | (2.22 to 15.9) *** | |
| 40–64 | 38 | 34,158 | 11.1 | 44 | 175,018 | 2.51 | 4.37 | (2.83 to 6.74) *** | 3.40 | (2.16 to 5.34) *** | |
| ≥65 | 55 | 23,315 | 23.6 | 165 | 141,385 | 11.67 | 1.99 | (1.47 to 2.70) *** | 1.68 | (1.23 to 2.30) ** | |
| Comorbidity | 0.54 | ||||||||||
| NO | 33 | 61,849 | 5.34 | 109 | 370,637 | 2.94 | 1.82 | (1.24 to 2.69) *** | 2.33 | (1.58 to 3.45) *** | |
| YES | 76 | 26,334 | 28.9 | 106 | 79,633 | 13.3 | 2.10 | (1.57 to 2.83) *** | 2.17 | (1.61 to 2.93) *** | |
| Follow-up time, months | 0.06 | ||||||||||
| ≤2 | 20 | 103 | 1950 | 2 | 26 | 775 | 0.70 | (0.31 to 1.58) | 0.71 | (0.31 to 1.67) | |
| 3–12 | 25 | 868 | 288 | 10 | 944 | 106 | 1.25 | (0.72 to 2.19) | 1.42 | (0.79 to 2.56) | |
| >12 | 64 | 87,212 | 7.34 | 97 | 449,300 | 2.16 | 1.84 | (1.38 to 2.44) *** | 1.75 | (1.31 to 2.34) *** | |
| PE | |||||||||||
| Gender | 0.06 | ||||||||||
| Female | 20 | 42,315 | 4.73 | 56 | 205,734 | 2.72 | 1.73 | (1.04 to 2.89) ** | 1.65 | (0.98 to 2.77) | |
| Male | 33 | 45,868 | 7.19 | 53 | 244,536 | 2.17 | 3.26 | (2.11 to 5.04) *** | 3.04 | (1.94 to 4.75) *** | |
| Age, years | 0.004 | ||||||||||
| 18–39 | 10 | 30,710 | 3.26 | 5 | 133,866 | 0.37 | 8.48 | (2.90 to 24.8) *** | 6.72 | (2.23 to 20.3) *** | |
| 40–64 | 20 | 34,158 | 5.86 | 23 | 175,018 | 1.31 | 4.39 | (2.41 to 8.00) *** | 3.28 | (1.75 to 6.13) *** | |
| ≥65 | 23 | 23,315 | 9.86 | 81 | 141,385 | 5.73 | 1.77 | (1.11 to 2.82 ) ** | 1.54 | (0.96 to 2.47) | |
| Comorbidity | <0.001 | ||||||||||
| NO | 24 | 61,849 | 3.88 | 58 | 370,637 | 1.56 | 2.47 | (1.54 to 3.98) *** | 3.05 | (1.89 to 4.92) *** | |
| YES | 29 | 26,334 | 11.01 | 51 | 79,633 | 6.40 | 1.70 | (1.08 to 2.68) ** | 1.74 | (1.10 to 2.77) * | |
| Follow-up time, months | 0.73 | ||||||||||
| ≤2 | 2 | 103 | 195 | 8 | 26 | 3100 | 1.16 | (0.25 to 5.38) | 1.12 | (0.22 to 5.73) | |
| 3–12 | 10 | 868 | 115 | 15 | 944 | 159 | 1.94 | (0.87 to 4.33) | 2.41 | (1.04 to 5.55) * | |
| >12 | 97 | 87,212 | 11.1 | 29 | 449,300 | 0.6 | 1.57 | (1.04 to 2.37) * | 1.56 | (1.03 to 2.38) * | |
| Variable | Event | Crude HR ‡ | Adjusted HR § | SHR | |||||
|---|---|---|---|---|---|---|---|---|---|
| n | PY | Rate† | (95% CI) | (95% CI) | (95% CI) | ||||
| Primary analysis (frequency matching) | |||||||||
| DVT | |||||||||
| non-NTS | 215 | 450,270 | 4.77 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 109 | 88,183 | 12.36 | 2.54 | (2.02 to 3.20) *** | 2.35 | (1.86 to 2.98) *** | 1.83 | (1.44 to 2.31) *** |
| PE | |||||||||
| non-NTS | 109 | 450,270 | 2.42 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 53 | 88,183 | 6.01 | 2.46 | (1.77 to 3.41) *** | 2.36 | (1.69 to 3.30) *** | 1.84 | (1.30 to 2.60) *** |
| Sensitivity analysis (PSM) | |||||||||
| DVT | |||||||||
| non-NTS | 256 | 425,265 | 6.02 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 102 | 88,109 | 11.6 | 1.90 | (1.51 to 2.38) *** | 2.17 | (1.72 to 2.73) *** | 1.69 | (1.34 to 2.13) *** |
| PE | |||||||||
| non-NTS | 113 | 425,265 | 2.66 | 1.00 | - | 1.00 | - | 1.00 | - |
| NTS | 47 | 88,109 | 5.33 | 1.98 | (1.41 to 2.78) *** | 2.16 | (1.53 to 3.05) *** | 1.71 | (1.21 to 2.40) ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, R.; Wu, D.-K.; Wei, J.C.-C.; Yip, H.-T.; Hung, Y.-M.; Hung, C.-H. The Risk of Subsequent Deep Vein Thrombosis and Pulmonary Embolism in Patients with Nontyphoidal Salmonellosis: A Nationwide Cohort Study. Int. J. Environ. Res. Public Health 2020, 17, 3567. https://doi.org/10.3390/ijerph17103567
Chang R, Wu D-K, Wei JC-C, Yip H-T, Hung Y-M, Hung C-H. The Risk of Subsequent Deep Vein Thrombosis and Pulmonary Embolism in Patients with Nontyphoidal Salmonellosis: A Nationwide Cohort Study. International Journal of Environmental Research and Public Health. 2020; 17(10):3567. https://doi.org/10.3390/ijerph17103567
Chicago/Turabian StyleChang, Renin, Den-Ko Wu, James Cheng-Chung Wei, Hei-Tung Yip, Yao-Min Hung, and Chih-Hsin Hung. 2020. "The Risk of Subsequent Deep Vein Thrombosis and Pulmonary Embolism in Patients with Nontyphoidal Salmonellosis: A Nationwide Cohort Study" International Journal of Environmental Research and Public Health 17, no. 10: 3567. https://doi.org/10.3390/ijerph17103567
APA StyleChang, R., Wu, D.-K., Wei, J. C.-C., Yip, H.-T., Hung, Y.-M., & Hung, C.-H. (2020). The Risk of Subsequent Deep Vein Thrombosis and Pulmonary Embolism in Patients with Nontyphoidal Salmonellosis: A Nationwide Cohort Study. International Journal of Environmental Research and Public Health, 17(10), 3567. https://doi.org/10.3390/ijerph17103567





