Phthalate Esters and Their Potential Risk in PET Bottled Water Stored under Common Conditions
Abstract
1. Introduction
2. Material and Methods
2.1. Consumer Survey and Brand Selection
2.2. Chemical Reagents
2.3. Measurement of PAEs in PET Bottles
2.4. Sample Treatment
2.4.1. Storage of PET Bottled Water under Normal Conditions
2.4.2. Effect of Temperature on the Concentration of PAEs in Commercial Bottled Water
2.4.3. Effect of Storage Temperature on the Concentrations of PAEs in Pure Water
2.5. Water Preparation
2.6. PAEs Analysis
2.7. Risk Assessment
2.8. Data Analysis
3. Results
3.1. Concentrations of PAEs in PET Bottles
3.2. Concentrations of PAEs in Commercial Bottled Water Stored under Common Conditions
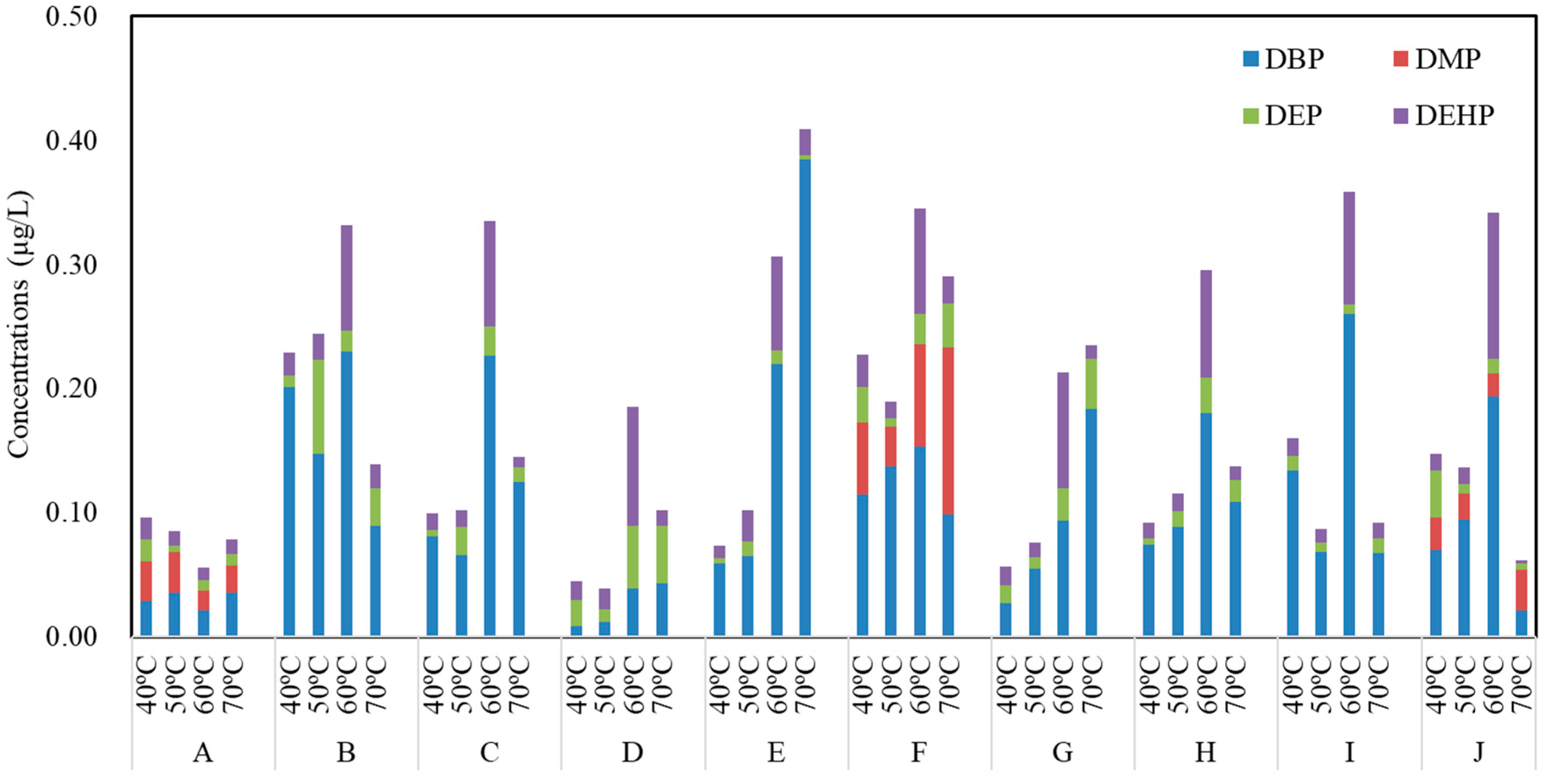
3.3. Concentrations of PAEs in Commercial Bottled Water Stored at Different Temperatures
3.4. Concentrations of PAEs in Pure Water Stored at Different Temperatures
3.5. Health Risk Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bottled Water Market. Global Industry Analysis, Size, Share, Growth, Trends and Forecast 2017–2024; Rep id: TMRGL658; Bottled Water Market: London, UK, 2018; p. 283. [Google Scholar]
- Larsen, S.T.; Hansen, J.S.; Hansen, E.W.; Clausen, P.A.; Nielsen, G.D. Airway inflammation and adjuvants effect after repeated airborne exposures to di-(2-ethylhexyl) phthalate and ovalbumin in BALB/c mice. Toxicology 2007, 235, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Diana, A.; Dimitra, V. Alkylphenols and phthalates in bottled waters. J. Hazard. Mater. 2011, 185, 281–286. [Google Scholar]
- Bach, C.; Dauchy, X.; Chagnon, M.C.; Etienne, S. Chemical compounds and toxicological assessments of drinking water stored in polyethylene terephthalate (PET) bottles: A source of controversy reviewed. Water Res. 2012, 46, 571–583. [Google Scholar] [CrossRef] [PubMed]
- Sejidov, F.T.; Mansoori, Y.; Goodarzi, N. Esterification reaction using solid heterogeneous acid catalysts under solvent-less condition. J. Mol. Catal. A 2005, 240, 186–190. [Google Scholar] [CrossRef]
- Cao, X.L. Determination of phthalates and adipate in bottled water by headspace solidphase microextraction and gas chromatography/mass spectrometry. J. Chromatogr. A 2008, 1178, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Casajuana, N.; Lacorte, S. Presence and release of phthalic esters and other endocrine disrupting compounds in drinking water. Chromatographia 2003, 57, 649–655. [Google Scholar] [CrossRef]
- Jeddi, M.Z.; Rastkari, N.; Ahmadkhaniha, R.; Yunesian, M. Concentrations of phthalates in bottled water under common storage conditions: Do they pose a health risk to children? Food Res. Int. 2015, 69, 256–265. [Google Scholar] [CrossRef]
- Bošnir, J.; Puntarić, D.; Galić, A.; Škes, I.; Dijanić, T.; Klarić, M.; Grgić, M.; Čurković, M.; Šmit, Z. Migration of phthalates from plastic containers into soft drinks and mineral water. Food Technol. Biotechnol. 2007, 45, 91–95. [Google Scholar]
- Li, H.; Li, C.M.; An, L.H.; Deng, C.; Su, H.; Wang, L.F.; Jiang, Z.J.; Zhou, J.; Wang, J.; Zhang, C.H.; et al. Phthalate esters in bottled drinking water and their human exposure in Beijing, China. Food Addit. Contam. B 2019, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.K.; Fung, L.C.; Pang, M. of six phthalates in polypropylene consumer products by sonication-assisted extraction/GC-MS methods. Malays. J. Anal. Sci. 2011, 15, 167–174. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality, 3rd ed.; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- Ministry of Health of the People’s Republic of China; Standardization Administration of the People’s Republic of China. Standard for Drinking Water Quality; GB 5749-2006; Ministry of Health of the People’s Republic of China: Beijing, China; Standardization Administration of the People’s Republic of China: Beijing, China, 2006.
- EPA. Drinking Water Contaminants; EPA 816-F-09-0004; U.S. Environmental Protection Agency: Washington, DC, USA, 2009.
- Wagner, M.; Oehlmann, J. Endocrine disruptors in bottled mineral water: Total estrogenic burden and migration from plastic bottles. Environ. Sci. Pollut. Res. 2009, 16, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Keresztes, S.; Tatár, E.; Czégény, Z.; Záray, G.; Mihucz, V.G. Study on the leaching of phthalates from polyethylene terephthalate bottles into mineral water. Sci. Total Environ. 2013, 458, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.J.; Hu, X.J.; Lin, S.B. Determination of 15 phthalate esters in drinking water by solid phase extraction-ultra performance liquid chromatography-tandem mass spectrometry. Chin. J. Anal. Chem. 2014, 42, 1281–1287. [Google Scholar]
- Fatoki, O.S.; Noma, A. Determination of phthalate esters in the aquatic environment. S. Afr. J. Chem. 2001, 54, 69–83. [Google Scholar]
- Zeng, F.; Wen, J.; Cui, K.; Wu, L.; Liu, M.; Li, Y.; Lin, Y.; Zhu, F.; Ma, Z.; Zeng, Z. Seasonal distribution of phthalate esters in surface water of the urban lakes in the subtropical city, Guangzhou, China. J. Hazard. Mater. 2009, 169, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Sun, J.; Chen, Z.; Chen, X.; Jing, J.; Liu, J.; Zhang, Y. Levels and sources of phthalate esters in shallow groundwater and surface water of Dongguan city, South China. Geochem. J. 2012, 46, 421–428. [Google Scholar] [CrossRef]
- Chidya, R.C.G.; Singano, L.; Chitedze, I.; Mourad, K.A. Standards compliance and health implications of bottled water in Malawi. Int. J. Environ. Res. Public Health 2019, 16, 951. [Google Scholar] [CrossRef] [PubMed]
- Ceretti, E.; Zani, C.; Zerbini, I.; Guzzella, L.; Scaglia, M.; Berna, V.; Donato, F.; Monarca, S.; Feretti, D. Comparative assessment of genotoxicity of mineral water packed in polyethylene terephthalate (PET) and glass bottles. Water Res. 2010, 44, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Z.; Yaniger, S.I.; Jordan, V.C.; Klein, D.J.; Bittner, G.D. Most plastic products release estrogenic chemicals: A potential health problem that can be solved. Environ. Health Perspect. 2011, 119, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Bittner, G.D.; Yang, C.Z.; Stoner, M.A. Estrogenic chemicals often leach from BPA-free plastic products that are replacements for BPA-containing polycarbonate products. Environ. Health 2014, 13, 41. [Google Scholar] [CrossRef] [PubMed]
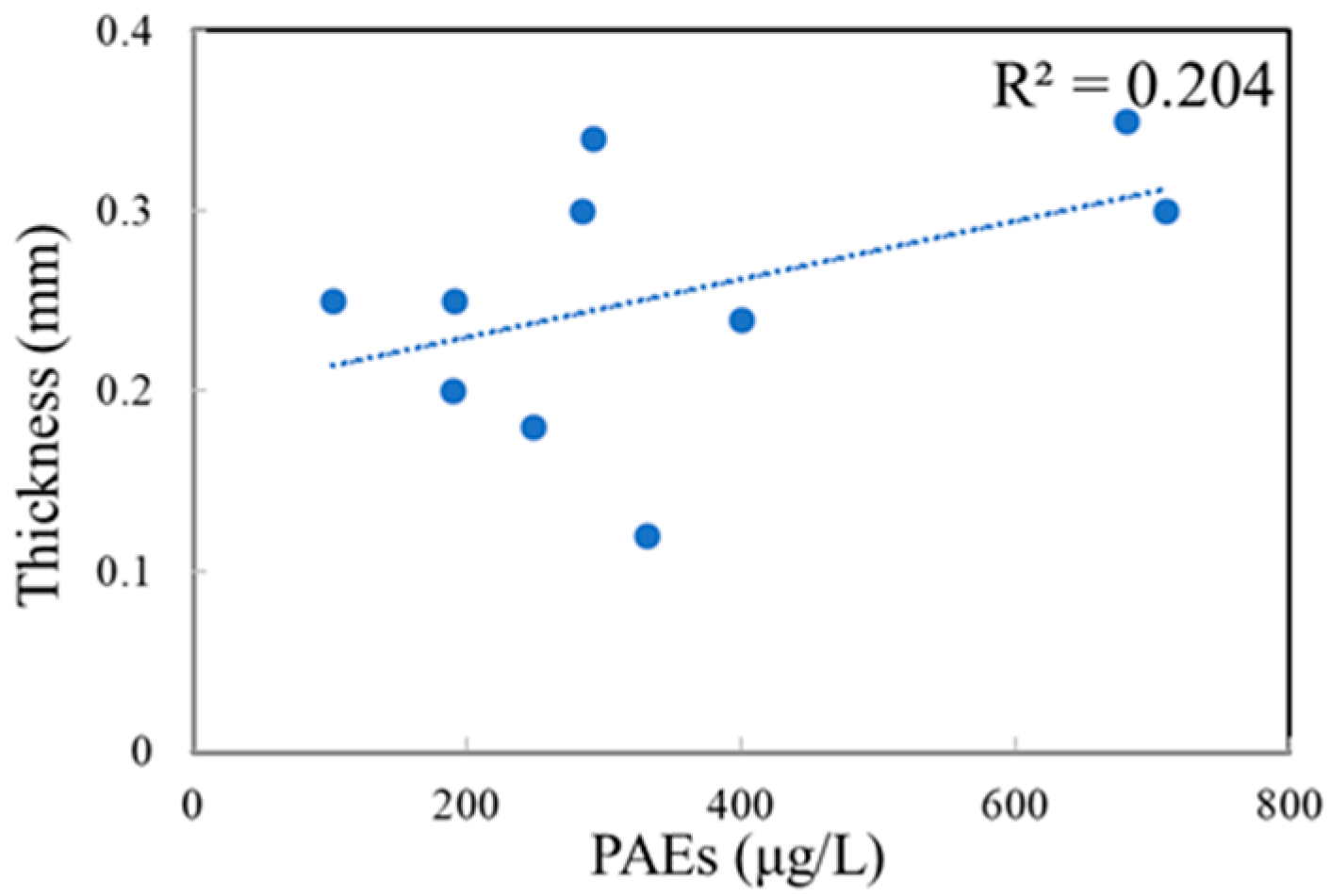
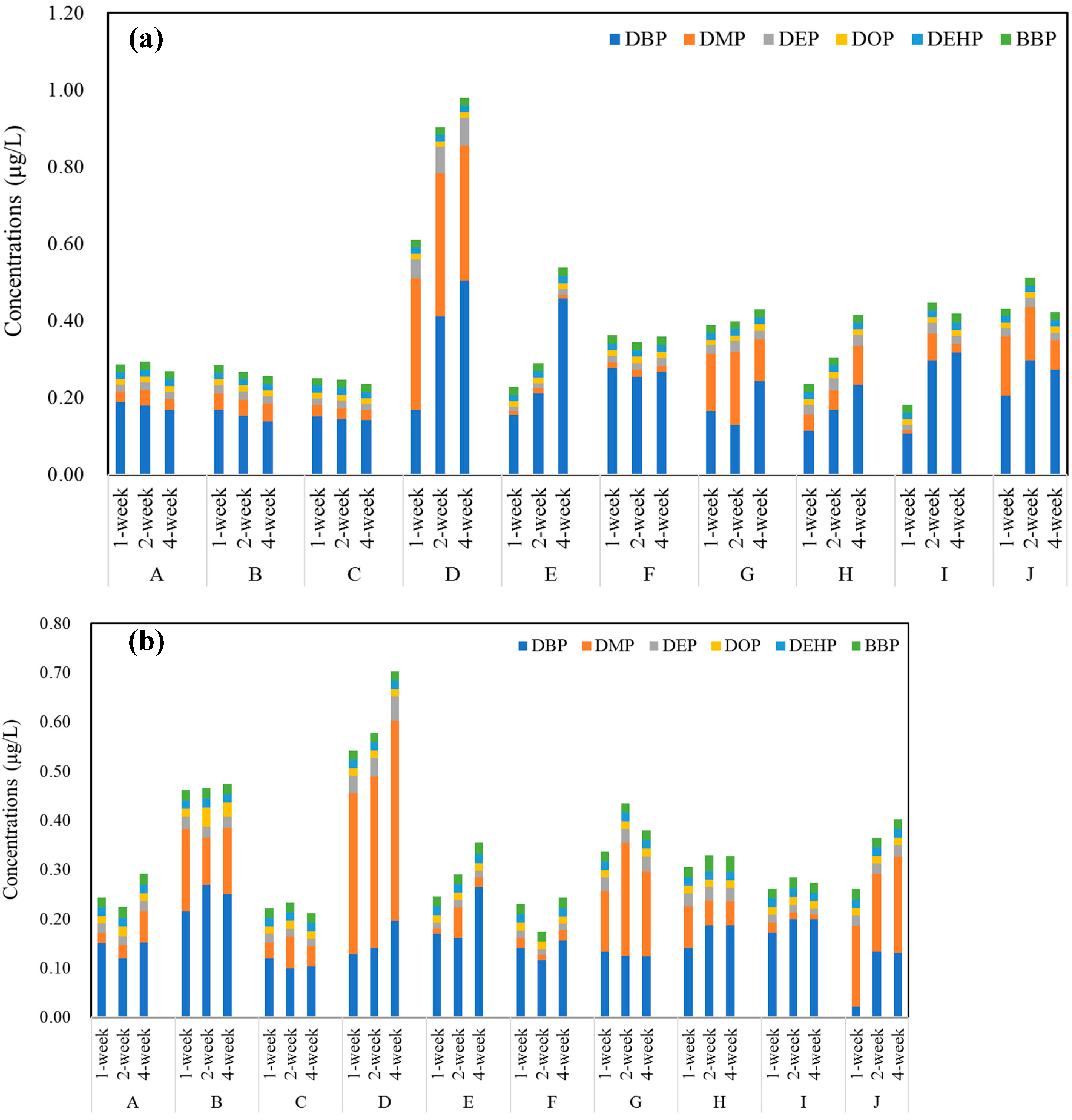
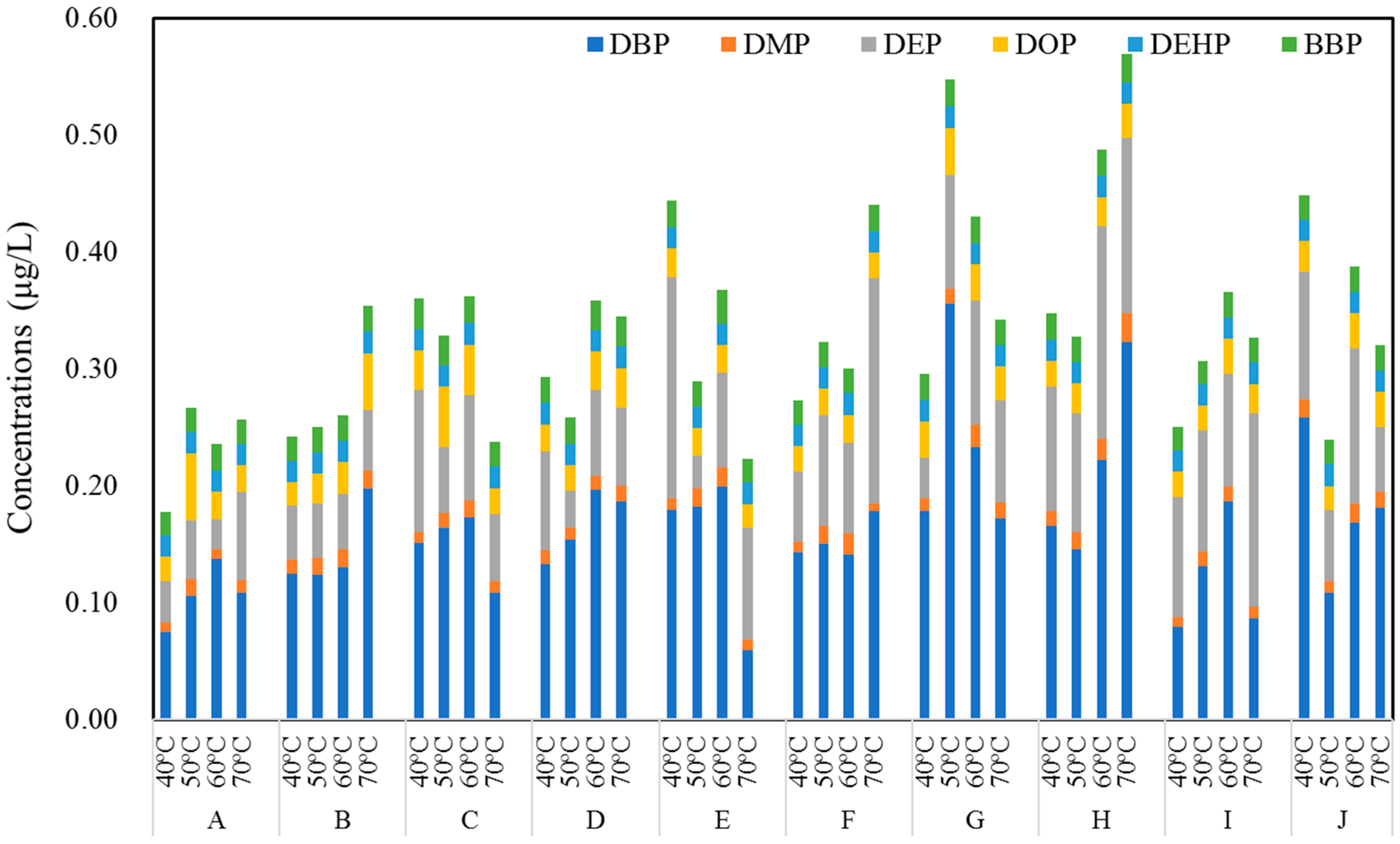
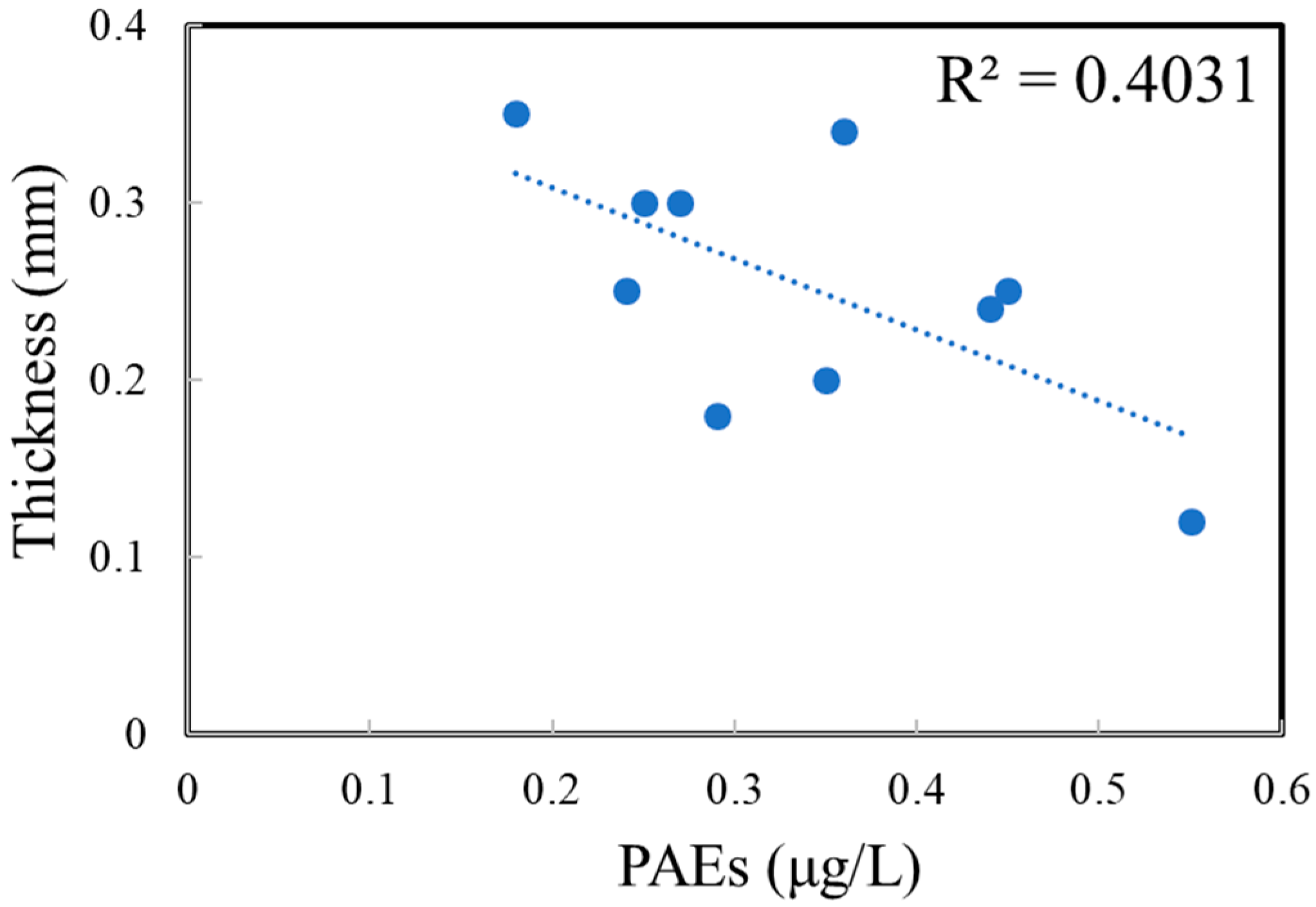
| Brands | Product Date | Bottle Size | Bottle Color | Bottle Thickness/mm | Bottled Water | Price (¥/bottle) |
|---|---|---|---|---|---|---|
| A | March, 2016 | 510 mL | Clear | 0.35 | Natural mineral water | 5.10 |
| B | March, 2016 | 570 mL | Light blue | 0.25 | Natural mineral water | 2.30 |
| C | February, 2016 | 600 mL | Clear | 0.34 | Natural mineral water | 2.80 |
| D | March, 2016 | 550 mL | Light blue | 0.18 | Purified drinking water | 2.70 |
| E | February, 2016 | 550 mL | Clear | 0.24 | Natural mineral water | 1.10 |
| F | February, 2016 | 500 mL | Clear | 0.30 | Natural mineral water | 2.30 |
| G | March, 2016 | 550 mL | Light blue | 0.12 | Purified drinking water | 2.10 |
| H | March, 2016 | 596 mL | Clear | 0.20 | Purified drinking water | 1.40 |
| I | March, 2016 | 555 mL | Clear | 0.30 | Purified drinking water | 1.70 |
| J | March, 2016 | 550 mL | Light blue | 0.25 | Purified drinking water | 1.40 |
| A | B | C | D | E | F | G | H | I | J | |
|---|---|---|---|---|---|---|---|---|---|---|
| DEP | 182.43 ± 5.03 | 29.92 ± 2.11 | 50.73 ± 4.12 | 49.47 ± 1.02 | 114.83 ± 4.23 | 22.88 ± 1.44 | 45.78 ± 3.02 | 34.83 ± 2.35 | 68.08 ± 5.66 | ND |
| DMP | 188.58 ± 8.20 | 34.12 ± 3.35 | 41.90 ± 2.15 | 16.12 ± 0.44 | 9.07 ± 0.24 | 58.10 ± 8.41 | 133.93 ± 9.54 | 19.50 ± 2.01 | 130.27 ± 11.30 | 39.07 ± 5.20 |
| DBP | 310.12 ± 15.22 | 126.20 ± 7.84 | 199.52 ± 9.55 | 334.82 ± 6.45 | 159.32 ± 14.55 | 167.20 ± 7.74 | 150.57 ± 11.45 | 134.42 ± 15.44 | 511.52 ± 22.07 | 62.90 ± 8.04 |
| Total | 681.13 | 190.23 | 292.15 | 400.40 | 283.22 | 248.18 | 330.28 | 188.75 | 709.87 | 101.97 |
| Storage at Indoor (24 ± 1 °C) | Storage at Outdoor (22.5–44.4 °C) | |||||||
|---|---|---|---|---|---|---|---|---|
| DEHP | DEP | DBP | BBP | DEHP | DEP | DBP | BBP | |
| Maximum Concentration | 0.02 | 0.05 | 0.26 | 0.03 | 0.02 | 0.07 | 0.51 | 0.03 |
| EDI a | 0.0003 | 0.0008 | 0.0043 | 0.0005 | 0.0003 | 0.0012 | 0.0085 | 0.0005 |
| RfD b | 20 | 800 | 100 | 200 | 20 | 800 | 100 | 200 |
| HQ c | 1.67 × 10−5 | 1.04 × 10−6 | 4.33 × 10−5 | 2.50 × 10−6 | 1.67 × 10−5 | 1.46 × 10−6 | 8.50 × 10−5 | 2.50 × 10−6 |
| Storage at 40 °C | Storage at 70 °C | |||||||
| DEHP | DEP | DEHP | DEP | DEHP | DEP | DEHP | DEP | |
| Maximum Concentration | 0.02 | 0.19 | 0.02 | 0.19 | 0.02 | 0.19 | 0.02 | 0.19 |
| EDI a | 0.0003 | 0.0032 | 0.0003 | 0.0032 | 0.0003 | 0.0032 | 0.0003 | 0.0032 |
| RfD b | 20 | 800 | 20 | 800 | 20 | 800 | 20 | 800 |
| HQ c | 1.67 × 10−5 | 3.96 × 10−6 | 1.67 × 10−5 | 3.96 × 10−6 | 1.67 × 10−5 | 3.96 × 10−6 | 1.67 × 10−5 | 3.96 × 10−6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, X.; Zhou, G.; Lei, K.; LeBlanc, G.A.; An, L. Phthalate Esters and Their Potential Risk in PET Bottled Water Stored under Common Conditions. Int. J. Environ. Res. Public Health 2020, 17, 141. https://doi.org/10.3390/ijerph17010141
Xu X, Zhou G, Lei K, LeBlanc GA, An L. Phthalate Esters and Their Potential Risk in PET Bottled Water Stored under Common Conditions. International Journal of Environmental Research and Public Health. 2020; 17(1):141. https://doi.org/10.3390/ijerph17010141
Chicago/Turabian StyleXu, Xiangqin, Gang Zhou, Kun Lei, Gerald A. LeBlanc, and Lihui An. 2020. "Phthalate Esters and Their Potential Risk in PET Bottled Water Stored under Common Conditions" International Journal of Environmental Research and Public Health 17, no. 1: 141. https://doi.org/10.3390/ijerph17010141
APA StyleXu, X., Zhou, G., Lei, K., LeBlanc, G. A., & An, L. (2020). Phthalate Esters and Their Potential Risk in PET Bottled Water Stored under Common Conditions. International Journal of Environmental Research and Public Health, 17(1), 141. https://doi.org/10.3390/ijerph17010141






