Spatially Filtered Multilevel Analysis on Spatial Determinants for Malaria Occurrence in Korea
Abstract
1. Introduction
2. Study Area, Data, and Methodology
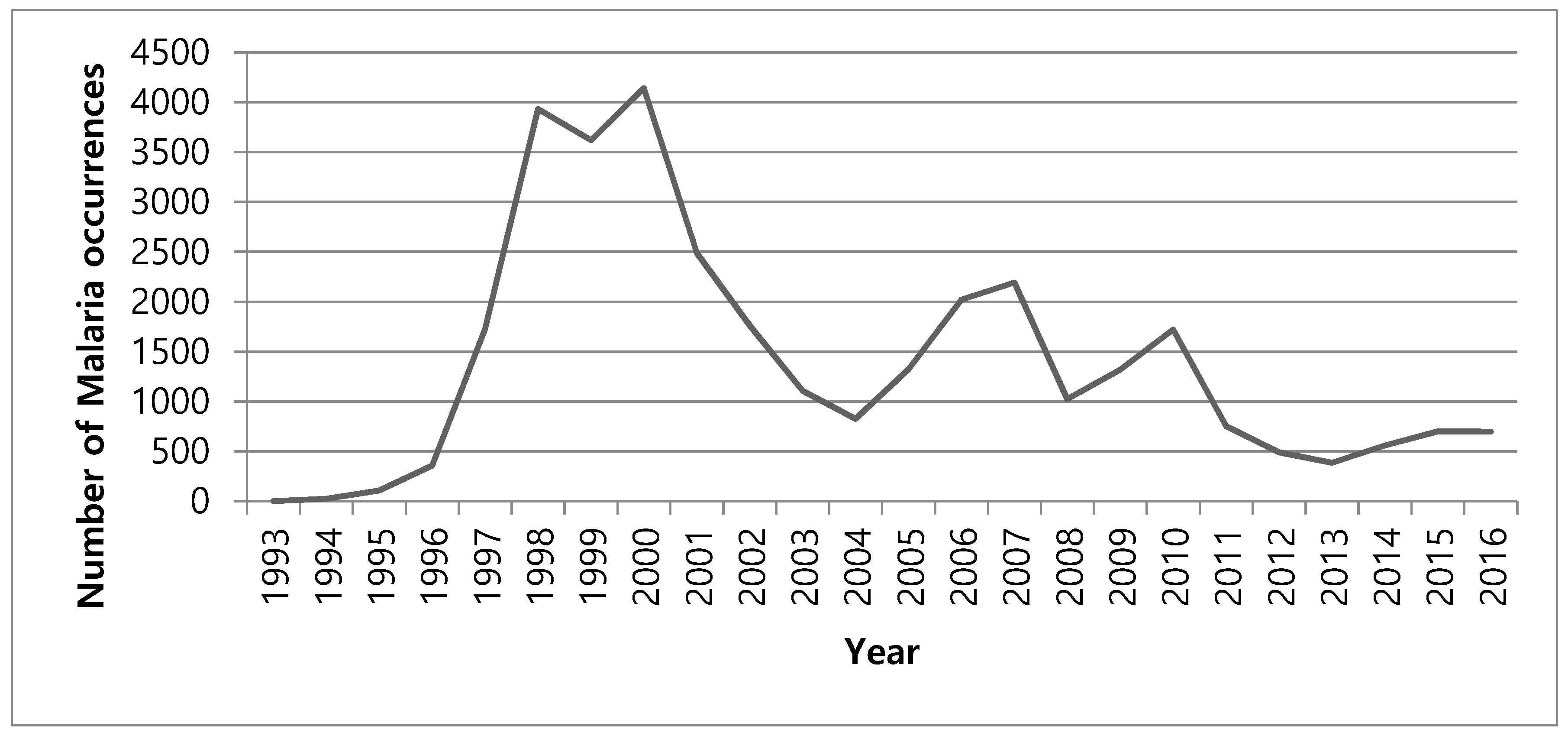
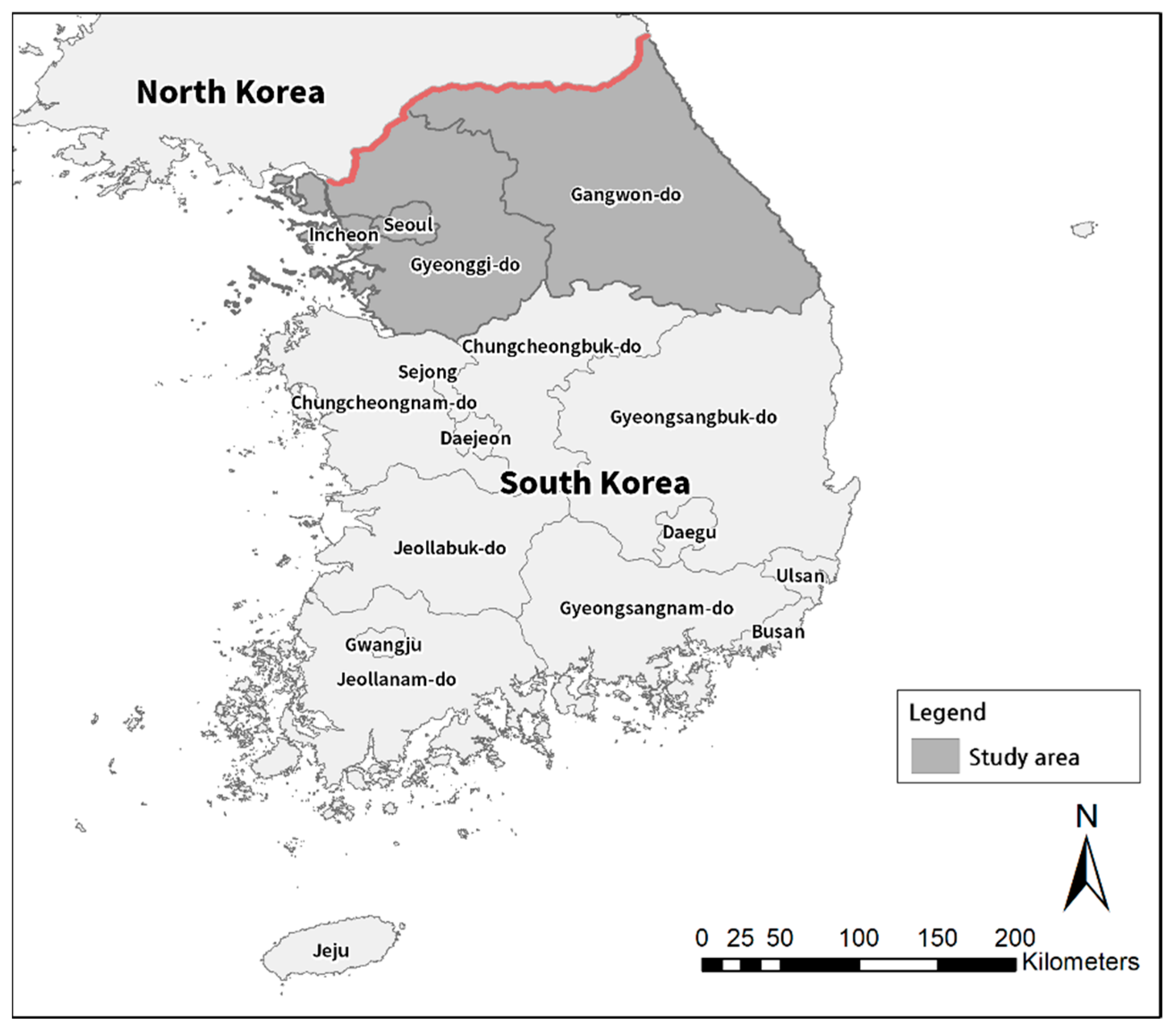
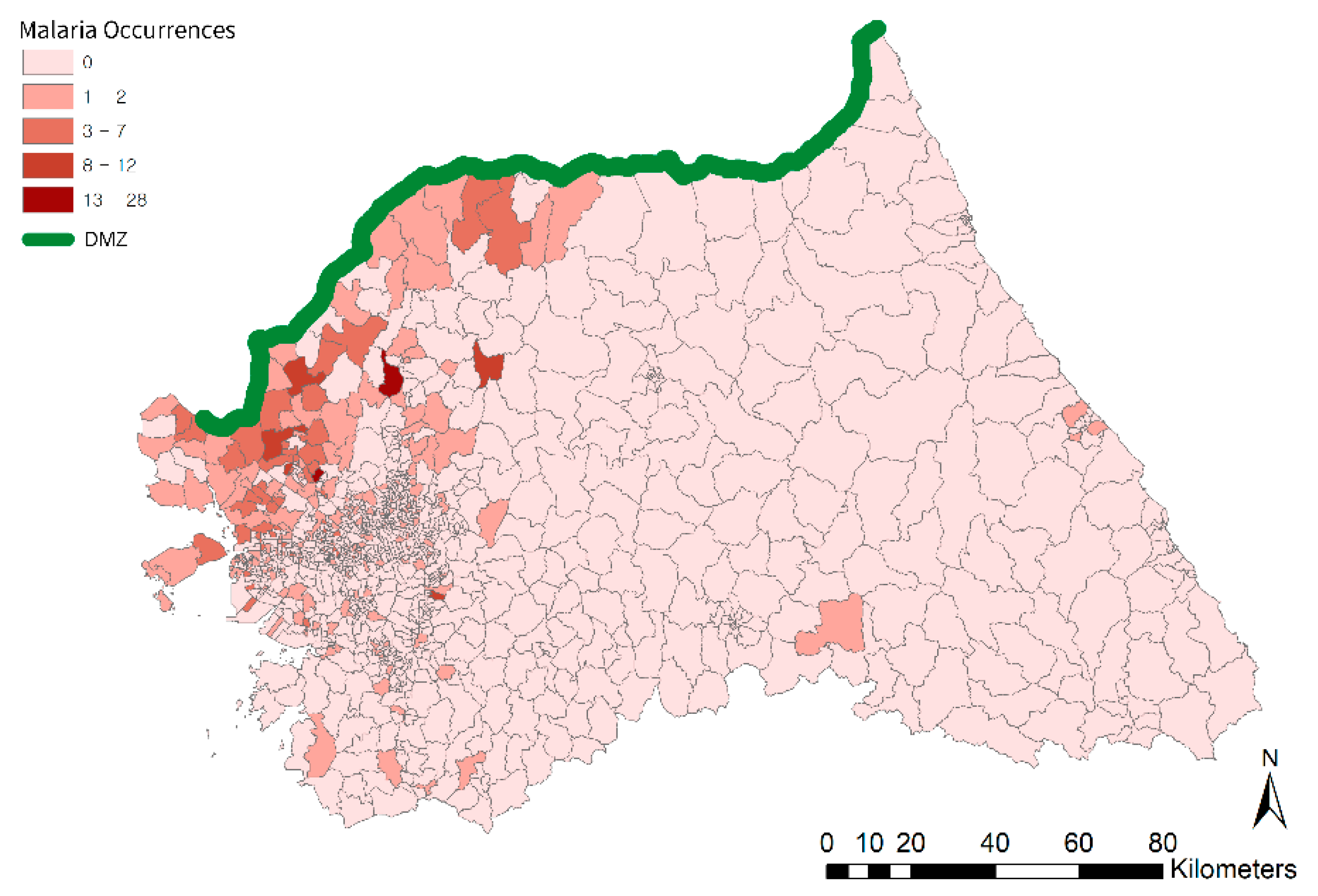
2.1. Study Area and Data
2.2. Analysis Methods
2.2.1. Multilevel Model
2.2.2. Spatially Filtered Multilevel Model
3. Results
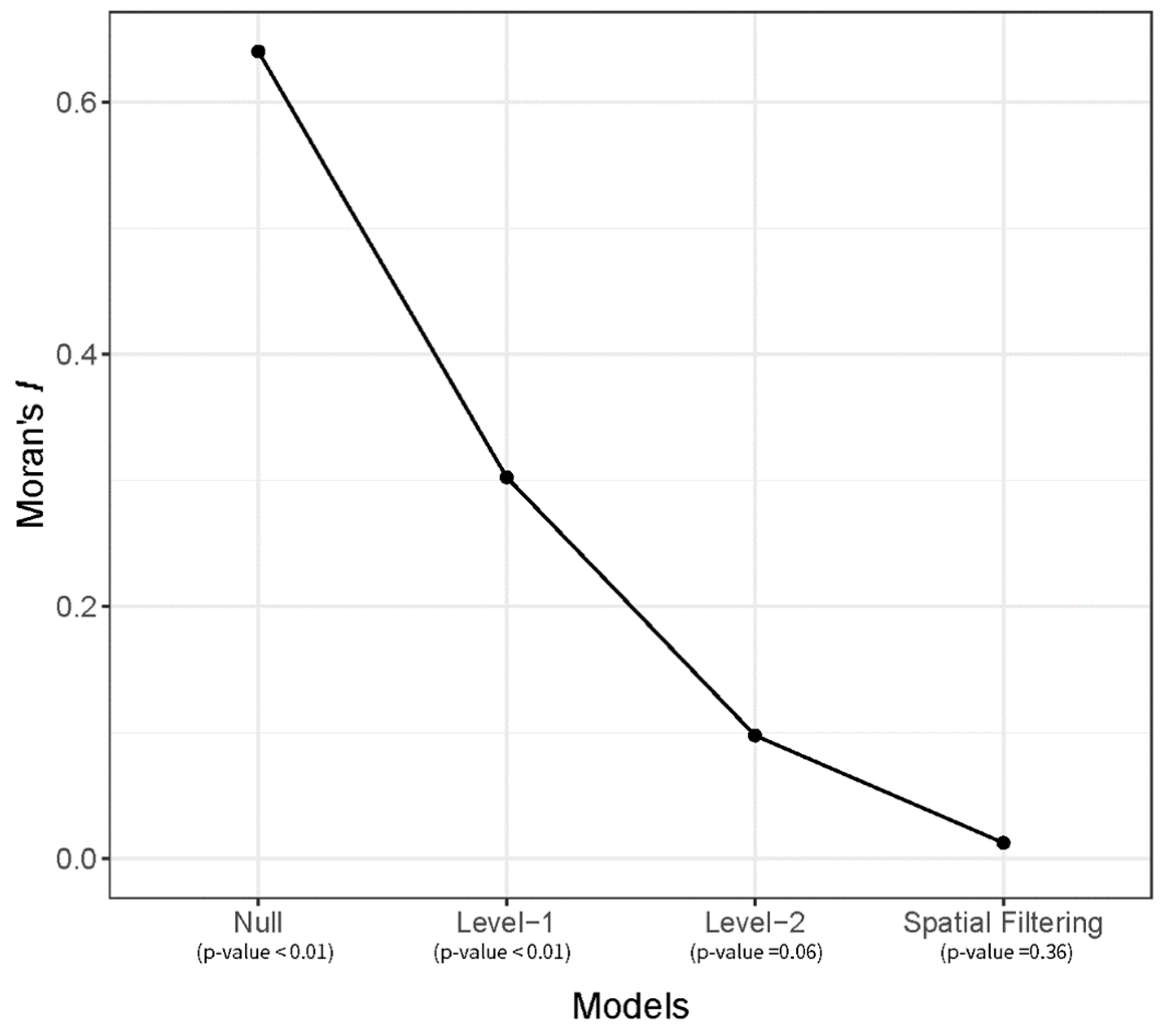
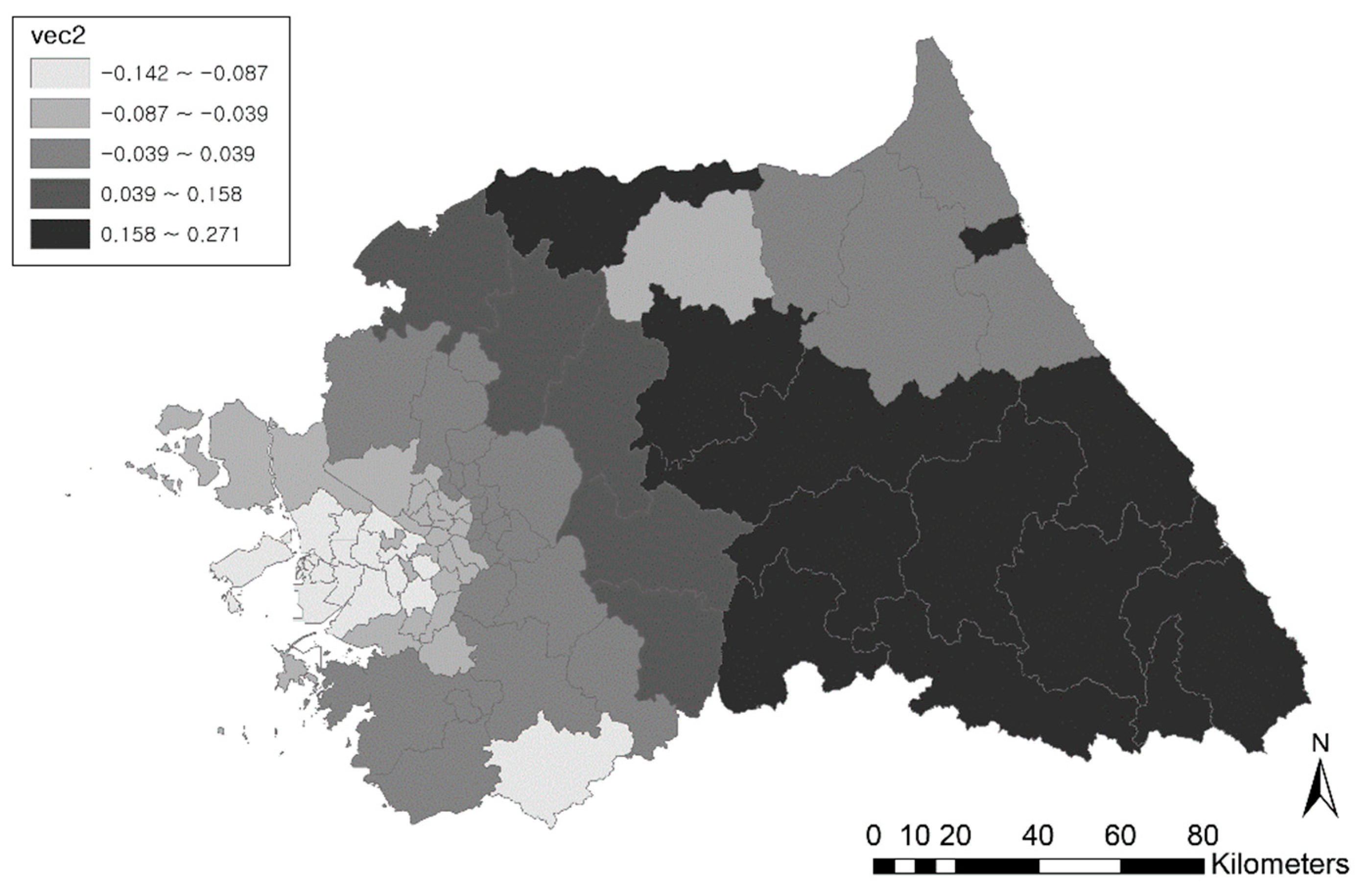
3.1. Spatial Filtering Process
3.2. Modeling Results
3.3. Results Interpretation—Spatially Filtered Multilevel Model
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Synopsis of the world malaria situation. Wkly. Epidemiol. Rec. 1979, 56, 145–149. [Google Scholar]
- Warrell, D.A.; Gilles, H.M. Essential Malariology, 4th ed.; CRC Press: New York, NY, USA, 2002; pp. 85–105. ISBN 9780340740644. [Google Scholar]
- Reisen, W.K. Landscape Epidemiology of Vector-Borne Diseases. Annu. Rev. Entomol. 2010, 55, 461–483. [Google Scholar] [CrossRef]
- Chai, J.Y. Re-emerging Plasmodium vivax malaria in the Republic of Korea. Korean J. Parasitol. 1999, 37, 129–143. [Google Scholar] [CrossRef]
- Yeom, J.-S.; Park, J.-W. Status of Vivax Malaria after Re-emergence in South Korea. Infect. Chemother. 2008, 40, 191. [Google Scholar] [CrossRef]
- Ree, H.-I. Unstable vivax malaria in Korea. Korean J. Parasitol. 2000, 38, 119. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ministry of Health and Welfare & Korea Centers for Disease Control and Prevention. Malaria Administrative Guideline; Human Culture Arirang: Yangpyeong, Korea, 2016; ISBN 9791159671708. [Google Scholar]
- Bousema, T.; Griffin, J.T.; Sauerwein, R.W.; Smith, D.L.; Churcher, T.S.; Takken, W.; Ghani, A.; Drakeley, C.; Gosling, R. Hitting Hotspots: Spatial Targeting of Malaria for Control and Elimination. PLoS Med. 2012, 9, e1001165. [Google Scholar] [CrossRef]
- Mauny, F. Multilevel modelling and malaria: A new method for an old disease. Int. J. Epidemiol. 2004, 33, 1337–1344. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Peterson, I.; Borrell, L.N.; El-Sadr, W.; Teklehaimanot, A. Individual and Household Level Factors Associated with Malaria Incidence in a Highland Region of Ethiopia: A Multilevel Analysis. Am. J. Trop. Med. Hyg. 2009, 80, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Messina, J.P.; Taylor, S.M.; Meshnick, S.R.; Linke, A.M.; Tshefu, A.K.; Atua, B.; Mwandagalirwa, K.; Emch, M. Population, behavioural and environmental drivers of malaria prevalence in the Democratic Republic of Congo. Malar. J. 2011, 10, 161. [Google Scholar] [CrossRef] [PubMed]
- Woyessa, A.; Deressa, W.; Ali, A.; Lindtjørn, B. Malaria risk factors in Butajira area, south-central Ethiopia: A multilevel analysis. Malar. J. 2013, 12, 273. [Google Scholar] [CrossRef]
- Samadoulougou, S.; Maheu-Giroux, M.; Kirakoya-Samadoulougou, F.; De Keukeleire, M.; Castro, M.C.; Robert, A. Multilevel and geo-statistical modeling of malaria risk in children of Burkina Faso. Parasit. Vectors 2014, 7, 350. [Google Scholar] [CrossRef]
- Incardona, S.; Vong, S.; Chiv, L.; Lim, P.; Nhem, S.; Sem, R.; Khim, N.; Doung, S.; Mercereau-Puijalon, O.; Fandeur, T. Large-scale malaria survey in Cambodia: Novel insights on species distribution and risk factors. Malar. J. 2007, 6, 37. [Google Scholar] [CrossRef]
- Ren, Z.; Wang, D.; Hwang, J.; Bennett, A.; Sturrock, H.J.W.; Ma, A.; Huang, J.; Xia, Z.; Feng, X.; Wang, J. Spatial-Temporal Variation and Primary Ecological Drivers of Anopheles sinensis Human Biting Rates in Malaria Epidemic-Prone Regions of China. PLoS ONE 2015, 10, e0116932. [Google Scholar] [CrossRef]
- Noh, M.; Lee, Y.; Oh, S.; Chu, C.; Gwack, J.; Youn, S.-K.; Cho, S.H.; Lee, W.J.; Huh, S. Spatial and Temporal Distribution of Plasmodium vivax Malaria in Korea Estimated with a Hierarchical Generalized Linear Model. Osong Public Health Res. Perspect. 2012, 3, 192–198. [Google Scholar] [CrossRef]
- Snijders, T.A.; Bosker, R. Multilevel Analysis: An Introduction to Basic and Applied Multilevel Analysis; Sage: London, UK, 1999; pp. 41–73. ISBN 978-1-84920-200-8. [Google Scholar]
- Jerrett, M.; Gale, S.; Kontgis, C. Spatial modeling in environmental and public health research. Int. J. Environ. Res. Public Health 2010, 7, 1302–1329. [Google Scholar] [CrossRef]
- Corrado, L.; Fingleton, B. Multilevel Modelling with Spatial Effects; No. 2011-13; Scottish Institute for Research in Economics (SIRE): Edinburgh, Scotland, 2011. [Google Scholar]
- Langford, I.H.; Leyland, A.H.; Rasbash, J.; Goldstein, H. Multilevel Modelling of the Geographical Distributions of Diseases. J. R. Stat. Soc. Ser. C Appl. Stat. 1999, 48, 253–268. [Google Scholar] [CrossRef]
- Griffith, D.A. A comparison of six analytical disease mapping techniques as applied to West Nile Virus in the coterminous United States. Int. J. Health Geogr. 2005, 4, 18. [Google Scholar] [CrossRef]
- Park, Y.; Kim, Y. A spatially filtered multilevel model to account for spatial dependency: Application to self-rated health status in South Korea. Int. J. Health Geogr. 2014, 13, 6. [Google Scholar] [CrossRef]
- Koram, K.A.; Bennett, S.; Adiamah, J.H.; Greenwood, B.M. Socio-economic risk factors for malaria in a peri-urban area of The Gambia. Trans. R. Soc. Trop. Med. Hyg. 1995, 89, 146–150. [Google Scholar] [CrossRef]
- Lee, J.S.; Lee, W.J.; Cho, S.H.; Ree, H.-I. Outbreak of vivax malaria in areas adjacent to the demilitarized zone, South Korea, 1998. Am. J. Trop. Med. Hyg. 2002, 66, 13–17. [Google Scholar] [CrossRef]
- Sithiprasasna, R.; Ja Lee, W.; Ugsang, D.M.; Linthicum, K.J. Identification and characterization of larval and adult anopheline mosquito habitats in the Republic of Korea: Potential use of remotely sensed data to estimate mosquito distributions. Int. J. Health Geogr. 2005, 4, 17. [Google Scholar] [CrossRef]
- Ree, H.-I. Studies on Anopheles sinensis, the vector species of vivax malaria in Korea. Korean J. Parasitol. 2005, 43, 75. [Google Scholar] [CrossRef]
- Yeom, J.-S. Current status and outlook of mosquito-borne diseases in Korea. J. Korean Med. Assoc. 2017, 60, 468. [Google Scholar] [CrossRef]
- Im, J.H.; Kwon, H.Y.; Baek, J.; Park, S.W.; Durey, A.; Lee, K.H.; Chung, M.-H.; Lee, J.-S. Severe Plasmodium vivax infection in Korea. Malar. J. 2017, 16. [Google Scholar] [CrossRef]
- Chang, K.S.; Yoo, D.-H.; Ju, Y.R.; Lee, W.G.; Roh, J.Y.; Kim, H.-C.; Klein, T.A.; Shin, E.-H. Distribution of malaria vectors and incidence of vivax malaria at Korean army installations near the demilitarized zone, Republic of Korea. Malar. J. 2016, 15. [Google Scholar] [CrossRef]
- Kim, H.-C.; Pacha, L.A.; Lee, W.-J.; Lee, J.-K.; Gaydos, J.C.; Sames, W.J.; Lee, H.-C.S.; Bradley, K.; Jeung, G.-G.; Tobler, S.K.; et al. Malaria in the Republic of Korea, 1993–2007. Variables Related to Re-emergence and Persistence of Plasmodium vivax Among Korean Populations and U.S. Forces in Korea. Mil. Med. 2009, 174, 762–769. [Google Scholar] [CrossRef]
- Park, J.-W.; Klein, T.A.; Lee, H.-C.; Pacha, L.A.; Ryu, S.-H.; Yeom, J.-S.; Moon, S.-H.; Kim, T.-S.; Chai, J.-Y.; Oh, M.-D.; et al. Vivax malaria: A continuing health threat to the Republic of Korea. Am. J. Trop. Med. Hyg. 2003, 69, 159–167. [Google Scholar] [CrossRef]
- Consul, P.C.; Jain, G.C. A generalization of the Poisson distribution. Technometrics 1973, 15, 791–799. [Google Scholar] [CrossRef]
- Wakefield, J. Disease mapping and spatial regression with count data. Biostatistics 2007, 8, 158–183. [Google Scholar] [CrossRef]
- Finch, W.H.; Bolin, J.E.; Kelley, K. Multilevel Modeling Using R; CRC Press: New York, NY, USA, 2014; pp. 141–166. ISBN 978-1-4665-1586-4. [Google Scholar]
- Raudenbush, S.W.; Bryk, A.S. Hierarchical Linear Models: Applications and Data Analysis Methods; Sage: Thousand Oaks, CA, USA, 2002; pp. 309–316. ISBN 978-0761919049. [Google Scholar]
- Bivand, R.S.; Pebesma, E.J.; Gomez-Rubio, V.; Pebesma, E.J. Applied Spatial Data Analysis with R; Springer: New York, NY, USA, 2008; ISBN 978-1-4614-7618-4. [Google Scholar]
- Casman, E.A.; Dowlatabadi, H. The Contextual Determinants of Malaria; Resources for the Future: Washington, DC, USA, 2002; pp. 93–110. ISBN 1-891853-19-8. [Google Scholar]
| Categories | Variables | Unit | Mean | Standard Deviation | References | |
|---|---|---|---|---|---|---|
| Dependent variable | Malaria occurrences | Count | 0.376 | 1.415 | ||
| Independent variables | District level (Eup, Myeon, and Dong) | Housing cost | 1000 | 1580.0 | 1360.3 | [2,9,10,11,12,13,14,23] |
| Sex ratio | - | 1.019 | 0.092 | [2,9,12,14,16] | ||
| Average age | - | 41.26 | 4.559 | [10,13,14,15,16] | ||
| Rice paddy area ratio | % | 4.1 | 8.756 | [15,24,25] | ||
| Distance from DMZ | km | 4.482 | 2.663 | [7,24,26,27,28] | ||
| City and county level (Si, Gun, and Gu) | Health budget per capita | \ | 47,337 | 29,278 | [2,11,13,14] | |
| Military base area | % | 16.78 | 25.21 | [7,24,26,27,29,30] | ||
| Categories | Multilevel Models | Spatially Filtered Multilevel Model | ||
|---|---|---|---|---|
| Null Model | Level-1 Multilevel | Level-2 Multilevel | ||
| Fixed effects | ||||
| Level-1 Variables | ||||
| Housing costs | - | −0.03 | −0.17 | −0.24 * |
| Sex ratio | - | 0.06 | 0.07 | 0.07 |
| Average age | - | −0.25 ** | −0.21 ** | −0.20 ** |
| Rice paddy ratio | - | 0.15 ** | 0.15 ** | 0.15 ** |
| DMZ distance | - | −1.31 ** | −1.31 ** | −1.20 ** |
| Level-2 Variables | ||||
| Health budget per capita | - | - | −0.91 ** | −0.82 ** |
| Military base area | - | - | 0.22 ** | 0.32 ** |
| Random effects | ||||
| Group-level variance | 1.74 | 0.50 | 0.25 | 0.20 |
| Constant | −1.94 | −2.06 | −2.27 | −2.31 |
| AIC | 1998.8 | 1901.5 | 1882.0 | 1879.3 |
| Log-likelihood | −997.4 | −943.8 | −932.0 | −929.6 |
| Moran’s I of random effect | 0.64 ** | 0.30 ** | 0.10 (p = 0.06) | 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Kim, Y. Spatially Filtered Multilevel Analysis on Spatial Determinants for Malaria Occurrence in Korea. Int. J. Environ. Res. Public Health 2019, 16, 1250. https://doi.org/10.3390/ijerph16071250
Kim S, Kim Y. Spatially Filtered Multilevel Analysis on Spatial Determinants for Malaria Occurrence in Korea. International Journal of Environmental Research and Public Health. 2019; 16(7):1250. https://doi.org/10.3390/ijerph16071250
Chicago/Turabian StyleKim, Sehyeong, and Youngho Kim. 2019. "Spatially Filtered Multilevel Analysis on Spatial Determinants for Malaria Occurrence in Korea" International Journal of Environmental Research and Public Health 16, no. 7: 1250. https://doi.org/10.3390/ijerph16071250
APA StyleKim, S., & Kim, Y. (2019). Spatially Filtered Multilevel Analysis on Spatial Determinants for Malaria Occurrence in Korea. International Journal of Environmental Research and Public Health, 16(7), 1250. https://doi.org/10.3390/ijerph16071250




