Factors Associated with Persistent Lower Respiratory Symptoms or Asthma among Residents Exposed to a Sulphur Stockpile Fire Incident
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.2.1. Definition of Cases
- Macassar resident at the time of the fire incident
- Eighteen years or older at the time of the fire incident
- Underwent a medical evaluation at the Macassar disaster project clinic
- Free of persistent LRS, asthma and other chronic respiratory illness such as pneumonia or chronic obstructive pulmonary disease at the time of the disaster
- Absence of pulmonary TB (PTB) at least one year prior to the disaster
- Reported persistent respiratory symptoms/asthma at year 1 (Yr1) and year 6 (Yr6) after the incident which in the opinion of the medical panel (MRP) was probably related to inhaling SO2 vapours at the time of the fire incident
2.2.2. Definition of Controls
2.2.3. Definition of Persistent Lower Respiratory Symptoms or Asthma Attributable to the Fire
2.3. Data Collection
2.3.1. Health Outcome Assessment
2.3.2. Environmental Exposure Assessment
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, R.; Huang, W.; Wong, C.M.; Wang, Z.; Thach, T.Q.; Chen, B.; Kan, H. Short-term exposure to sulfur dioxide and daily mortality in 17 Chinese cities: The China air pollution and health effects study (CAPES). Environ. Res. 2012, 118, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.A.; Demers, P.A.; Karr, C.J.; Koehoorn, M.; Lencar, C.; Tamburic, L.; Brauer, M. Effect of early life exposure to air-pollution on development of childhood asthma. Environ. Health Perspect. 2010, 118, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.M.; Gokhale, J.; Shofer, S.; Kuschner, W.G. Outdoor air pollution: Nitrogen dioxide, sulfur dioxide, and carbon monoxide health effects. Am. J. Med. Sci. 2007, 333, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zhang, Z.; Wang, Z.; Ferm, M.; Liang, Y.; Norbäck, D. Asthmatic symptoms among pupils in relation to winter indoor and outdoor air pollution in schools in Taiyuan. China Environ. Health Perspect. 2008, 116, 90–97. [Google Scholar] [CrossRef]
- Kim, K.H.; Jahan, S.A.; Kabir, E. A review on human health perspective of air pollution with respect to allergies and asthma. Environ. Int. 2013, 59, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Burra, T.A.; Moineddin, R.; Agha, M.M.; Glazier, R.H. Social disadvantage, air pollution, and asthma physician visits in Toronto, Canada. Environ. Res. 2009, 109, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Samoli, E.; Nastos, P.T.; Paliatsos, A.G.; Katsouyanni, K.; Priftis, K.N. Acute effects of air pollution on pediatric asthma exacerbation: Evidence of association and effect modification. Environ. Res. 2011, 111, 418–424. [Google Scholar] [CrossRef]
- Zhang, J.J.; Smith, K.R. Household air pollution from coal and biomass fuels in China: Measurements, health impacts, and interventions. Environ. Health Perspect. 2007, 115, 848–855. [Google Scholar] [CrossRef]
- Hansell, A.L.; Horwell, C.J.; Oppenheimer, C. The health hazards of volcanoes and geothermal areas. Occup. Environ. Med. 2006, 63, 149–156. [Google Scholar] [CrossRef]
- Hansell, A.; Oppenheimer, C. Health hazards from volcanic gases: A systematic literature review. Arch. Environ. Health. 2004, 59, 628–639. [Google Scholar] [CrossRef]
- Amaral, A.F.S.; Rodrigues, A.S. Chronic exposure to volcanic environments and chronic bronchitis incidence in the Azores, Portugal. Environ. Res. 2007, 103, 419–423. [Google Scholar] [CrossRef]
- Demeter, S.L.; Cordasco, E.M.; Guidotti, T.L. Permanent respiratory impairment and upper airway symptoms despite clinical improvement in patients with reactive airways dysfunction syndrome. Sci. Total Environ. 2001, 270, 49–55. [Google Scholar] [CrossRef]
- Piirilä, P.L.; Nordman, H.; Korhonen, O.S.; Winblad, I. A thirteen –year follow-up of respiratory effects of acute exposure to sulphur dioxide. Scand. J. Work Environ. Health 1996, 22, 191–196. [Google Scholar] [CrossRef]
- Andersson, E.; Knutsson, A.; Hagberg, S.; Nilsson, T.; Karlsson, B.; Alfredsson, L.; Torén, K. Incidence of asthma among workers exposed to sulphur dioxide and other irritant gases. Eur. Respir. J. 2006, 27, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Kochi, T.; Iwasawa, S.; Nakano, M.; Tsuboi, T.; Tanaka, S.; Kitamura, H.; Wilson, D.J.; Takebayashi, T.; Omae, K. Influence of sulfur dioxide on the respiratory system of Miyakejima adult residents 6 years after returning to the island. J. Occup. Health 2017, 59, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Batterman, S.A.; Cairncross, E.; Huang, Y.L. Estimate and Evaluation of Exposure from a large Sulphur Fire in South Africa. Environ. Res. Sect. 1999, A8, 316–333. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, R.; Viegi, G.; Brusasco, V.; Crapo, R.O.; Burgos, F.; Casaburi, R.; Coates, A.; van der Grinten, C.P.; Gustafsson, P.; Hankinson, J.; et al. Interpretative strategies for lung function tests. Eur. Respir. J. 2005, 26, 948–968. [Google Scholar] [CrossRef]
- Quanjer, P.H.; Tammeling, G.J.; Cotes, J.E.; Pedersen, O.F.; Peslin, R.; Yernault, J.C. Lung volumes and forced ventilatory flows. Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Official Statement of the European Respiratory Society. Eur. Respir. J. 1993, 6, 5–40. [Google Scholar] [CrossRef]
- Industrial Source Complex Short Term Model, ISCST3. 2002 Support Centre for Regulatory Air models. Available online: http://www.epa.gov/scram001/mcbs/iscst3z9.txt (accessed on 1 February 2019).
- Desai Commission. Report of the Commission of Inquiry into the sulphur Fire at Somerset West; Desai Commission: Pretoria, South Africa, 1997. [Google Scholar]
- Prezant, D.J.; Levin, S.; Kelly, K.J.; Aldrich, T.K. Upper and lower respiratory diseases after occupational and environmental disasters. Mt. Sinai J. Med. 2008, 75, 89–100. [Google Scholar] [CrossRef]
- Herbert, R.; Moline, J.; Skloot, G.; Metzger, K.; Baron, S.; Luft, B.; Markowitz, S.; Udasin, I.; Harrison, D.; Stein, D.; et al. The World Trade Center disaster and the health of workers: Five year assessment of a unique medical screening program. Environ. Health Perspect. 2006, 114, 1853–1858. [Google Scholar] [CrossRef]
- Brackbill, R.M.; Hadler, J.L.; DiGrande, L.; Ekenga, C.C.; Farfel, M.R.; Friedman, S.; Perlman, S.E.; Stellman, S.D.; Walker, D.J.; Wu, D.; et al. Asthma and posttraumatic stress symptoms 5 to 6 years following exposure to the World Trade Center terrorist attack. JAMA 2009, 302, 502–516. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Jones, R.; Reibman, J.; Morse, D.; Hwang, S.A. Lower respiratory symptoms among residents living near the World Trade Center two and four years after 9/11. Int. J. Occup. Environ. Health 2010, 16, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Mauer, M.P.; Cummings, K.R.; Hoen, R. Long-term respiratory symptoms in World Trade Center responders. Occup. Med. 2010, 60, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Weakley, J.; Webber, M.P.; Gustave, J.; Kelly, K.; Cohen, H.W.; Hall, C.B.; Prezant, D.J. Trends in respiratory diagnoses and symptoms of firefighters exposed to the World Trade Center disaster: 2005–2010. Prev. Med. 2011, 53, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Wisnivesky, J.P.; Teitelbaum, S.L.; Todd, A.C.; Boffetta, P.; Crane, M.; Crowley, L.; de la Hoz, R.E.; Dellenbaugh, C.; Harrison, D.; Herbert, R.; et al. Persistence of multiple illnesses in World Trade Center rescue and recovery workers: A cohort study. Lancet 2011, 378, 888–897. [Google Scholar] [CrossRef]
- Friedman, S.M.; Farfel, M.R.; Maslow, C.; Jordan, H.T.; Li, J.; Alper, H.; Cone, J.E.; Stellman, S.D.; Brackbill, R.M. Risk factors for and consequences of persistent lower respiratory symptoms among World Trade Center Health Registrants 10 years after the disaster. Occup. Environ. Med. 2016, 73, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Meggs, W.J. RADS and RUDS–the toxic induction of asthma and rhinitis. J. Toxicol. Clin. Toxicol. 1994, 32, 487–501. [Google Scholar] [CrossRef]
- Moscato, G.; Vandenplas, O.; Gerth Van Wijk, R.; Malo, J.L.; Quirce, S.; Walusiak, J.; Castano, R.; De Groot, H.; Folletti, I.; Gautrin, D.; et al. Occupational rhinitis. Allergy 2008, 63, 969–980. [Google Scholar]
- Vandenplas, O.; Wiszniewska, M.; Raulf, M.; de Blay, F.; Gerth van Wijk, R.; Moscato, G.; Nemery, B.; Pala, G.; Quirce, S.; Sastre, J.; et al. EAACI position paper: Irritant-induced asthma. Allergy 2014. [Google Scholar] [CrossRef]
- Delclos, G.L.; Gimeno, D.; Arif, A.A.; Burau, K.D.; Carson, A.; Lusk, C.; Stock, T.; Symanski, E.; Whitehead, L.W.; Zock, J.P.; et al. Occupational risk factors and asthma among health care professionals. Am. J. Respir. Crit. Care Med. 2007, 175, 667–675. [Google Scholar] [CrossRef]
- Vizcaya, D.; Mirabelli, M.C.; Anto, J.M.; Orriols, R.; Burgos, F.; Arjona, L.; Zock, J.P. A workforce-based study of occupational exposures and asthma symptoms in cleaning workers. Occup. Environ. Med. 2011, 68, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Lillienberg, L.; Andersson, E.; Janson, C.; Dahlman-Hoglund, A.; Forsberg, B.; Holm, M.; Glslason, T.; Jögi, R.; Omenaas, E.; Schlünssen, V.; et al. Occupational exposure and new-onset asthma in a population-based study in Northern Europe (RHINE). Ann. Occup. Hyg. 2013, 57, 482–492. [Google Scholar] [PubMed]
- Brooks, S.M.; Weiss, M.A.; Bernstein, I.L. Reactive airway dysfunction syndrome (RADS): Persistent asthma syndrome after high-level irritant exposure. Chest 1985, 88, 376–384. [Google Scholar] [CrossRef]
- Bhérer, L.; Cushman, R.; Courteau, J.P.; Quévillon, M.; Côté, G.; Bourbeau, J.; L’Archevêque, J.; Cartier, A.; Malo, J.L. Survey of construction workers repeatedly exposed to chlorine over a three to six-month period in a pulpmill: II. Follow up of affected workers by questionnaire, spirometry, and assessment of bronchial responsiveness 18 to 24 months after exposure ended. Occup. Environ. Med. 1994, 51, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Malo, J.L.; L’Archeveque, J.; Castellanos, L.; Lavoie, K.; Ghezzo, H.; Maghni, K. Long-term outcomes of acute irritant-induced asthma. Am. J. Respir. Crit. Care Med. 2009, 179, 923–928. [Google Scholar] [CrossRef]
- Ehrlich, R.I.; White, N.; Norman, R.; Laubscher, R.; Steyn, K.; Lombard, C.; Bradshaw, D. Wheeze, asthma diagnosis and medication use: A national adult survey in a developing country. Thorax 2005, 60, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Jithoo, A. Respiratory Symptoms and Chronic Obstructive Pulmonary Disease. Prevalence and Risk Factor in a Predominantly Low-Income Urban Area of Cape Town, South Africa. Ph.D. Thesis, University of Cape Town, Cape Town, South Africa, 2006; pp. 1–284. [Google Scholar]
- Hnizdo, E.; Singh, T.; Churchyard, G.J. Chronic pulmonary function impairment caused by initial and recurrent pulmonary tuberculosis following treatment. Thorax 2000, 55, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, R.I.; Adams, S.; Baatjies, R.; Jeebhay, M.F. Chronic airflow obstruction and respiratory symptoms following tuberculosis: A review of South African studies. Int. J. Tuberc. Lung Dis. 2011, 15, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Amaral, A.F.; Coton, S.; Kato, B.; Tan, W.C.; Studnicka, M.; Janson, C.; Gislason, T.; Mannino, D.; Bateman, E.D.; Buist, S.; et al. Tuberculosis associates with both airflow obstruction and low lung function: BOLD results. Eur. Respir. J. 2015, 46, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.B.; Liu, X.; Zeig-Owens, R.; Webber, M.P.; Aldrich, T.K.; Weakley, J.; Schwartz, T.; Cohen, H.W.; Glaser, M.S.; Olivieri, B.L.; et al. The Duration of an Exposure Response Gradient between Incident Obstructive Airways Disease and Work at the World Trade Center Site: 2001–2011. PLoS Curr. 2015. [Google Scholar] [CrossRef]
- de la Hoz, R.E.; Shohet, M.R.; Chasan, R.; Bienenfeld, L.A.; Afilaka, A.A.; Levin, S.M.; Herbert, R. Occupational toxicant inhalation injury: The World Trade Center (WTC) experience. Int. Arch. Occup. Environ. Health 2008, 81, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Koksal, N.; Hasanoglu, H.C.; Gokirmak, M.; Yildirim, Z.; Gultek, A. An Occupation that induces an asthma-like syndrome in agricultural environments. Am. Ind. Med. 2003, 43, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitch, S.; Greyson, N.D.; Weiser, W.; Hoffstein, V. Clinical and laboratory features of acute sulphur dioxide: Two-year follow-up. Am. Rev. Respir. Dis. 1989, 139, 556–558. [Google Scholar] [CrossRef]
- Ishigami, A.; Kikuchi, Y.; Iwasawa, S.; Nishiwaki, Y.; Takebayashi, T.; Tanaka, S.; Omae, K. Volcanic sulfur dioxide and acute respiratory symptoms in Miyakejima Island. Occup. Environ. Med. 2008, 65, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Batterman, A.S.; White, N. Exposure and Health Effects from a Large Sulphur fire in South Africa. In Proceedings of the Air and Waste Health Management Association’s Annual Meeting and Exhibition, St. Louis, MO, USA, 20–24 June 1999; pp. 99–785. [Google Scholar]
| Demographic Characteristics | Case | Controls |
|---|---|---|
| (n = 76) | (n = 180) | |
| Age (yrs) | 43 ± 12 | 41 ± 13 |
| Gender | ||
| Males | 23 (30%) | 63 (35%) |
| Females | 53 (70%) | 117 (65%) |
| Smoking status | ||
| Non-smoker | 33 (43.4%) | 73 (40.5%) |
| Ex-smoker | 29 (38.2%) | 55 (30.5%) |
| Current smoker | 14 (18.4%) | 52 (29%) |
| Previous tuberculosis * | 13 (17%) | 10 (6%) |
| Obstructive Lung Disease Based on Spirometry | Prevalence (%) |
|---|---|
| (n = 76) | |
| FEV1 increase post-bronchodilator (≥12% and ≥200 ml) | 28 (37%) |
| FEV1/FVC ratio ≤70% * | 18 (24%) |
| Impaired lung function: FEV1 <80% predicted * | 48 (63%) |
| Mild: 50–79% predicted | 42 (55%) |
| Moderate: 30–49% predicted | 4 (5%) |
| Severe: <30% predicted | 2 (3%) |
| Cases (n = 76) | Controls (n = 180) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Exposure Metric | Mean ± S.D. | Range | Median | IQR | Mean ± S.D. | Range | Median | IQR | p-Value |
| Total hours of exposure | 8.43 ± 6.33 | 1–21 | 6 | 6.5 | 8.54 ± 6.11 | 1–21 | 6 | 7 | 0.896 |
| Peak exposure (ppm) | 46.46 ± 86.67 | 0–444.06 | 6.94 | 64.51 | 40.63 ± 95.91 | 0–540.64 | 4.01 | 41.17 | 0.649 |
| Cumulative exposure (ppm×hrs) | 104.22 ± 168.46 | 0–821.97 | 13.37 | 163.78 | 101.40 ± 187.70 | 0–884.23 | 7.74 | 106.95 | 0.910 |
| Outside dwelling at the time of the fire | 26 (34%) | 63 (35%) | 0.904 | ||||||
| Host Factors | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| Age | 1.01 | 0.99–1.03 | 0.458 |
| Gender | |||
| - Female | 1.24 | 0.70–2.21 | 0.464 |
| Smoking status | |||
| - Non-smokers | 1.00 | ||
| - Ex-smokers | 1.20 | 0.65–2.21 | 0.554 |
| - Current smokers | 0.61 | 0.30–1.26 | 0.186 |
| Previous Pulmonary TB * | 3.49 | 1.46–8.35 | 0.005 |
| Self-reported acute symptoms: | |||
| Ocular-nasal symptoms | |||
| - burning eyes | 0.77 | 0.45–1.33 | 0.345 |
| - burning/sore nose | 0.91 | 0.50–1.66 | 0.770 |
| - burning/sore throat | 0.93 | 0.49–1.79 | 0.836 |
| Lower respiratory symptoms | |||
| - burning/sore chest | 1.47 | 0.79–2.74 | 0.218 |
| - cough | 1.17 | 0.65–2.09 | 0.602 |
| - shortness of breath | 1.97 | 0.95–4.09 | 0.070 |
| - tight chest | 9.93 | 5.15–19.11 | <0.001 |
| Constitutional symptoms | |||
| - headache | 0.27 | 0.08–0.92 | 0.036 |
| Gastro-intestinal symptoms | |||
| - nausea/vomiting | 0.60 | 0.25–1.45 | 0.257 |
| - diarrhoea | 0.79 | 0.08–7.69 | 0.837 |
| Using Logistic Regression Models | O.R. | 95% CI | p-Value |
|---|---|---|---|
| Unadjusted models | |||
| Location at time of incident | |||
| Outside v/s inside | 0.97 | 0.55–1.70 | 0.904 |
| Total hours of exposure (hrs) a | |||
| 5–6 hrs | 1.48 | 0.72–3.05 | 0.291 |
| 7–11 hrs | 0.95 | 0.44–2.04 | 0.900 |
| >11 | 1.06 | 0.50–2.26 | 0.880 |
| Cumulative exposure (ppm×hours) b | |||
| 6–50 ppm | 0.79 | 0.39–1.61 | 0.509 |
| >50 ppm | 1.40 | 0.76–2.57 | 0.282 |
| Peak exposure (ppm) c | |||
| 2–20 | 1.02 | 0.50–2.01 | 0.964 |
| >20 | 1.42 | 0.77–2.62 | 0.267 |
| Hourly exposures | |||
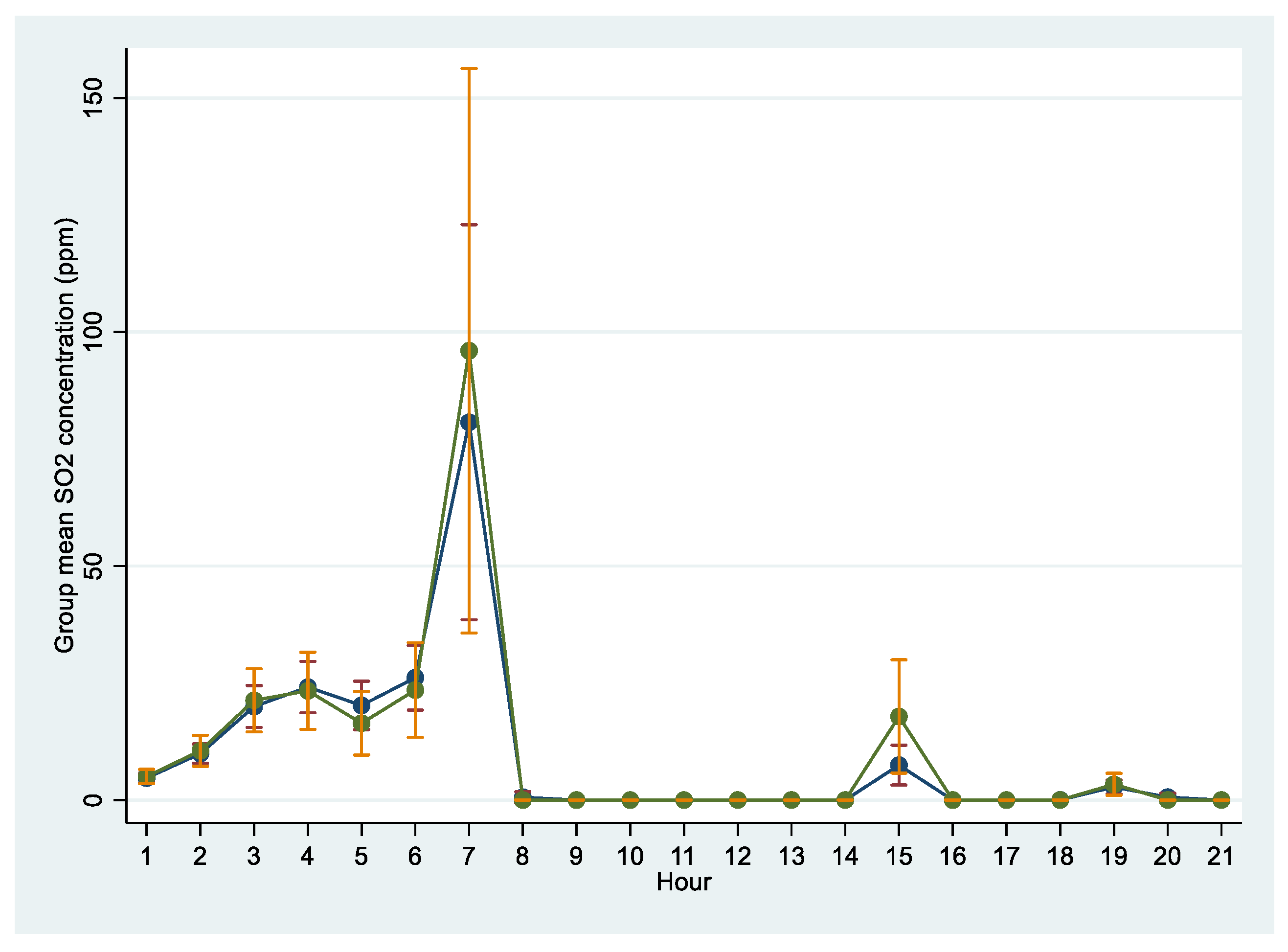
| Exposure at hour 7 (hr7) | 1.00 | 0.99–1.00 | 0.695 |
| Exposure at hour 15 (hr15) | 1.03 | 0.99–1.06 | 0.060 |
| Adjusted models * | |||
| Hourly exposures | |||
| Exposure at hour 7 (hr7) | 1.00 | 0.99–1.00 | 0.338 |
| Exposure at hour 15 (hr15) | 1.04 | 1.01–1.07 | 0.021 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baatjies, R.; Adams, S.; Cairncross, E.; Omar, F.; Jeebhay, M.F. Factors Associated with Persistent Lower Respiratory Symptoms or Asthma among Residents Exposed to a Sulphur Stockpile Fire Incident. Int. J. Environ. Res. Public Health 2019, 16, 438. https://doi.org/10.3390/ijerph16030438
Baatjies R, Adams S, Cairncross E, Omar F, Jeebhay MF. Factors Associated with Persistent Lower Respiratory Symptoms or Asthma among Residents Exposed to a Sulphur Stockpile Fire Incident. International Journal of Environmental Research and Public Health. 2019; 16(3):438. https://doi.org/10.3390/ijerph16030438
Chicago/Turabian StyleBaatjies, Roslynn, Shahieda Adams, Eugene Cairncross, Faieza Omar, and Mohamed F. Jeebhay. 2019. "Factors Associated with Persistent Lower Respiratory Symptoms or Asthma among Residents Exposed to a Sulphur Stockpile Fire Incident" International Journal of Environmental Research and Public Health 16, no. 3: 438. https://doi.org/10.3390/ijerph16030438
APA StyleBaatjies, R., Adams, S., Cairncross, E., Omar, F., & Jeebhay, M. F. (2019). Factors Associated with Persistent Lower Respiratory Symptoms or Asthma among Residents Exposed to a Sulphur Stockpile Fire Incident. International Journal of Environmental Research and Public Health, 16(3), 438. https://doi.org/10.3390/ijerph16030438





