Standard Expected Years of Life Lost Due to Malignant Neoplasms in Poland, 2000–2014
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vallin, J.; Meslé, F. Convergences and divergences in mortality. A new approach to health transition. Demogr. Res. 2004, 10, 11–44. [Google Scholar] [CrossRef]
- Zatoński, W.A.; Bhala, N. Changing trends of diseases in Eastern Europe: Closing the gap. Public Health 2012, 126, 248–522. [Google Scholar] [CrossRef] [PubMed]
- Powles, J.W.; Zatoński, W.; Vander Hoorn, S.; Ezzati, M. The contribution of leading diseases and risk factors to excess losses of healthy life in Eastern Europe: Burden of disease study. BMC Public Health 2005, 5, 116. [Google Scholar] [CrossRef] [PubMed]
- Maniecka-Bryła, I.; Pikala, M.; Bryła, M. Life years lost due to cardiovascular diseases. Kardiol. Pol. 2013, 71, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Pikala, M.; Maniecka-Bryła, I. Years of life lost due to malignant neoplasms characterized by the highest mortality rate. Arch. Med. Sci. 2014, 10, 999–1006. [Google Scholar] [CrossRef]
- White, A.; de Sousa, B.; de Visser, R.; Houston, R.; Madsen, S.A.; Makara, P.; Richardson, N.; Zatoński, W. The State of Men’s in Europe. Report, Directorate-General for Heath & Consumers. Available online: https://ec.europa.eu/health/sites/health/files/state/docs/men_health_extended_en.pdf (accessed on 16 June 2019).
- Jemal, A.; Siegel, R.; Ward, E.; Murray, T.; Xu, J.; Thun, M.J. Cancer Statistics 2007. CA Cancer J. Clin. 2009, 57, 43–66. [Google Scholar] [CrossRef]
- Stahl, T.; Wismar, M.; Ollila, E.; Lahtinen, E.; Leppo, K. Health in All Policies: Prospects and Potentials; Ministry of Social Affairs and Health: Helsinki, Finland, 2006.
- Marmot, M. Review of Social Determinants and the Health Divide in the WHO European Region; Final Report; WHO Regional Office for Europe; UCL Institute of Health Equity: Copenhagen, Denmark, 2013. [Google Scholar]
- Doll, R.; Peto, R. The causes of cancer: Quantitative estimates of avoidable risks of cancer in the United States today. J. Natl. Cancer Inst. 1981, 66, 1191–1308. [Google Scholar] [CrossRef]
- Boyle, P.; Levin, B. World Cancer Report 2008; International Agency for Research on Cancer: Lyon, France, 2008. [Google Scholar]
- Hashim, D.; Boffetta, P.; La Vecchia, C.; Rota, M.; Bertuccio, P.; Malvezzi, M.; Negri, E. The global decrease in cancer mortality: Trends and disparities. Ann. Oncol. 2016, 27, 926–933. [Google Scholar] [CrossRef]
- Wojtyś, P.; Godlewski, D.; Antczak, A. Predictions of cancer incidence in Poland in 2019. Centr. Eur. J. Med. 2013, 8, 185. [Google Scholar] [CrossRef]
- Jemal, A.; Center, M.M.; DeSantis, C.; Ward, E.M. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1893–1907. [Google Scholar] [CrossRef]
- Causes of Death Statistics. Eurostat Statistics Explained. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Causes_of_death_statistics (accessed on 16 March 2019).
- La Vecchia, C.; Rota, M.; Malvezzi, M.; Negri, E. Potential for improvement in cancer management: Reducing mortality in the European Union. Oncologist 2015, 20, 495–498. [Google Scholar] [CrossRef] [PubMed]
- Popat, K.; McQueen, K.; Feeley, T.W. The global burden of cancer. Best Pract. Res. Clin. Anaesthesiol. 2013, 27, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Maniecka-Bryła, I.; Bryła, M.; Bryła, P.; Pikala, M. The burden of premature mortality in Poland analysed with the use of standard expected years of life lost. BMC Public Health 2015, 5, 101. [Google Scholar] [CrossRef] [PubMed]
- Plass, D.; Chau, P.Y.; Thach, T.Q.; Jahn, H.J.; Lai, P.C.; Wong, C.M.; Kraemer, A. Quantifying the burden of disease due to premature mortality in Hong Kong using standard expected years of life lost. BMC Public Health 2013, 13, 863. [Google Scholar] [CrossRef] [PubMed]
- Pikala, M.; Maniecka-Bryła, I. Fifteen-year mortality trends in Poland analysed with the use of standard expected years of life lost, 2000–2014. Sci. Rep. 2017, 7, 8730. [Google Scholar] [CrossRef]
- Castillo-Rodríguez, L.; Díaz-Jiménez, D.; Castañeda-Orjuela, C.; De la Hoz-Restrepo, F. Years of Life Lost (YLL) in Colombia 1998–2011: Overall and avoidable causes of death analysis. PLoS ONE 2015, 10, e0125456. [Google Scholar] [CrossRef]
- Murray, C.J.; Lopez, A.D. Global Burden of Diseases; University Medical Publishing House Vesalius: Warsaw, Poland, 2000; ISBN 8385688633. [Google Scholar]
- Revision of the European Standard Population. Available online: https://ec.europa.eu/eurostat/documents/3859598/5926869/KS-RA-13-028-EN.PDF/e713fa79-1add-44e8-b23d-5e8fa09b3f8f (accessed on 16 June 2018).
- Lopez, A.D.; Mathers, C.D.; Ezzati, M.; Jamison, D.T.; Murray, C.J. Global Burden of Diseases and Risk Factors; The World Bank and Oxford University Press: Washington, WA, USA, 2006; ISBN 0821362633. [Google Scholar]
- WHO Methods and Data Sources for Global Burden of Disease Estimates 2000–2011. Available online: https://www.who.int/healthinfo/statistics/GlobalDALYmethods_2000_2011.pdf (accessed on 8 June 2018).
- Marshall, R.J. Standard Expected Years of Life Lost as a measure of disease burden: An investigation of its presentation, meaning and interpretation. In Handbook of Disease Burdens and Quality of Life Measures; Preedy, V.R., Watson, R.R., Eds.; Springer: New York, NY, USA, 2010; pp. 401–413. ISBN 9780387786643. [Google Scholar]
- Kim, H.; Fay, M.P.; Feuer, E.J.; Midthune, D.N. Permutation tests for joinpoint regression with applications to cancer rates. Stat. Med. 2000, 19, 335–351. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Murphy, S.L.; Kochanek, K.D.; Xu, J.; Heron, M. Deaths: Final Data for 2012. Natl. Vital Stat. Rep. 2015, 63, 100–117. [Google Scholar]
- The World Health Report 2004—Changing History. Available online: https://www.who.int/whr/2004/en/report04_en.pdf (accessed on 18 July 2018).
- 10 Facts on Noncommunicable Diseases. Available online: https://www.who.int/features/factfiles/noncommunicable_diseases/en/ (accessed on 5 June 2018).
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics, 2002. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Bosetti, C.; Bertuccio, P.; Malvezzi, M.; Levi, F.; Chatenoud, L.; Negri, E.; La Vecchia, C. Cancer mortality in Europe, 2005-2009, and an overview of trends since 1980. Ann. Oncol. 2013, 24, 2657–2671. [Google Scholar] [CrossRef] [PubMed]
- Chatenoud, L.; Bertuccio, P.; Bosetti, C.; Malvezzi, M.; Levi, F.; Negri, E.; La Vecchia, C. Trends in mortality from major cancers in the Americas: 1980–2010. Ann. Oncol. 2014, 25, 1843–1853. [Google Scholar] [CrossRef]
- Ferro, A.; Peleteiro, B.; Malvezzi, M.; Bosetti, C.; Bertuccio, P.; Levi, F.; Negri, E.; La Vecchia, C.; Lunet, N. Worldwide trends in gastric cancer mortality (1980–2011), with predictions to 2015, and incidence by subtype. Eur. J. Cancer 2014, 50, 1330–1344. [Google Scholar] [CrossRef]
- Pikala, M.; Burzyńska, M.; Pikala, R.; Bryła, M.; Maniecka-Bryła, I. Educational inequalities in premature mortality in Poland, 2002–2011: A population-based cross-sectional study. BMJ Open 2016, 6, e011501. [Google Scholar] [CrossRef]
- Malvezzi, M.; Bertuccio, P.; Rosso, T.; Rota, M.; Levi, F.; La Vecchia, C.; Negri, E. European cancer mortality predictions for the year 2015: Does lung cancer have the highest death rate in EU women? Ann. Oncol. 2015, 26, 779–786. [Google Scholar] [CrossRef]
- Zatoński, W.; Przewoźniak, K.; Sulkowska, U.; West, R.; Wojtyła, A. Tobacco smoking in countries of the European Union. Ann. Agric. Environ. Med. 2012, 19, 181–192. [Google Scholar]
- Rachtan, J. Smoking, passive smoking and lung cancer cell types among women in Poland. Lung Cancer 2002, 35, 129–136. [Google Scholar] [CrossRef]
- Zatoński, W.; Przewoźniak, K. Tobacco smoking in Poland in the years 1923-1987. Pol. Popul. Rev. 1993, 3, 103–124. [Google Scholar]
- Zatoński, W.; Zatoński, M.; Przewoźniak, K. Health improvement in Poland is contingent on continued extensive tobacco control measures. Ann. Agric. Environ. Med. 2013, 20, 405–411. [Google Scholar] [PubMed]
- Carioli, G.; Malvezzi, M.; Rodriguez, T.; Bertuccio, P.; Negri, E.; La Vecchia, C. Trends and predictions to 2020 in breast cancer mortality in Europe. Breast 2017, 36, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Wojciechowska, U.; Didkowska, J. Incidence and Mortality from Malignant Tumours in Poland. Polish National Cancer Registry. Available online: http://onkologia.org.pl/raporty/ (accessed on 20 June 2019).
- Local Data Bank. Available online: http://www.stat.gov.pl (accessed on 29 January 2019).
- Freimanis, R.I.; Yacobozzi, M. Breast cancer screening. N. Carol. Med. J. 2014, 75, 117–120. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Berry, D.A.; Cronin, K.A.; Plevritis, S.K.; Fryback, D.G.; Clarke, L.; Zelen, M.; Mandelblatt, J.S.; Yakovlev, A.Y.; Habbema, J.D.; Feuer, E.J. Effect of screening and adjuvant therapy on mortality from breast cancer. N. Engl. J. Med. 2005, 353, 1784–1792. [Google Scholar] [CrossRef]
- DeSantis, C.; Siegel, R.; Bandi, P.; Jemal, A. Breast cancer statistics, 2011. CA Cancer J. Clin. 2011, 61, 409–418. [Google Scholar] [CrossRef]
- Wong, M.C.; Goggins, W.B.; Wang, H.H.; Fung, F.D.; Leung, C.; Wong, S.Y.; Ng, C.F.; Sung, J.J. Global incidence and mortality for prostate cancer: Analysis of temporal patterns and trends in 36 countries. Eur. Urol. 2016, 70, 862–874. [Google Scholar] [CrossRef]
- Zhou, C.K.; Check, D.P.; Lortet-Tieulent, J.; Laversanne, M.; Jemal, A.; Ferlay, J.; Bray, F.; Cook, M.B.; Devesa, S.S. Prostate cancer incidence in 43 populations worldwide: An analysis of time trends overall and by age. Int. J. Cancer 2016, 138, 1388–1400. [Google Scholar] [CrossRef]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef]
- Waśkiewicz, A.; Szcześniewska, D.; Szostak-Wegierek, D.; Kwaśniewska, M.; Pająk, A.; Stępniak, U.; Kozakiewicz, K.; Tykarski, A.; Zdrojewski, T.; Zujko, M.E.; et al. Are dietary habits of the Polish population consistent with the recommendations for prevention of cardiovascular disease? WOBASZ II Project. Kardiol. Pol. 2016, 74, 969–977. [Google Scholar] [CrossRef]
- The State Agency for the Prevention of Alcohol-Related Problems (PARPA). Available online: https://www.parpa.pl/index.php/badania-i-informacje-statystyczne/statystyki (accessed on 17 June 2019).
- Program Badań Przesiewowych Raka Jelita Grubego. Available online: http://pbp.org.pl/ (accessed on 3 August 2019).
- Pimenta, J.M.; Galindo, C.; Jenkins, D.; Taylor, S.M. Estimate of the global burden of cervical adenocarcinoma and potential impact of prophylactic human papillomavirus vaccination. BMC Cancer 2013, 13, 553. [Google Scholar] [CrossRef]
- Ponti, A.; Anttila, A.; Ronco, G.; Senore, C. Cancer Screening in the European Union; International Agency for Research on Cancer: Lyon, France, 2017. [Google Scholar]
- Human papillomavirus vaccines: WHO position paper, May 2017—Recommendations. Vaccine 2017, 35, 5753–5755. [CrossRef] [PubMed]
- Parkin, D.M. The global health burden of infection-associated cancers in the year 2002. Int. J. Cancer 2006, 118, 3030–3044. [Google Scholar] [CrossRef] [PubMed]
- Peleteiro, B.; La Vecchia, C.; Lunet, N. The role of Helicobacter pylori infection in the web of gastric cancer causation. Eur. J. Cancer Prev. 2012, 21, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Suh, M. Screening for gastric cancer: The usefulness of endoscopy. Clin. Endosc. 2014, 47, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Cierniak, M. Na co Umarł Pacjent—Czyli, co Jest Wpisywane na Kartach Zgonów? Available online: http://stat.gov.pl/download/gfx/portalinformacyjny/pl/defaultaktualnosci/5746/1/1/2/l_na_co_umarl_pacjent.pdf (accessed on 19 March 2019).
| Causes of Death | 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Men | |||||||||||||||
| Malignant neoplasms overall | 47.24 | 47.96 | 48.43 | 47.40 | 47.76 | 46.82 | 46.52 | 46.34 | 45.48 | 44.75 | 43.29 | 42.30 | 42.32 | 41.07 | 40.51 |
| Bladder cancer | 2.28 | 2.21 | 2.31 | 2.48 | 2.42 | 2.25 | 2.26 | 2.39 | 2.33 | 2.41 | 2.33 | 2.37 | 2.30 | 2.38 | 2.25 |
| Brain cancer | 0.97 | 0.99 | 0.78 | 0.83 | 0.92 | 1.09 | 1.07 | 1.07 | 1.11 | 1.14 | 1.06 | 1.04 | 1.12 | 1.05 | 1.03 |
| Colorectal cancer | 4.68 | 4.82 | 4.93 | 4.88 | 4.92 | 4.89 | 5.01 | 5.20 | 5.26 | 5.20 | 5.27 | 5.16 | 5.23 | 5.14 | 5.20 |
| Leukaemia | 1.13 | 1.23 | 0.99 | 1.03 | 1.06 | 1.31 | 1.31 | 1.32 | 1.29 | 1.24 | 1.21 | 1.31 | 1.24 | 1.26 | 1.23 |
| Liver cancer | 1.01 | 1.05 | 0.98 | 0.99 | 0.96 | 0.85 | 0.94 | 0.86 | 0.86 | 0.87 | 0.88 | 0.83 | 0.91 | 0.86 | 0.82 |
| Lung cancer | 15.18 | 15.61 | 12.21 | 11.91 | 12.11 | 14.33 | 14.14 | 13.90 | 13.94 | 13.28 | 12.94 | 12.43 | 12.32 | 11.94 | 11.72 |
| Pancreatic cancer | 1.78 | 1.76 | 1.80 | 1.82 | 1.79 | 1.79 | 1.80 | 1.84 | 1.87 | 1.90 | 1.82 | 1.73 | 1.82 | 1.77 | 1.83 |
| Prostate cancer | 4.16 | 4.46 | 4.57 | 4.32 | 4.46 | 4.36 | 4.38 | 4.50 | 4.31 | 4.34 | 4.08 | 4.17 | 4.20 | 4.15 | 4.11 |
| Stomach cancer | 3.95 | 3.77 | 3.78 | 3.60 | 3.53 | 3.34 | 3.23 | 3.23 | 3.11 | 2.98 | 2.95 | 2.86 | 2.78 | 2.66 | 2.63 |
| Woman | |||||||||||||||
| Malignant neoplasms overall | 24.11 | 24.12 | 23.94 | 24.06 | 23.72 | 23.82 | 23.68 | 23.65 | 23.36 | 22.98 | 22.49 | 22.08 | 22.45 | 21.98 | 21.90 |
| Bladder cancer | 0.35 | 0.37 | 0.35 | 0.35 | 0.35 | 0.38 | 0.36 | 0.37 | 0.39 | 0.37 | 0.36 | 0.38 | 0.36 | 0.39 | 0.39 |
| Brain cancer | 0.71 | 0.65 | 0.59 | 0.62 | 0.66 | 0.79 | 0.85 | 0.83 | 0.80 | 0.86 | 0.81 | 0.75 | 0.80 | 0.76 | 0.69 |
| Breast cancer | 3.05 | 3.05 | 3.01 | 3.03 | 2.94 | 3.06 | 3.04 | 3.01 | 3.03 | 2.91 | 2.85 | 2.94 | 2.96 | 3.04 | 3.05 |
| Cervix uteri cancer | 1.20 | 1.08 | 1.09 | 1.06 | 1.03 | 1.00 | 1.01 | 1.05 | 0.95 | 0.94 | 0.92 | 0.87 | 0.87 | 0.86 | 0.82 |
| Colorectal cancer | 2.87 | 2.92 | 2.87 | 2.87 | 2.80 | 2.81 | 2.72 | 2.68 | 2.76 | 2.72 | 2.73 | 2.63 | 2.77 | 2.67 | 2.56 |
| Leukaemia | 0.67 | 0.70 | 0.60 | 0.,62 | 0.62 | 0.73 | 0.75 | 0.73 | 0.67 | 0.69 | 0.67 | 0.69 | 0.64 | 0.66 | 0.63 |
| Liver cancer | 0.74 | 0.73 | 0.68 | 0.64 | 0.61 | 0.57 | 0.57 | 0.56 | 0.59 | 0.54 | 0.53 | 0.51 | 0.48 | 0.47 | 0.43 |
| Lung cancer | 2.56 | 2.72 | 2.40 | 2.48 | 2.44 | 2.93 | 2.99 | 3.18 | 3.17 | 3.29 | 3.36 | 3.36 | 3.40 | 3.44 | 3.73 |
| Ovarian cancer | 1.27 | 1.32 | 1.31 | 1.36 | 1.33 | 1.37 | 1.36 | 1.41 | 1.40 | 1.38 | 1.38 | 1.38 | 1,28 | 1.35 | 1.37 |
| Pancreatic cancer | 1.26 | 1.23 | 1.23 | 1.29 | 1.20 | 1.21 | 1.28 | 1.29 | 1.33 | 1.25 | 1.25 | 1.22 | 1.29 | 1.24 | 1.29 |
| Stomach cancer | 1.48 | 1.47 | 1.37 | 1.35 | 1.28 | 1.20 | 1.19 | 1.20 | 1.18 | 1.06 | 1.05 | 0.97 | 1.00 | 0.99 | 0.97 |
| Causes of Death | SEYLLp (per 10,000) | AAPC | 95% CI | SEYLLd | AAPC | 95% CI | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2000 | 2014 | 2000 | 2014 | |||||||
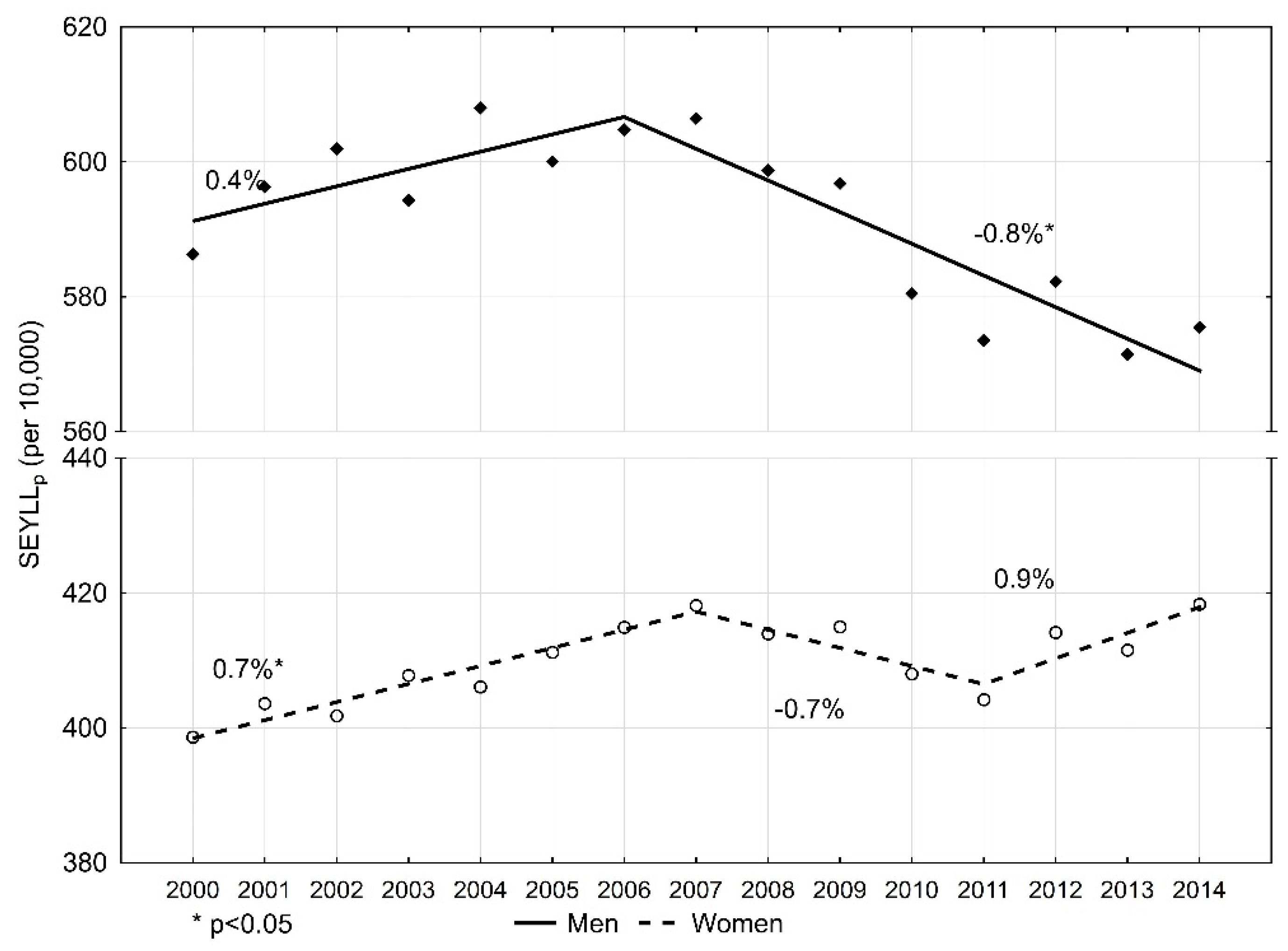
| Malignant neoplasms overall | 586.3 | 575.5 | −0.3 | −0.5 | 0.0 | 22.6 | 20.3 | −0.8 * | −0.9 | −0.7 |
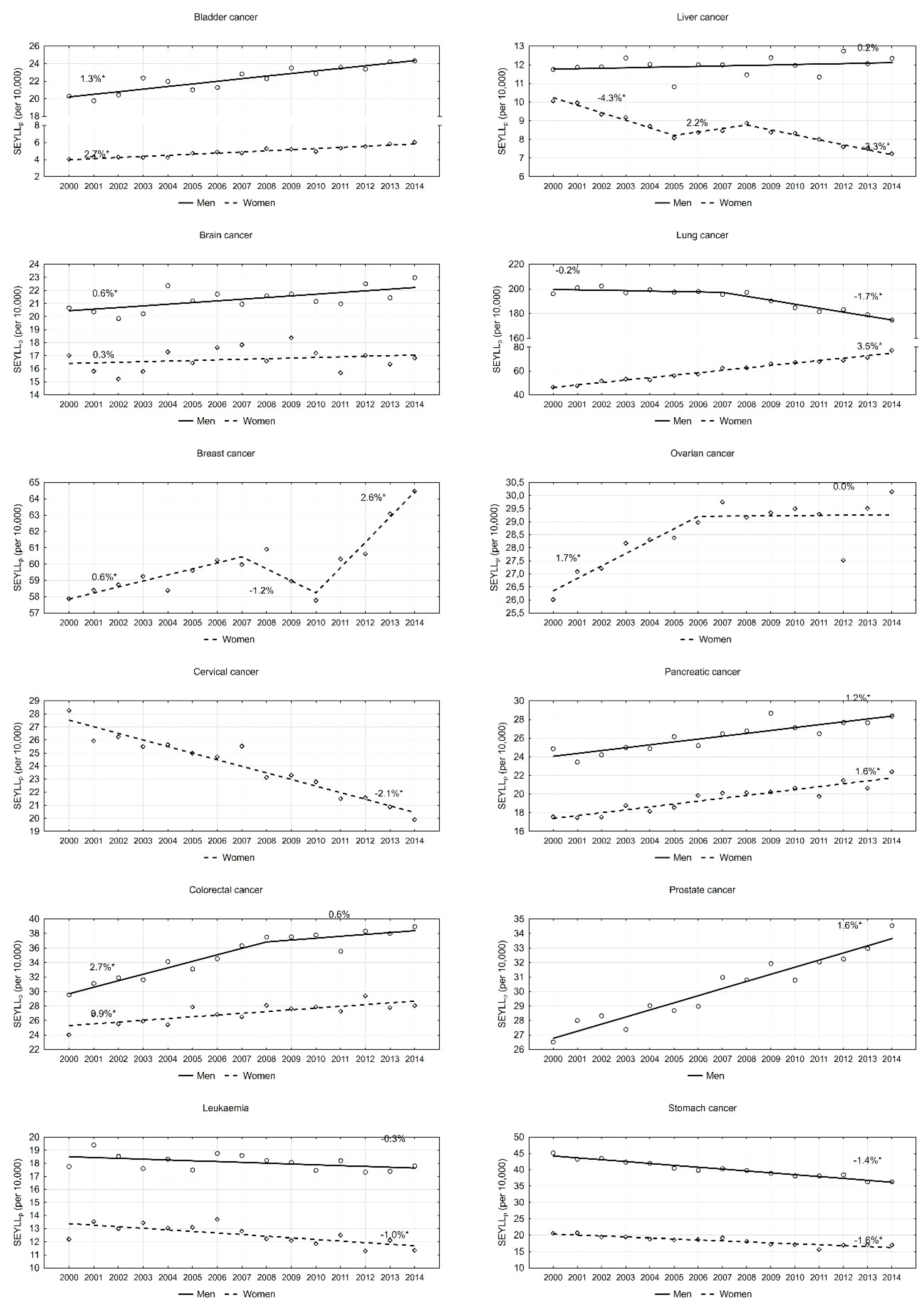
| Bladder cancer | 20.3 | 24.3 | 1.3 * | 1.0 | 1.7 | 18.8 | 17.0 | −0.7 * | −0.8 | −0.6 |
| Brain cancer | 20.7 | 23.0 | 0.6 * | 0.2 | 1.0 | 31.4 | 27.7 | −0.9 * | −1.2 | −0.6 |
| Colorectal cancer | 29.5 | 39.0 | 1.8 * | 1.1 | 2.5 | 20.2 | 18.3 | −0.7 * | −1.0 | −0.4 |
| Leukaemia | 17.8 | 17.8 | −0.3 | −0.7 | 0.0 | 26.9 | 21.4 | −1.6 * | −1.9 | −1.3 |
| Liver cancer | 11.8 | 12.3 | 0.2 | −0.3 | 0.7 | 21.9 | 21.2 | −0.2 * | −0.5 | 0 |
| Lung cancer | 196.1 | 174.7 | −0.9 * | −1.3 | −0.6 | 22.7 | 20.5 | −0.7 * | −0.8 | −0.6 |
| Pancreatic cancer | 24.9 | 28.4 | 1.2 * | 0.8 | 1.6 | 24.0 | 21.4 | −0.7 * | −0.8 | −0.5 |
| Prostate cancer | 26.5 | 34.5 | 1.6 * | 1.4 | 1.9 | 15.6 | 14.5 | −0.5 * | −0.7 | −0.3 |
| Stomach cancer | 45.2 | 36.3 | −1.4 * | −1.6 | −1.2 | 21.6 | 20.0 | −0.5 * | −0.6 | −0.4 |
| Causes of Death | SEYLLp (per 10,000) | AAPC | 95% CI | SEYLLd | AAPC | 95% CI | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2000 | 2014 | 2000 | 2014 | |||||||
| Malignant neoplasms overall | 398.6 | 418.3 | 0.3 * | 0.1 | 2.0 | 21.5 | 19.4 | −0.7 * | −0.8 | −0.6 |
| Bladder cancer | 4.0 | 6.0 | 2.7 * | 2.2 | 3.2 | 16.2 | 15.7 | −0.2 | −0.4 | 0.1 |
| Brain cancer | 17.0 | 16.8 | 0.3 | −0.4 | 1.0 | 28.7 | 24.7 | −1.3 * | −1.9 | −0.7 |
| Breast cancer | 57.9 | 64.5 | 0.8 * | 0.2 | 1.3 | 24.2 | 21.4 | −0.9 * | −1.0 | −0.8 |
| Cervix uteri cancer | 28.3 | 19.9 | −2.1 * | −2.5 | −1.8 | 28.0 | 24.3 | −1.1 * | −1.5 | −0.7 |
| Colorectal cancer | 24.0 | 28.0 | 0.9 * | 0.5 | 1.3 | 17.9 | 16.4 | −0.6 * | −0.8 | −0.5 |
| Leukaemia | 12.2 | 11.3 | −1.0 * | −1.5 | 0.4 | 22.9 | 18.5 | −1.7 * | −2.0 | −1.4 |
| Liver cancer | 10.1 | 7.2 | −2.5 * | −3.4 | −1.6 | 18.4 | 17.1 | −0.6 * | −0.7 | −0.5 |
| Lung cancer | 46.5 | 77.3 | 3.5 * | 3.2 | 3.8 | 22.8 | 20.9 | −0.5 * | −0.6 | −0.4 |
| Ovarian cancer | 26.0 | 30.1 | 0.7 * | 0.1 | 1.4 | 25.2 | 22.4 | −0.9 * | −1.0 | −1.1 |
| Pancreatic cancer | 17.6 | 22.4 | 1.6 * | 1.2 | 2.0 | 18.7 | 17.6 | −0.5 * | −0.6 | −0.3 |
| Stomach cancer | 20.5 | 17.0 | −1.6 * | −2.0 | −1.1 | 18.7 | 18.0 | −0.3 * | −0.4 | −0.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pikala, M.; Burzyńska, M.; Maniecka-Bryła, I. Standard Expected Years of Life Lost Due to Malignant Neoplasms in Poland, 2000–2014. Int. J. Environ. Res. Public Health 2019, 16, 4898. https://doi.org/10.3390/ijerph16244898
Pikala M, Burzyńska M, Maniecka-Bryła I. Standard Expected Years of Life Lost Due to Malignant Neoplasms in Poland, 2000–2014. International Journal of Environmental Research and Public Health. 2019; 16(24):4898. https://doi.org/10.3390/ijerph16244898
Chicago/Turabian StylePikala, Małgorzata, Monika Burzyńska, and Irena Maniecka-Bryła. 2019. "Standard Expected Years of Life Lost Due to Malignant Neoplasms in Poland, 2000–2014" International Journal of Environmental Research and Public Health 16, no. 24: 4898. https://doi.org/10.3390/ijerph16244898
APA StylePikala, M., Burzyńska, M., & Maniecka-Bryła, I. (2019). Standard Expected Years of Life Lost Due to Malignant Neoplasms in Poland, 2000–2014. International Journal of Environmental Research and Public Health, 16(24), 4898. https://doi.org/10.3390/ijerph16244898





