Bio-Capture of Solid Pollutants by Vegetation Canopy Cave in Shallow Water Flow
Abstract
1. Introduction
2. Experimental Set-Up and Methodology
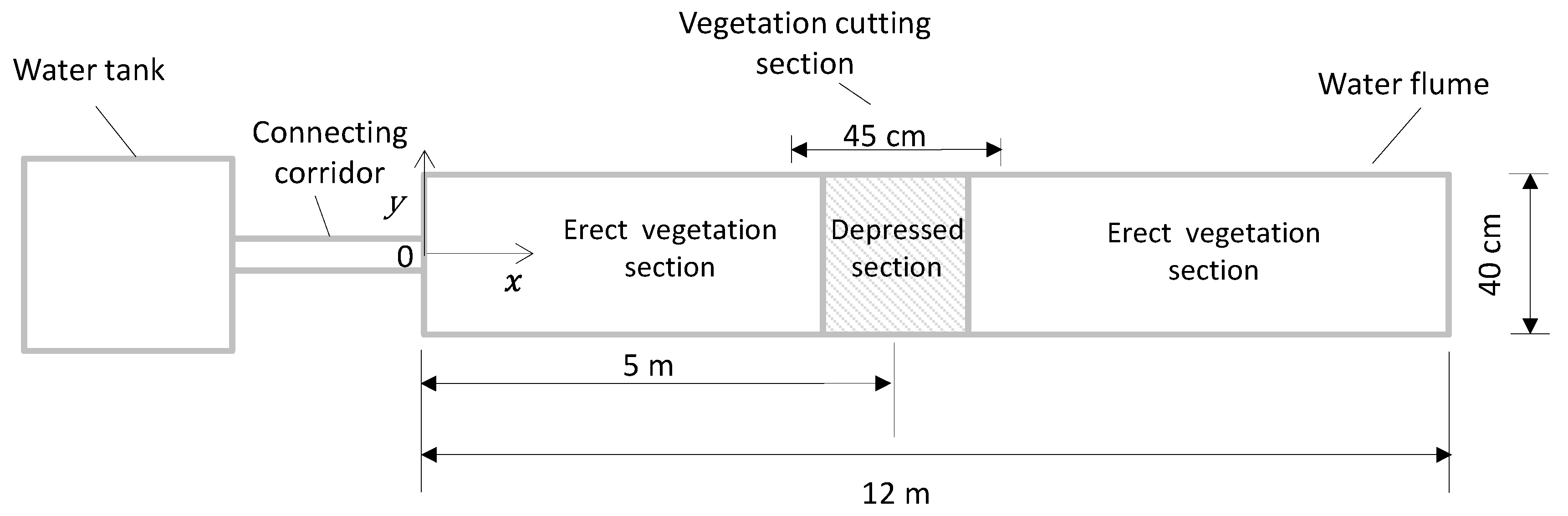
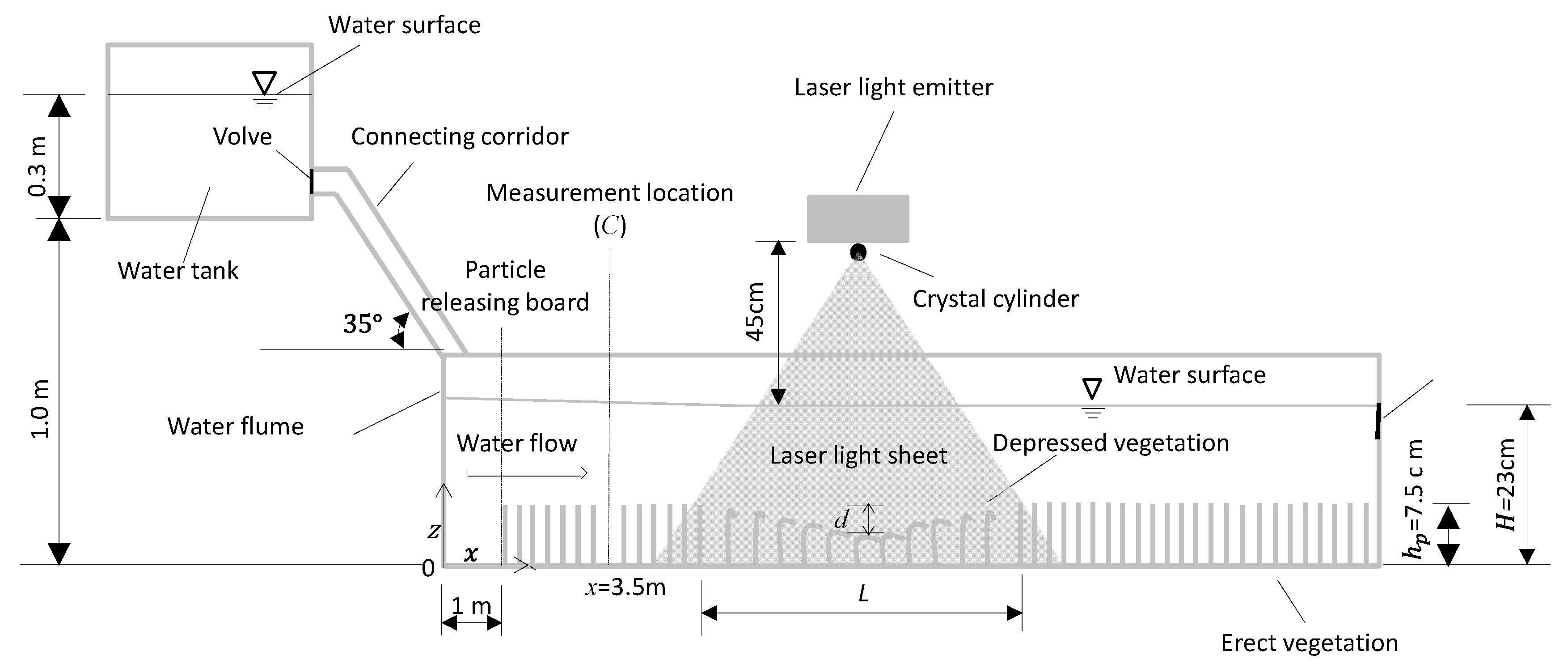
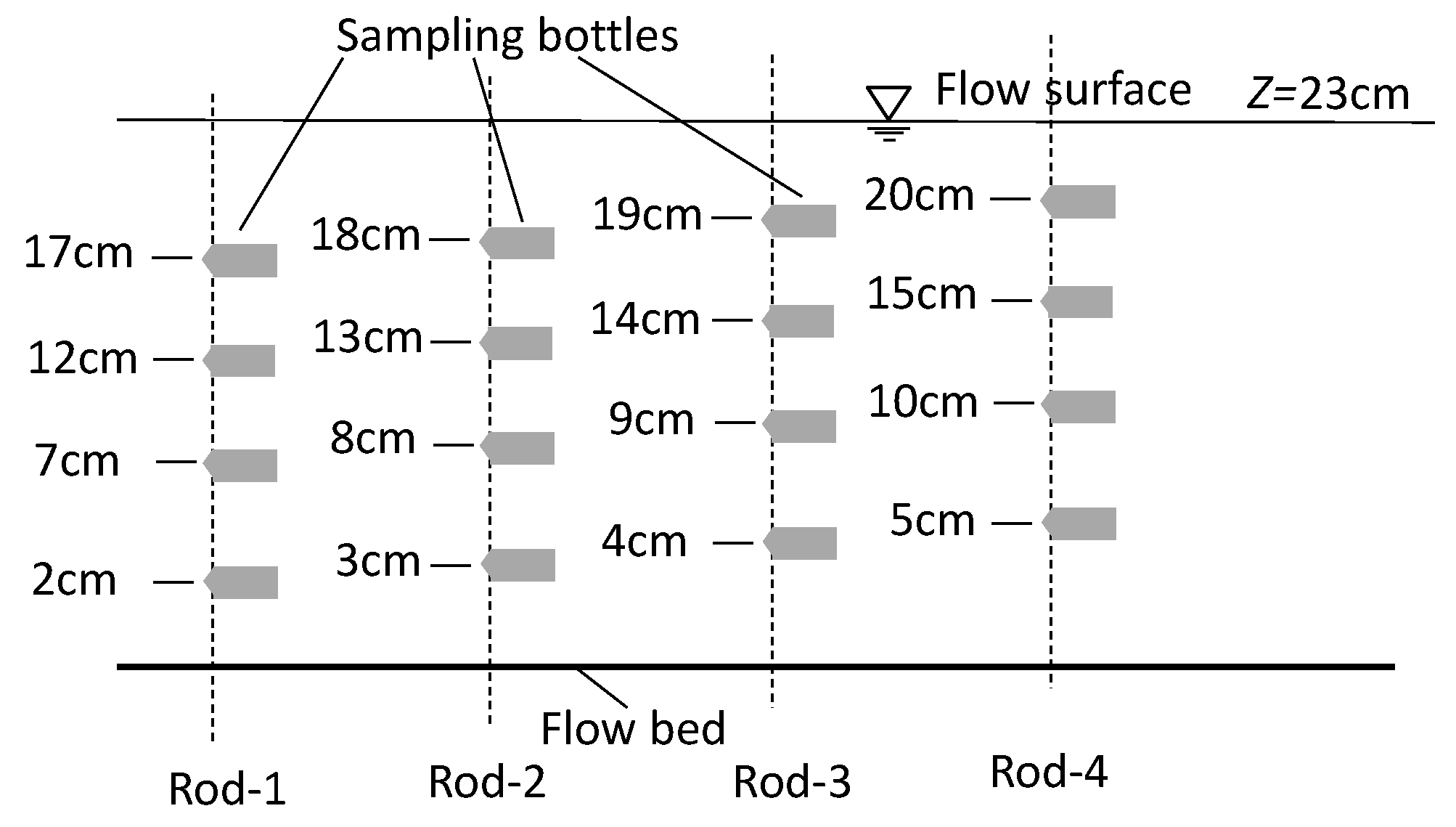
2.1. Water Flume and Coordinate System
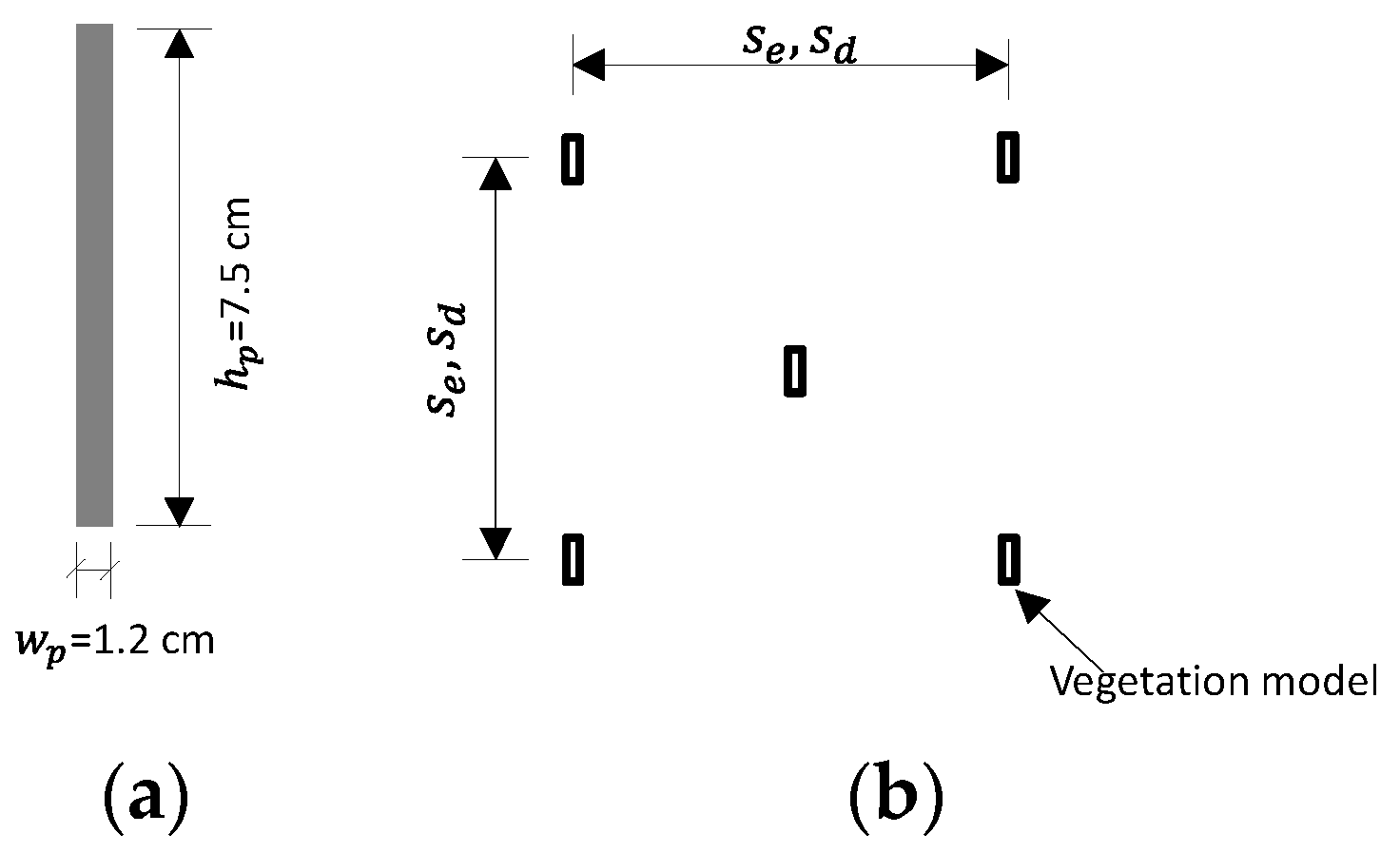
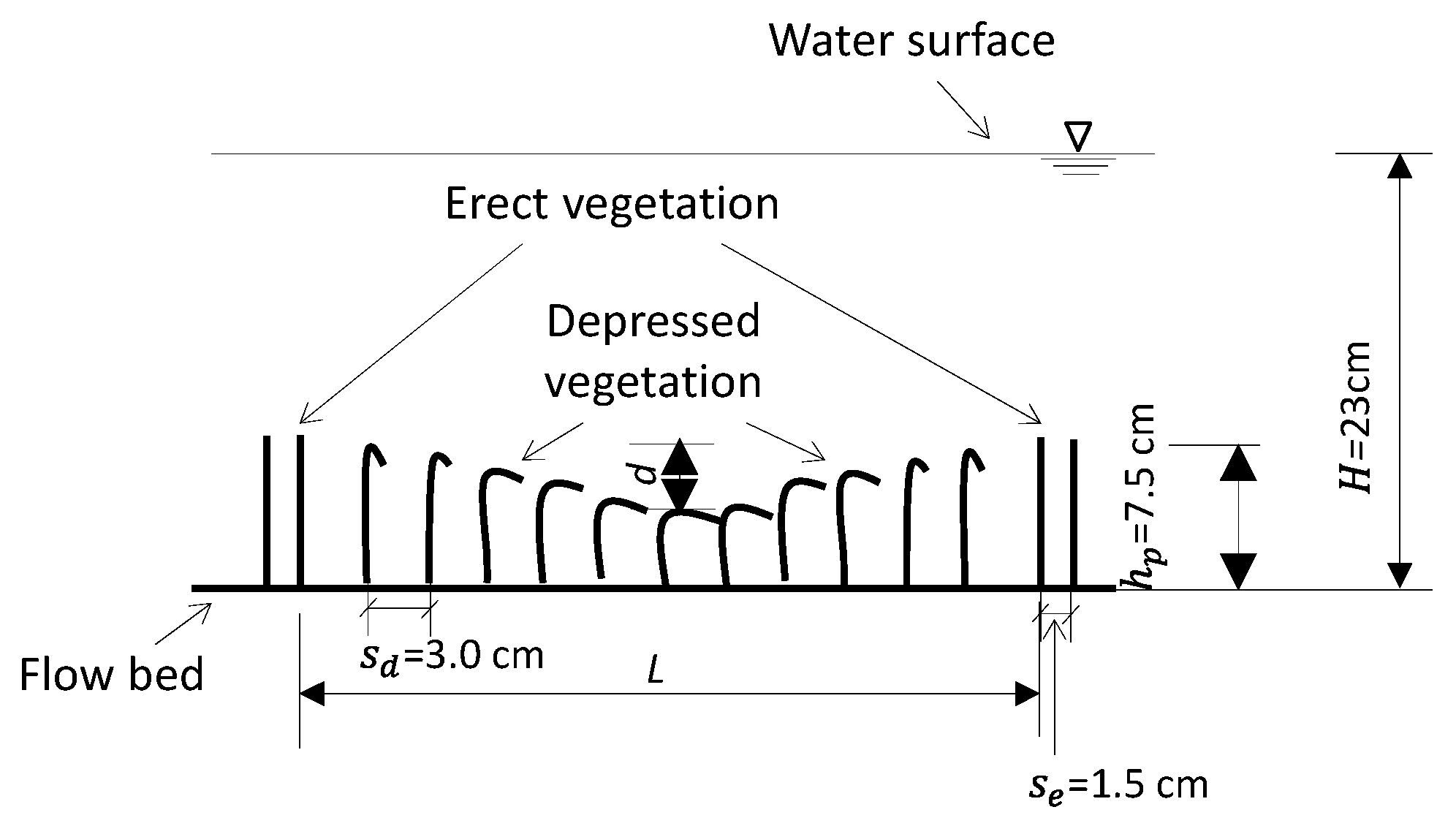
2.2. Flow Conditions and Vegetation Canopy Configurations
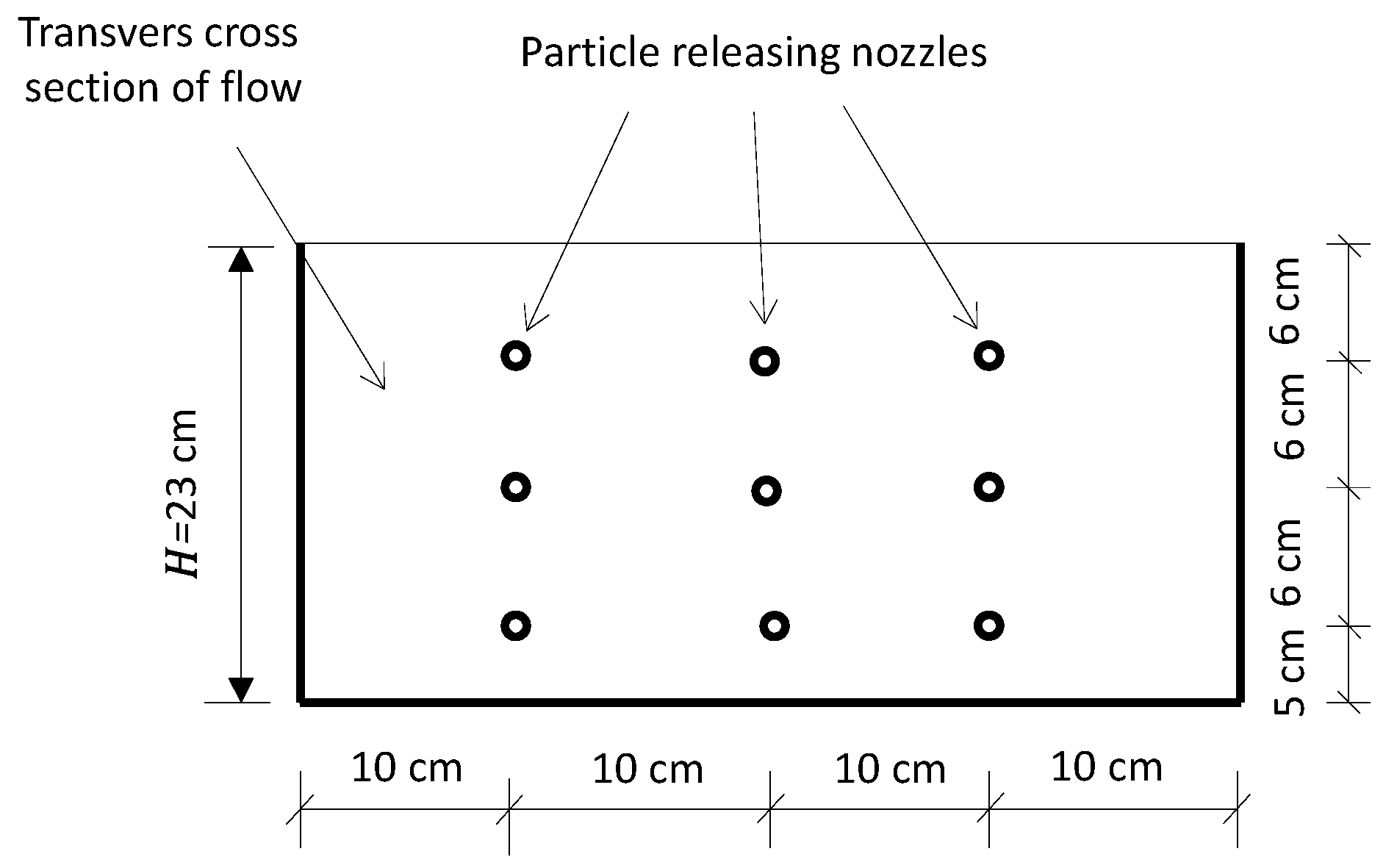
2.3. Fine Solid Pollutants and Observation Method
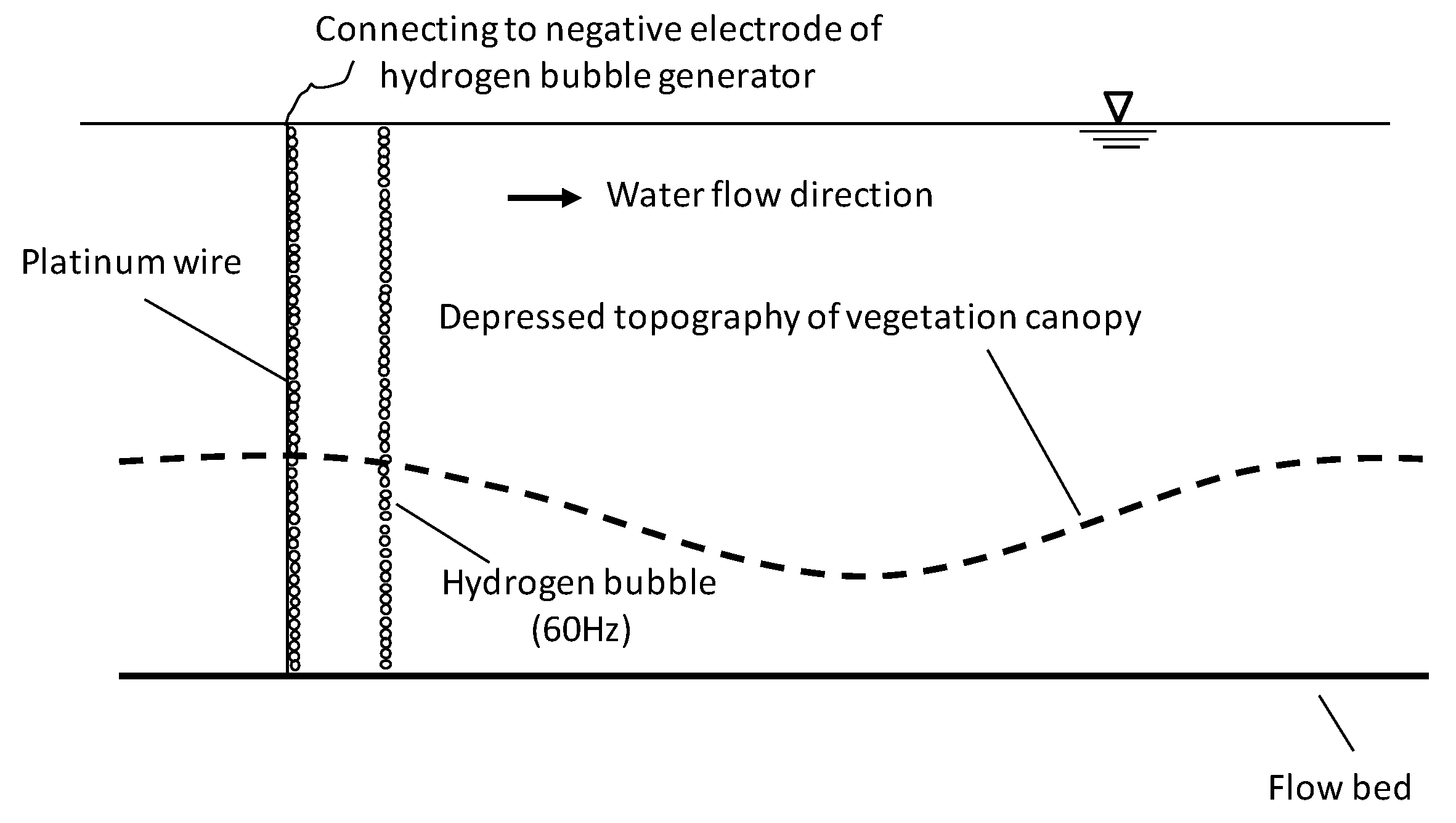
2.4. Flow Field Observation
2.5. Calculation of Vorticity and Shear Velocity
3. Result
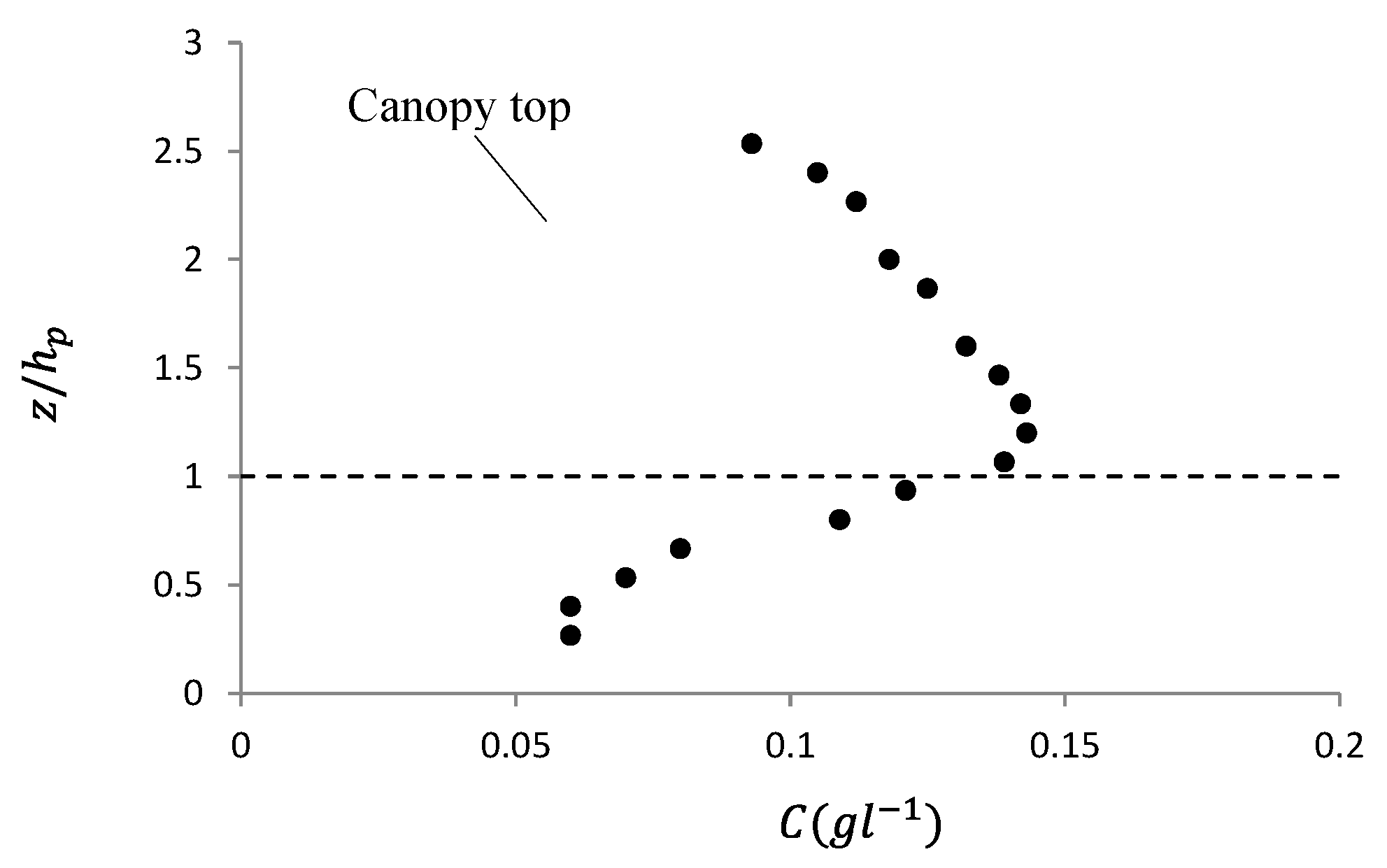
3.1. Vertical Profile of Fine Solid Pollutants in Front of the Caves
3.2. Total Amount of Foliage Captured Solid Pollutants in the Canopy Cave Regions
3.3. Hydrodynamic Mechanisms of Foliage Capture of Fine Solid Pollutants
3.3.1. Flow Velocity Field around the Canopy Caves
3.3.2. Streamlines around the Canopy Caves
3.3.3. Evolution of Vorticities in the Canopy Caves
3.3.4. Normal and Shear Turbulent Momentums around the Canopy Caves
4. Discussions
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhao, Z.; Liu, G.; Liu, Q.; Huang, C.; Li, H. Studies on Spatiotemporal Variability of River Water Quality and Its Relationships with Soil and Precipitation: A Case Study of the Mun River Basin in Thailand. Int. J. Environ. Res. Public Health 2018, 15, 2466. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shen, H.; Zhao, Y.; Cao, W.; Hu, C.; Sun, C. Distribution and Potential Ecological Risk of Heavy Metals in Water, Sediments, and Aquatic Macrophytes: A Case Study of the Junction of Four Rivers in Linyi City, China. Int. J. Environ. Res. Public Health 2019, 16, 2861. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Wang, P.; Wang, C.; Wang, X.; Hu, B. Assessment of the Multi-Objective Reservoir Operation for Maintaining the Turbidity Maximum Zone in the Yangtze River Estuary. Int. J. Environ. Res. Public Health 2018, 15, 2118. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Sun, T.; Su, M. Short-term Response of Aquatic Ecosystem Metabolism to Turbidity Disturbance in Experimental Estuarine Wetlands. Ecol. Eng. 2019, 136, 55–61. [Google Scholar] [CrossRef]
- Tunçsiper, B. Combined Natural Wastewater Treatment Systems for Removal of Organic Matter and Phosphorus from Polluted Streams. J. Clean. Prod. 2019, 228, 1368–1376. [Google Scholar] [CrossRef]
- Guo, C.; Cui, Y.; Shi, Y.; Luo, Y.; Liu, F.; Wan, D.; Ma, Z. Improved Test to Determine Design Parameters for Optimization of Free Surface Flow Constructed Wetlands. Bioresour. Technol. 2019, 280, 199–212. [Google Scholar] [CrossRef]
- Dagenais, D.; Brisson, J.; Fletcher, T.D. The Role of Plants in Bioretention Systems: Does the Science Underpin Current Guidance? Ecol. Eng. 2018, 120, 532–545. [Google Scholar] [CrossRef]
- Elzein, Z.; Abdou, A.; ElGawad, I.A. Constructed Wetlands as a Sustainable Wastewater Treatment Method in Communities. Procedia Environ. Sci. 2016, 34, 605–617. [Google Scholar] [CrossRef]
- Alemu, T.; Bahrndorff, S.; Alemayehu, E.; Ambelu, A. Agricultural Sediment Reduction Using Natural Herbaceous Buffer Strips: A Case Study of the East African Highland. Water Environ. J. 2017, 31, 522–527. [Google Scholar] [CrossRef]
- Abdelhakeem, S.G.; Aboulroos, S.A.; Kamel, M.M. Performance of a Vertical Subsurface Flow Constructed Wetland under Different Operational Conditions. J. Adv. Res. 2016, 7, 803–814. [Google Scholar] [CrossRef]
- Leguédois, S.; Ellis, T.W.; Hairsine, P.B.; Tongway, D.J. Sediment Trapping by a Tree Belt: Processes and Consequences for Sediment Delivery. Hydrol. Process. 2008, 22, 3523–3534. [Google Scholar] [CrossRef]
- Ünlü, S.; Alpar, B. Ecological Risk Assessment of HCH and DDT Residues in a Sediment Core from the Küçükçekmece Lagoon, Turkey. Bull. Environ. Contam. Toxicol. 2018, 101, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Adrian, R.J. Particle-Imaging Techniques for Experimental Fluid Mechanics. Annu. Rev. Fluid Mech. 1991, 23, 261–304. [Google Scholar] [CrossRef]
- Keane, R.D.; Adrain, R.J. Theory of Cross Correlation Analysis of PIV Image. Appl. Sci. Res. 1992, 49, 191–215. [Google Scholar] [CrossRef]
- Kolerski, T.; Wielgat, P. Velocity Field Characteristics at the Inlet to a Pipe Culvert. Arch. Hydro-Eng. Environ. Mech. 2014, 61, 127–140. [Google Scholar] [CrossRef]
- Ghisalberti, M.; Nepf, H.M. Mixing Layers and Coherent Structures in Vegetated Aquatic Flows. J. Phys. Res. 2002, 107. [Google Scholar] [CrossRef]
- Li, D.; Yang, Z.; Sun, Z.; Huai, W.; Liu, J. Theoretical Model of Suspended Sediment Concentration in a Flow with Submerged Vegetation. Water 2018, 10, 1656. [Google Scholar] [CrossRef]
- Wang, X.Y.; Xie, W.M.; Zhang, D.; He, Q. Wave and Vegetation Effects on Flow and Suspended Sediment Characteristics: A flume Study. Estuar. Coast. Shelf Sci. 2016, 182, 1–11. [Google Scholar] [CrossRef]
- Okamoto, T.; Nezu, I.; Ikeda, H. Vertical Mass and Momentum Transport in Open-Channel Flows with Submerged Vegetations. J. Hydro-Environ. Res. 2012, 6, 297. [Google Scholar] [CrossRef]
- Nezu, I.; Sanjou, M. Turbulence Structure and Coherent Motion in Vegetated Canopy Open-Channel Flows. J. Hydro-Environ. Res. 2008, 2, 62–90. [Google Scholar] [CrossRef]
- Kuriata-Potasznik, A.; Szymczyk, S.; Pilejczyk, D. Effect of Bottom Sediments on The Nutrient and Metal Concentration in Macrophytes of River-Lake Systems. Ann. Limnol.-Int. J. Limnol. 2018, 54, 1. [Google Scholar] [CrossRef]
- Madsen, J.D.; Chambers, P.A.; James, W.F.; Koch, E.W.; Westlake, D.F. The Interaction Between Water Movement, Sediment Dynamics and Submersed Macrophytes. Hydrobiologia 2001, 444, 71–84. [Google Scholar] [CrossRef]
- Griffin, E.R.; Perignon, M.C.; Friedman, J.M.; Tucker, G.E. Effects of Woody Vegetation on Overbank Sand Transport during A Large Flood, Rio Puerco, New Mexico. Geomorphology 2014, 207, 30–50. [Google Scholar] [CrossRef]
- Hansen, J.C.R.; Reidenbach, M.A. Seasonal Growth and Senescence of a Zostera Marina Seagrass Meadow Alters Wave-Dominated Flow and Sediment Suspension within A Coastal Bay. Estuar. Coasts 2013, 36, 1099–1114. [Google Scholar] [CrossRef]
- Kleeberg, A.; Kohler, J.; Sukhodolova, T.; Sukhodolov, A. Effects of Aquatic Macrophytes on Organic Matter Deposition, Resuspension and Phosphorus Entrainment in a Lowland River. Freshw. Biol. 2010, 55, 326–345. [Google Scholar] [CrossRef]
- Liu, J.H.; Yang, S.L.; Zhu, Q.; Zhang, J. Controls on Suspended Sediment Concentration Profiles in The Shallow and Turbid Yangtze Estuary. Cont. Shelf Res. 2014, 90, 96–108. [Google Scholar] [CrossRef]
- Lupker, M.; France-Lanord, C.; Lavé, J.; Bouchez, J.; Galy, V.; Métivier, F.; Gaillardet, J.; Lartiges, B.; Mugnier, L.J. A Rouse-based Method to Integrate the Chemical Composition of River Sediments: Application to the Ganga Basin. J. Geophy. Res. 2011, 116, 1–24. [Google Scholar] [CrossRef]
- Neary, V.; Constantinescu, S.; Bennett, S.; Diplas, P. Effects of Vegetation on Turbulence, Sediment Transport, and Stream Morphology. J. Hydraul. Eng. 2012, 138, 765–776. [Google Scholar] [CrossRef]
- Wang, F.; Lu, T.; Sikora, W. Intertidal Marsh Suspended Sediment Transport Processes, Terrebonne Bay, Louisiana, USA. J. Coast. Res. 1993, 9, 209–220. [Google Scholar]
- Wilkie, L.; O’Hare, M.T.; Davidson, I.; Dudley, B.; Paterson, D.M. Particle Trapping and Retention by Zostera Noltii: A Flume and Field Study. Aquat. Bot. 2012, 102, 15–22. [Google Scholar] [CrossRef]
- Branković, S.; Pavlović-Muratspahić, D.; Topuzović, M.; Glišić, R.; Milivojević, J.; Đekić, V. Metals Concentration and Accumulation in Several Aquatic Macrophytes. Biotechnol. Biotechnol. Equip. 2012, 26, 2731–2736. [Google Scholar] [CrossRef][Green Version]
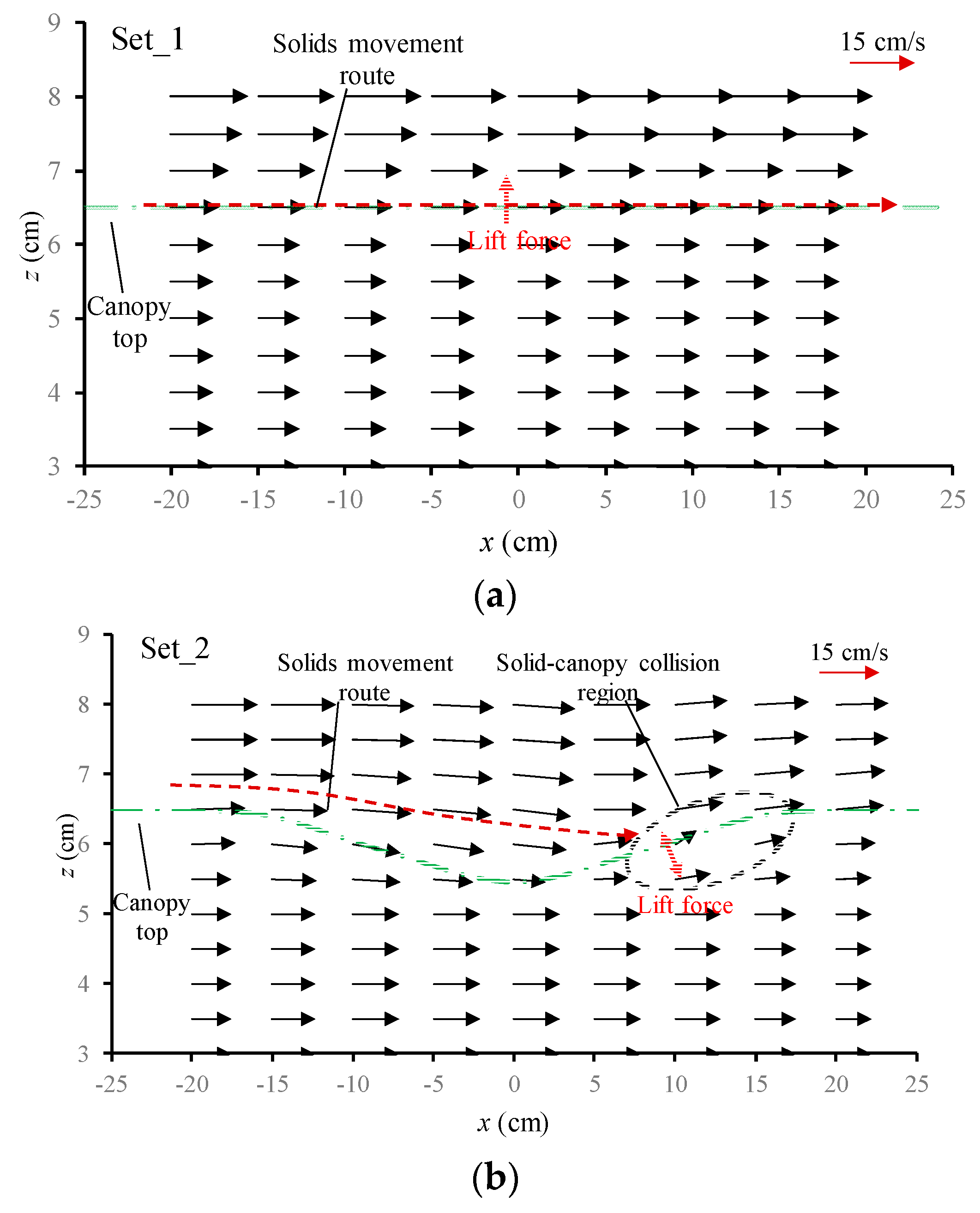
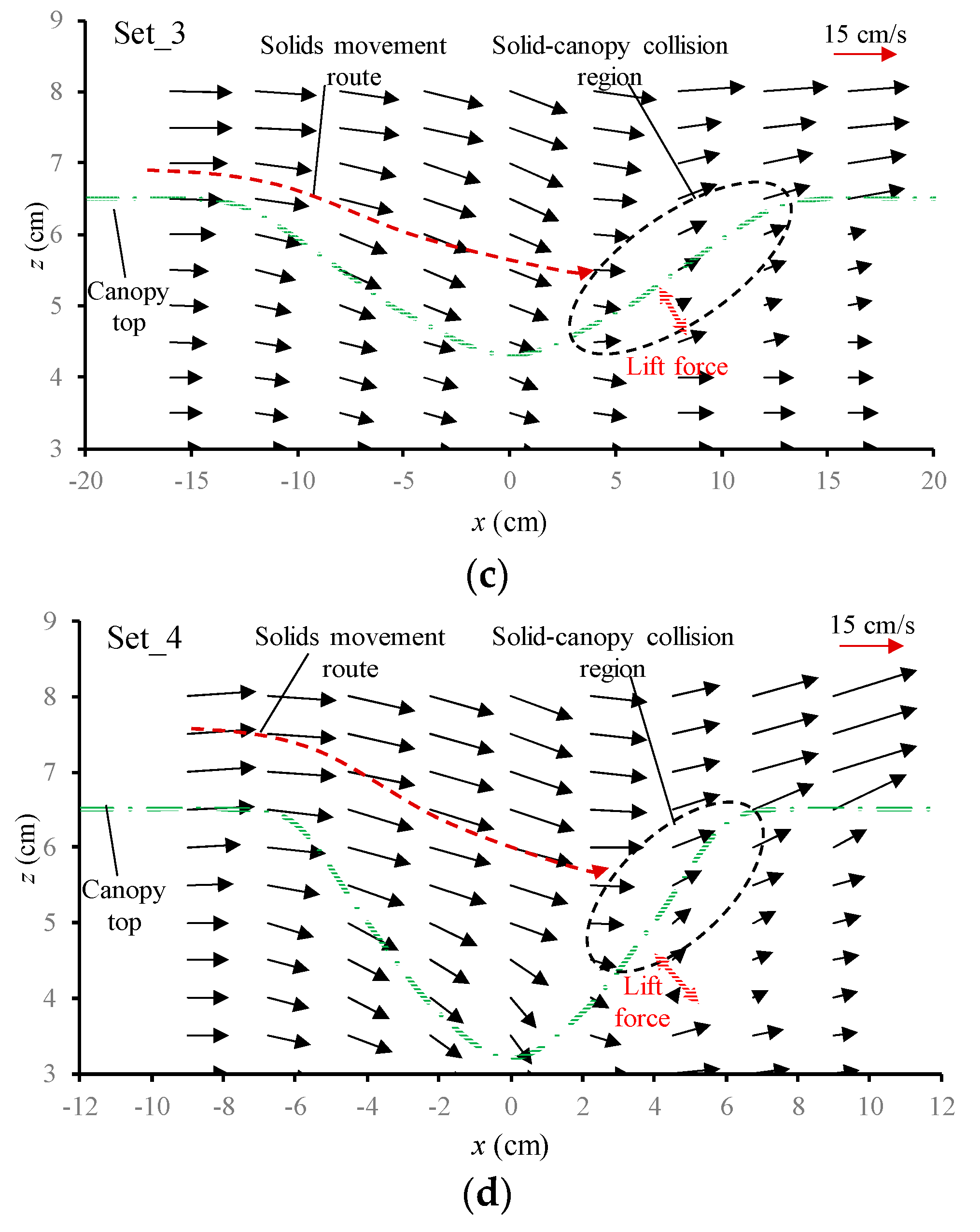
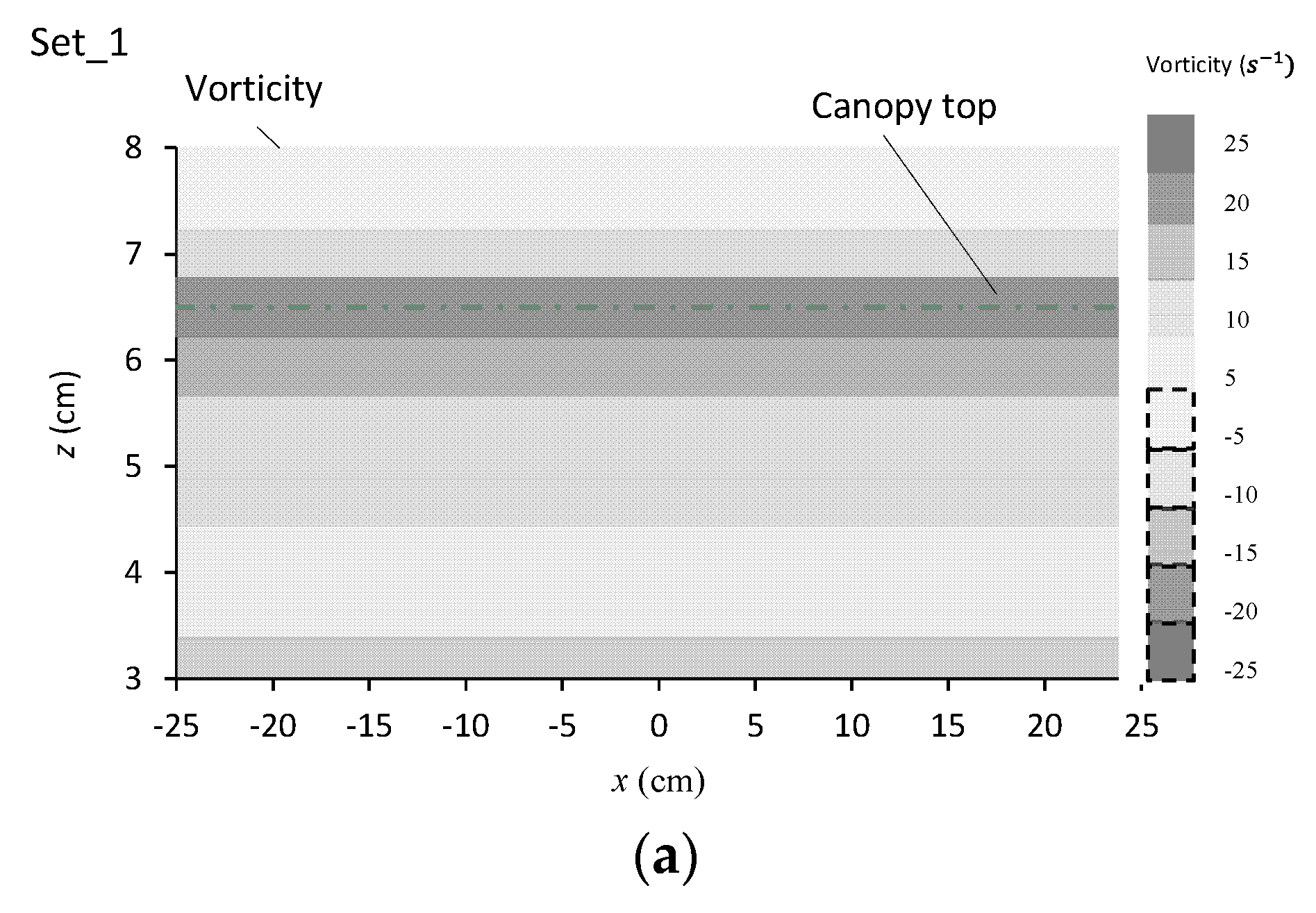
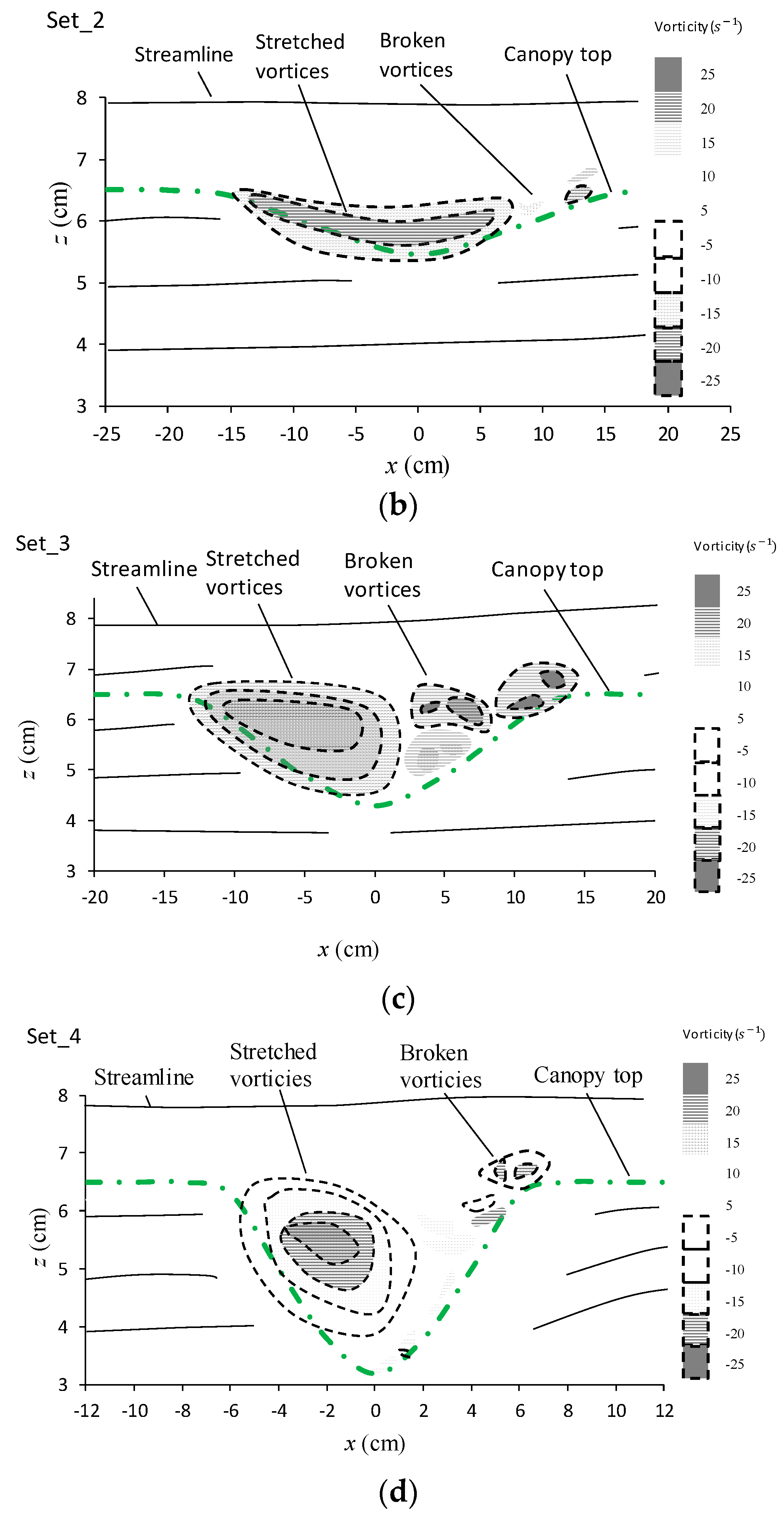
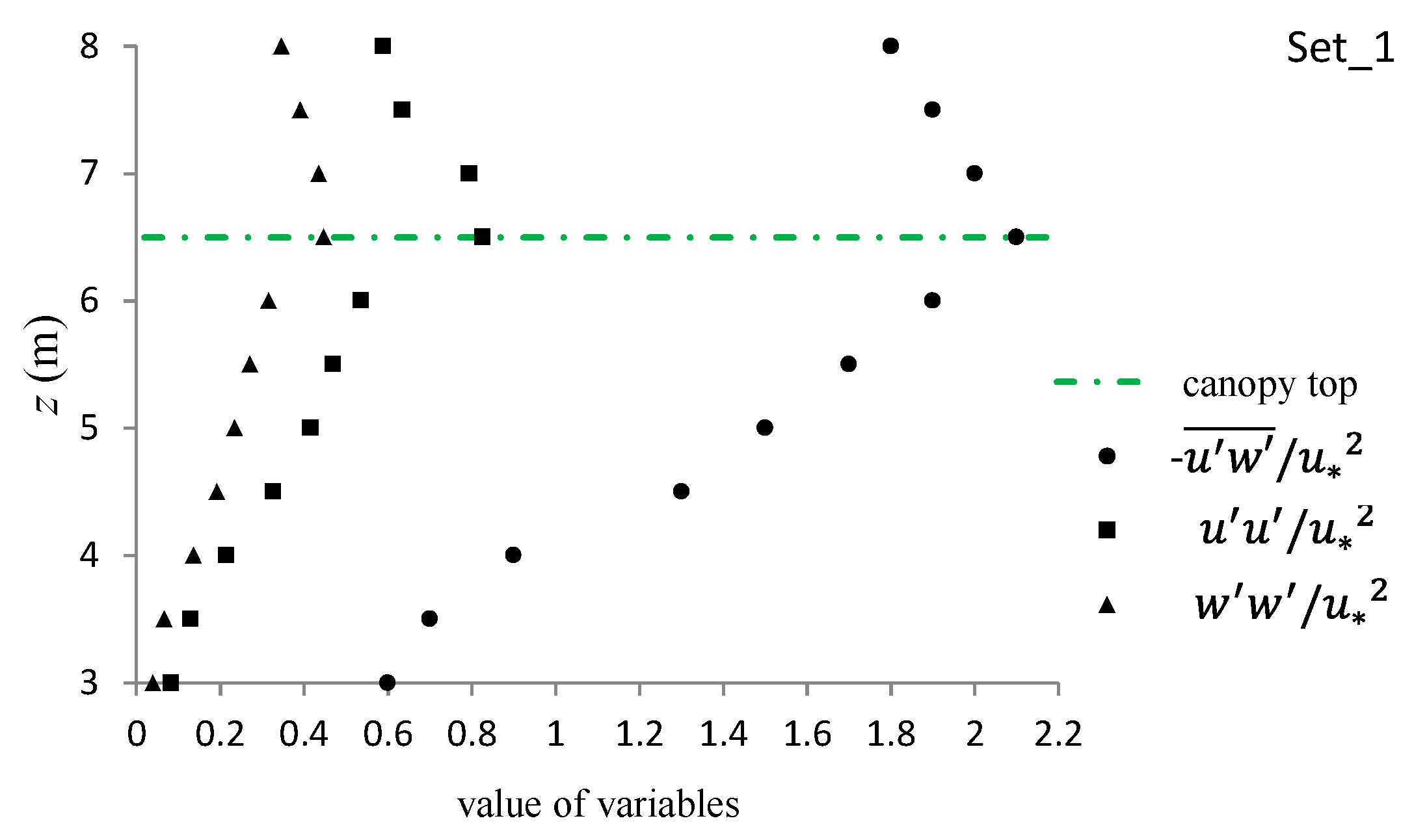
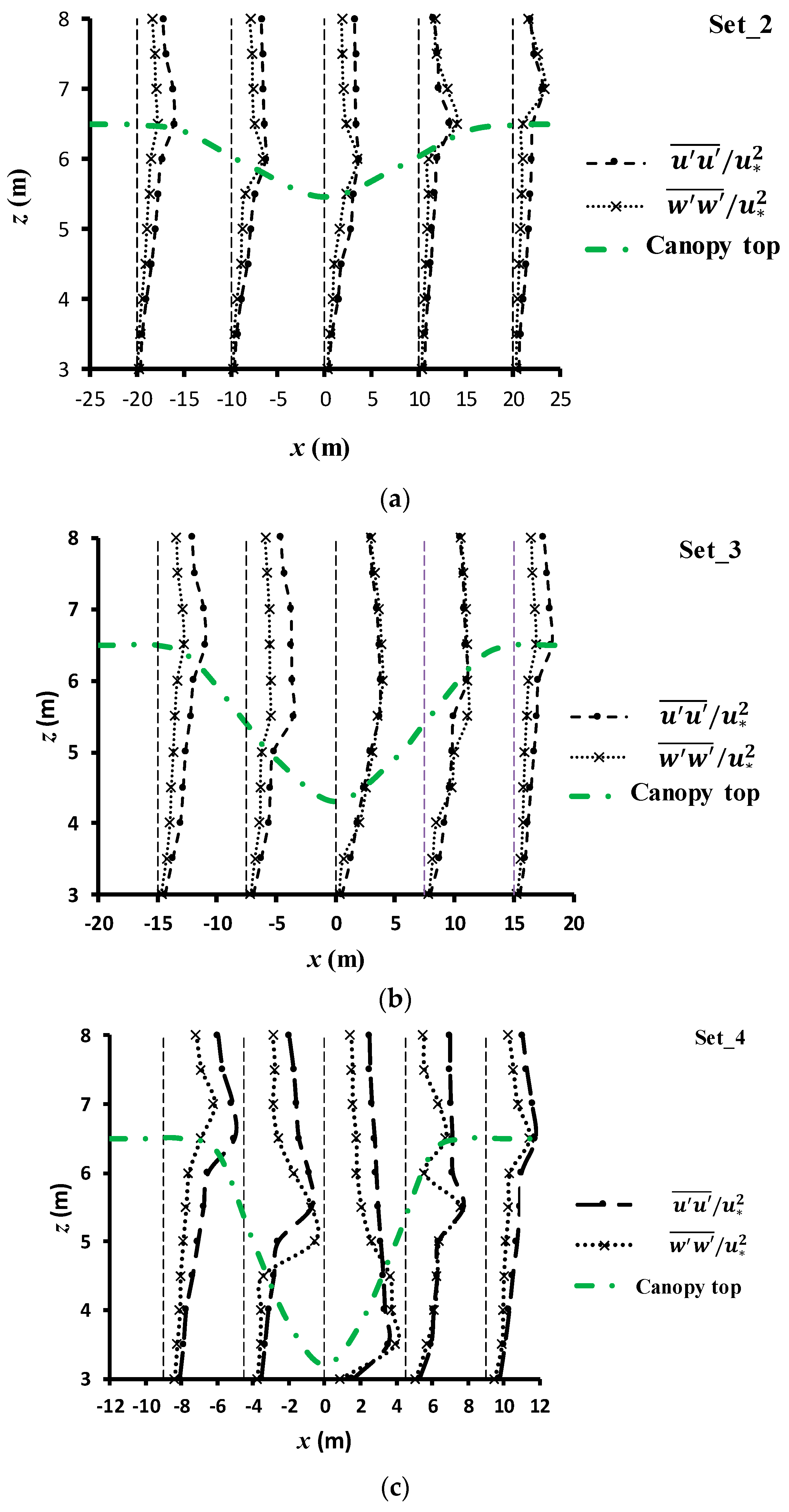
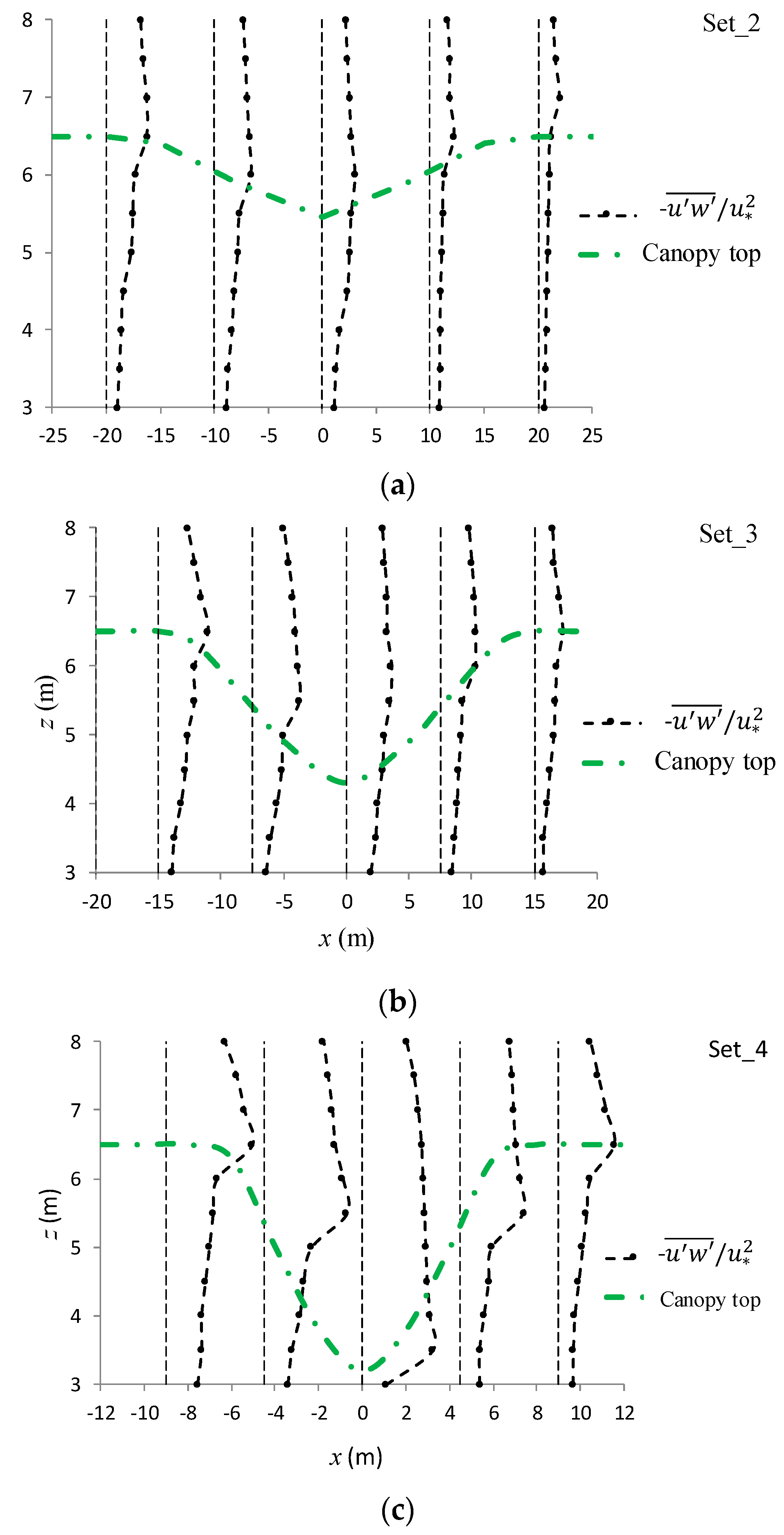
| Experiment No. | Depression Length L (cm) | Depression Depth d (cm) | Aspect Ratio L/d |
|---|---|---|---|
| Set_1 | - | - | Flat canopy top |
| Set_2 | 36 | 1.0 | 36 |
| Set_3 | 30 | 2.0 | 15 |
| Set_4 | 24 | 3.0 | 8 |
| Experiment No. | Aspect Ratio L/d | Captured Solid Pollutants (g) | Enhance Rate of Foliage Capture (Times) |
|---|---|---|---|
| Set_1 | Flat canopy top | 0.65 | 1 |
| Set_2 | 36 | 3.12 | 4.8 |
| Set_3 | 15 | 5.39 | 8.3 |
| Set_4 | 8 | 8.56 | 13.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Xie, L.; Su, T.-c. Bio-Capture of Solid Pollutants by Vegetation Canopy Cave in Shallow Water Flow. Int. J. Environ. Res. Public Health 2019, 16, 4846. https://doi.org/10.3390/ijerph16234846
Li Y, Xie L, Su T-c. Bio-Capture of Solid Pollutants by Vegetation Canopy Cave in Shallow Water Flow. International Journal of Environmental Research and Public Health. 2019; 16(23):4846. https://doi.org/10.3390/ijerph16234846
Chicago/Turabian StyleLi, Yanhong, Liquan Xie, and Tsung-chow Su. 2019. "Bio-Capture of Solid Pollutants by Vegetation Canopy Cave in Shallow Water Flow" International Journal of Environmental Research and Public Health 16, no. 23: 4846. https://doi.org/10.3390/ijerph16234846
APA StyleLi, Y., Xie, L., & Su, T.-c. (2019). Bio-Capture of Solid Pollutants by Vegetation Canopy Cave in Shallow Water Flow. International Journal of Environmental Research and Public Health, 16(23), 4846. https://doi.org/10.3390/ijerph16234846






