Target, Suspect and Non-Target Screening of Silylated Derivatives of Polar Compounds Based on Single Ion Monitoring GC-MS
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Stock Solutions
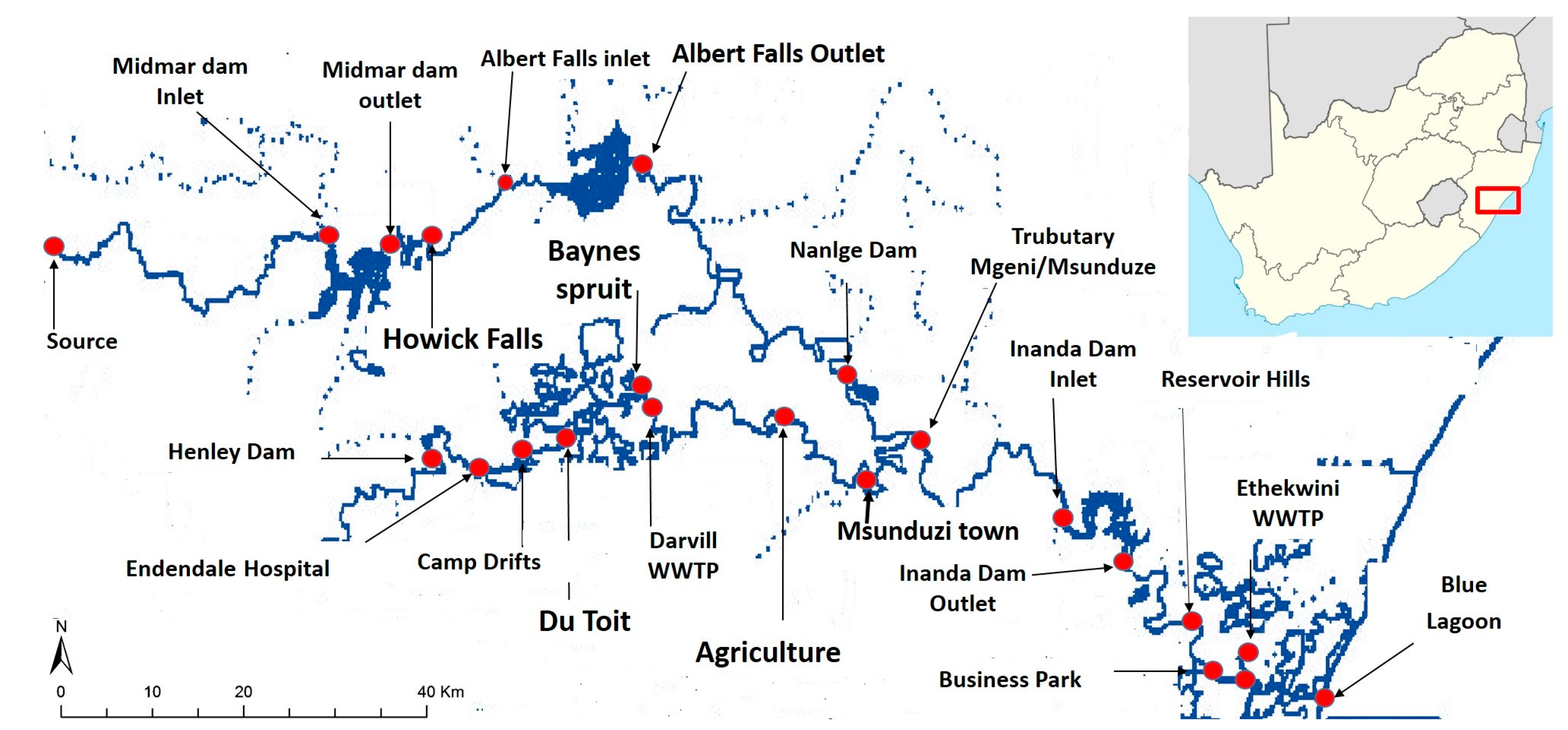
2.2. Sampling of Sediments and Water
2.3. Sampling of Biosolids
2.4. Sample Extraction
2.4.1. Water
2.4.2. Sediments and Biosolids
2.4.3. Recoveries and Procedural Blanks
2.5. Derivatization
2.6. GC-MS Analysis
2.7. Data Analysis (Including Software)
3. Confirmation of Standards
4. Results and Discussion
4.1. Analysis of Environmental Samples: Target Analysis
4.1.1. Quantification Analysis
4.1.2. Qualitative Analysis
4.2. Suspect Analysis
4.3. Non-Target Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agunbiade, F.O.; Moodley, B. Pharmaceuticals as emerging organic contaminants in Umgeni River water system, KwaZulu-Natal, South Africa. Environ. Monit. Assess. 2014, 186, 7273–7291. [Google Scholar] [CrossRef] [PubMed]
- Agunbiade, F.O.; Moodley, B. Occurrence and distribution pattern of acidic pharmaceuticals in surface water, wastewater, and sediment of the Msunduzi River, Kwazulu-Natal, South Africa. Environ. Toxicol. Chem. 2016, 35, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Albero, B.; Sánchez-Brunete, C.; García-Valcárcel, A.I.; Pérez, R.A.; Tadeo, J.L. Ultrasound-assisted extraction of emerging contaminants from environmental samples. Trends Anal. Chem. 2015, 71, 110–118. [Google Scholar] [CrossRef]
- Dafouz, R.; Caceres, N.; Rodriguez-Gil, J.L.; Mastroianni, N.; de Alda, M.L.; Barcelo, D.; de Miguel, A.G.; Valcarcel, Y. Does the presence of caffeine in the marine environment represent an environmental risk? A regional and global study. Sci. Total Environ. 2018, 615, 632–642. [Google Scholar] [CrossRef]
- Sopilniak, A.; Elkayam, R.; Rossin, A.V.; Lev, O. Emerging organic pollutants in the vadose zone of a soil aquifer treatment system: Pore water extraction using positive displacement. Chemosphere 2018, 190, 383–392. [Google Scholar] [CrossRef]
- Gavrilescu, M.; Demnerova, K.; Aamand, J.; Agathoss, S.; Fava, F. Emerging pollutants in the environment: Present and future challenges in biomonitoring, ecological risks and bioremediation. New Biotech. 2015, 32, 147–156. [Google Scholar] [CrossRef]
- Prasse, C.; Wagner, M.; Schulz, R.; Ternes, T.A. Biotransformation of the Antiviral Drugs Acyclovir and Penciclovir in Activated Sludge Treatment. Environ. Sci. Technol. 2011, 45, 2761–2769. [Google Scholar] [CrossRef]
- Souchier, M.; Benali-Raclot, D.; Benanou, D.; Boireau, V.; Gomez, E.; Casellas, C.; Chiron, S. Screening triclocarban and its transformation products in river sediment using liquid chromatography and high resolution mass spectrometry. Sci. Total Environ. 2015, 502, 199–205. [Google Scholar] [CrossRef]
- Haakh, F. Emerging organic pollutants-A new challenge for drinking water supply? Wasser Abwasser 2010, 151, 740–748. [Google Scholar]
- Gago-Ferrero, P.; Schymanski, E.L.; Bletsou, A.A.; Aalizadeh, R.; Hollender, J.; Thomaidis, N.S. Extended Suspect and Non-Target Strategies to Characterize Emerging Polar Organic Contaminants in Raw Wastewater with LC-HRMS/MS. Environ. Sci. Technol. 2015, 49, 12333–12341. [Google Scholar] [CrossRef]
- Hernandez, F.; Ibanez, M.; Portoles, T.; Cervera, M.I.; Sancho, J.V.; Lopez, F.J. Advancing towards universal screening for organic pollutants in waters. J. Hazard. Mater. 2015, 282, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Sursyakova, V.V.; Burmakina, G.V.; Rubaylo, A.I. Strategy for non-target ionic analysis by capillary electrophoresis with ultraviolet detection. Anal. Bioanal. Chem. 2017, 409, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Gosetti, F.; Mazzucco, E.; Gennaro, M.C.; Marengo, E. Non-Target UHPLC/MS Analysis Emergency Contaminants Water; Springer International Publishing: Basel, Switzerland, 2015. [Google Scholar]
- Hauler, C.; Vetter, W. A non-targeted gas chromatography/electron capture negative ionization mass spectrometry selected ion monitoring screening method for polyhalogenated compounds in environmental samples. Rapid Commun. Mass Spectrom. 2015, 29, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Plassmann, M.M.; Tengstrand, E.; Åberg, K.M.; Benskin, J.P. Non-target time trend screening: A data reduction strategy for detecting emerging contaminants in biological samples. Anal. Bioanal. Chem. 2016, 408, 4203–4208. [Google Scholar] [CrossRef] [PubMed]
- Loos, R.; Gawlik, B.M.; Locoro, G.; Rimaviciute, E.; Contini, S.; Bidoglio, G. EU-wide survey of polar organic persistent pollutants in European river waters. Environ. Pollut. 2009, 157, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Schymanski, E.L.; Singer, H.P.; Slobodnik, J.; Ipolyi, I.M.; Oswald, P.; Krauss, M.; Schulze, T.; Haglund, P.; Letzel, T.; Grosse, S.; et al. Non-target screening with high-resolution mass spectrometry: Critical review using a collaborative trial on water analysis. Anal. Bioanal. Chem. 2015, 407, 6237–6255. [Google Scholar] [CrossRef] [PubMed]
- Bade, R.; Causanilles, A.; Emke, E.; Bijlsma, L.; Sancho, J.V.; Hernandez, F.; de Voogt, P. Facilitating high resolution mass spectrometry data processing for screening of environmental water samples: An evaluation of two deconvolution tools. Sci. Total Environ. 2016, 569, 434–441. [Google Scholar] [CrossRef]
- Solliec, M.; Roy-Lachapelle, A.; Sauve, S. Development of a suspect and non-target screening approach to detect veterinary antibiotic residues in a complex biological matrix using liquid chromatography/high-resolution mass spectrometry. Rapid Commun. Mass Spectrom. 2015, 29, 2361–2373. [Google Scholar] [CrossRef]
- Jernberg, J.; Pellinen, J.; Rantalainen, A.L. Qualitative nontarget analysis of landfill leachate using gas chromatography time-of-flight mass spectrometry. Talanta 2013, 103, 384–391. [Google Scholar] [CrossRef]
- Baduel, C.; Mueller, J.F.; Tsai, H.H.; Ramos, M.J.G. Development of sample extraction and clean-up strategies for target and non-target analysis of environmental contaminants in biological matrices. J. Chromatogr. A 2015, 1426, 33–47. [Google Scholar] [CrossRef]
- Castro, G.; Roca, M.; Rodriguez, I.; Ramil, M.; Cela, R. Identification and determination of chlorinated azoles in sludge using liquid chromatography quadrupole time-of-flight and triple quadrupole mass spectrometry platforms. J. Chromatogr. A 2016, 1476, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.N.; Xu, Y.; Li, F.; Jmaiff, L.; Hrudey, S.E.; Li, X.F. Nontargeted identification of peptides and disinfection byproducts in water. J. Environ. Sci. 2016, 42, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Gumbi, B.P.; Moodley, B.; Birungi, G.; Ndungu, P.G. Detection and quantification of acidic drug residues in South African surface water using gas chromatography-mass spectrometry. Chemosphere 2017, 168, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Gumbi, B.P.; Moodley, B.; Birungi, G.; Ndungu, P.G. Assessment of nonsteroidal anti-inflammatory drugs by ultrasonic-assisted extraction and GC-MS in Mgeni and Msunduzi river sediments, KwaZulu-Natal, South Africa. Environ. Sci. Pollut. Res. 2017, 24, 20015–20028. [Google Scholar] [CrossRef]
- Matongo, S.; Birungi, G.; Moodley, B.; Ndungu, P. Pharmaceutical residues in water and sediment of Msunduzi River, KwaZulu-Natal, South Africa. Chemosphere 2015, 134, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Ruiz, R.; Andres-Costa, M.J.; Andreu, V.; Pico, Y. Simultaneous determination of traditional and emerging illicit drugs in sediments, sludges and particulate matter. J. Chromatogr. A 2015, 1405, 103–115. [Google Scholar] [CrossRef]
- Antonić, J.; Heath, E. Determination of NSAIDs in river sediment samples. Anal. Bioanal. Chem. 2007, 387, 1337–1342. [Google Scholar] [CrossRef]
- Archana, G.; Dhodapkar, R.; Kumar, A. Offline solid-phase extraction for preconcentration of pharmaceuticals and personal care products in environmental water and their simultaneous determination using the reversed phase high-performance liquid chromatography method. Environ. Monit. Assess. 2016, 188, 10616–10661. [Google Scholar] [CrossRef]
- Castiglioni, S.; Zuccato, E.; Chiabrando, C.; Fanelli, R.; Bagnati, R. Mass spectrometric analysis of illicit drugs in wastewater and surface water. Mass Spectrom. Rev. 2008, 27, 378–394. [Google Scholar] [CrossRef]
- González-Mariño, I.; Quintana, J.B.; Rodríguez, I.; Cela, R. Determination of drugs of abuse in water by solid-phase extraction, derivatisation and gas chromatography–ion trap-tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 1748–1760. [Google Scholar] [CrossRef]
- Wu, J.-W.; Chen, H.-C.; Ding, W.-H. Ultrasound-assisted dispersive liquid–liquid microextraction plus simultaneous silylation for rapid determination of salicylate and benzophenone-type ultraviolet filters in aqueous samples. J. Chromatogr. A 2013, 1302, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.; Ellison, S.L.; Wood, R. Harmonized guidelines for single-laboratory validation of methods of analysis (IUPAC Technical Report). Pure Appl. Chem. 2002, 74, 835–855. [Google Scholar] [CrossRef]
- Gonzalez, A.G.; Herrador, M.A. A practical guide to analytical method validation, including measurement uncertainty and accuracy profiles. Trends Anal. Chem. 2007, 26, 227–238. [Google Scholar] [CrossRef]
- Togola, A.; Budzinski, H. Analytical development for analysis of pharmaceuticals in water samples by SPE and GC-MS. Anal. Bioanal. Chem. 2007, 388, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Basaglia, G.; Pietrogrande, M.C. Optimization of a SPME/GC/MS Method for the Simultaneous Determination of Pharmaceuticals and Personal Care Products in Waters. Chromatographia 2012, 75, 361–370. [Google Scholar] [CrossRef]
- Helenkar, A.; Sebok, A.; Zaray, G.; Molnar-Perl, I.; Vasanits-Zsigrai, A. The role of the acquisition methods in the analysis of the non-steroidal anti-inflammatory drugs in Danube River by gas chromatography-mass spectrometry. Talanta 2010, 82, 600–607. [Google Scholar] [CrossRef]
- Lee, S.; Song, G.J.; Kannan, K.; Moon, H.B. Occurrence of PBDEs and other alternative brominated flame retardants in sludge from wastewater treatment plants in Korea. Sci. Total Environ. 2014, 470, 1422–1429. [Google Scholar] [CrossRef]
- Molina-Fernandez, N.; Perez-Conde, C.; Rainieri, S.; Sanz-Landaluze, J. Method for quantifying NSAIDs and clofibric acid in aqueous samples, lumpfish (Cyclopterus lumpus) roe, and zebrafish (Danio rerio) eleutheroembryos and evaluation of their bioconcentration in zebrafish eleutheroembryos. Environ. Sci. Pollut. Res. 2016, 24, 10907–10918. [Google Scholar] [CrossRef]
- Vila, M.; Celeiro, M.; Lamas, J.P.; Garcia-Jares, C.; Dagnac, T.; Llompart, M. Simultaneous in-vial acetylation solid-phase microextraction followed by gas chromatography tandem mass spectrometry for the analysis of multiclass organic UV filters in water. J. Hazard. Mater. 2017, 323, 45–55. [Google Scholar] [CrossRef]
- Wang, D.-G.; Zheng, Q.-D.; Wang, X.-P.; Du, J.; Tian, C.-G.; Wang, Z.; Ge, L.-K. Illicit drugs and their metabolites in 36 rivers that drain into the Bohai Sea and north Yellow Sea, north China. Environ. Sci. Pollut. Res. 2016, 23, 16495–16503. [Google Scholar] [CrossRef]
- Weigel, S.; Kallenborn, R.; Hühnerfuss, H. Simultaneous solid-phase extraction of acidic, neutral and basic pharmaceuticals from aqueous samples at ambient (neutral) pH and their determination by gas chromatography–mass spectrometry. J. Chromatogr. A 2004, 1023, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Schymanski, E.L.; Jeon, J.; Gulde, R.; Fenner, K.; Ruff, M.; Singer, H.P.; Hollender, J. Identifying small molecules via high resolution mass spectrometry: Communicating confidence. Environ. Sci. Technol. 2014, 48, 2097–2098. [Google Scholar] [CrossRef] [PubMed]
| Target Analytes TMS Derivative | Type | Chemical Formula Trimethylsilyl (TMS) | Retention Time Minutes | Fragment Pattern TMS Derivative (m/z) | Selected Ion Monitored (m/z) | Similarity Index |
|---|---|---|---|---|---|---|
| Methamphetamine | Illicit drug | C10H15N | 5.450 | 58, 91, 134, 148 | 58, 91 | 84 |
| Salicylic acid | NSAID | C7H6O3 | 6.345 | 73, 135, 193, 209, 267 | 135, 267 | 82 |
| Acetylsalicylic acid | NSAID | C9H8O4 | 6.467 | 65, 73, 120, 195, 210, 268 | 120, 195 | 95 |
| Nalidixic acid | Antibiotic | C12H12N2O3 | 6.914 | 73, 116, 162, 180, 236, 301 | 180, 236 | 89 |
| Ibuprofen | NSAID | C13H18O2 | 7.105 | 73, 117, 160, 191, 263, 278 | 117, 160 | 80 |
| Propylparaben | Antifungal agent | C10H12O3 | 7.458 | 73, 116, 162, 180, 236, 301, 251 | 162, 236 | 80 |
| Phenacetin | Analgesic | C10H13NO2 | 7.800 | 53, 109, 137, 179, 209 | 109, 179 | 96 |
| Acetaminophen | NSAID | C8H9NO2 | 8.000 | 73, 106, 166, 181, 223 | 181, 223 | 88 |
| Phenoxyphenol | Standard | C12H10O2 | 8.250 | 73, 122, 150, 185, 258 | 150, 258 | 91 |
| Morphine | Opioid analgesic | C17H19NO3 | 8.450 | 75, 103, 119, 174, 204, 232, 285 | 232, 204 | 81 |
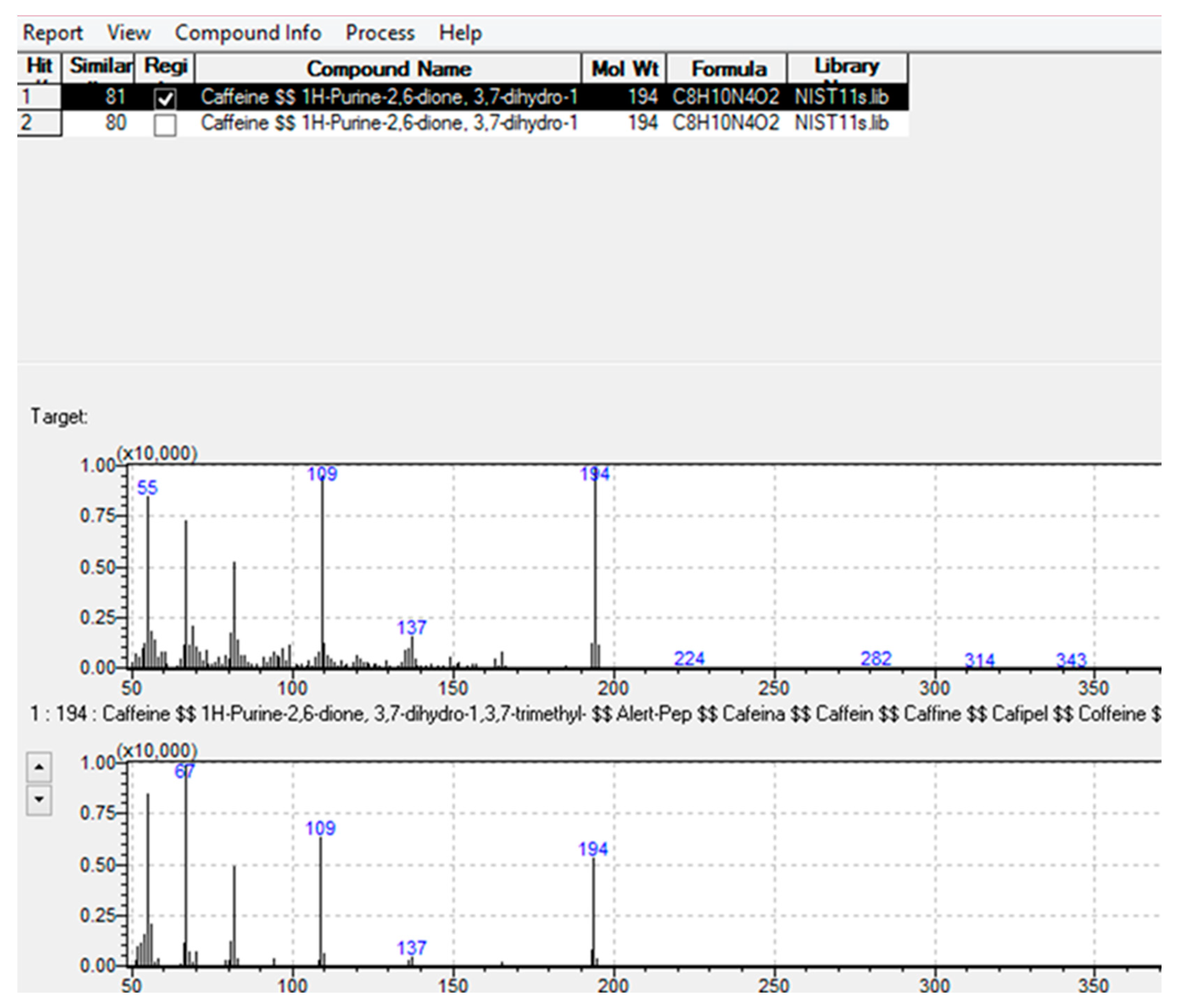
| Caffeine | Stimulant | C8H10N4O2 | 8.912 | 109, 194 | 109, 194 | 81 |
| Naproxen | NSAID | C14H14O3 | 10.685 | 173, 41, 185, 243, 302 | 185, 243 | 88 |
| procaine | Anaesthetic | C13H20N2O2 | 10.950 | 58, 86, 164 | 58, 86 | 81 |
| Triclosan | Disinfectant | C12H7Cl3O2 | 11.250 | 109, 185, 200 | 109, 200 | 95 |
| Meclofenamic acid | NSAID | C14H11Cl2NO2 | 11.990 | 73, 152, 208, 223, 313, 180 | 223, 313 | 95 |
| Ketoprofen | NSAID | C16H14O3 | 12.105 | 73, 105, 165, 179, 253, 282, 311 | 282, 311 | 88 |
| Diclofenac | NSAID | C14H11Cl2NO2 | 12.806 | 73, 93, 151, 214, 277, 367 | 214, 367 | 91 |
| Carbamazepine | Anticonvulsant | C15H12N2O | 13.654 | 63, 96, 165, 193, 236 | 193, 236 | 88 |
| Chloramphenicol | Antibiotic | C11H12Cl2N2O5 | 13.921 | 73, 93, 147, 208, 225, 361, 451 | 208, 225 | 82 |
| Cocaine | Illicit drug | C17H21NO4 | 14.530 | 77, 82, 152, 182, 272, 303 | 82, 182 | 91 |
| Procainamide | Transformation | C11H26NO2 | 15.450 | 85, 99, 192 | 86, 99 | 88 |
| 2-phenylindolizine | Metabolite | C14H11N | 15.960 | 63, 96, 165, 193 | 165, 193 | 80 |
| Sulfamethoxazole | Antibiotic | C10H11N3O3S | 16.500 | 65, 92, 156, 189, 253 | 92, 156 | 91 |
| Chlorpromazine | Antipsychotic | C17H19ClN2S | 17.605 | 58, 214, 232, 272, 315 | 58, 214 | 94 |
| Lactose | Metabolite | C36H86O16 | 18.560 | 73, 103, 147, 204, 243, 319, 521 | 204, 243 | 81 |
| Sulfamethazine | Antibiotic | C12H14N4O2S | 20.052 | 92, 108, 156, 213, 277 | 92, 213 | 95 |
| Clozapine | Antipsychotic | C18H19ClN4 | 21.750 | 70, 99, 164, 192, 243, 268, 326 | 192, 243 | 93 |
| Target Analytes | %Recovery | Limit of Detection (LOD) | Limit of Quantification (LOQ) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| River Water % | Wastewater % | Sediments % | Biosolids % | River Water ng/L | Wastewater ng/L | Sediments ng/g | Biosolids ng/g | River Water ng/L | Wastewater ng/L | Sediments ng/g | Biosolids ng/g | |
| Salicylic acid | 70 | 65 | 100 | 105 | 41 | 51 | 0.04 | 0.17 | 135 | 164 | 0.15 | 0.56 |
| Acetylsalicylic acid | 99 | 90 | 91 | 102 | 285 | 403 | 0.02 | 0.09 | 950 | 1333 | 0.07 | 0.03 |
| Ibuprofen | 99 | 96 | 92 | 102 | 143 | 160 | 0.05 | 0.03 | 477 | 533 | 0.16 | 0.08 |
| Propylparaben | 98 | 102 | 94 | 105 | 1000 | 1500 | 0.10 | 0.14 | 4000 | 3000 | 0.3 | 0.42 |
| Phenacetin | 105 | 115 | 120 | 98 | 345 | 432 | 0.08 | 0.18 | 1151 | 1444 | 0.26 | 0.59 |
| Caffeine | 96 | 104 | 92 | 107 | 100 | 400 | 0.35 | 0.1 | 300 | 1100 | 1.07 | 0.33 |
| Naproxen | 82 | 80 | 66 | 112 | 75 | 101 | 0.08 | 0.03 | 248 | 333 | 0.280 | 0.104 |
| Triclosan | 100 | 94 | 91 | 108 | 89 | 100 | 0.08 | 0.1 | 270 | 290 | 0.25 | 0.36 |
| Meclofenamic acid | 103 | 106 | 85 | 121 | 82 | 111 | 0.11 | 0.14 | 272 | 368 | 0.38 | 0.46 |
| Diclofenac | 90 | 93 | 103 | 98 | 484 | 559 | 0.09 | 0.55 | 1614 | 1864 | 0.31 | 1.8 |
| Carbamazepine | 95 | 80 | 91 | 66 | 140 | 200 | 0.11 | 1.0 | 290 | 650 | 0.32 | 3.4 |
| Chloramphenicol | 98 | 102 | 98 | 102 | 100 | 500 | 1.8 | 2.5 | 250 | 1400 | 5.5 | 7.6 |
| Target Analytes | Mgeni River | Msunduzi River | ||||||
|---|---|---|---|---|---|---|---|---|
| River Water (ng/L) | Sediments (ng/g) | River Aater (ng/L) | Sediments (ng/g) | |||||
| No. of Samples = 24 | No. of Samples = 48 | No. of Samples = 14 | No. of Samples = 28 | |||||
| Range | Median | Range | Median | Range | Median | Range | Median | |
| Salicylic acid | ND–D | D | ND–40 | 1.4 | ND | ND | ND–3.43 | 0.28 |
| Acetylsalicylic acid | ND–1130 | 70 | ND–200 | 32 | ND–D | D | ND–163 | 8.0 |
| Ibuprofen | ND–2570 | 3870 | ND–13 | 2.3 | ND–D | D | ND–1.3 | 0.50 |
| Propylparaben | ND–D | D | ND–13 | 1.1 | ND–22,000 | 7000 | ND–31 | 5.0 |
| Phenacetin | ND–68,300 | 2300 | ND–0.32 | 0.15 | ND–2170 | 10 | ND–0.67 | 0.30 |
| Caffeine | ND–D | D | ND–128 | 2.0 | ND–15,000 | 4500 | ND–89 | 1.9 |
| Naproxen | ND–59,000 | 2300 | ND–15 | 0.98 | ND–2380 | 580 | ND–D | D |
| Triclosan | ND–5000 | 2000 | ND–79 | 3.3 | ND–20,000 | 2500 | ND–43 | 4.9 |
| Meclofenamic acid | ND–23,800 | 4201 | ND–4.0 | 0.98 | ND | ND | ND–2.8 | 1.0 |
| Diclofenac | ND–1010 | 370 | ND–3.75 | 0.91 | ND | ND | ND–8.0 | 1.9 |
| Carbamazepine | ND–D | D | ND–12 | 1.3 | ND–D | D | ND–4.7 | 2.0 |
| Chloramphenicol | ND–D | D | ND–5.0 | 0.54 | ND–D | D | ND–19 | 5.0 |
| Target Analytes | Wastewater Treatment Plants | |||
|---|---|---|---|---|
| Wastewater (ng/L) | Biosolids (ng/g) | |||
| No. of Samples = 16 | No. of Samples = 16 | |||
| Range | Median | Range | Median | |
| Salicylic acid | ND–66,000 | 820 | ND–55 | 2.3 |
| Acetylsalicylic acid | ND | ND | ND–221 | 29 |
| Ibuprofen | ND–17,600 | 3000 | ND–27 | 2.5 |
| Propylparaben | ND–12,000 | 6200 | ND–28 | 4.0 |
| Phenacetin | ND–19,500 | 10 | ND–40 | 5.0 |
| Caffeine | D–15,000 | 7000 | ND–173 | 24 |
| Naproxen | ND–D | D | ND–13 | 3.0 |
| Triclosan | ND–30,000 | 5000 | ND–3.2 | 0.94 |
| Meclofenamic acid | ND–2380 | 580 | ND–86 | 8.0 |
| Diclofenac | ND–10,200 | 250 | ND–206 | 12 |
| Carbamazepine | ND–D | D | ND–5.5 | 1.1 |
| Chloramphenicol | ND–D | D | ND–16 | 5.3 |
| Target Analytes | Schymanski Assessment Level [43] | River Water | Wastewater | Sediments | Biosolids |
|---|---|---|---|---|---|
| Acetaminophen | 1 | Detected | Detected | Detected | Detected |
| Ketoprofen | 1 | Detected | Detected | Detected | Detected |
| Sulfamethoxazole | 1 | Detected | - | - | - |
| Nalidixic acid | 1 | Detected | Detected | - | - |
| Sulfamethazine | 1 | - | - | - | Detected |
| Chlorpromazine | 1 | - | - | - | Detected |
| Clozapine | 1 | - | - | - | Detected |
| Procaine | 1 | - | - | - | Detected |
| Cocaine | 1 | - | Detected | - | - |
| Methamphetamine | 1 | Detected | Detected | Detected | Detected |
| Morphine | 1 | Detected | Detected | Detected | Detected |
| 2-phenylindolizine | 1 | - | Detected | - | Detected |
| Lactose | 1 | - | - | - | Detected |
| Procainamide | 1 | - | - | - | - |
| Suspect Analytes | Schymanski Assessment Level [43] | Chemical Formula | Fragment Pattern m/z | River Water | Wastewater | Sediments | Biosolids | Similarity Index |
|---|---|---|---|---|---|---|---|---|
| Clofibric acid | 2 | C10H11ClO3 | 39, 99, 128, 130, 214 | Detected | Detected | - | Detected | 75 |
| Codeine | 2 | C18H21NO3 | 115, 162, 214, 229, 299 | - | Detected | - | - | 80 |
| Oxazepam | 2 | C15H11ClN2O2 | 77, 205, 233, 239, 268 | - | Detected | - | - | 76 |
| Trimethoprim | 1 | C14H18N4O3 | 123, 200, 243, 259, | - | - | - | Detected | 91 |
| Nicotine | 2 | C10H14N2 | 42, 84, 161 | - | - | - | Detected | 75 |
| Amphetamine | 1 | C9H13N | 44, 65, 91, 120 | - | Detected | - | - | 95 |
| Benzoylecgonine | 1 | C16H19NO4 | 77, 82, 94, 124, 138, | - | Detected | - | - | 95 |
| Benzocaine | 2 | C16H11NO2 | 65, 92, 120, 137, 165 | - | - | Detected | - | 80 |
| Cotinine | 2 | C16H19NO4 | 98, 176 | - | - | - | - | 79 |
| Propranolol | 2 | C16H21NO2 | 30, 72, 115, 144, 331 | Detected | - | - | - | 80 |
| Azelaic acid | 2 | C9H14Cl2O2 | 55, 83, 124, 152, 367 | Detected | - | - | - | 82 |
| 4-Oxoisophorone | 2 | C9H12O2 | 39, 68, 96, 152 | - | Detected | - | - | 80 |
| Musk xylene | 2 | C12H15N3O6 | 43, 282 | - | - | Detected | - | 80 |
| 2-Pyrrolidone | 2 | C4H7NO | 73, 142, 157 | - | Detected | - | 81 | |
| 2-Phenoxyethanol | 2 | C8H10O2 | 77, 94, 138 | Detected | - | - | 79 |
| Non-Target Analytes | Source or Origin | Chemical Formula | Fragment Pattern m/z | River Water | Wastewater | Sediments | Biosolids | Similarity Index |
|---|---|---|---|---|---|---|---|---|
| Butyldiglycol | Paints | C8H18O3 | 57, 100, 132 | Detected | Detected | - | - | 79 |
| 2-propanol, 1-[1-methyl-2-(properyloxy)ether | - | C9H18O3 | 59, 103, 174 | detected | - | - | - | 80 |
| Nicotinic acid | Vitamin | C6H5NO2 | 51, 91, 136, 195 | - | Detected | - | - | 72 |
| Phenylmalonic acid | - | C9H8O4 | 69, 91, 136 | - | - | 88 | ||
| 2-ethyl-3-hydroxyhexyl 2-methyl propanoate | - | C12H24O3 | 71, 95, 99, 143, 174 | Detected | Detected | - | - | 79 |
| Oxindole | Human metabolite | C8H7NO | 78, 104, 133 | Detected | Detected | - | - | 81 |
| 2,6-Dimethylphenyl isocyanide | Cyanobacteria | C9H9NO | 51, 118, 147 | Detected | Detected | - | - | 60 |
| Obtusifoliol | Hormone | C30H50O | 75, 215, 355, 370, 429 | - | Detected | - | - | 75 |
| Cholesterol | Hormone | C27H46O | 129, 329, 353, 368, 458 | - | Detected | - | - | 88 |
| Metolachlor | Herbicide, pesticide | C15H22ClNO2 | 91, 162, 238 | Detected | - | - | 95 | |
| Bisphenol A | Plasticizer | C15H16O2 | 119, 213, 228, 372 | - | - | - | - | 96 |
| Triethyl phosphate | Plasticizer | C6H15O4P | 81, 99, 109, 155, 182 | Detected | Detected | Detected | Detected | 81 |
| Triethyl citrate | Plasticizer | C12H20O7 | 115, 157, 203, 348 | Detected | Detected | Detected | Detected | 82 |
| Oxybenzone | UV filters | C14H12O3 | 51, 77, 151, 227, 300 | Detected | - | Detected | 80 | |
| Tris(2-chloroethyl) phosphate | Flame retardant | C6H12Cl3O4P | 63, 143, 205, 249, 253 | Detected | - | - | - | 79 |
| Triphenyl phosphate | Flame retardant | C18H15O4P | 77,169, 233, 233, 326 | - | - | - | Detected | 90 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gumbi, B.P.; Moodley, B.; Birungi, G.; Ndungu, P.G. Target, Suspect and Non-Target Screening of Silylated Derivatives of Polar Compounds Based on Single Ion Monitoring GC-MS. Int. J. Environ. Res. Public Health 2019, 16, 4022. https://doi.org/10.3390/ijerph16204022
Gumbi BP, Moodley B, Birungi G, Ndungu PG. Target, Suspect and Non-Target Screening of Silylated Derivatives of Polar Compounds Based on Single Ion Monitoring GC-MS. International Journal of Environmental Research and Public Health. 2019; 16(20):4022. https://doi.org/10.3390/ijerph16204022
Chicago/Turabian StyleGumbi, Bhekumuzi Prince, Brenda Moodley, Grace Birungi, and Patrick Gathura Ndungu. 2019. "Target, Suspect and Non-Target Screening of Silylated Derivatives of Polar Compounds Based on Single Ion Monitoring GC-MS" International Journal of Environmental Research and Public Health 16, no. 20: 4022. https://doi.org/10.3390/ijerph16204022
APA StyleGumbi, B. P., Moodley, B., Birungi, G., & Ndungu, P. G. (2019). Target, Suspect and Non-Target Screening of Silylated Derivatives of Polar Compounds Based on Single Ion Monitoring GC-MS. International Journal of Environmental Research and Public Health, 16(20), 4022. https://doi.org/10.3390/ijerph16204022





