Feasibility and Preliminary Efficacy of an m-Health Intervention Targeting Physical Activity, Diet, and Sleep Quality in Shift-Workers
Abstract
1. Introduction
- To evaluate the feasibility of an intervention aimed at improving physical activity, diet, and sleep quality not tailored to shift-workers (Move, Eat, and Sleep [29]) in a shift-worker population and receive feedback on improvements to increase acceptability.
- To estimate the effect of the shift-worker Move, Eat, and Sleep intervention on improving physical activity, diet quality, and sleep quality in shift-workers.
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Intervention Development
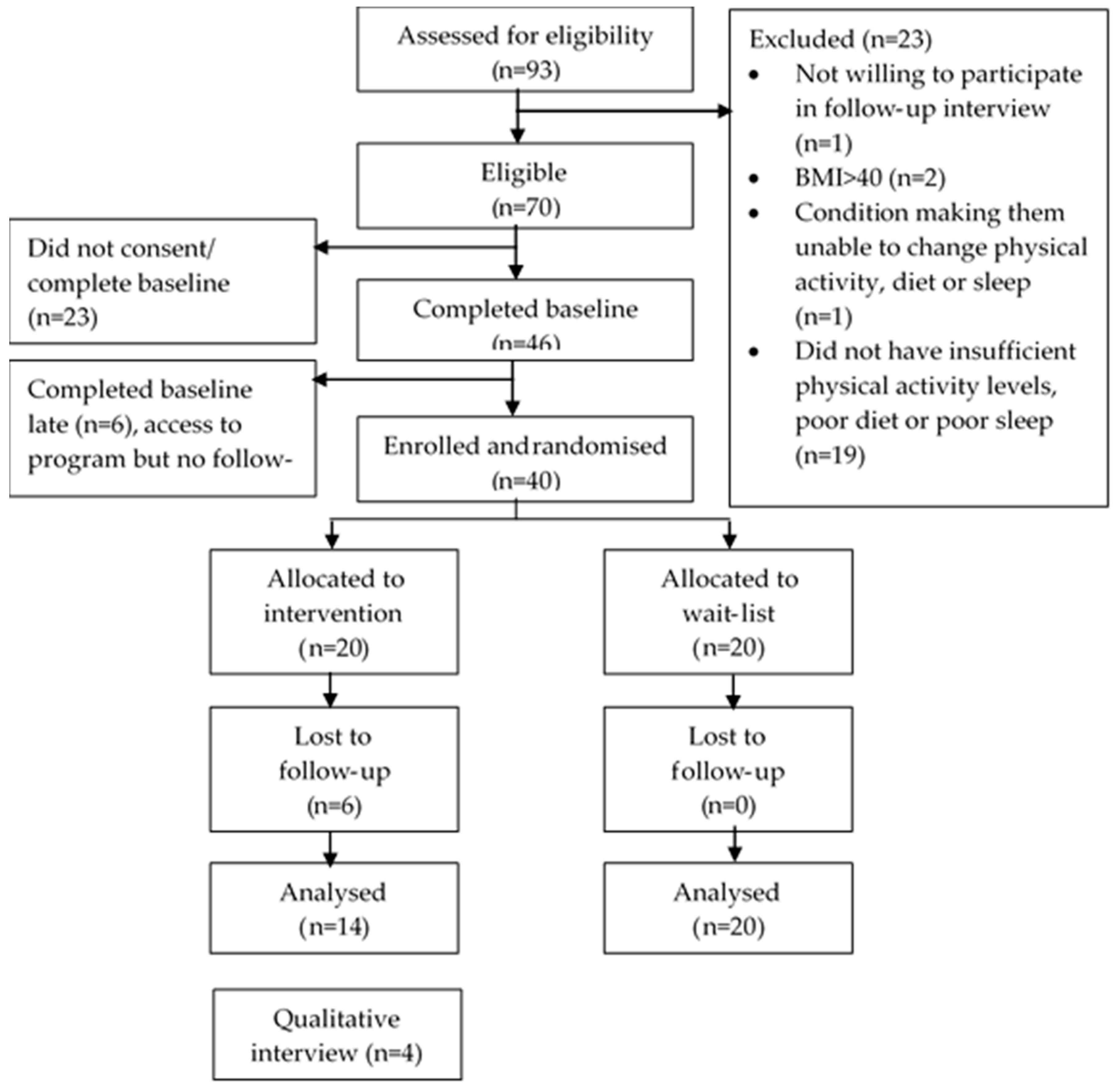
2.3. Participants and Recruitment
2.4. Wait-List Group
2.5. Intervention Group
2.5.1. Balanced App
2.5.2. Weekly Summary Reports
2.5.3. The Shift-Worker Move, Eat, and Sleep Handbook
2.5.4. Weekly SMS
2.6. Data Collection
2.7. Feasibility Measures
2.8. Implementation Outcomes
2.9. Estimation of Treatment Effect
2.10. Sample Size
2.11. Qualitative Analysis
2.12. Statistical Analysis
3. Results
3.1. Feasibility of Research Procedures
3.1.1. Recruitment
3.1.2. Reminders
3.1.3. Capacity and Resources
3.2. Implementation Outcomes
3.2.1. User Engagement and Retention
3.2.2. Acceptability of Overall Intervention and Its Components
3.2.3. Qualitative Interviews
3.3. Estimation of Treatment Effect
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Agnes, P.T.; Isabella, B.; Jorge, C.; Oscar, V.L.; Greet, V.; Aleksandra, W.; Mathijn, W. Sixth European Working Conditions Survey—Overview Report; Eurofound: Brussels, Belgium, 2016. [Google Scholar]
- Australian Bureau of Statistics. Working Time Arrangements, Australia, November 2012; ABS: Canberra, Australia, 2012.
- Wang, F.; Zhang, L.; Zhang, Y.; Zhang, B.; He, Y.; Xie, S.; Li, M.; Miao, X.; Chan, E.Y.; Tang, J.L.; et al. Meta-analysis on night shift work and risk of metabolic syndrome. Obes. Rev. 2014, 15, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Yang, C.; Tong, X.; Sun, H.; Cong, Y.; Yin, X.; Li, L.; Cao, S.; Dong, X.; Gong, Y.; et al. Shift work and diabetes mellitus: A meta-analysis of observational studies. Occup. Environ. Med. 2015, 72, 72. [Google Scholar] [CrossRef] [PubMed]
- Vogel, M.; Braungardt, T.; Meyer, W.; Schneider, W. The effects of shift work on physical and mental health. J. Neural Transm. 2012, 119, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Shan, Z.; Li, Y.; Zong, G.; Guo, Y.; Li, J.; Manson, J.E.; Hu, F.B.; Willett, W.C.; Schernhammer, E.S.; Bhupathiraju, S.N. Rotating night shift work and adherence to unhealthy lifestyle in predicting risk of type 2 diabetes: Results from two large US cohorts of female nurses. BMJ 2018, 363, k4641. [Google Scholar] [CrossRef] [PubMed]
- Torquati, L.; Mielke, G.I.; Brown, W.J.; Kolbe-Alexander, T. Shift work and the risk of cardiovascular disease. A systematic review and meta-analysis including dose-response relationship. Scand. J. Work Environ. Health 2018. [Google Scholar] [CrossRef]
- McEwen, B.S. Stress, adaptation, and disease. Allostasis and allostatic load. Ann. N. Y. Acad. Sci. 1998, 840, 33–44. [Google Scholar] [CrossRef]
- Winkler, M.R.; Mason, S.; Laska, M.N.; Christoph, M.J.; Neumark-Sztainer, D. Does non-standard work mean non-standard health? Exploring links between non-standard work schedules, health behavior, and well-being. SSM Popul. Health 2018, 4, 135–143. [Google Scholar] [CrossRef]
- Dixon, J.; Carey, G.; Strazdins, L.; Banwell, C.; Woodman, D.; Burgess, J.; Bittman, M.; Venn, D.; Sargent, G. Contemporary contestations over working time: Time for health to weigh in. BMC Public Health 2014, 14, 1068. [Google Scholar] [CrossRef][Green Version]
- Niu, S.F.; Chung, M.H.; Chen, C.H.; Hegney, D.; O’Brien, A.; Chou, K.R. The effect of shift rotation on employee cortisol profile, sleep quality, fatigue, and attention level: A systematic review. J. Nurs. Res. 2011, 19, 68–81. [Google Scholar] [CrossRef]
- Åkerstedt, T.; Wright, K.P. Sleep Loss and Fatigue in Shift Work and Shift Work Disorder. Sleep Med. Clin. 2009, 4, 257–271. [Google Scholar] [CrossRef]
- St-Onge, M.P.; Grandner, M.A.; Brown, D.; Conroy, M.B.; Jean-Louis, G.; Coons, M.; Bhatt, D.L. Sleep Duration and Quality: Impact on Lifestyle Behaviors and Cardiometabolic Health: A Scientific Statement From the American Heart Association. Circulation 2016, 134, e367–e386. [Google Scholar] [CrossRef] [PubMed]
- Kecklund, G.; Axelsson, J. Health consequences of shift work and insufficient sleep. BMJ 2016, 355, 5210. [Google Scholar] [CrossRef] [PubMed]
- Rüger, M.; Scheer, F.A.J.L. Effects of circadian disruption on the cardiometabolic system. Rev. Endocr. Metab. Disord. 2009, 10, 245–260. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D. The effects of shift work on free-living physical activity and sedentary behavior. Prev. Med. 2015, 76, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Bonnell, E.K.; Huggins, C.E.; Huggins, C.T.; McCaffrey, T.A.; Palermo, C.; Bonham, M.P. Influences on Dietary Choices during Day versus Night Shift in Shift Workers: A Mixed Methods Study. Nutrients 2017, 9, 193. [Google Scholar] [CrossRef] [PubMed]
- van de Langenberg, D.; Vlaanderen, J.J.; Dolle, M.E.T.; Rookus, M.A.; van Kerkhof, L.W.M.; Vermeulen, R.C.H. Diet, Physical Activity, and Daylight Exposure Patterns in Night-Shift Workers and Day Workers. Ann. Work Expo. Health 2019, 63, 9–21. [Google Scholar] [CrossRef]
- Antunes, L.C.; Levandovski, R.; Dantas, G.; Caumo, W.; Hidalgo, M.P. Obesity and shift work: Chronobiological aspects. Nutr. Res. Rev. 2010, 23, 155–168. [Google Scholar] [CrossRef]
- de Assis, M.A.; Nahas, M.V.; Bellisle, F.; Kupek, E. Meals, snacks and food choices in Brazilian shift workers with high energy expenditure. J. Hum. Nutr. Diet. 2003, 16, 283–289. [Google Scholar] [CrossRef]
- Johnston, J.D.; Ordovás, J.M.; Scheer, F.A.; Turek, F.W. Circadian Rhythms, Metabolism, and Chrononutrition in Rodents and Humans. Adv. Nutr. 2016, 7, 399–406. [Google Scholar] [CrossRef]
- McHill, A.W.; Wright, K.P., Jr. Role of sleep and circadian disruption on energy expenditure and in metabolic predisposition to human obesity and metabolic disease. Obes. Rev. 2017, 18, 15–24. [Google Scholar] [CrossRef]
- Nea, F.M.; Kearney, J.; Livingstone, M.B.E.; Pourshahidi, L.K.; Corish, C.A. Dietary and lifestyle habits and the associated health risks in shift workers. Nutr. Res. Rev. 2015, 28, 143–166. [Google Scholar] [CrossRef] [PubMed]
- Kolbe-Alexander, T.L.; Gomersall, S.; Clark, B.; Torquati, L.; Pavey, T.; Brown, W.J. A hard day’s night: Time use in shift workers. BMC Public Health 2019, 19, 452. [Google Scholar] [CrossRef] [PubMed]
- Demou, E.; MacLean, A.; Cheripelli, L.J.; Hunt, K.; Gray, C.M. Group-based healthy lifestyle workplace interventions for shift workers: A systematic review. Scand. J. Work Environ. Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Papantoniou, K.; Vetter, C.; Schernhammer, E.S. Shift work practices and opportunities for intervention. Occup. Environ. Med. 2017, 74, 2. [Google Scholar] [CrossRef] [PubMed]
- Neil-Sztramko, S.E.; Pahwa, M.; Demers, P.A.; Gotay, C.C. Health-related interventions among night shift workers: A critical review of the literature. Scand. J. Work Environ. Health 2014, 40, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Nea, F.M.; Pourshahidi, L.K.; Kearney, J.; Livingstone, M.B.E.; Bassul, C.; Corish, C.A. A Qualitative Exploration of the Shift Work Experience: The Perceived Barriers and Facilitators to a Healthier Lifestyle and the Role of the Workplace Environment. J. Occup. Environ. Med. 2017, 59, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Duncan, M.J.; Brown, W.J.; Burrows, T.L.; Collins, C.E.; Fenton, S.; Glozier, N.; Kolt, G.S.; Morgan, P.J.; Hensley, M.; Holliday, E.G.; et al. Examining the efficacy of a multicomponent m-Health physical activity, diet and sleep intervention for weight loss in overweight and obese adults: Randomised controlled trial protocol. BMJ Open 2018, 8, e026179. [Google Scholar] [CrossRef]
- Eldridge, S.M.; Chan, C.L.; Campbell, M.J.; Bond, C.M.; Hopewell, S.; Thabane, L.; Lancaster, G.A. CONSORT 2010 statement: Extension to randomised pilot and feasibility trials. BMJ 2016, 355, 5239. [Google Scholar] [CrossRef]
- Murawski, B.; Plotnikoff, R.C.; Rayward, A.T.; Vandelanotte, C.; Brown, W.J.; Duncan, M.J. Randomised controlled trial using a theory-based m-health intervention to improve physical activity and sleep health in adults: The Synergy Study protocol. BMJ Open 2018, 8, e018997. [Google Scholar] [CrossRef]
- Rayward, A.T.; Murawski, B.; Plotnikoff, R.C.; Vandelanotte, C.; Brown, W.J.; Holliday, E.G.; Duncan, M.J. A randomised controlled trial to test the efficacy of an m-health delivered physical activity and sleep intervention to improve sleep quality in middle-aged adults: The Refresh Study Protocol. Contemp. Clin. Trials 2018, 73, 36–50. [Google Scholar] [CrossRef]
- Murawski, B.; Plotnikoff, R.C.; Rayward, A.T.; Oldmeadow, C.; Vandelanotte, C.; Brown, W.J.; Duncan, M.J. Efficacy of an m-Health Physical Activity and Sleep Health Intervention for Adults: A Randomized Waitlist-Controlled Trial. Am. J. Prev. Med. 2019, 57, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Abraham, C.; Michie, S. A taxonomy of behavior change techniques used in interventions. Health Psychol. 2008, 27, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Bandura, A. The primacy of self-regulation in health promotion. Appl. Psychol. Int. Rev. Psychol. Appl. Rev. Int. 2005, 54, 245–254. [Google Scholar] [CrossRef]
- Harkin, B.; Webb, T.L.; Chang, B.P.I.; Prestwich, A.; Conner, M.; Kellar, I.; Benn, Y.; Sheeran, B. Does monitoring goal progress promote goal attainment? A meta-analysis of the experimental evidence. Psychol. Bull. 2016, 142, 198–229. [Google Scholar] [CrossRef] [PubMed]
- Michie, S.; Ashford, S.; Sniehotta, F.F.; Dombrowski, S.U.; Bishop, A.; French, D.P. A refined taxonomy of behaviour change techniques to help people change their physical activity and healthy eating behaviours: The CALO-RE taxonomy. Psychol. Health 2011, 26, 1479–1498. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. The Active Australia Survey: A Guide and Manual for Implementation, Analysis and Reporting; AIHW: Canberra, Austrilia, 2003.
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Duncan, M.J.; Vandelanotte, C.; Trost, S.G.; Rebar, A.L.; Rogers, N.; Burton, N.W.; Murawski, B.; Rayward, A.; Fenton, S.; Brown, W.J. Balanced: A randomised trial examining the efficacy of two self-monitoring methods for an app-based multi-behaviour intervention to improve physical activity, sitting and sleep in adults. BMC Public Health 2016, 16, 670. [Google Scholar] [CrossRef]
- Proctor, E.; Silmere, H.; Raghavan, R.; Hovmand, P.; Aarons, G.; Bunger, A.; Griffey, R.; Hensley, M. Outcomes for Implementation Research: Conceptual Distinctions, Measurement Challenges, and Research Agenda. Adm. Policy Ment. Health 2011, 38, 65–76. [Google Scholar] [CrossRef]
- Jordan, P.; Jordan, J.W.; McClelland, I. Usability Evaluation in Industry; Taylor & Francis: London, UK, 1996. [Google Scholar]
- Collins, C.E.; Burrows, T.L.; Rollo, M.E.; Boggess, M.M.; Watson, J.F.; Guest, M.; Duncanson, K.; Pezdirc, K.; Hutchesson, M.J. The comparative validity and reproducibility of a diet quality index for adults: The Australian Recommended Food Score. Nutrients 2015, 7, 785–798. [Google Scholar] [CrossRef]
- National Health and Medical Research Council. Australian Dietary Guidelines; National Health and Medical Research Council: Canberra, Austrilia, 2013.
- Mollayeva, T.; Thurairajah, P.; Burton, K.; Mollayeva, S.; Shapiro, C.M.; Colantonio, A. The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: A systematic review and meta-analysis. Sleep Med. Rev. 2016, 25, 52–73. [Google Scholar] [CrossRef]
- Lohse, B.; Satter, E.; Horacek, T.; Gebreselassie, T.; Oakland, M.J. Measuring eating competence: Psychometric properties and validity of the ecSatter Inventory. J. Nutr. Educ. Behav. 2007, 39, S154–S166. [Google Scholar] [CrossRef] [PubMed]
- Netzer, N.C.; Stoohs, R.A.; Netzer, C.M.; Clark, K.; Strohl, K.P. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann. Intern. Med. 1999, 131, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Mastin, D.F.; Bryson, J.; Corwyn, R. Assessment of sleep hygiene using the Sleep Hygiene Index. J. Behav. Med. 2006, 29, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. CHAPTER 10—Technical Appendix: Computational Procedures. In Statistical Power Analysis for the Behavioral Sciences; Cohen, J., Ed.; Academic Press: New York, NY, USA, 1977; pp. 455–463. [Google Scholar] [CrossRef]
- Lee, M.; Lee, H.; Kim, Y.; Kim, J.; Cho, M.; Jang, J.; Jang, H. Mobile App-Based Health Promotion Programs: A Systematic Review of the Literature. Int. J. Environ. Res. Public Health 2018, 15, 2838. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, S.A.; Williams, A.J.; Morrissey, K.; Price, L.; Harrison, J. Mobile health interventions to promote physical activity and reduce sedentary behaviour in the workplace: A systematic review. Digit. Health 2019, 5, 2055207619839883. [Google Scholar] [CrossRef] [PubMed]
- Schoeppe, S.; Alley, S.; Van Lippevelde, W.; Bray, N.A.; Williams, S.L.; Duncan, M.J.; Vandelanotte, C. Efficacy of interventions that use apps to improve diet, physical activity and sedentary behaviour: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2016, 13, 127. [Google Scholar] [CrossRef] [PubMed]
- Yardley, L.; Spring, B.J.; Riper, H.; Morrison, L.G.; Crane, D.H.; Curtis, K.; Merchant, G.C.; Naughton, F.; Blandford, A. Understanding and Promoting Effective Engagement With Digital Behavior Change Interventions. Am. J. Prev. Med. 2016, 51, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Edwards, E.A.; Lumsden, J.; Rivas, C.; Steed, L.; Edwards, L.A.; Thiyagarajan, A.; Sohanpal, R.; Caton, H.; Griffiths, C.J.; Munafò, M.R.; et al. Gamification for health promotion: Systematic review of behaviour change techniques in smartphone apps. BMJ Open 2016, 6, e012447. [Google Scholar] [CrossRef]
- Tong, H.L.; Laranjo, L. The use of social features in mobile health interventions to promote physical activity: A systematic review. Digit. Med. 2018, 1, 43. [Google Scholar] [CrossRef]
- Australian Government. Australias Physical Activity and Sedentary Behaviour Guidelines for Adults (16–64 Years); Department of Health: Canberra, Austrilia, 2014.
- Australian Bureau of Statistics. Australian Health Survey: Physical Activity, 2011–2012. Available online: http://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/4364.0.55.0042011-12?OpenDocument (accessed on 20 August 2019).
- Murawski, B.; Wade, L.; Plotnikoff, R.C.; Lubans, D.R.; Duncan, M.J. A systematic review and meta-analysis of cognitive and behavioral interventions to improve sleep health in adults without sleep disorders. Sleep Med. Rev. 2018, 40, 160–169. [Google Scholar] [CrossRef]
- Seyffert, M.; Lagisetty, P.; Landgraf, J.; Chopra, V.; Pfeiffer, P.N.; Conte, M.L.; Rogers, M.A.M. Internet-Delivered Cognitive Behavioral Therapy to Treat Insomnia: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0149139. [Google Scholar] [CrossRef] [PubMed]
- Burton, J. WHO Healthy Workplace Framework and Model: Background Document and Supporting Literature and Practices Organisation; World Health Organisation: Geneva, Switzerland, 2010. [Google Scholar]
| Variable and Measurement Scale/Category | Intervention | Wait-List | Total | |
|---|---|---|---|---|
| Mean (SD) | ||||
| Age | Years | 34.9 (9.2) | 36.6 (10.1) | 35.7 (9.5) |
| Education | Years | 16.7 (2.5) | 15.4 (3.2) | 16.0 (2.9) |
| Body mass index | Weight, kg/height, m2 | 26.3 (4.3) | 29.9 (5.4) | 28.1 (5.2) |
| Shifts last week | Count | 4.1 (1.2) | 4.2 (1.6) | 4.2 (1.4) |
| Length of shifts | Hours | 10.0 (2.1) | 9.9 (2.0) | 9.9 (2.0) |
| Depression Anxiety and Stress Scale-21 | Depression (0–42) 1 | 9.6 (9.3) | 8.0 (7.7) | 8.8 (8.5) |
| Anxiety (0–42) 2 | 4.7 (4.6) | 5.5 (5.8) | 5.1 (5.2) | |
| Stress (0–42) 3 | 12.0 (8.0) | 13.8 (7.3) | 12.9 (7.6) | |
| Count (%) | ||||
| Sex | Female | 12 (60) | 9 (45) | 21 (52.5) |
| Male | 8 (40) | 11 (55) | 19 (47.5) | |
| Ethnic background | Caucasian | 18 (90) | 18 (90 | 36 (90) |
| Aboriginal, Torres Strait, or Pacific Islander | - | 1 (5) | 1 (2.5) | |
| Asian | 1 (5) | 1 (5) | 2 (5) | |
| Middle Eastern | 1 (5) | - | 1 (2.5) | |
| Marital status | Partnered | 15 (75) | 14 (70) | 29 (72.5) |
| Not partnered | 5 (25) | 6 (30) | 11 (27.5) | |
| Individual income per annum (gross) | ≤AUD$70 k/annum | 8 (30) | 9 (45) | 16 (40) |
| >AUD$70 k/annum | 12 (70) | 10 (50) | 13 (57.5) | |
| Do not know | - | 1 (5) | 1 (2.5) | |
| Employment status | Full time | 12 (60) | 14 (70) | 26 (65) |
| Part time | 5 (25) | 5 (25) | 11 (27.5) | |
| Causal | 1 (5) | - | 1 (2.5) | |
| Other | 1 (5) | 1 (5) | 2 (5) | |
| Live away from home for work | Yes | 1 (5) | 3 (15) | 4 (10) |
| No | 19 (95) | 17 (85) | 36 (90) | |
| Work pattern | Mainly night | 2 (10) | - | 2 (5) |
| Rotating | 13 (65) | 14 (70) | 27 (67.5) | |
| Some days, afternoons, and nights | 4 (20) | 5 (25) | 9 (22.5) | |
| Some days and afternoons/mainly afternoons | 1 (5) | 1 (5) | 2 (5) | |
| Length of time as shift-worker | ≤2 years | 4 (20) | 2 (10) | 6 (20) |
| 3–6 years | 4 (20) | 7 (35) | 11 (27.5) | |
| ≥7 years | 10 (50) | 11 (45) | 21 (52.5) | |
| Hours worked last week (total) | ≤30 h | 4 (20) | 3 (15) | 7 (17.5) |
| 31 to 50 h | 13 (65) | 15 (75) | 28 (70) | |
| >50 h | 3 (15) | 2 (10) | 5 (12.5) | |
| Berlin questionnaire 4 | High risk of sleep apnea | 6 (30) | 9 (45) | 15 (37.5) |
| Low risk sleep apnea | 14 (70) | 11 (55) | 25 (62.5) | |
| Fallen asleep while driving | Yes, 1–2 times per month | 3 (15) | 2 (10) | 5 (12.5) |
| Yes, but “nearly never” | 5 (25) | 4 (20) | 9 (22.5) | |
| Not fallen asleep | 12 (60) | 14 (70) | 26 (65) | |
| Chronic disease | Yes | 7 (35) | 10 (50) | 17 (42.5) |
| No | 13 (65) | 10 (50) | 23 (57.5) | |
| Self-rated health | Poor to fair | 8 (40) | 7 (35) | 15 (37.5) |
| Very good to excellent | 12 (60) | 13 (65) | 25 (62.5) | |
| Variable and Measurement Scale/Category | Intervention (n = 20) | Wait-List (n = 20) | Total (n = 40) | |
|---|---|---|---|---|
| Mean (SD) | ||||
| Average moderate to vigorous physical activity (MVPA) 1 | Average minutes | 1462 (300) | 1198 (450) | 1330 (401) |
| Australian Recommended Food Score (ARFS) | Average score | 31.2 (7.0) | 32.7 (7.2) | 31.9 (7.0) |
| Take-away (breakfast, lunch, dinner, and/or snacks) | Average times per week | 4.2 (2.7) | 5.7 (2.7) | 4.9 (2.8) |
| Discretionary foods 2 | Average score per item 3 | 2.5 (1.1) | 2.9 (1.0) | 2.7 (1.0) |
| Total score 4 | 12.6 (5.3) | 14.3 (4.9) | 13.4 (5.1) | |
| Sitting time (hours) 5 | Average per day | 6.4 (3.7) | 7.7 (4.6) | 7.1 (4.2) |
| Pittsburgh Sleep Quality Index score | Total score | 7.5 (4.3) | 7.9 (3.3) | 7.7 (3.8) |
| Sleep Hygiene Index 6 | Total score (0–60) | 39.5 (6.4) | 36.4 (5.9) | 37.9 (6.3) |
| Eating competence score 8 | Total score (0–48) | 27.4 (7.3) | 25.1 (8.8) | 26.2 (8.1) |
| n (%) | ||||
| Meet physical activity recommendations 7 | No | 11 (55) | 11 (55) | 22 (55) |
| Yes | 9 (45) | 9(45) | 18 (45) | |
| Sitting time category | >8 h per day | 8 (40) | 9 (45) | 17 (42.5) |
| ≤8 h per day | 12 (60) | 11 (55) | 23 (57.5) | |
| Eating competence category 9 | Low eating competence | 29 (72.5) | 25 (73.5) | 54 (73.0) |
| Eating competent | 11 (27.5) | 9 (26.5) | 20 (27.0) | |
| Diet quality category (ARFS categories) | Needs work (0–33) | 11 (57.9) | 9 (50.0) | 20 (54.5) |
| Getting there (34–38) | 5 (26.3) | 6 (33.3) | 11 (29.7) | |
| Excellent (39–46) | 3 (16.7) | 3 (15.8) | 6 (16.2) | |
| Outstanding (47+) | - | - | - | |
| Pittsburgh Sleep Quality Index category 10 | Poor sleep quality | 14 (70) | 17 (85) | 31 (77.5) |
| Good sleep quality | 6 (30) | 3 (15) | 9 (22.5) | |
| Physical Activity: How Useful Was Shift-Worker Move, Eat, and Sleep in Helping You (n = 13) | Slightly to Moderately Useful (n (%)) |
|---|---|
| Increase your confidence for engaging in regular physical activity over the past 4 weeks? | 10 (76.9) |
| Overcome barriers to participating in physical activity over the past 4 weeks? | 10 (76.9) |
| To plan for physical activity over the past 4 weeks? | 9 (69.2) |
| To stay motivated to participate in physical activity over the past 4 weeks? | 10 (76.9) |
| Food: How useful was shift-worker Move, Eat, and Sleep in helping you (n = 13) | |
| Increase your confidence for engaging in healthy food habits over the past 4 weeks? | 11 (84.6) |
| Overcome barriers to engaging in healthy food habits over the past 4 weeks? | 9 (69.2) |
| To plan for healthy food habits over the past 4 weeks? | 9 (69.2) |
| To stay motivated to engage in healthy food habits over the past 4 weeks? | 10 (76.9) |
| Sleep: How useful was shift-worker Move, Eat, and Sleep in helping you (n = 13) | |
| Feel confident in prioritizing my sleep needs in the past 4 weeks? | 6 (46.2) |
| Overcome barriers to healthy sleep habits in the past 4 weeks? | 10 (76.9) |
| Plan for healthy sleep habits over the past 4 weeks? | 11 (84.6) |
| Stay motivated to engage in healthy sleep habits over the past 4 weeks? | 10 (76.9) |
| Handbook items (n = 13 as n = 1 did not read any of the handbook) | Agree to Strongly Agree |
| The handbook was useful | 9 (69.2) |
| The physical activity information was useful | 9 (69.2) |
| The physical activity action plan was useful | 6 (46.2) |
| The information on food variety was useful | 9 (69.2) |
| The tools for increasing food variety were useful | 8 (61.5) |
| The hunger/fullness awareness was useful | 7 (53.9) |
| The hunger/fullness diary was useful | 6 (46.2) |
| The information on food planning was useful | 4 (30.8) |
| The meal planning tool was useful | 4 (30.8) |
| The action plan for eating habits and food variety tool was useful | 5 (38.5) |
| The sleep information was useful | 8 (61.5) |
| The action plan for sleep was useful | 5 (38.5) |
| The relaxation and stress management section was useful | 7 (58.3) |
| The goal-setting information was useful | 7 (53.9) |
| App items (n = 12 as n = 2 did not download the app) | |
| The Balanced App was useful | 7 (58.3) |
| Physical activity section of app was useful | 6 (50.0) |
| It was easy to personalize my activity goals | 8 (66.7) |
| It was easy to track my physical activity | 7 (58.3) |
| Food section of app was useful | 6 (50.0) |
| It was easy to personalize my food goals | 7 (58.3) |
| It was easy to track my food goals | 7 (58.3) |
| Sleep section of app was useful | 5 (41.7) |
| It was easy to personalize my sleep goals | 7 (58.3) |
| It was easy to track my sleep goals | 7 (58.3) |
| Progress graph in app useful | 3 (25.0) |
| Resources in app were useful | 4 (33.3) |
| Other components (n = 13) | |
| The weekly reports were useful | 7 (53.8) |
| The weekly SMSs were useful | 9 (69.2) |
| The multiple parts of the intervention were not overwhelming | 10 (76.9) |
| The program was able to be personalized enough to match my goals | 9 (69.2) |
| There was enough contact with the study coordinators | 8 (61.5) |
| System Usability Scale | |
| System Usability Scale score (0–100 points) (n = 13) | 62.7 (12.7) |
| Baseline | Follow-Up | Mean Change from Baseline (95% CI) | Mean Difference between Groups (95% CI) | Group × Time p-Value | Effect Size (Cohen’s d) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Outcomes | Wait-List (n = 20) | Intervention (n = 20) | Wait-List (n = 20) | Intervention (n = 14) | Wait-List | Intervention | |||
| Moderate to vigorous physical activity minutes 1 | 1120 (390 to 1680) | 1462 (660 to 1680) | 1273 (360 to 1680) | 1248 (330 to 1680) | 75 (−144 to 294) | −232 (−480 to 16) | −307 (−638 to 24) | 0.069 | 0.67 |
| Australian Recommended Food Score, total score | 33.2 (29.7 to 36.6) | 31.0 (27.6 to 34.5) | 33.2 (29.7 to 36.6) | 35.6 (31.6 to 39.5) | 0.01 (−2.8 to 2.8) | 4.5 (1.1 to 7.9) | 4.5 (0.1 to 8.9) | 0.047 | 0.76 |
| Pittsburgh Sleep Quality Index, total score | 7.9 (3 to 15) | 7.5 (2 to 18) | 7.1 (3 to 13) | 7 (2 to 13) | −0.8 (−2.1 to 0.5) | −1.0 (−2.5 to 0.4) | 0.2 (−0.7 to 2.2) | 0.806 | 0.19 |
| Proportion meeting physical activity guidelines 2 | 9 (45.0) | 10 (50.0) | 9 (45.0) | 9 (64.3) | No change | +14.3% 3 | - | - | - |
| Eating competence | 25.1 (8 to 42) | 27.1 (14 to 43) | 27.4 (10 to 41) | 31.1 (18 to 42) 4 | 2.1 (−2.3 to 6.4) | 3.7 (−0.3 to −7.2) | −1.5 (−8.1 to 5.1) | 0.650 | - |
| Frequency of fast food | 5.7 (0 to 10) | 4.2 (0 to 10) | 6.2 (1 to 10) | 3.6 (0 to 10) | 0.5 (−0.6 to 1.6) | −0.4 (−1.8 to 0.9) | 0.9 (−0.8 to 2.7) | 0.298 | - |
| Frequency of discretionary foods | 2.9 (1 to 4.8) | 2.5 (0.4 to 4.6) | 2.9 (0.8 to 4.8) | 2.2 (0.4 to 3.6) | 0.0 (−0.32 to 0.31) | −0.3 (−0.7 to 0.1) | −0.3 (−0.8 to 0.2) | 0.196 | - |
| Sleep Hygiene Index | 36.4 (5.9 (21 to 45) | 39.5 (29 to 51) | 37.6 (20 to 54) | 39.9 (29 to 53) | 1.6 (−0.9 to 4.0) | 0.6 (−2.2 to 3.5) | 0.9 (−2.8 to 4.7) | 0.623 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oftedal, S.; Burrows, T.; Fenton, S.; Murawski, B.; Rayward, A.B.; Duncan, M.J. Feasibility and Preliminary Efficacy of an m-Health Intervention Targeting Physical Activity, Diet, and Sleep Quality in Shift-Workers. Int. J. Environ. Res. Public Health 2019, 16, 3810. https://doi.org/10.3390/ijerph16203810
Oftedal S, Burrows T, Fenton S, Murawski B, Rayward AB, Duncan MJ. Feasibility and Preliminary Efficacy of an m-Health Intervention Targeting Physical Activity, Diet, and Sleep Quality in Shift-Workers. International Journal of Environmental Research and Public Health. 2019; 16(20):3810. https://doi.org/10.3390/ijerph16203810
Chicago/Turabian StyleOftedal, Stina, Tracy Burrows, Sasha Fenton, Beatrice Murawski, Anna B. Rayward, and Mitch J. Duncan. 2019. "Feasibility and Preliminary Efficacy of an m-Health Intervention Targeting Physical Activity, Diet, and Sleep Quality in Shift-Workers" International Journal of Environmental Research and Public Health 16, no. 20: 3810. https://doi.org/10.3390/ijerph16203810
APA StyleOftedal, S., Burrows, T., Fenton, S., Murawski, B., Rayward, A. B., & Duncan, M. J. (2019). Feasibility and Preliminary Efficacy of an m-Health Intervention Targeting Physical Activity, Diet, and Sleep Quality in Shift-Workers. International Journal of Environmental Research and Public Health, 16(20), 3810. https://doi.org/10.3390/ijerph16203810







