Breast Cancer and Exposure to Organochlorines in the CECILE Study: Associations with Plasma Levels Measured at the Time of Diagnosis and Estimated during Adolescence
Abstract
1. Introduction
2. Methods
2.1. Recruitment of Cases and Controls
2.2. Data and Sample Collection
2.3. Organochlorine Quantification
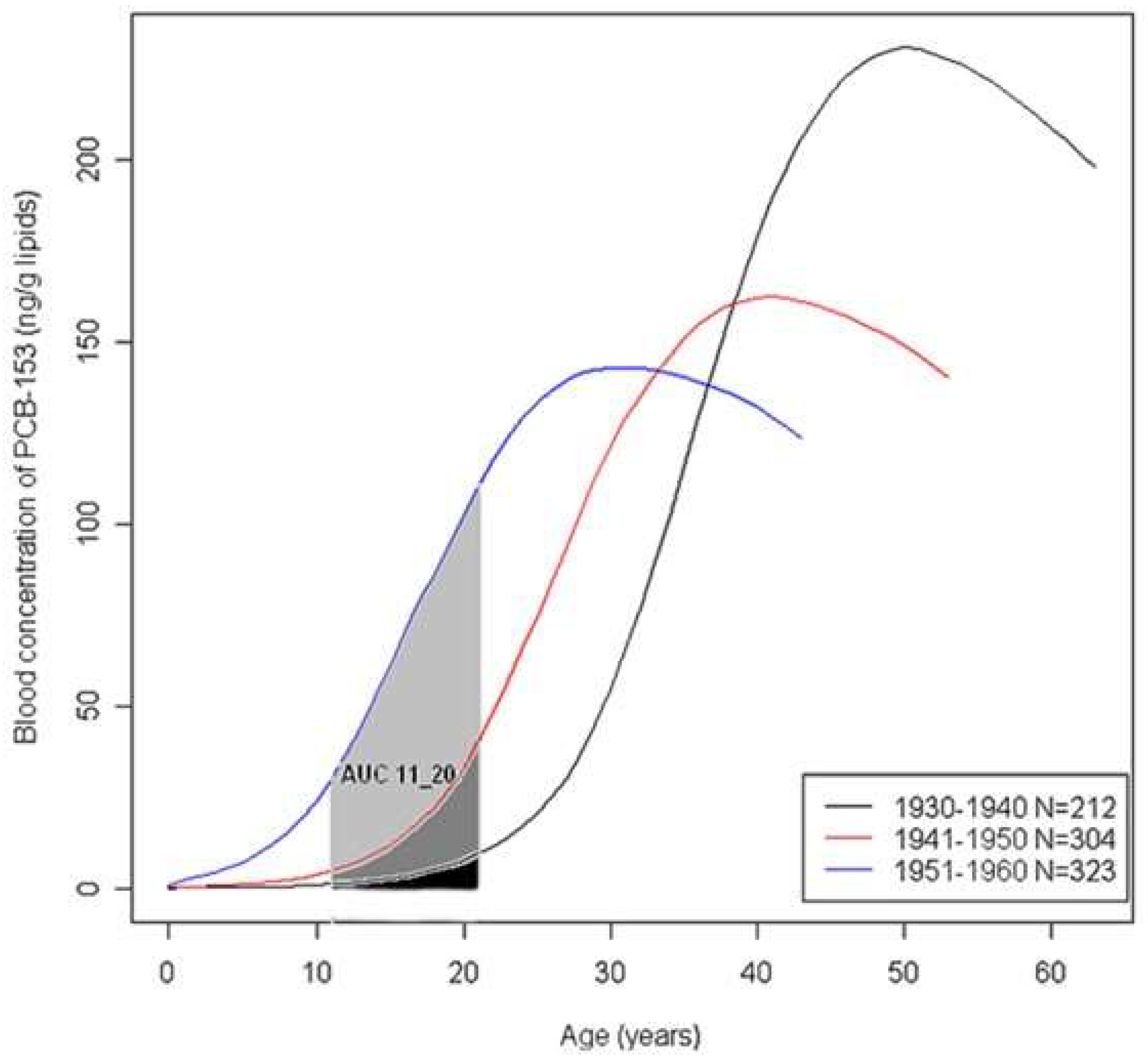
2.4. PCB153 Lifetime Toxicokinetic Profiles
2.5. Statistical Analyses
3. Results
4. Discussion
4.1. PBPK Model for Critical Exposure Windows
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Imputation of Values below the Limit of Detection (LOD)
Appendix B. The Daily Oral Dose
- A first simulation of the toxicokinetic profile is done by arbitrarily setting a maximal dose (MD1). We thus obtain a PCB153 level estimated at the time of enrollment (PCB153estimated)
- The maximal dose in the definitive model (MD2) is calculated as follows:where PCB153measured is the measured level of PCB153 at the time of enrollment.
References
- Ferlay, J.; Soerjomataram, I.; Ervik, M. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide; International Agency for Research on Cancer: Lyon, France, 2013. [Google Scholar]
- Belot, A.; Grosclaude, P.; Bossard, N.; Jougla, E.; Benhamou, E.; Delafosse, P.; Guizard, A.-V.; Molinié, F.; Danzon, A.; Bara, S.; et al. Cancer incidence and mortality in France over the period 1980–2005. Rev. DÉpidémiologie Santé Publique 2008, 56, 159–175. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, K.M.; Udesky, J.O.; Rudel, R.A.; Brody, J.G. Environmental chemicals and breast cancer: An updated review of epidemiological literature informed by biological mechanisms. Environ. Res. 2018, 160, 152–182. [Google Scholar] [CrossRef] [PubMed]
- WHO. State of the Science of Endocrine Disrupting Chemicals. 2012. Available online: http://www.who.int/ceh/publications/endocrine/en/ (accessed on 4 January 2019).
- Hiatt, R.A.; Brody, J.G. Environmental Determinants of Breast Cancer. Annu. Rev. Public Health 2018, 39, 113–133. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Opinion of the Scientific Panel on Contaminants in the Food Chain on a Request from the Commission Related to DDT as an Undesirable Substance in Animal Feed; EFSA: Parma, Italy, 2006.
- Desvignes, V.; Mahé, A.; Laffray, X.; Vigreux-Besret, C.; Feidt, C.; Badot, P.-M.; Volatier, J.-L.; Rivière, G. Polychlorobiphenyls in freshwater fish: A new strategy to set maximum contamination limits. Food Addit. Contam. Part Chem. Anal. Control Expo. Risk Assess. 2017, 34, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Rudel, R.A.; Fenton, S.E.; Ackerman, J.M.; Euling, S.Y.; Makris, S.L. Environmental exposures and mammary gland development: State of the science, public health implications, and research recommendations. Environ. Health Perspect. 2011, 119, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Fenton, S.E. Endocrine-disrupting compounds and mammary gland development: Early exposure and later life consequences. Endocrinology 2006, 147, S18–S24. [Google Scholar] [CrossRef] [PubMed]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Polychlorinated Biphenyls and Polybrominated Biphenyls; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Lyon, France, 2016. [Google Scholar]
- Loomis, D.; Guyton, K.; Grosse, Y.; El Ghissasi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K. Carcinogenicity of lindane, DDT, and 2,4-dichlorophenoxyacetic acid. Lancet Oncol. 2015, 16, 891–892. [Google Scholar] [CrossRef]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. DDT, Lindane, and 2,4-D; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Lyon, France, 2018. [Google Scholar]
- Cohn, B.A.; La Merrill, M.; Krigbaum, N.Y.; Yeh, G.; Park, J.-S.; Zimmermann, L.; Cirillo, P.M. DDT Exposure in Utero and Breast Cancer. J. Clin. Endocrinol. Metab. 2015, 100, 2865–2872. [Google Scholar] [CrossRef]
- Cohn, B.A.; Wolff, M.S.; Cirillo, P.M.; Sholtz, R.I. DDT and breast cancer in young women: New data on the significance of age at exposure. Environ. Health Perspect. 2007, 115, 1406–1414. [Google Scholar] [CrossRef]
- Cohn, B.A.; Terry, M.B.; Plumb, M.; Cirillo, P.M. Exposure to polychlorinated biphenyl (PCB) congeners measured shortly after giving birth and subsequent risk of maternal breast cancer before age 50. Breast Cancer Res. Treat. 2012, 136, 267–275. [Google Scholar] [CrossRef]
- Verner, M.-A.; Charbonneau, M.; López-Carrillo, L.; Haddad, S. Physiologically based pharmacokinetic modeling of persistent organic pollutants for lifetime exposure assessment: A new tool in breast cancer epidemiologic studies. Environ. Health Perspect. 2008, 116, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Bachelet, D.; Truong, T.; Verner, M.-A.; Arveux, P.; Kerbrat, P.; Charlier, C.; Guihenneuc-Jouyaux, C.; Guénel, P. Determinants of serum concentrations of 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene and polychlorinated biphenyls among French women in the CECILE study. Environ. Res. 2011, 111, 861–870. [Google Scholar] [CrossRef] [PubMed]
- INSEE; DARES. Nomenclatures des Professions et Catégories Socio-Professionnelles des Emplois Salariés D’entreprise; INSEE (Institut National de la Statistique et des Etudes Economiques): Paris, France, 2003. [Google Scholar]
- Phillips, D.L.; Pirkle, J.L.; Burse, V.W.; Bernert, J.T.; Henderson, L.O.; Needham, L.L. Chlorinated hydrocarbon levels in human serum: Effects of fasting and feeding. Arch. Environ. Contam. Toxicol. 1989, 18, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Verner, M.-A.; Bachelet, D.; McDougall, R.; Charbonneau, M.; Guénel, P.; Haddad, S. A case study addressing the reliability of polychlorinated biphenyl levels measured at the time of breast cancer diagnosis in representing early-life exposure. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2011, 20, 281–286. [Google Scholar] [CrossRef]
- OCDE. Rapport sur la Mise en Application par les Pays Membres de la Décision du Conseil sur la Protection de L’environnment par le Contrôle des Diphényles Polychlorés; OCDE Organisation for Economic Cooperation and Development: Paris, France, 1982. [Google Scholar]
- Baars, A.J.; Bakker, M.I.; Baumann, R.A.; Boon, P.E.; Freijer, J.I.; Hoogenboom, L.A.P.; Hoogerbrugge, R.; van Klaveren, J.D.; Liem, A.K.D.; Traag, W.A.; et al. Dioxins, dioxin-like PCBs and non-dioxin-like PCBs in foodstuffs: Occurrence and dietary intake in The Netherlands. Toxicol. Lett. 2004, 151, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Barr, D.B.; Weihe, P.; Davis, M.D.; Needham, L.L.; Grandjean, P. Serum polychlorinated biphenyl and organochlorine insecticide concentrations in a Faroese birth cohort. Chemosphere 2006, 62, 1167–1182. [Google Scholar] [CrossRef] [PubMed]
- Gascon, M.; Vrijheid, M.; Garí, M.; Fort, M.; Grimalt, J.O.; Martinez, D.; Torrent, M.; Guxens, M.; Sunyer, J. Temporal trends in concentrations and total serum burdens of organochlorine compounds from birth until adolescence and the role of breastfeeding. Environ. Int. 2015, 74, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Ingber, S.Z.; Buser, M.C.; Pohl, H.R.; Abadin, H.G.; Murray, H.E.; Scinicariello, F. DDT/DDE and breast cancer: A meta-analysis. Regul. Toxicol. Pharmacol. RTP 2013, 67, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-H.; Cha, E.S.; Ko, Y.; Hwang, M.-S.; Hong, J.-H.; Lee, W.J. Exposure to Dichlorodiphenyltrichloroethane and the Risk of Breast Cancer: A Systematic Review and Meta-analysis. Osong Public Health Res. Perspect. 2014, 5, 77–84. [Google Scholar] [CrossRef]
- Leng, L.; Li, J.; Luo, X.-M.; Kim, J.-Y.; Li, Y.-M.; Guo, X.-M.; Chen, X.; Yang, Q.-Y.; Li, G.; Tang, N.-J. Polychlorinated biphenyls and breast cancer: A congener-specific meta-analysis. Environ. Int. 2016, 88, 133–141. [Google Scholar] [CrossRef]
- Wielsøe, M.; Kern, P.; Bonefeld-Jørgensen, E.C. Serum levels of environmental pollutants is a risk factor for breast cancer in Inuit: A case control study. Environ. Health Glob. Access Sci. Source 2017, 16, 56. [Google Scholar] [CrossRef] [PubMed]
- Gladen, B.C.; Longnecker, M.P.; Schecter, A.J. Correlations among polychlorinated biphenyls, dioxins, and furans in humans. Am. J. Ind. Med. 1999, 35, 15–20. [Google Scholar] [CrossRef]
- Itoh, H.; Iwasaki, M.; Hanaoka, T.; Kasuga, Y.; Yokoyama, S.; Onuma, H.; Nishimura, H.; Kusama, R.; Tsugane, S. Serum organochlorines and breast cancer risk in Japanese women: A case-control study. Cancer Causes Control CCC 2009, 20, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Arrebola, J.P.; Belhassen, H.; Artacho-Cordón, F.; Ghali, R.; Ghorbel, H.; Boussen, H.; Perez-Carrascosa, F.M.; Expósito, J.; Hedhili, A.; Olea, N. Risk of female breast cancer and serum concentrations of organochlorine pesticides and polychlorinated biphenyls: A case-control study in Tunisia. Sci. Total Environ. 2015, 520, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Holford, T.R.; Zheng, T.; Mayne, S.T.; Zahm, S.H.; Tessari, J.D.; Boyle, P. Joint effects of nine polychlorinated biphenyl (PCB) congeners on breast cancer risk. Int. J. Epidemiol. 2000, 29, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Holford, T.R.; Tessari, J.; Mayne, S.T.; Owens, P.H.; Ward, B.; Carter, D.; Boyle, P.; Dubrow, R.; Archibeque-Engle, S.; et al. Breast cancer risk associated with congeners of polychlorinated biphenyls. Am. J. Epidemiol. 2000, 152, 50–58. [Google Scholar] [CrossRef] [PubMed]
- McGlynn, K.A.; Quraishi, S.M.; Graubard, B.I.; Weber, J.-P.; Rubertone, M.V.; Erickson, R.L. Polychlorinated biphenyls and risk of testicular germ cell tumors. Cancer Res. 2009, 69, 1901–1909. [Google Scholar] [CrossRef]
- Emeville, E.; Giusti, A.; Coumoul, X.; Thomé, J.-P.; Blanchet, P.; Multigner, L. Associations of plasma concentrations of dichlorodiphenyldichloroethylene and polychlorinated biphenyls with prostate cancer: A case-control study in guadeloupe (French west indies). Environ. Health Perspect. 2015, 123, 317–323. [Google Scholar] [CrossRef]
- Bonefeld-Jørgensen, E.C.; Andersen, H.R.; Rasmussen, T.H.; Vinggaard, A.M. Effect of highly bioaccumulated polychlorinated biphenyl congeners on estrogen and androgen receptor activity. Toxicology 2001, 158, 141–153. [Google Scholar] [CrossRef]
- Oh, S.M.; Ryu, B.T.; Lee, S.K.; Chung, K.H. Antiestrogenic potentials of ortho-PCB congeners by single or complex exposure. Arch. Pharm. Res. 2007, 30, 199–209. [Google Scholar] [CrossRef]
- Plísková, M.; Vondrácek, J.; Canton, R.F.; Nera, J.; Kocan, A.; Petrík, J.; Trnovec, T.; Sanderson, T.; van den Berg, M.; Machala, M. Impact of polychlorinated biphenyls contamination on estrogenic activity in human male serum. Environ. Health Perspect. 2005, 113, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Wyrick, K.L.; Meadows, G.G.; Wills, T.B.; Vorderstrasse, B.A. Activation of the aryl hydrocarbon receptor by TCDD inhibits mammary tumor metastasis in a syngeneic mouse model of breast cancer. Toxicol. Sci. Off. J. Soc. Toxicol. 2011, 124, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Viel, J.-F.; Clément, M.-C.; Hägi, M.; Grandjean, S.; Challier, B.; Danzon, A. Dioxin emissions from a municipal solid waste incinerator and risk of invasive breast cancer: A population-based case-control study with GIS-derived exposure. Int. J. Health Geogr. 2008, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Chevrier, J.; Dewailly, E.; Ayotte, P.; Mauriège, P.; Després, J.P.; Tremblay, A. Body weight loss increases plasma and adipose tissue concentrations of potentially toxic pollutants in obese individuals. Int. J. Obes. Relat. Metab. Disord. J. Int. Assoc. Study Obes. 2000, 24, 1272–1278. [Google Scholar] [CrossRef]
- Fréry, N.; Guldner, L.; Saoudi, A.; Garnier, R.; Zeghnoun, A.; Bidondo, M.-L. Exposition de la Population Française aux Substances Chimiques de L’environnement; Institut de Veille Sanitaire: Paris, France, 2003. [Google Scholar]
| Organochlorine a | Detection Frequency (%) | 25th Percentile | 50th Percentile | 75th Percentile | 90th Percentile | Max |
|---|---|---|---|---|---|---|
| Controls | ||||||
| <50 years (N = 357) | ||||||
| p.p’-DDE | 38.1 | <LOD | <LOD | 131.5 | 209.1 | 839.6 |
| PCB138 | 7.0 | <LOD | <LOD | <LOD | <LOD | 230.7 |
| PCB153 | 36.1 | <LOD | <LOD | 103.1 | 165.1 | 496.8 |
| PCB180 | 12.6 | <LOD | <LOD | <LOD | 131.7 | 353.0 |
| ≥50 years (N = 698) | ||||||
| p.p’-DDE | 61.7 | <LOD | 105.7 | 196.0 | 342.3 | 2440.0 |
| PCB138 | 26.1 | <LOD | <LOD | 79.1 | 155.0 | 748.0 |
| PCB153 | 70.9 | <LOD | 106.2 | 161.7 | 239.4 | 1218.0 |
| PCB180 | 27.8 | <LOD | <LOD | 100.0 | 170.8 | 654.2 |
| Cases | ||||||
| <50 years (N = 162) | ||||||
| p.p’-DDE | 40.1 | <LOD | <LOD | 141.3 | 288.0 | 2051.0 |
| PCB138 | 11.1 | <LOD | <LOD | <LOD | 113.0 | 224.5 |
| PCB153 | 42.0 | <LOD | <LOD | 110.6 | 210.3 | 440.8 |
| PCB180 | 15.4 | <LOD | <LOD | <LOD | 177.8 | 384.2 |
| ≥50 years (N = 533) | ||||||
| p.p’-DDE | 58.2 | <LOD | 93.7 | 201.4 | 374.2 | 2196.0 |
| PCB138 | 21.6 | <LOD | <LOD | <LOD | 140.3 | 756.2 |
| PCB153 | 66.2 | <LOD | 94.7 | 146.2 | 205.4 | 559.0 |
| PCB180 | 23.3 | <LOD | <LOD | <LOD | 158.0 | 798.1 |
| Matching Variables and Breast Cancer Risk Factors | Cases (n = 695) | Controls (n = 1055) | OR a | 95% CI | |||
|---|---|---|---|---|---|---|---|
| n | % | n | % | ||||
| Age | <40 | 28 | 4 | 114 | 11 | ||
| (Years) | 40–49 | 134 | 19 | 243 | 23 | ||
| 50–59 | 245 | 35 | 318 | 30 | |||
| ≥60 | 288 | 41 | 380 | 36 | |||
| Study Area | Côte d’Or | 210 | 30 | 368 | 35 | ||
| Ille-et-Vilaine | 485 | 70 | 687 | 65 | |||
| Age at menarche | <14 | 434 | 63 | 652 | 62 | 1.00 | reference |
| (Years) | 14 | 131 | 19 | 204 | 20 | 0.91 | [0.74, 1.13] |
| 15+ | 121 | 18 | 189 | 18 | 0.89 | [0.71, 1.11] | |
| missing | 9 | 10 | |||||
| Parity | 0 | 76 | 11 | 72 | 7 | 1.00 | reference |
| 1 | 106 | 15 | 134 | 13 | 0.76 | [0.53, 1.08] | |
| 2 | 281 | 40 | 373 | 35 | 0.69 | [0.51, 0.94] | |
| 3 | 167 | 24 | 324 | 31 | 0.45 | [0.32, 0.62] | |
| 4+ | 65 | 9 | 152 | 14 | 0.34 | [0.23, 0.50] | |
| Age at first full term | <22 | 152 | 25 | 284 | 29 | 0.80 | [0.64, 1.01] |
| Pregnancy among parous | 22–24 | 189 | 31 | 297 | 30 | 1.00 | reference |
| Women (years) | 25–27 | 132 | 21 | 235 | 24 | 0.92 | [0.72, 1.17] |
| 28+ | 146 | 24 | 167 | 17 | 1.48 | [1.15, 1.90] | |
| Duration of breastfeeding | never breastfed | 301 | 49 | 458 | 47 | 1.00 | reference |
| Among parous women | <26 | 224 | 37 | 370 | 38 | 1.00 | [0.82, 1.21] |
| (Weeks) | 26–52 | 56 | 9 | 95 | 10 | 1.00 | [0.73, 1.36] |
| 53+ | 31 | 5 | 57 | 6 | 0.88 | [0.59, 1.30] | |
| missing | 7 | 3 | |||||
| Body mass index | <50 years | ||||||
| (kg/m²) | <18.5 | 11 | 7 | 8 | 2 | 2.61 | [1.16, 6.06] |
| 18.5–24.9 | 118 | 73 | 231 | 65 | 1.00 | reference | |
| 25–34.9 | 24 | 15 | 80 | 22 | 0.56 | [0.36, 0.86] | |
| 35+ | 9 | 6 | 37 | 10 | 0.46 | [0.23, 0.86] | |
| missing | 1 | ||||||
| ≥50 years | |||||||
| <18.5 | 14 | 3 | 16 | 2 | 1.12 | [0.6, 2.1] | |
| 18.5–24.9 | 284 | 54 | 358 | 51 | 1.00 | reference | |
| 25–34.9 | 153 | 29 | 217 | 31 | 0.89 | [0.71, 1.11] | |
| 35+ | 78 | 15 | 106 | 15 | 0.92 | [0.69, 1.22] | |
| missing | 4 | 1 | |||||
| Hormonal replacement therapy (current intake) | No | 570 | 87 | 904 | 90 | 1.00 | reference |
| Yes | 85 | 13 | 96 | 10 | 1.31 | [1.00, 1.72] | |
| missing | 40 | 55 | |||||
| Family history of breast | |||||||
| Cancer in first-degree | No | 574 | 83 | 953 | 90 | 1.00 | reference |
| Relatives | Yes | 121 | 17 | 102 | 10 | 1.93 | [1.51, 2.45] |
| Benign breast disease | No | 404 | 58 | 743 | 70 | 1.00 | reference |
| Yes | 290 | 42 | 311 | 29 | 1.62 | [1.37, 1.93] | |
| missing | 1 | 1 | |||||
| Cases | Controls | OR a | 95% CI | p-Trend | ||
|---|---|---|---|---|---|---|
| DDE (ng/g lipids) | ||||||
| All | <LOD | 310 | 480 | 1.00 | reference | 0.11 |
| 51.3–131.5 | 125 | 186 | 0.97 | [0.76, 1.24] | ||
| 131.5–212.4 | 91 | 185 | 0.64 | [0.49, 0.83] | ||
| ≥212.4 | 150 | 189 | 0.93 | [0.73, 1.18] | ||
| <50 years | <LOD | 96 | 215 | 1.00 | reference | 0.85 |
| 53.1–130.6 | 18 | 45 | 0.75 | [0.42, 1.30] | ||
| 130.6–181.1 | 15 | 45 | 0.71 | [0.39, 1.25] | ||
| ≥181.1 | 32 | 46 | 1.48 | [0.90, 2.41] | ||
| ≥50 years | <LOD | 214 | 265 | 1.00 | reference | 0.06 |
| 51.3–132.8 | 107 | 141 | 0.97 | [0.73, 1.28] | ||
| 132.8–222.2 | 80 | 140 | 0.63 | [0.47, 0.85] | ||
| ≥222.2 | 114 | 143 | 0.81 | [0.61, 1.07] | ||
| PCB153 (ng/g lipids) | ||||||
| All | <LOD | 265 | 424 | 1.00 | reference | 0.28 |
| 36.6–110.0 | 156 | 202 | 1.14 | [0.90, 1.46] | ||
| 110.0–162.6 | 133 | 204 | 0.89 | [0.69, 1.14] | ||
| ≥162.6 | 122 | 210 | 0.75 | [0.57, 0.97] | ||
| <50 years | <LOD | 93 | 223 | 1.00 | reference | 0.32 |
| 59.4–110.1 | 27 | 42 | 1.72 | [1.01, 2.90] | ||
| 110.1–155.4 | 13 | 42 | 0.65 | [0.33, 1.22] | ||
| ≥155.4 | 28 | 44 | 1.46 | [0.85, 2.49] | ||
| ≥50 years | <LOD | 172 | 201 | 1.00 | reference | 0.09 |
| 36.6–110.0 | 129 | 161 | 1.00 | [0.76, 1.32] | ||
| 110.0–163.4 | 116 | 161 | 0.85 | [0.64, 1.13] | ||
| ≥163.4 | 98 | 166 | 0.65 | [0.48, 0.89] |
| Birth Cohort | PCB153 at Diagnosis (ng/g lipids) | Cases | Controls | OR a | 95% CI | Calendar Years When Aged 11–20 Years | PCB153 at Age 11–20 Years b(ng/g lipids) | Cases | Controls | OR a | 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1930–1940 | <98.1 | 70 | 69 | 1.00 | reference | 1941–1960 | <1.5 | 88 | 69 | 1.00 | reference |
| 98.1–173.7 | 69 | 68 | 1.14 | [0.73, 1.75] | 1.5–2.8 | 59 | 68 | 0.51 | [0.32, 0.82] | ||
| ≥173.7 | 41 | 70 | 0.58 | [0.35, 0.89] | ≥2.8 | 33 | 70 | 0.24 | [0.13, 0.42] | ||
| 1941–1950 | <LOD | 73 | 102 | 1.00 | reference | 1952–1970 | <6.4 | 71 | 99 | 1.00 | reference |
| LOD–134.2 | 79 | 99 | 1.34 | [0.91, 1.97] | 6.4–12.5 | 63 | 98 | 0.65 | [0.42, 0.99] | ||
| ≥134.2 | 51 | 100 | 0.62 | [0.40, 0.94] | ≥12.5 | 69 | 104 | 0.53 | [0.32, 0.85] | ||
| 1951–1960 | <LOD | 103 | 138 | 1.00 | reference | 1962–1980 | <35.0 | 94 | 107 | 1.00 | reference |
| LOD–124.9 | 62 | 92 | 0.99 | [0.67, 1.46] | 35.0–63.0 | 69 | 106 | 0.67 | [0.45, 0.99] | ||
| ≥124.9 | 48 | 93 | 0.52 | [0.34, 0.79] | ≥63.0 | 50 | 110 | 0.38 | [0.24, 0.60] | ||
| 1961–1981 | <LOD | 51 | 150 | 1.00 | reference | 1972–1990 | <47.1 | 29 | 70 | 1.00 | reference |
| LOD–127.7 | 11 | 29 | 1.13 | [0.52, 2.41] | 47.1–83.6 | 23 | 66 | 0.73 | [0.39, 1.36] | ||
| ≥127.7 | 18 | 30 | 1.15 | [0.56, 2.30] | ≥83.6 | 28 | 73 | 0.71 | [0.38, 1.34] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bachelet, D.; Verner, M.-A.; Neri, M.; Cordina Duverger, É.; Charlier, C.; Arveux, P.; Haddad, S.; Guénel, P. Breast Cancer and Exposure to Organochlorines in the CECILE Study: Associations with Plasma Levels Measured at the Time of Diagnosis and Estimated during Adolescence. Int. J. Environ. Res. Public Health 2019, 16, 271. https://doi.org/10.3390/ijerph16020271
Bachelet D, Verner M-A, Neri M, Cordina Duverger É, Charlier C, Arveux P, Haddad S, Guénel P. Breast Cancer and Exposure to Organochlorines in the CECILE Study: Associations with Plasma Levels Measured at the Time of Diagnosis and Estimated during Adolescence. International Journal of Environmental Research and Public Health. 2019; 16(2):271. https://doi.org/10.3390/ijerph16020271
Chicago/Turabian StyleBachelet, Delphine, Marc-André Verner, Monica Neri, Émilie Cordina Duverger, Corinne Charlier, Patrick Arveux, Sami Haddad, and Pascal Guénel. 2019. "Breast Cancer and Exposure to Organochlorines in the CECILE Study: Associations with Plasma Levels Measured at the Time of Diagnosis and Estimated during Adolescence" International Journal of Environmental Research and Public Health 16, no. 2: 271. https://doi.org/10.3390/ijerph16020271
APA StyleBachelet, D., Verner, M.-A., Neri, M., Cordina Duverger, É., Charlier, C., Arveux, P., Haddad, S., & Guénel, P. (2019). Breast Cancer and Exposure to Organochlorines in the CECILE Study: Associations with Plasma Levels Measured at the Time of Diagnosis and Estimated during Adolescence. International Journal of Environmental Research and Public Health, 16(2), 271. https://doi.org/10.3390/ijerph16020271






