Effectiveness of an Online CBT-I Intervention and a Face-to-Face Treatment for Shift Work Sleep Disorder: A Comparison of Sleep Diary Data
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Di Milia, L.; Waage, S.; Pallesen, S.; Bjorvatn, B. Shift work disorder in a random population sample—Prevalence and comorbidities. PLoS ONE 2013, 8, e55306. [Google Scholar] [CrossRef] [PubMed]
- Beers, T.M. Flexible Schedules and Shift Work: Replacing the 9-to-5 Workday. Mon. Lab. Rev. 2000, 123, 33. [Google Scholar]
- Statista. Schichtarbeit—Anteil der Erwerbstätigen in Deutschland bis 2016. Available online: https://de.statista.com/statistik/daten/studie/360921/umfrage/anteil-der-erwerbstaetigen-in-deutschland-die-schichtarbeit-leisten/ (accessed on 28 August 2018).
- Das Europäische Parlament und der Rat der Europäischen Union. Richtlinie 2003/88/EG des Europäischen Parlaments und des Rates vom 4. November 2003 über bestimmte Aspekte der Arbeitszeitgestaltung. Available online: https://eur-lex.europa.eu/legal-content/DE/TXT/?uri=celex%3A32003L0088 (accessed on 28 August 2018).
- Richter, K.; Acker, J.; Kamcev, N.; Bajraktarov, S.; Piehl, A.; Niklewski, G. Recommendations for the prevention of breast cancer in shift workers. EPMA J. 2011, 2, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Puttonen, S.; Härmä, M.; Hublin, C. Shift work and cardiovascular disease—Pathways from circadian stress to morbidity. Scand. J. Work Environ. Health 2010, 36, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Matheson, A.; O’Brien, L.; Reid, J.-A. The impact of shiftwork on health: A literature review. J. Clin. Nurs. 2014, 23, 3309–3320. [Google Scholar] [CrossRef] [PubMed]
- Ohayon, M.M.; Lemoine, P.; Arnaud-Briant, V.; Dreyfus, M. Prevalence and consequences of sleep disorders in a shift worker population. J. Psychosom. Res. 2002, 53, 577–583. [Google Scholar] [CrossRef]
- Karasek, R.A. Job Demands, Job Decision Latitude, and Mental Strain: Implications for Job Redesign. Adm. Sci. Q. 1979, 24, 285. [Google Scholar] [CrossRef]
- Siegrist, J.; Starke, D.; Chandola, T.; Godin, I.; Marmot, M.; Niedhammer, I.; Peter, R. The measurement of effort—Reward imbalance at work: European comparisons. Soc. Sci. Med. 2004, 58, 1483–1499. [Google Scholar] [CrossRef]
- Peter, R.; Alfredsson, L.; Knutsson, A.; Siegrist, J.; Westerholm, P. Does a stressful psychosocial work environment mediate the effects of shift work on cardiovascular risk factors? Scand. J. Work Environ. Health 1999, 25, 376–381. [Google Scholar] [CrossRef]
- Dorrian, J.; Skinner, N. Alcohol consumption patterns of shiftworkers compared with dayworkers. Chronobiol. Int. 2012, 29, 610–618. [Google Scholar] [CrossRef]
- Stevens, R.G. Working against our endogenous circadian clock: Breast cancer and electric lighting in the modern world. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2009, 680, 106–108. [Google Scholar] [CrossRef]
- Hajak, G.; Zulley, J. Schlafstörungen bei Schichtarbeit—Wenn die Arbeit mit der inneren Uhr kollidiert. Psychoneuro 2008, 34, 70–77. [Google Scholar] [CrossRef]
- Czeisler, C.A.; Johnson, M.P.; Duffy, J.F.; Brown, E.N.; Ronda, J.M.; Kronauer, R.E. Exposure to bright light and darkness to treat physiologic maladaptation to night work. N. Engl. J. Med. 1990, 322, 1253–1259. [Google Scholar] [CrossRef]
- Drake, C.L.; Roehrs, T.; Richardson, G.; Walsh, J.K.; Roth, T. Shift Work Sleep Disorder: Prevalence and Consequences Beyond that of Symptomatic Day Workers. Sleep 2004, 27, 1453–1462. [Google Scholar] [CrossRef]
- Härmä, M.; Tenkanen, L.; Sjöblom, T.; Alikoski, T.; Heinsalmi, P. Combined effects of shift work and life-style on the prevalence of insomnia, sleep deprivation and daytime sleepiness. Scand. J. Work Environ. Health 1998, 24, 300–307. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Jehan, S.; Zizi, F.; Pandi-Perumal, S.R.; Myers, A.K.; Auguste, E.; Jean-Louis, G.; McFarlane, S.I. Shift Work and Sleep: Medical Implications and Management. Sleep Med. Disord. 2017, 1. [Google Scholar] [CrossRef]
- Wickwire, E.M.; Geiger-Brown, J.; Scharf, S.M.; Drake, C.L. Shift Work and Shift Work Sleep Disorder: Clinical and Organizational Perspectives. Chest 2017, 151, 1156–1172. [Google Scholar] [CrossRef]
- Knauth, P.; Hornberger, S. Preventive and compensatory measures for shift workers. Occup. Med. (Lond.) 2003, 53, 109–116. [Google Scholar] [CrossRef]
- Burgess, H.J.; Sharkey, K.M.; Eastman, C.I. Bright light, dark and melatonin can promote circadian adaptation in night shift workers. Sleep Med. Rev. 2002, 6, 407–420. [Google Scholar] [CrossRef]
- Crowley, S.J.; Lee, C.; Tseng, C.Y.; Fogg, L.F.; Eastman, C.I. Combinations of bright light, scheduled dark, sunglasses, and melatonin to facilitate circadian entrainment to night shift work. J. Biol. Rhythm. 2003, 18, 513–523. [Google Scholar] [CrossRef]
- Arendt, J.; Skene, D.J.; Middleton, B.; Lockley, S.W.; Deacon, S. Efficacy of melatonin treatment in jet lag, shift work, and blindness. J. Biol. Rhythm. 1997, 12, 604–617. [Google Scholar] [CrossRef]
- Auld, F.; Maschauer, E.L.; Morrison, I.; Skene, D.J.; Riha, R.L. Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders. Sleep Med. Rev. 2017, 34, 10–22. [Google Scholar] [CrossRef]
- Wright, S.W.; Lawrence, L.M.; Wrenn, K.D.; Haynes, M.L.; Welch, L.W.; Schlack, H.M. Randomized Clinical Trial of Melatonin After Night-Shift Work: Efficacy and Neuropsychologic Effects. Ann. Emerg. Med. 1998, 32, 334–340. [Google Scholar] [CrossRef]
- Reid, K.J.; Abbott, S.M. Jet Lag and Shift Work Disorder. Sleep Med. Clin. 2015, 10, 523–535. [Google Scholar] [CrossRef]
- Järnefelt, H.; Lagerstedt, R.; Kajaste, S.; Sallinen, M.; Savolainen, A.; Hublin, C. Cognitive behavioral therapy for shift workers with chronic insomnia. Sleep Med. 2012, 13, 1238–1246. [Google Scholar] [CrossRef]
- Lee, K.A.; Gay, C.L.; Alsten, C.R. Home-based behavioral sleep training for shift workers: A pilot study. Behav. Sleep Med. 2014, 12, 455–468. [Google Scholar] [CrossRef]
- Morin, C.M. Cognitive-behavioral approaches to the treatment of insomnia. J. Clin. Psychiatry 2004, 65 (Suppl. 16), 33–40. [Google Scholar]
- Müller, T.; Paterok, B. Schlaftraining. Ein Therapiemanual zur Behandlung von Schlafstörungen; Hogrefe: Göttingen, Germany, 2010. [Google Scholar]
- Riemann, D.; Perlis, M.L. The treatments of chronic insomnia: A review of benzodiazepine receptor agonists and psychological and behavioral therapies. Sleep Med. Rev. 2009, 13, 205–214. [Google Scholar] [CrossRef]
- Morgenthaler, T.; Kramer, M.; Alessi, C.; Friedman, L.; Boehlecke, B.; Brown, T.; Coleman, J.; Kapur, V.; Lee-Chiong, T.; Owens, J.; et al. Practice parameters for the psychological and behavioral treatment of insomnia: An update. An american academy of sleep medicine report. Sleep 2006, 29, 1415–1419. [Google Scholar]
- Circadian Information LP. Shiftwork Lifestyle Training. Available online: https://www.circadian.com/images/pdf/shiftwork_lifestyle_training.pdf (accessed on 10 July 2019).
- Espie, C.A.; Kyle, S.D.; Williams, C.; Ong, J.C.; Douglas, N.J.; Hames, P.; Brown, J.S.L. A randomized, placebo-controlled trial of online cognitive behavioral therapy for chronic insomnia disorder delivered via an automated media-rich web application. Sleep 2012, 35, 769–781. [Google Scholar] [CrossRef]
- Vincent, N.; Lewycky, S. Logging on for Better Sleep: RCT of the Effectiveness of Online Treatment for Insomnia. Sleep 2009, 32, 807–815. [Google Scholar] [CrossRef]
- Ye, Y.-Y.; Chen, N.-K.; Chen, J.; Liu, J.; Lin, L.; Liu, Y.-Z.; Lang, Y.; Li, X.-J.; Yang, X.-J.; Jiang, X.-J. Internet-based cognitive-behavioural therapy for insomnia (ICBT-I): A meta-analysis of randomised controlled trials. BMJ Open 2016, 6, e010707. [Google Scholar] [CrossRef]
- Barnes, C.M.; Miller, J.A.; Bostock, S. Helping employees sleep well: Effects of cognitive behavioral therapy for insomnia on work outcomes. J. Appl. Psychol. 2017, 102, 104–113. [Google Scholar] [CrossRef]
- Horsch, C.H.; Lancee, J.; Griffioen-Both, F.; Spruit, S.; Fitrianie, S.; Neerincx, M.A.; Beun, R.J.; Brinkman, W.-P. Mobile Phone-Delivered Cognitive Behavioral Therapy for Insomnia: A Randomized Waitlist Controlled Trial. J. Med. Int. Res. 2017, 19, e70. [Google Scholar] [CrossRef]
- Thorndike, F.P.; Ritterband, L.M.; Gonder-Frederick, L.A.; Lord, H.R.; Ingersoll, K.S.; Morin, C.M. A randomized controlled trial of an internet intervention for adults with insomnia: Effects on comorbid psychological and fatigue symptoms. J. Clin. Psychol. 2013, 69, 1078–1093. [Google Scholar] [CrossRef]
- Blom, K.; Tarkian Tillgren, H.; Wiklund, T.; Danlycke, E.; Forssén, M.; Söderström, A.; Johansson, R.; Hesser, H.; Jernelöv, S.; Lindefors, N.; et al. Internet- vs. group-delivered cognitive behavior therapy for insomnia: A randomized controlled non-inferiority trial. Behav. Res. Ther. 2015, 70, 47–55. [Google Scholar] [CrossRef]
- Seyffert, M.; Lagisetty, P.; Landgraf, J.; Chopra, V.; Pfeiffer, P.N.; Conte, M.L.; Rogers, M.A.M. Internet-Delivered Cognitive Behavioral Therapy to Treat Insomnia: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0149139. [Google Scholar] [CrossRef]
- Currie, S.R.; Clark, S.; Hodgins, D.C.; El-Guebaly, N. Randomized controlled trial of brief cognitive-behavioural interventions for insomnia in recovering alcoholics. Addiction 2004, 99, 1121–1132. [Google Scholar] [CrossRef]
- Jernelöv, S.; Lekander, M.; Blom, K.; Rydh, S.; Ljótsson, B.; Axelsson, J.; Kaldo, V. Efficacy of a behavioral self-help treatment with or without therapist guidance for co-morbid and primary insomnia: A randomized controlled trial. BMC Psychiatry 2012, 12, 5. [Google Scholar] [CrossRef]
- Psychiatric Research Unit, WHO Collaborating Center for Mental Health. WHO (Fünf): Fragebogen zum Wohlbefinden. Available online: https://www.psykiatri-regionh.dk/who-5/Documents/WHO5_German.pdf (accessed on 28 August 2018).
- Bastien, C. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med. 2001, 2, 297–307. [Google Scholar] [CrossRef]
- Johns, M.W. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. BDI-II: Beck Depression Inventory; Pearson: San Antonio, TX, USA, 1996. [Google Scholar]
- Montgomery, S.A.; Asberg, M. A new depression scale designed to be sensitive to change. Br. J. Psychiatry 1979, 134, 382–389. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Richter, K. Schlafen Sie gut! Ihr Schlafprogramm für aufgeweckte Tage und erholsame Nächte; Trias: Stuttgart, Germany, 2015; ISBN 9783830482253. [Google Scholar]
- Deutsche Gesellschaft für Schlafmedizin. Schlaftagebücher in der Diagnostik von Insomnien. Available online: https://www.dgsm.de/fachinformationen_frageboegen_schlaftagebuecher.php?language=german (accessed on 10 September 2018).
- IBM Corp. IBM SPSS Statistics for Windows, Version 25.0, IBM Corp.: Armonk, NY, USA, 2017.
- Frobeen, A.; Basler, H.D.; Rehfisch, H.-P.; Andreasson, M. Wirksam entspannen: Progressive Muskelentspannung zum Download. Available online: https://www.tk.de/techniker/magazin/life-balance/aktiv-entspannen/progressive-muskelentspannung-zum-download-2021142 (accessed on 28 August 2018).
- Espie, C.A.; Emsley, R.; Kyle, S.D.; Gordon, C.; Drake, C.L.; Siriwardena, A.N.; Cape, J.; Ong, J.C.; Sheaves, B.; Foster, R.; et al. Effect of digital cognitive behavioral therapy for insomnia on health, psychological well-being, and sleep-related quality of life: A randomized clinical trial. JAMA Psychiatry 2018. [Google Scholar] [CrossRef]
- Kyle, S.D.; Miller, C.B.; Rogers, Z.; Siriwardena, A.N.; Macmahon, K.M.; Espie, C.A. Sleep restriction therapy for insomnia is associated with reduced objective total sleep time, increased daytime somnolence, and objectively impaired vigilance: Implications for the clinical management of insomnia disorder. Sleep 2014, 37, 229–237. [Google Scholar] [CrossRef]
- Kyle, S.D.; Morgan, K.; Spiegelhalder, K.; Espie, C.A. No pain, no gain: An exploratory within-subjects mixed-methods evaluation of the patient experience of sleep restriction therapy (SRT) for insomnia//No pain, no gain: An exploratory within-subjects mixed-methods evaluation of the patient experience of sleep restriction therapy (SRT) for insomnia. Sleep Med. 2011, 12, 735–747. [Google Scholar] [CrossRef]
- Rhodes, A.E.; Goering, P.N.; To, T.; Williams, J.I. Gender and outpatient mental health service use. Soc. Sci. Med. 2002, 54, 1–10. [Google Scholar] [CrossRef]
- Toscos, T.; Carpenter, M.; Drouin, M.; Roebuck, A.; Kerrigan, C.; Mirro, M. College Students’ Experiences with, and Willingness to Use, Different Types of Telemental Health Resources: Do Gender, Depression/Anxiety, or Stress Levels Matter? Telemed. J. E Health 2018. [Google Scholar] [CrossRef]
- Fisher, E.; Farina, A. Attitudes toward seeking professional psychological help: Development and research utility of an attitude scale. J. Coll. Stud. Dev. 1995, 36, 368–373. [Google Scholar]
- Okujava, N.; Malashkhia, N.; Shagidze, S.; Tsereteli, A.; Arevadze, B.; Chikhladze, N.; de Weerd, A.; van Straten, A. Digital cognitive behavioral therapy for insomnia—The first Georgian version. Can we use it in practice? Int. Interv. 2019, 17, 100244. [Google Scholar] [CrossRef]
| Question | Example Answer |
|---|---|
| Evening questions | |
| Have you had daytime sleep today? If so, please report total sleep time. | Yes, 30 min |
| Did you take medication for sleep today? If so, what kind? | Yes, Mirtazapine |
| Have you had alcohol today? If so, what kind and how much? | Yes, 3 glasses of wine |
| How was your mood before going to bed? (scale of 1 = great to 6 = horrible) | 2 |
| How tired were you before going to bed? (scale of 1 = not tired at all to 6 = dead tired) | 4 |
| Morning questions | |
| At what time did you go to bed? | 23:00 |
| When did you turn of the lights? | 23:30 |
| How many minutes did it take you to fall asleep? | 20 |
| How often did you wake up last night? | Twice |
| How many minutes did you lie awake? Don’t count the minutes it took you to fall asleep. | 40 |
| What time did you last wake up tonight? | 6:00 |
| When did you finally get up? | 6:30 |
| How long did you sleep in total? | 5:15 |
| How restful was your sleep? (scale of 1 = perfectly restful to 6 = catastrophic) | 4 |
| How was your mood after getting up? (scale of 1 = great to 6 = horrible) | 5 |
| Session | Online | Outpatient |
|---|---|---|
| 1 | Sleep restriction Instruction via semi-standardized email | Initial examination, diagnosis Semi-standardized clinical interview during face to face session |
| Interim | - | Actometry (14 days) |
| 2 | Psychoeducation, sleep hygiene Information via semi-standardized email | Sleep restriction Instruction and discussion during face to face session |
| 3 | Relaxation techniques Information via standardized email, link to online audio resources for progressive muscle relaxation [54] | Psychoeducation, sleep hygiene Information and discussion during face to face session |
| Interim | - | Bright light therapy at home with a loan device |
| 4 | Concluding remarks, recommendations for the future via semi-standardized email | Relaxation techniques Information and training during face to face session |
| 5 | - | Cognitive restructuring Information, discussion and training during face to face session |
| 6 | - | Concluding remarks, recommendations for the future Information and discussion during face to face session |
| Pre- | Post- | p-Value (One-Tailed) | |
|---|---|---|---|
| Online sample (n = 21) | |||
| World Health Organization wellbeing questionnaire (WHO-5) | 10.9 ± 3.8 | 13.9 ± 4.2 | 0.004 |
| Insomnia Severity Index (ISI) | 13.9 ± 3.8 | 10.6 ± 5.6 | 0.002 |
| Epworth Sleepiness Scale (ESS) | 9.1 ± 3.7 | 8.2 ± 3.7 | 0.109 |
| Total sleep time | 386.1 ± 58.6 | 395.1 ± 76.1 | 0.195 |
| Total bed time | 483.7 ± 52.2 | 458.1 ± 67.1 | 0.041 |
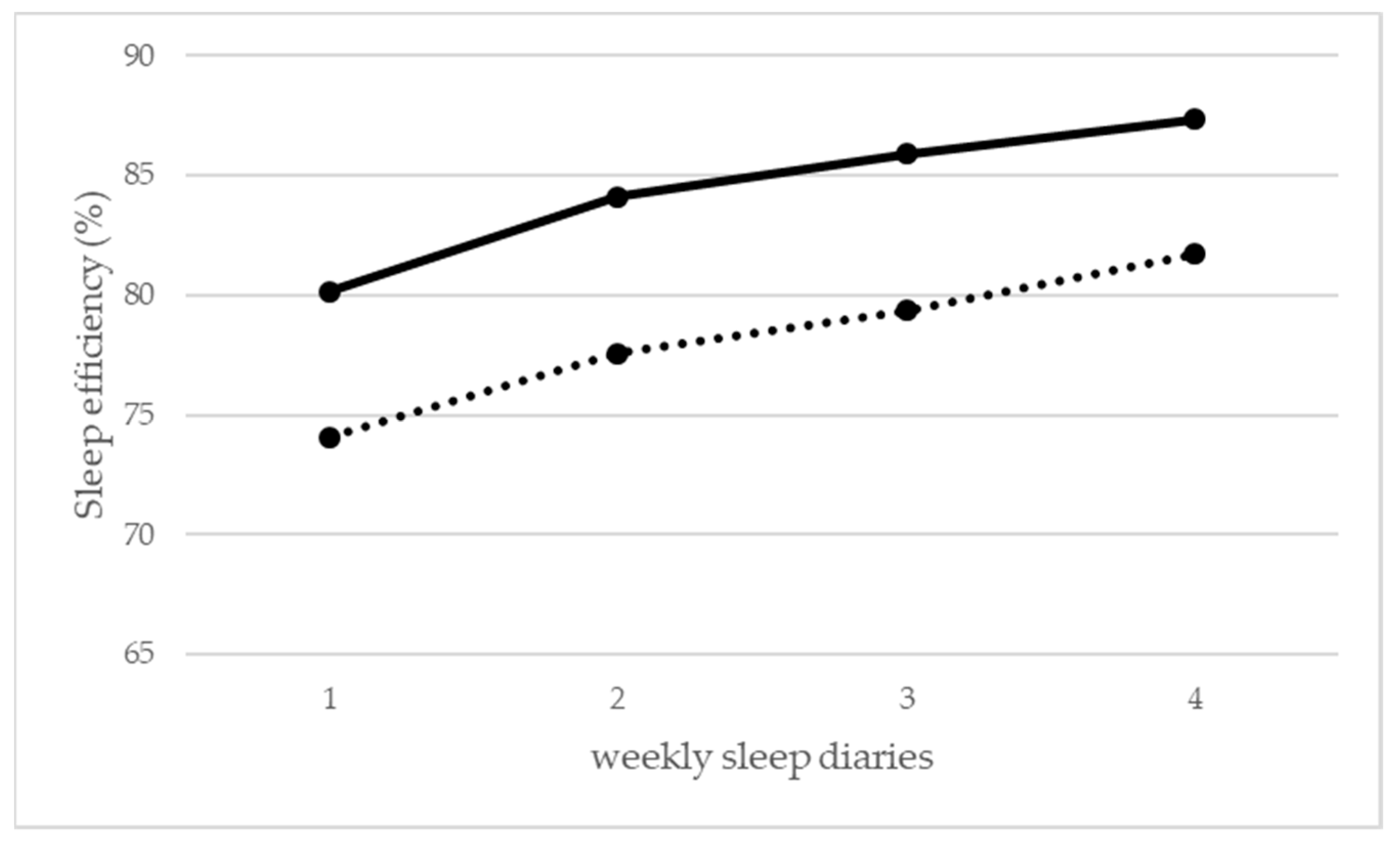
| Sleep efficiency | 80.1% ± 11.6% | 87.3% ± 11.1% | 0.001 |
| Outpatient sample (n = 12) | |||
| Beck Depression Inventory (BDI-II) | 14.4 ± 10.6 | 7.3 ± 7.3 | 0.007 |
| Montgomery–Åsberg Depression Rating Scale (MADRS) | 19.2 ± 5.6 | 8.2 ± 6.5 | <0.001 |
| Pittsburgh Sleep Quality Index (PSQI) | 11.5 ± 3.9 | 9.0 ± 4.3 | 0.007 |
| ESS | 9.6 ± 4.6 | 7.8 ± 3.9 | 0.124 |
| Total sleep time | 349.3 ± 60.9 | 354.2 ± 64.0 | 0.377 |
| Total bed time | 480.4 ± 61.0 | 435.8 ± 55.3 | 0.019 |
| Sleep efficiency | 74.1% ± 16.6% | 81.7% ± 11.3% | 0.026 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peter, L.; Reindl, R.; Zauter, S.; Hillemacher, T.; Richter, K. Effectiveness of an Online CBT-I Intervention and a Face-to-Face Treatment for Shift Work Sleep Disorder: A Comparison of Sleep Diary Data. Int. J. Environ. Res. Public Health 2019, 16, 3081. https://doi.org/10.3390/ijerph16173081
Peter L, Reindl R, Zauter S, Hillemacher T, Richter K. Effectiveness of an Online CBT-I Intervention and a Face-to-Face Treatment for Shift Work Sleep Disorder: A Comparison of Sleep Diary Data. International Journal of Environmental Research and Public Health. 2019; 16(17):3081. https://doi.org/10.3390/ijerph16173081
Chicago/Turabian StylePeter, Lukas, Richard Reindl, Sigrid Zauter, Thomas Hillemacher, and Kneginja Richter. 2019. "Effectiveness of an Online CBT-I Intervention and a Face-to-Face Treatment for Shift Work Sleep Disorder: A Comparison of Sleep Diary Data" International Journal of Environmental Research and Public Health 16, no. 17: 3081. https://doi.org/10.3390/ijerph16173081
APA StylePeter, L., Reindl, R., Zauter, S., Hillemacher, T., & Richter, K. (2019). Effectiveness of an Online CBT-I Intervention and a Face-to-Face Treatment for Shift Work Sleep Disorder: A Comparison of Sleep Diary Data. International Journal of Environmental Research and Public Health, 16(17), 3081. https://doi.org/10.3390/ijerph16173081




