Effects of Maternal Prenatal Multi-Micronutrient Supplementation on Growth and Development until 3 Years of Age
Abstract
1. Introduction
2. Materials and Methods
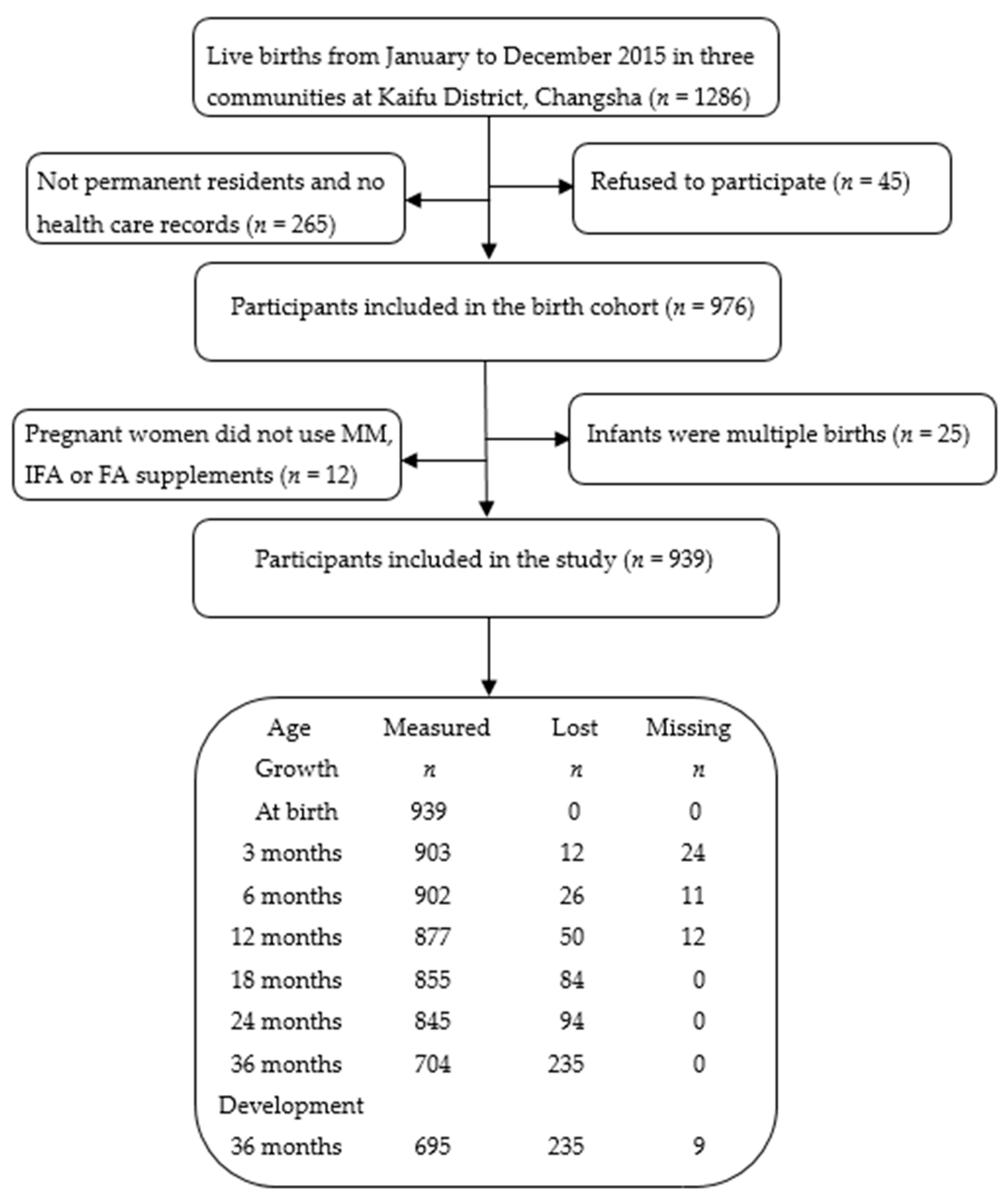
2.1. Study Design and Population
2.2. Data Collection and Procedure
2.3. Measurement of Prenatal Micronutrient Supplementation Status
2.4. Measurement of Infant Growth and Development
2.5. Measurement of Covariates
2.6. Statistical Analysis
2.7. Ethical Considerations
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haider, B.A.; Bhutta, Z.A. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2017, 4, CD004905. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: Use of Multiple Micronutrient Powders for Home Fortification of Foods Consumed by Pregnant Women; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Christian, P.; Stewart, C.P. Maternal micronutrient deficiency, fetal development, and the risk of chronic disease. J. Nutr. 2010, 140, 437–445. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guideline: WHO Recommendations on Antenatal Care for a Positive Pregnancy Experience; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization; United Nations Children’s Fund. Reaching Optimal Iodine Nutrition in Pregnant and Lactating Women and Young Children: A Joint Statement by the World Health Organization and the United Nations Children’s Fund; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Yu, X.; Chen, W.; Wei, Z.; Ren, T.; Yang, X.; Yu, X. Effects of maternal mild zinc deficiency and different ways of zinc supplementation for offspring on learning and memory. Food Nutr. Res. 2016, 60, 29467. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.C.; Schulze, K.J.; Khatry, S.K.; De Luca, L.M.; West, K.P.J. Maternal vitamin A supplementation increases natural antibody concentrations of preadolescent offspring in rural Nepal. Nutrition 2015, 31, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Zerfu, T.A.; Ayele, H.T. Micronutrients and pregnancy; effect of supplementation on pregnancy and pregnancy outcomes: A systematic review. Nutr. J. 2013, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- West, K.P.J.; Shamim, A.A.; Mehra, S.; Labrique, A.B.; Ali, H.; Shaikh, S.; Klemm, R.D.; Wu, L.S.; Mitra, M.; Haque, R.; et al. Effect of maternal multiple micronutrient vs. iron-folic acid supplementation on infant mortality and adverse birth outcomes in rural Bangladesh: The JiVitA-3 randomized trial. JAMA 2014, 312, 2649–2658. [Google Scholar] [CrossRef]
- Devakumar, D.; Fall, C.H.; Sachdev, H.S.; Margetts, B.M.; Osmond, C.; Wells, J.C.; Costello, A.; Osrin, D. Maternal antenatal multiple micronutrient supplementation for long-term health benefits in children: A systematic review and meta-analysis. BMC Med. 2016, 14, 90. [Google Scholar] [CrossRef]
- Dennison, E.; Fall, C.; Cooper, C.; Barker, D. Prenatal factors influencing long-term outcome. Horm. Res. 1997, 48, 25–29. [Google Scholar] [CrossRef]
- Barker, D.J.; Gluckman, P.D.; Godfrey, K.M.; Harding, J.E.; Owens, J.A.; Robinson, J.S. Fetal nutrition and cardiovascular disease in adult life. Lancet 1993, 341, 938–941. [Google Scholar] [CrossRef]
- Black, M.M.; Walker, S.P.; Fernald, L.C.H.; Andersen, C.T.; DiGirolamo, A.M.; Lu, C.; McCoy, D.C.; Fink, G.; Shawar, Y.R.; Shiffman, J.; et al. Lancet Early Childhood Development Series Steering Committee. Early childhood development coming of age: Science through the life course. Lancet 2017, 389, 77–90. [Google Scholar] [CrossRef]
- Christian, P.; Kim, J.; Mehra, S.; Shaikh, S.; Ali, H.; Shamim, A.A.; Wu, L.; Klemm, R.; Labrique, A.B.; West, K.P.J. Effects of prenatal multiple micronutrient supplementation on growth and cognition through 2 y of age in rural Bangladesh: The JiVitA-3 Trial. Am. J. Clin. Nutr. 2016, 104, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Huy, N.D.; Le Hop, T.; Shrimpton, R.; Hoa, C.V. An effectiveness trial of multiple micronutrient supplementation during pregnancy in Vietnam: Impact on birthweight and on stunting in children at around 2 years of age. Food Nutr. Bull. 2009, 30, S506–S516. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.; Saville, N.; Shrestha, B.P.; Costello, A.M.; Manandhar, D.S.; Osrin, D. Effects of antenatal multiple micronutrient supplementation on children’s weight and size at 2 years of age in Nepal: Follow-up of a double-blind randomised controlled trial. Lancet 2008, 371, 492–499. [Google Scholar] [CrossRef]
- Roberfroid, D.; Huybregts, L.; Lanou, H.; Ouedraogo, L.; Henry, M.C.; Meda, N.; Kolsteren, P. MISAME study group. Impact of prenatal multiple micronutrients on survival and growth during infancy: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yan, H.; Zeng, L.; Cheng, Y.; Wang, D.; Li, Q. No effect of maternal micronutrient supplementation on early childhood growth in rural western China: 30 month follow-up evaluation of a double blind, cluster randomized controlled trial. Eur. J. Clin. Nutr. 2012, 66, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Yan, H.; Zeng, L.; Cheng, Y.; Liang, W.; Dang, S.; Wang, Q.; Tsuji, I. Effects of maternal multimicronutrient supplementation on the mental development of infants in rural western China: Follow-up evaluation of a double-blind, randomized, controlled trial. Pediatrics 2009, 123, e685–e692. [Google Scholar] [CrossRef] [PubMed]
- Sha, T.; Gao, X.; Chen, C.; Li, L.; He, Q.; Wu, X.; Cheng, G.; Tian, Q.; Yang, F.; Yan, Y. Associations of Pre-Pregnancy BMI, Gestational Weight Gain and Maternal Parity with the Trajectory of Weight in Early Childhood: A Prospective Cohort Study. Int. J. Environ. Res. Public Health 2019, 16, 1110. [Google Scholar] [CrossRef]
- Wu, X.; Gao, X.; Sha, T.; Zeng, G.; Liu, S.; Li, L.; Chen, C.; Yan, Y. Modifiable Individual Factors Associated with Breastfeeding: A Cohort Study in China. Int. J. Environ. Res. Public Health 2019, 16, 820. [Google Scholar] [CrossRef]
- Schlesselman, J.J. Sample size requirements in cohort and case-control studies of disease. Am. J. Epidemiol. 1974, 99, 381–384. [Google Scholar] [CrossRef]
- World Health Organization. WHO Child Growth Standards: Length/Height-Forage, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Squires, J.; Bricker, D. Ages & Stages Questionnaires (ASQ-3), 3rd ed.; Brookes: Baltimore, MD, USA, 2009. [Google Scholar]
- Wei, M.; Bian, X.; Squires, J.; Yao, G.; Wang, X.; Xie, H.; Song, W.; Lu, J.; Zhu, C.; Yue, H. Studies of the norm and psychometrical properties of the ages and stages questionnaires, third edition, with a Chinese national sample. Zhonghua Er Ke Za Zhi 2015, 53, 913–918. [Google Scholar] [CrossRef]
- Xie, H.; Clifford, J.; Squires, J.; Chen, C.Y.; Bian, X.; Yu, Q. Adapting and validating a developmental assessment for chinese infants and toddlers: The ages & stages questionnaires: Inventory. Infant Behav. Dev. 2017, 49, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.F. Cooperative Meta-Analysis Group of the Working Group on Obesity in China. Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults–study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed. Environ. Sci. 2002, 15, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Lange, S.; Probst, C.; Quere, M.; Rehm, J.; Popova, S. Alcohol use, smoking and their co-occurrence during pregnancy among Canadian women, 2003 to 2011/12. Addict. Behav. 2015, 50, 102–109. [Google Scholar] [CrossRef]
- Lee, K.J.; Carlin, J.B. Multiple imputation for missing data: Fully conditional specification versus multivariate normal imputation. Am. J. Epidemiol. 2010, 171, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Spratt, M.; Carpenter, J.; Sterne, J.A.; Carlin, J.B.; Heron, J.; Henderson, J.; Tilling, K. Strategies for multiple imputation in longitudinal studies. Am. J. Epidemiol. 2010, 172, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Cheng, Y.; Pei, L.; Jiang, Y.; Lei, F.; Zeng, L.; Wang, Q.; Li, Q.; Kang, Y.; Shen, Y.; et al. Maternal iron intake during pregnancy and birth outcomes: A cross-sectional study in Northwest China. Br. J. Nutr. 2017, 117, 862–871. [Google Scholar] [CrossRef] [PubMed]
- United Nations International Children’s Emergency Fund, World Health Organization, United Nations Children’s Fund. Composition of a Multi-Micronutrient Supplement to be Used in Pilot Programmes Among Pregnant Women in Developing Countries; United Nations Children’s Fund: New York, NY, USA, 1999. [Google Scholar]
- Prado, E.L.; Sebayang, S.K.; Apriatni, M.; Adawiyah, S.R.; Hidayati, N.; Islamiyah, A.; Siddiq, S.; Harefa, B.; Lum, J.; Alcock, K.J.; et al. Maternal multiple micronutrient supplementation and other biomedical and socioenvironmental influences on children’s cognition at age 9–12 years in Indonesia: Follow-up of the SUMMIT randomised trial. Lancet Glob. Health 2017, 5, e217–e228. [Google Scholar] [CrossRef]
- Zeng, L.; Dibley, M.J.; Cheng, Y.; Dang, S.; Chang, S.; Kong, L.; Yan, H. Impact of micronutrient supplementation during pregnancy on birth weight, duration of gestation, and perinatal mortality in rural western China: Double blind cluster randomised controlled trial. BMJ 2008, 337, a2001. [Google Scholar] [CrossRef]
- Kaestel, P.; Michaelsen, K.F.; Aaby, P.; Friis, H. Effects of prenatal multimicronutrient supplements on birth weight and perinatal mortality: A randomised, controlled trial in Guinea-Bissau. Eur. J. Clin. Nutr. 2005, 59, 1081–1089. [Google Scholar] [CrossRef]
- Fall, C.H.; Fisher, D.J.; Osmond, C.; Margetts, B.M. Multiple micronutrient supplementation during pregnancy in low-income countries: A meta-analysis of effects on birth size and length of gestation. Food Nutr. Bull. 2009, 30, S533–S546. [Google Scholar] [CrossRef]
- Tanner, J.M. Catch-up growth in man. Br. Med. Bull. 1981, 37, 233–238. [Google Scholar] [CrossRef]
- Bhargava, A. Protein and Micronutrient Intakes Are Associated with Child Growth and Morbidity from Infancy to Adulthood in the Philippines. J. Nutr. 2016, 146, 133–141. [Google Scholar] [CrossRef]
- Adamo, A.M.; Oteiza, P.I. Zinc deficiency and neurodevelopment: The case of neurons. Biofactors 2010, 36, 117–124. [Google Scholar] [CrossRef]
- Skeaff, S.A. Iodine Deficiency in Pregnancy: The Effect on Neurodevelopment in the Child. Nutrients 2011, 3, 265–273. [Google Scholar] [CrossRef]
- McGrath, N.; Bellinger, D.; Robins, J.; Msamanga, G.I.; Tronick, E.; Fawzi, W.W. Effect of maternal multivitamin supplementation on the mental and psychomotor development of children who are born to HIV-1–infected mothers in Tanzania. Pediatrics 2006, 117, e216–e225. [Google Scholar] [CrossRef]
- Sanstead, H.H. W.O. Atwater memorial lecture. Zinc: Essentiality for brain development and function. Nutr. Rev. 1985, 43, 129–137. [Google Scholar] [CrossRef]
- Dong, J.; Yin, H.; Liu, W.; Wang, P.; Jiang, Y.; Chen, J. Congenital iodine deficiency and hypothyroidism impair LTP and decrease C-fos and C-jun expression in rat hippocampus. Neurotoxicology 2005, 26, 417–426. [Google Scholar] [CrossRef]
- Navarro, D.; Alvarado, M.; Navarrete, F.; GIner, M.; Obregon, M.J.; Manzanares, I.; Berbel, P. Gestational and early postnatal hypothyroidism alters VGLuT1 and VGAT bouton distribution in the neocortex and hippocampus, and behavior in rats. Front. Neuroanat. 2015, 17, 9. [Google Scholar] [CrossRef]
- Guilarte, T.R.; Wagner, H.N.; Frost, J.J. Effects of perinatal vitamin B6 deficiency on dopaminergic neurochemistry. J. Neurochem. 1987, 48, 432–439. [Google Scholar] [CrossRef]
- Potdar, R.D.; Sahariah, S.A.; Gandhi, M.; Kehoe, S.H.; Brown, N.; Sane, H.; Dayama, M.; Jha, S.; Lawande, A.; Coakley, P.J.; et al. Improving women’s diet quality preconceptionally and during gestation: Effects on birth weight and prevalence of low birth weight–a randomized controlled efficacy trial in India (Mumbai maternal nutrition project). Am. J. Clin. Nutr. 2014, 100, 1257–1268. [Google Scholar] [CrossRef]
| Characteristics | MM (n = 254) | IFA (n = 343) | FA (n = 342) | p |
|---|---|---|---|---|
| Maternal characteristics | ||||
| Age (years) | 0.618 | |||
| <25 | 10(3.9) | 14(4.1) | 23(6.7) | |
| 25–29 | 128(50.4) | 159(46.4) | 159(46.5) | |
| 30–34 | 86(33.9) | 123(35.9) | 117(34.2) | |
| ≥35 | 30(11.8) | 47(13.7) | 43(12.6) | |
| Education | 0.013 * | |||
| Junior school or below | 6(2.4) | 12(3.5) | 15(4.4) | |
| Senior high school | 23(9.1) | 38(11.1) | 59(17.3) | |
| College or above | 225(88.6) | 293(85.4) | 268(78.4) | |
| Average monthly household income (RMB) | 0.079 | |||
| ≤ 2000 | 13(5.1) | 10(2.9) | 13(3.8) | |
| 2001–5000 | 134(52.8) | 165(48.1) | 195(57.0) | |
| > 5000 | 107(42.1) | 168(49.0) | 134(39.2) | |
| Postpartum BMI (kg/m2) | 0.642 | |||
| <18.5 | 6(2.4) | 7(2.0) | 6(1.8) | |
| 18.5–23.9 | 144(56.7) | 207(60.3) | 188(55.0) | |
| 24–27.9 | 82(32.3) | 106(30.9) | 112(32.7) | |
| ≥28 | 22(8.7) | 23(6.7) | 36(10.5) | |
| Height (m) | 1.60 ± 0.08 | 1.60 ± 0.07 | 1.60 ± 0.04 | 0.687 |
| Parity | 0.014 * | |||
| Primiparous | 194(76.4) | 224(65.3) | 238(69.6) | |
| Multiparous | 60(23.6) | 119(34.7) | 104(30.4) | |
| Infant characteristics | ||||
| Gender | 0.259 | |||
| Male | 142(55.9) | 117(51.6) | 168(49.1) | |
| Female | 112(44.1) | 166(48.4) | 174(50.9) | |
| Birthweight (g) | 0.775 | |||
| <2500 | 3(1.2) | 5(1.5) | 8(2.3) | |
| 2500–3999 | 232(91.3) | 317(92.4) | 311(90.9) | |
| ≥4000 | 19(7.5) | 21(6.1) | 23(6.7) | |
| Length at birth (cm) | 50.01 ± 1.00 | 49.98 ± 0.77 | 49.89 ± 0.92 | 0.194 |
| Gestational age (weeks) | 0.732 | |||
| <37 | 9(3.2) | 11(3.1) | 13(3.8) | |
| 37–41 | 243(96.3) | 325(96.3) | 325(95.3) | |
| ≥42 | 2(0.5) | 7(0.6) | 4(0.9) |
| Outcomes | Unadjusted Analysis | Model 1 a | Model 2 b | |||
|---|---|---|---|---|---|---|
| β | 95% CI | β | 95% CI | β | 95% CI | |
| LAZ | ||||||
| At birth | 0.01 | (−0.15, 0.17) | 0.03 | (−0.13, 0.19) | 0.04 | (−0.13, 0.21) |
| 3 months | −0.13 | (−0.29, 0.04) | −0.12 | (−0.28, 0.05) | −0.11 | (−0.28, 0.06) |
| 6 months | −0.07 | (−0.23, 0.09) | −0.07 | (−0.23, 0.10) | −0.10 | (−0.27, 0.08) |
| 12 months | −0.05 | (−0.22, 0.12) | −0.04 | (−0.21, 0.13) | −0.04 | (−0.22, 0.14) |
| 18 months | −0.02 | (−0.19, 0.15) | −0.02 | (−0.20, 0.15) | −0.02 | (−0.21, 0.16) |
| 24 months | 0.05 | (−0.12, 0.23) | 0.03 | (−0.15, 0.20) | 0.03 | (−0.16, 0.21) |
| 36 months | 0.00 | (−0.16, 0.17) | −0.01 | (−0.18, 0.15) | −0.01 | (−0.20, 0.19) |
| WAZ | ||||||
| At birth | 0.03 | (−0.13, 0.19) | 0.02 | (−0.15, 0.18) | 0.14 | (−0.04, 0.32) |
| 3 months | −0.24 | (−0.40, −0.08) ** | −0.22 | (−0.38, −0.05) ** | −0.23 | (−0.40, −0.06) ** |
| 6 months | 0.08 | (−0.17, 0.16) | 0.00 | (−0.16, 0.17) | −0.03 | (−0.20, 0.14) |
| 12 months | 0.06 | (−1.11, 0.22) | 0.07 | (−0.10, 0.24) | 0.05 | (−0.13, 0.22) |
| 18 months | −0.07 | (−0.24, 0.10) | −0.07 | (−0.25, 0.10) | −0.11 | (−0.29, 0.08) |
| 24 months | 0.06 | (−0.11, 0.23) | 0.04 | (−0.13, 0.22) | 0.01 | (−0.17, 0.19) |
| 36 months | 0.05 | (−0.13, 0.22) | 0.04 | (−0.14, 0.22) | −0.01 | (−0.20, 0.19) |
| WLZ | ||||||
| At birth | −0.03 | (−0.19, 0.13) | −0.02 | (−0.18, 0.14) | 0.10 | (−0.08, 0.28) |
| 3 months | −0.20 | (−0.36, −0.03) * | −0.18 | (−0.35, −0.02) * | −0.20 | (−0.37, −0.02) * |
| 6 months | 0.05 | (−0.11, 0.21) | 0.06 | (−0.11, 0.22) | 0.04 | (−0.13, 0.21) |
| 12 months | 0.10 | (−0.07, 0.27) | 0.11 | (−0.06, 0.28) | 0.08 | (−0.10, 0.25) |
| 18 months | −0.07 | (−0.24, 0.10) | −0.07 | (−0.24, 0.10) | −0.11 | (−0.39, 0.17) |
| 24 months | 0.03 | (−0.15, 0.20) | 0.02 | (−0.16, 0.19) | −0.02 | (−0.20, 0.16) |
| 36 months | 0.07 | (−0.14, 0.27) | 0.07 | (−0.13, 0.27) | −0.01 | (−0.20, 0.19) |
| Development score at 36 months | ||||||
| Communication | 0.42 | (0.61, 0.23) *** | 0.35 | (0.54, 0.16) *** | 0.41 | (0.61, 0.21) *** |
| Gross motor | 0.56 | (0.39, 0.77) *** | 0.64 | (0.46, 0.83) *** | 0.68 | (0.49, 0.88) *** |
| Fine motor | 1.79 | (1.60, 1.98) *** | 1.80 | (1.61, 1.99) *** | 1.64 | (1.45, 1.84) *** |
| Problem solving | 0.23 | (0.04, 0.42) * | 0.23 | (0.04, 0.42) * | 0.29 | (0.10, 0.49) ** |
| Personal social | 0.74 | (0.55, 0.93) *** | 0.92 | (0.73, 1.11) *** | 0.90 | (0.70, 1.10) *** |
| Outcomes | Unadjusted Analysis | Model 1 a | Model 2 b | |||
|---|---|---|---|---|---|---|
| β | 95% CI | β | 95% CI | β | 95% CI | |
| LAZ | ||||||
| 3 to 12 months | −0.08 | (−0.22, 0.06) | −0.08 | (−0.21, 0.06) | −0.08 | (−0.21, 0.06) |
| 3 to 24 months | 0.00 | (−0.10, 0.10) | −0.01 | (−0.10, 0.09) | −0.03 | (−0.12, 0.07) |
| 3 to 36 months | 0.00 | (−0.09, 0.09) | −0.01 | (−0.10, 0.08) | −0.03 | (−0.12, 0.06) |
| WAZ | ||||||
| 3 to 12 months | −0.07 | (−0.21, 0.07) | −0.06 | (−0.20, 0.07) | −0.09 | (−0.22, 0.04) |
| 3 to 24 months | −0.04 | (−0.14, 0.07) | −0.03 | (−0.14, 0.07) | −0.07 | (−0.17, 0.03) |
| 3 to 36 months | −0.04 | (−0.13, 0.06) | −0.04 | (−0.13, 0.06) | −0.07 | (−0.17, 0.03) |
| WLZ | ||||||
| 3 to 12 months | −0.02 | (−0.15, 0.12) | −0.01 | (−0.15, 0.13) | −0.04 | (−0.18, 0.09) |
| 3 to 24 months | −0.03 | (−0.13, 0.08) | −0.02 | (−0.12, 0.09) | −0.06 | (−0.16, 0.05) |
| 3 to 36 months | −0.02 | (−0.12, 0.08) | −0.01 | (−0.11, 0.09) | −0.05 | (−0.16, 0.05) |
| Outcomes | At Birth | 3 Months | 6 Months | 12 Months | 18 Months | 24 Months | 36 Months |
|---|---|---|---|---|---|---|---|
| Underweight | |||||||
| Unadjusted | 0.34(0.04, 3.04) | 3.87(0.41, 36.97) | 3.77(0.89, 6.53) | 1.56(0.82, 2.96) | 3.01(0.56, 16.29) | 1.63(0.84, 3.16) | 1.47(0.75, 2.86) |
| Model 1 a | 0.31(0.04, 2.80) | 3.84(0.40, 36.87) | 3.69(0.85, 6.37) | 1.53(0.80, 2.92) | 3.02(0.56, 16.44) | 1.55(0.80, 3.01) | 1.61(0.82, 3.15) |
| Model 2 b | 0.22(0.02, 2.24) | 4.72(0.49, 45.74) | 3.52(0.74, 6.04) | 1.43(0.75, 2.72) | 3.83(0.63, 23.24) | 1.46(0.75, 2.85) | 1.48(0.73, 2.92) |
| Stunting | |||||||
| Unadjusted | 0.68(0.13, 3.70) | 1.29(0.33, 5.11) | 2.61(0.24, 28.64) | 1.29(0.18, 9.12) | 0.60(0.12, 3.07) | 2.87(0.73, 11.34) | 1.41(0.09, 22.39) |
| Model 1 a | 0.69(0.13, 3.79) | 1.28(0.32, 5.05) | 2.52(0.23, 27.36) | 1.07(0.15, 7.45) | 0.65(0.13, 3.23) | 3.11(0.79, 12.35) | 1.30(0.08, 20.58) |
| Model 2 b | 1.12(0.15, 8.25) | 1.36(0.34, 5.49) | 3.30(0.27, 41.13) | 1.22(0.13, 11.34) | 0.54(0.09, 3.26) | 5.55(1.19, 26.03) | 1.35(0.09, 19.82) |
| Wasting | |||||||
| Unadjusted | 1.05(0.47, 2.36) | 1.27(0.26, 6.25) | 1.43(0.67, 3.09) | 1.68(0.71, 4.01) | 1.51(0.38, 5.95) | 2.15(0.36, 12.76) | 0.47(0.05, 4.48) |
| Model 1 a | 1.02(0.46, 2.29) | 1.29(0.26, 6.37) | 1.49(0.69, 3.22) | 1.54(0.64, 3.67) | 1.53(0.39, 6.04) | 1.70(0.28, 10.40) | 0.36(0.04, 3.58) |
| Model 2 b | 0.89(0.38, 2.07) | 1.12(0.23, 5.60) | 1.35(0.64, 2.96) | 1.34(0.56, 3.22) | 1.92(0.44, 8.38) | 2.77(0.25, 31.08) | 0.53(0.06, 5.03) |
| Overweight | |||||||
| Unadjusted | 0.57(0.20, 1.59) | 0.50(0.31, 0.81) ** | 0.85(0.53, 1.35) | 0.92(0.49, 1.76) | 1.20(0.58, 2.52) | 1.18(0.50, 2.79) | 1.22(0.60, 2.51) |
| Model 1 a | 0.58(0.20, 1.62) | 0.51(0.32, 0.81) ** | 0.87(0.55, 1.39) | 1.00(0.53, 1.89) | 1.24(0.59, 2.59) | 1.21(0.52, 2.86) | 1.17(0.57, 2.41) |
| Model 2 b | 0.68(0.23, 2.00) | 0.52(0.32, 0.84) ** | 0.93(0.57, 1.50) | 0.89(0.44, 1.78) | 1.40(0.65, 3.04) | 1.04(0.41, 2.64) | 1.16(0.54, 2.48) |
| Obesity | |||||||
| Unadjusted | 0.29(0.11, 0.74) * | 0.54(0.29, 1.01) | 0.92(0.56, 1.51) | 0.97(0.51, 1.86) | 1.15(0.57, 2.32) | 0.61(0.24, 1.57) | 1.76(0.84, 3.68) |
| Model 1 a | 0.28(0.11, 0.73) ** | 0.54(0.29, 1.01) | 0.95(0.58, 1.56) | 1.02(0.53, 1.94) | 1.18(0.59, 2.38) | 0.61(0.24, 1.57) | 1.70(0.81, 3.56) |
| Model 2 b | 0.30(0.11, 0.78) * | 0.54(0.29, 1.02) | 0.99(0.59, 1.65) | 1.09(0.54, 2.17) | 1.18(0.58, 2.42) | 0.55 (0.20, 1.53) | 1.36(0.64, 2.91) |
| Outcomes | Unadjusted Analysis | Model 1 a | Model 2 b | |||
|---|---|---|---|---|---|---|
| RR | 95% CI | RR | 95% CI | RR | 95% CI | |
| Underweight | ||||||
| 3 to 12 months | 3.90 | (0.41, 37.13) | 3.89 | (0.41, 36.90) | 3.85 | (0.40, 37.19) |
| 3 to 24 months | 2.39 | (0.71, 8.07) | 2.41 | (0.72, 8.11) | 2.47 | (0.63, 9.70) |
| 3 to 36 months | 1.93 | (0.62, 6.01) | 1.97 | (0.65, 6.08) | 1.86 | (0.53, 6.56) |
| Stunting | ||||||
| 3 to 12 months | 1.48 | (0.51, 4.25) | 1.47 | (0.51, 4.24) | 1.26 | (0.41, 3.83) |
| 3 to 24 months | 1.41 | (0.66, 3.01) | 1.40 | (0.66, 2.97) | 1.32 | (0.68, 2.58) |
| 3 to 36 months | 1.48 | (0.69, 3.15) | 1.48 | (0.70, 3.14) | 1.39 | (0.64, 3.05) |
| Wasting | ||||||
| 3 to 12 months | 0.92 | (0.35, 2.37) | 0.99 | (0.37, 2.60) | 0.85 | (0.32, 2.37) |
| 3 to 24 months | 0.90 | (0.34, 2.36) | 0.95 | (0.36, 2.51) | 0.75 | (0.26, 2.19) |
| 3 to 36 months | 0.83 | (0.34, 2.02) | 0.85 | (0.35, 2.07) | 0.73 | (0.29, 1.85) |
| Overweight | ||||||
| 3 to 12 months | 0.69 | (0.47, 1.00) | 0.71 | (0.49, 1.03) | 0.62 | (0.43, 0.89) * |
| 3 to 24 months | 0.76 | (0.54, 1.07) | 0.79 | (0.56, 1.12) | 0.77 | (0.55, 1.08) |
| 3 to 36 months | 0.78 | (0.55, 1.10) | 0.80 | (0.57, 1.13) | 0.72 | (0.51, 1.02) |
| Obesity | ||||||
| 3 to 12 months | 0.77 | (0.51, 1.16) | 0.80 | (0.54, 1.20) | 0.78 | (0.53, 1.15) |
| 3 to 24 months | 0.78 | (0.54, 1.11) | 0.82 | (0.58, 1.17) | 0.78 | (0.55, 1.11) |
| 3 to 36 months | 0.87 | (0.63, 1.21) | 0.91 | (0.66, 1.26) | 0.82 | (0.60, 1.12) |
| Outcomes | Unadjusted Analysis | Model 1 a | Model 2 b | |||
|---|---|---|---|---|---|---|
| RR | 95% CI | RR | 95% CI | RR | 95% CI | |
| Communication | 1.24 | (0.47, 3.25) | 1.16 | (0.44, 3.03) | 0.98 | (0.34, 2.81) |
| Gross motor | 0.52 | (0.19, 1.44) | 0.48 | (0.17, 1.33) | 0.48 | (0.17, 1.38) |
| Fine motor | 0.58 | (0.26, 1.33) | 0.62 | (0.27, 1.40) | 0.52 | (0.22, 1.21) |
| Problem solving | 0.93 | (0.49, 1.77) | 0.93 | (0.49, 1.78) | 1.06 | (0.53, 2.10) |
| Personal social | 0.75 | (0.47, 1.19) | 0.73 | (0.46, 1.16) | 0.66 | (0.41, 1.09) |
| Total development | 0.80 | (0.56, 1.12) | 0.78 | (0.56, 1.10) | 0.77 | (0.54, 1.11) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, G.; Sha, T.; Gao, X.; Wu, X.; Tian, Q.; Yang, F.; Yan, Y. Effects of Maternal Prenatal Multi-Micronutrient Supplementation on Growth and Development until 3 Years of Age. Int. J. Environ. Res. Public Health 2019, 16, 2744. https://doi.org/10.3390/ijerph16152744
Cheng G, Sha T, Gao X, Wu X, Tian Q, Yang F, Yan Y. Effects of Maternal Prenatal Multi-Micronutrient Supplementation on Growth and Development until 3 Years of Age. International Journal of Environmental Research and Public Health. 2019; 16(15):2744. https://doi.org/10.3390/ijerph16152744
Chicago/Turabian StyleCheng, Gang, Tingting Sha, Xiao Gao, Xialing Wu, Qianling Tian, Fan Yang, and Yan Yan. 2019. "Effects of Maternal Prenatal Multi-Micronutrient Supplementation on Growth and Development until 3 Years of Age" International Journal of Environmental Research and Public Health 16, no. 15: 2744. https://doi.org/10.3390/ijerph16152744
APA StyleCheng, G., Sha, T., Gao, X., Wu, X., Tian, Q., Yang, F., & Yan, Y. (2019). Effects of Maternal Prenatal Multi-Micronutrient Supplementation on Growth and Development until 3 Years of Age. International Journal of Environmental Research and Public Health, 16(15), 2744. https://doi.org/10.3390/ijerph16152744





