Whitefly Control Strategies against Tomato Leaf Curl New Delhi Virus in Greenhouse Zucchini
Abstract
1. Introduction
2. Materials and Methods
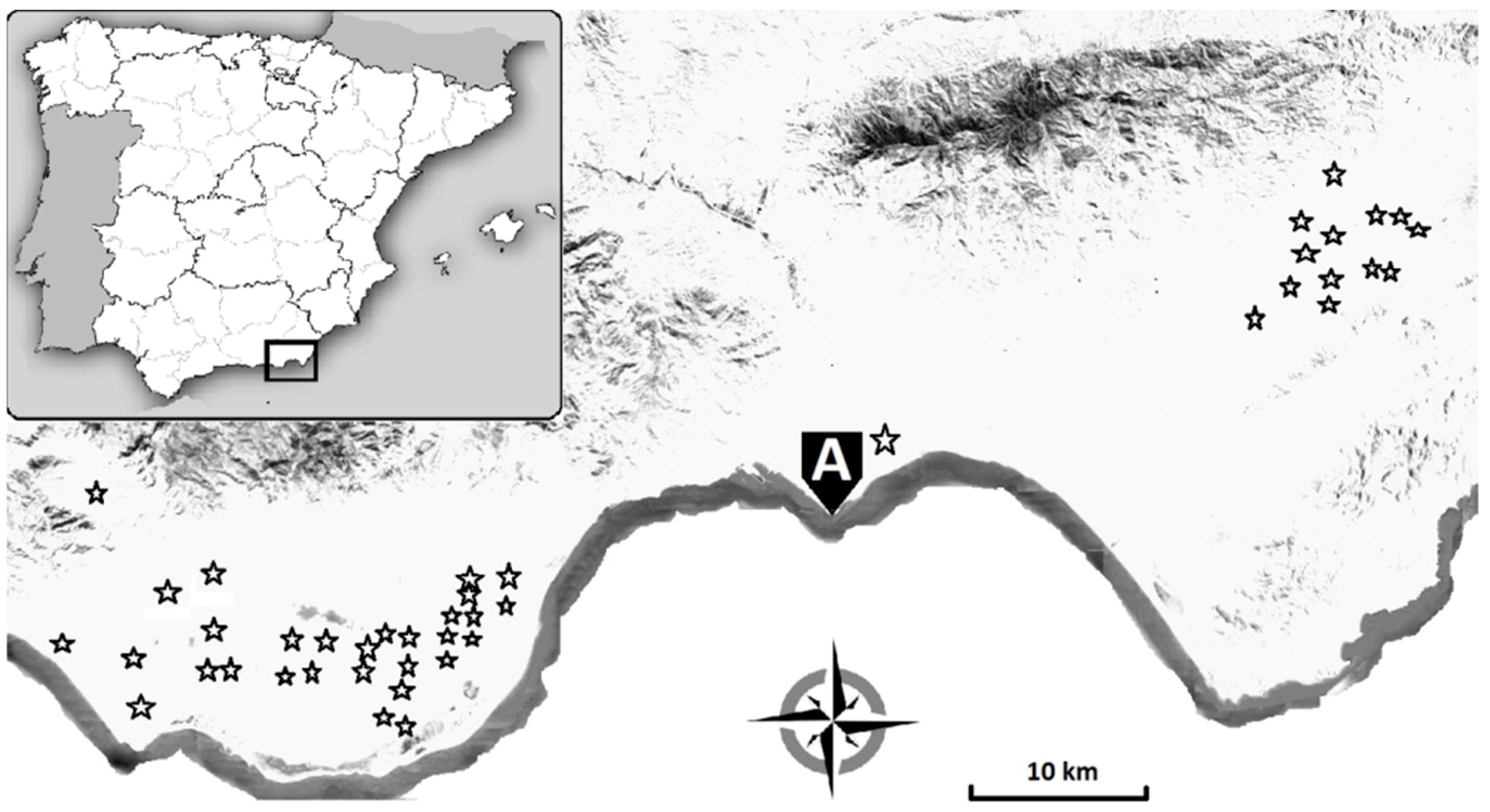
2.1. Commercial Zucchini Crop Monitoring
2.2. General Experimental Procedures
2.3. Monitoring B. tabaci and ToLCNDV Symptom Evolution in Experimental Greenhouse Compartments
2.4. Data Analysis
3. Results
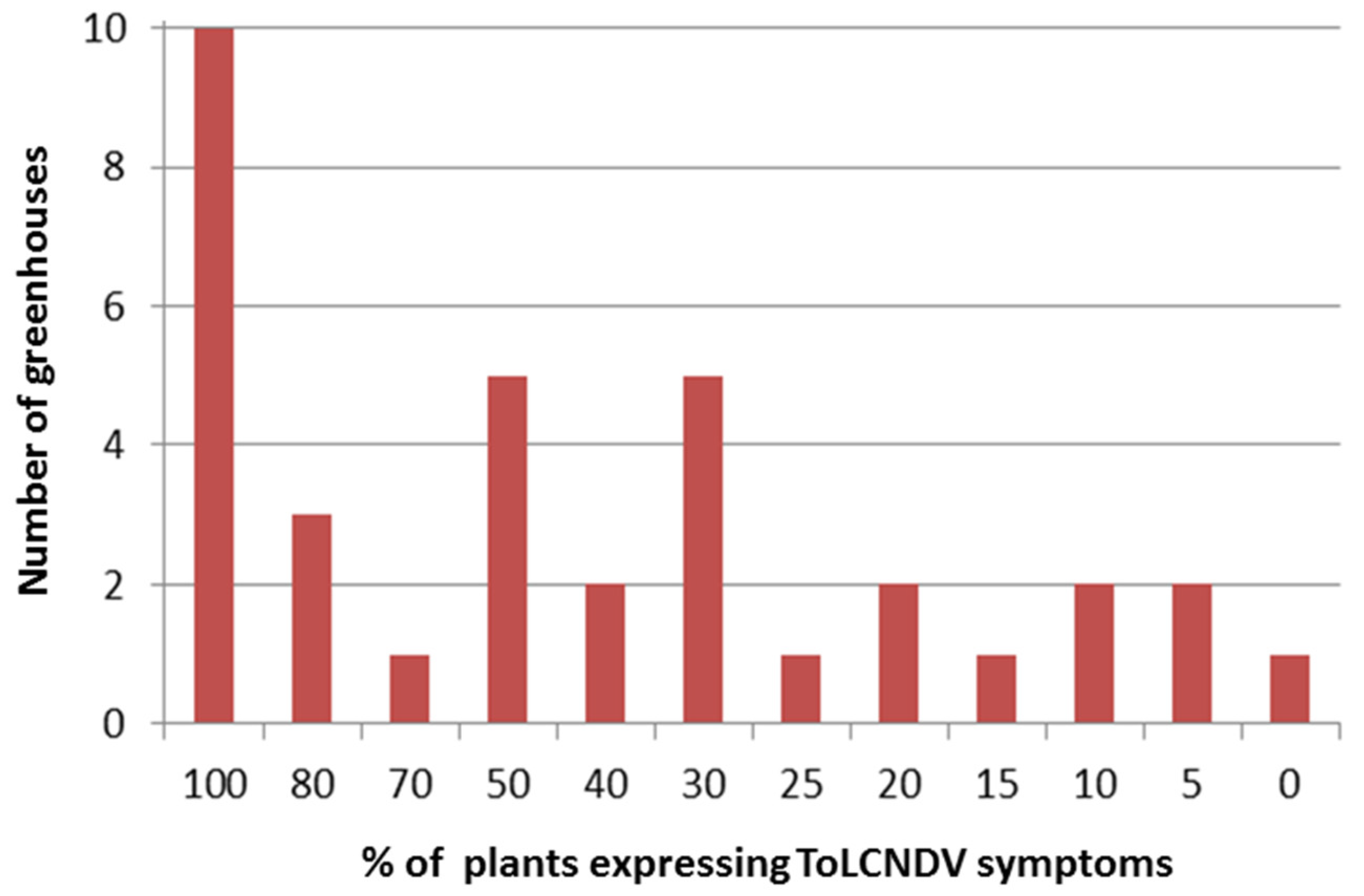
3.1. Phytosanitary Balance, Pest Management, and ToLCNDV Incidence in Commercial Zucchini Crops
3.2. Whitefly and ToLCNDV Evolution in Experimental Greenhouse Compartments
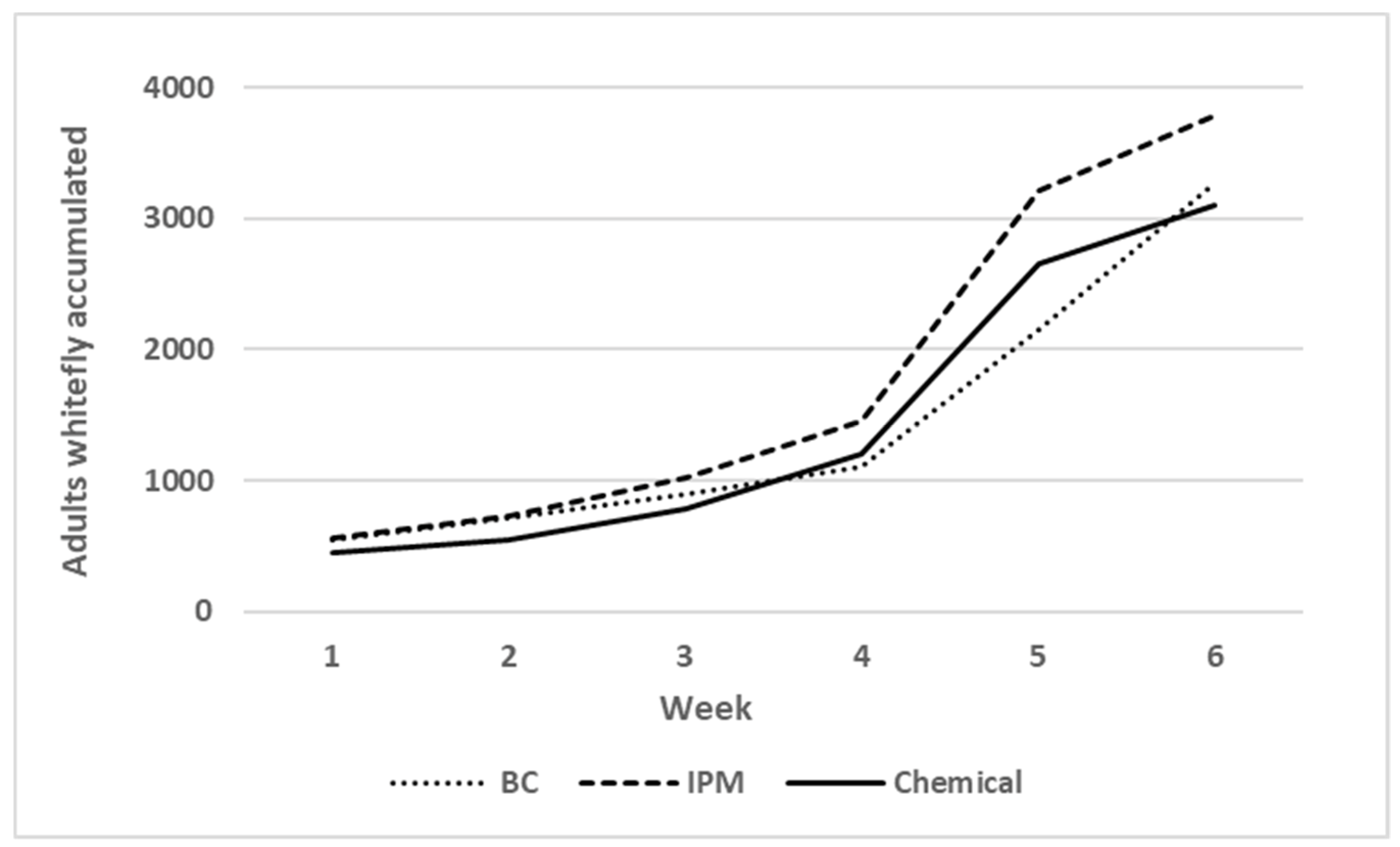
3.2.1. Whitefly
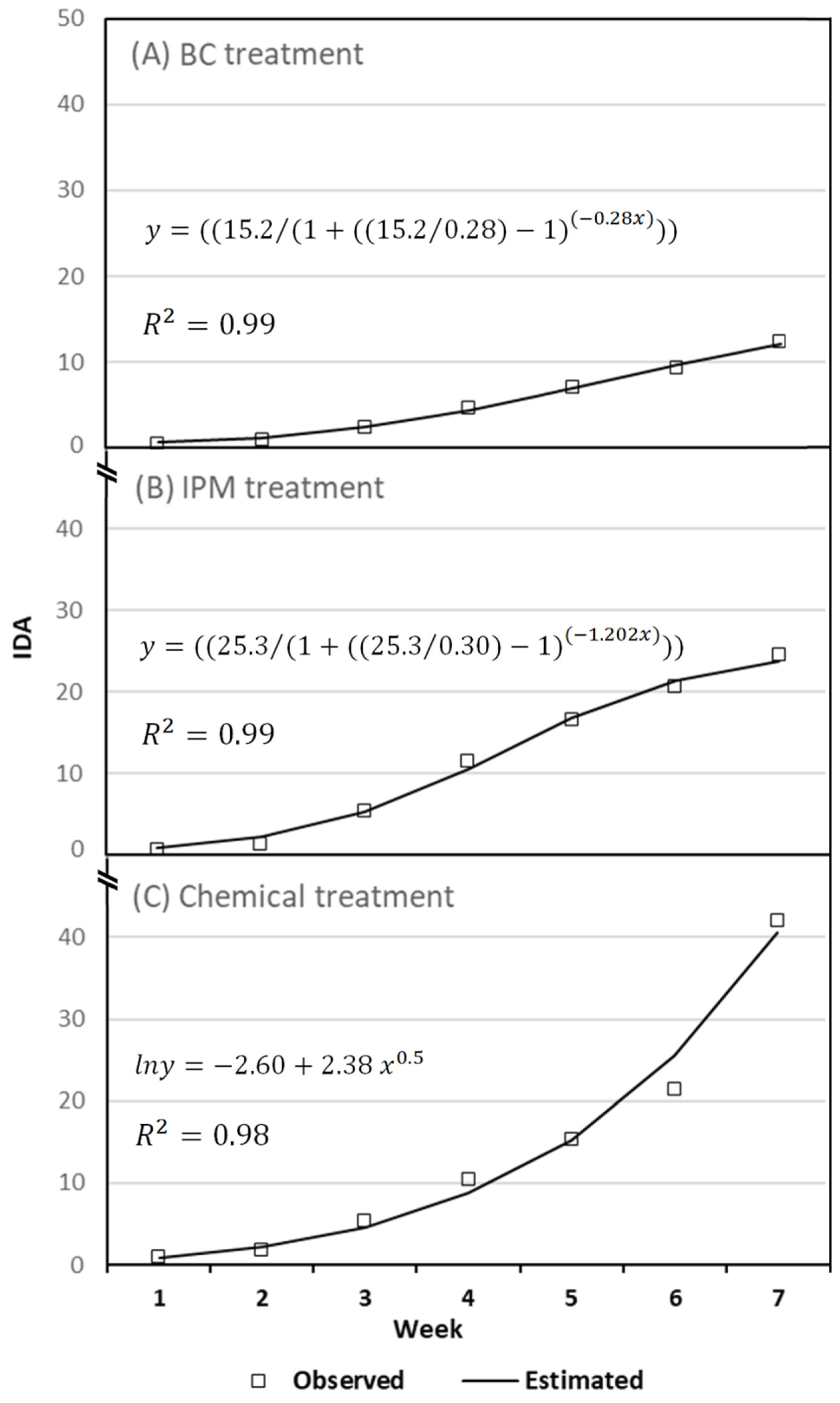
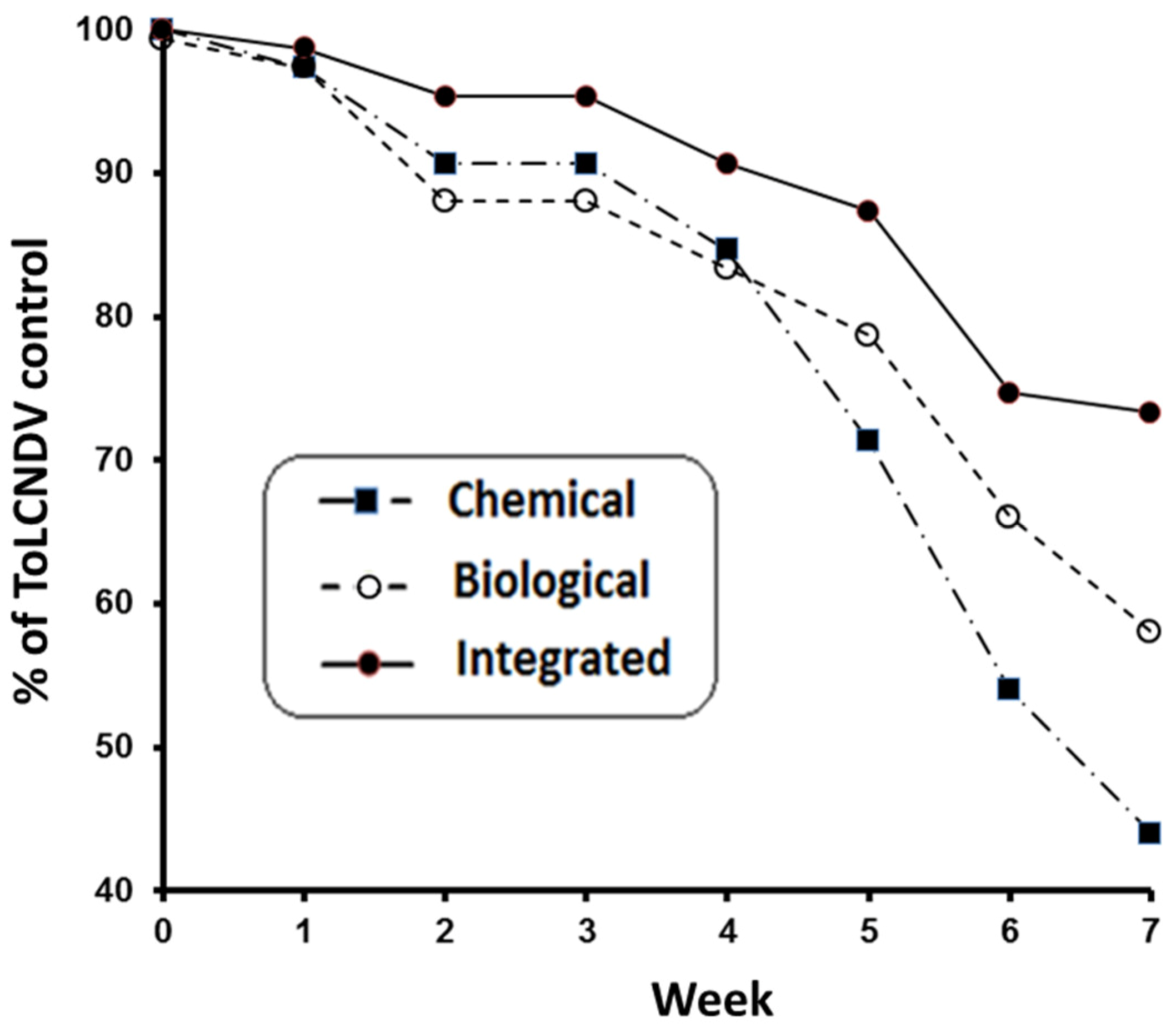
3.2.2. ToLCNDV Symptom Evolution
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oliveira, M.R.V.; Henneberry, T.J.; Anderson, P. History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot. 2001, 20, 709–723. [Google Scholar] [CrossRef]
- Polston, J.E.; De Barro, P.; Boykin, L.M. Transmission specificities of plant viruses with the newly identified species of the Bemisia tabaci species complex. Pest Manag. Sci. 2014, 70, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, S.; Briddon, R.W.; Zafar, Y.; Stanley, J. Geminivirus disease complexes: An emerging threat. Trends Plant Sci. 2006, 8, 128–134. [Google Scholar] [CrossRef]
- Navas-Castillo, J.; Fiallo-Olivé, E.; Sánchez-Campos, S. Emerging Virus Diseases Transmitted by Whiteflies. Annu. Rev. Phytopathol. 2011, 49, 219–248. [Google Scholar] [CrossRef]
- Janssen, D.; Simon, A.; Crespo, O.; Ruiz, L. Genetic population structure of Bemisia tabaci in Spain associated with Tomato leaf curl New Delhi virus. Plant Prot. Sci. 2017, 53, 25–31. [Google Scholar]
- Guirao, P.; Beitia, F.; Cenis, J.L. Biotype determination of Spanish populations of Bemisia tabaci (Hemiptera: Aleyrodidae). Bull. Entomol. Res. 1997, 87, 587–593. [Google Scholar] [CrossRef]
- Luo, C.; Jones, C.M.; Devine, G.; Zhang, F.; Denholm, I.; Gorman, K. Insecticide resistance in Bemisia tabaci biotype Q (Hemiptera: Aleyrodidae) from China. Crop. Prot. 2010, 29, 429–434. [Google Scholar] [CrossRef]
- Yao, F.-L.; Zheng, Y.; Huang, X.-Y.; Ding, X.-L.; Zhao, J.-W.; Desneux, N.; He, Y.-X.; Weng, Q.-Y. Dynamics of Bemisia tabaci biotypes and insecticide resistance in Fujian province in China during 2005–2014. Sci. Rep. 2017, 7, 40803. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, A.R.; Ishaaya, I. Dynamics of biotypes B and Q of the whitefly Bemisia tabaci and its impact on insecticide resistance. Pest Manag. Sci. 2014, 70, 1568–1572. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Grávalos, C.; Haro, J.P.; Cifuentes, D.; Bielza, P. Insecticide resistance status of Bemisia tabaci Q-biotype in south-eastern Spain. Pest Manag. Sci. 2009, 65, 885–891. [Google Scholar] [CrossRef]
- Juarez, M.; Tovar, R.; Fiallo-Olivé, E.; Aranda, M.A.; Gosálvez, B.; Castillo, P.; Moriones, E.; Navas-Castillo, J. First detection of Tomato leaf curl new Delhi virus infecting zucchini in Spain. Plant Dis. 2014, 98, 857. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.; Ruiz, L.; Velasco, L.; Janssen, D. Absolute quantification of Tomato leaf curl New Delhi virus Spain strain, ToLCNDV-ES: Virus accumulation in a host-specific manner. Plant Dis. 2017, 102, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, A.; Pascale, P. Consumers’ Behaviours and Attitudes toward Healthy Food Products: The case of Organic and Functional foods. In Proceedings of the 113th EAAE Seminar A Resilient European Food Industry and Food Chain in A Challenging World, Chania, Greece, 3–6 September 2009. [Google Scholar]
- Nomikou, M.; Janssen, A.; Schraag, R.; Sabelis, M.W. Phytoseiid predators as potential biological control agents for Bemisia tabaci. Exp. Appl. Acarol. 2001, 25, 271–291. [Google Scholar] [CrossRef]
- Calvo, F.J.; Bolckmans, K.; Belda, J. Controlling the tobacco Whitefly Bemisia tabaci (Genn.) (Hom.: Aleyrodidae) in horticultural crops with the predatory mite Amblyseius swirskii (Athias-Henriot). J. Insect Sci. 2008, 8, 11–12. [Google Scholar]
- Calvo, F.J.; Bolckmans, K.; Belda, J.E. Control of Bemisia tabaci and Frankliniella occidentalis in cucumber by Amblyseius swirskii. Biocontrol 2011, 56, 185–192. [Google Scholar] [CrossRef]
- Calvo, F.J.; Bolckmans, K.; Belda, J.E. Biological control-based IPM in sweet pepper greenhouses using Amblyseius swirskii (Acari: Phytoseiidae). Biocontrol Sci. Technol. 2012, 22, 1398–1416. [Google Scholar] [CrossRef]
- Calvo, F.J.; Knapp, M.; van Houten, Y.M.; Hoogerbrugge, H.; Belda, J.E. Amblyseius swirskii: What made this predatory mite such a successful biocontrol agent? Exp. Appl. Acarol. 2014, 65, 419–433. [Google Scholar] [CrossRef]
- Téllez, M.M. Estrategia de control biológico mediante la utilización de Amblyseius swirskii Athias-Henriot en cultivo de pepino. Phytoma 2015, 272, 20–24. [Google Scholar]
- Téllez, M.M.; Simon, A.; Rodriguez, E.; Janssen, D. Control of tomato leaf curl New Delhi virus in zucchini using the predatory mite Amblyseius swirskii. Biol. Control 2017, 114, 106–113. [Google Scholar] [CrossRef]
- Ruiz, L.; Simon, A.; Velasco, L.; Janssen, D. Biological characterization of Tomato leaf curl New Delhi virus from Spain. Plant Pathol. 2017, 66, 376–382. [Google Scholar] [CrossRef]
- Junta de Andalucía 2019. Available online: http://www.juntadeandalucia.es/agriculturaypesca/raif (accessed on 1 February 2019).
- Gueguen, G.; Vavre, F.; Gnankine, O.; Peterschmitt, M.; Charif, D.; Chiel, E.; Gottlieb, Y.; Ghanim, M.; Zchori-Fein, E.; Fleury, F. Endosymbiont metacommunities, mtDNA diversity and the evolution of the Bemisia tabaci (Hemiptera: Aleyrodidae) species complex. Mol. Ecol. 2010, 19, 4365–4376. [Google Scholar] [CrossRef] [PubMed]
- Ruppel, R.F. Cumulative insect-days as an index of crop protection. J. Econ. Entomol. 1983, 76, 375–377. [Google Scholar] [CrossRef]
- IBM Corp. Released 2017. IBM SPSS Statistics for Windows; Version 25.0; IBM Corp.: Armonk, NY, USA, 2017. [Google Scholar]
- Therneau, T.; Grambsch, P.M. Modeling Survival Data-Extending the Cox Model; Springer: New York, NY, USA, 2000. [Google Scholar]
- Campbell, C.L.; Madden, L.V. Introduction to Plant Disease Epidemiology; John Wiley & Sons: New York, NY, USA, 1990. [Google Scholar]
- Tello, J.C.; Camacho, F. Organisms for the Control of Pathogens in Protected Crops. Cultural Practices for Sustainable Agriculture; Fundación Cajamar: Almería, Spain, 2014. [Google Scholar]
- Nomikou, M.; Janssen, A.; Schraag, R.; Sabelis, M.W. Vulnerability of Bemisia tabaci immatures to phytoseiid predators: Consequences for oviposition and influence of alternative food. Entomol. Exp. Appl. 2004, 110, 95–102. [Google Scholar] [CrossRef]
- Messelink, G.J.; van Maanen, R.; van Steenpaal, S.E.F.; Janssen, A. Biological control of thrips and whiteflies by a shared predator: Two pests are better than one. Biol. Control 2008, 44, 372–379. [Google Scholar] [CrossRef]
- Palumbo, J.C.; Horowitz, A.R.; Prabhaker, N. Insecticidal control and resistance management for Bemisia tabaci. Crop Prot. 2001, 20, 739–765. [Google Scholar] [CrossRef]
- Grávalos, C.; Fernández, E.; Belando, A.; Moreno, I.; Ros, C.; Bielza, P. Cross-resistance and baseline susceptibility of Mediterranean strains of Bemisia tabaci to cyantraniliprole. Pest Manag. Sci. 2015, 71, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Van der Blom, J. Applied entomology in Spanish greenhouse horticulture. Proc. Neth. Entomol. Soc. Meet. 2010, 21, 9–17. [Google Scholar]
- Janssen, D.; Ruiz, L.; Garcia, C. Viruses transmitted by the whitefly Bemisia tabaci in organic greenhouse crops: Current situation and risks in Europe. BioGreenhouse 2017. [Google Scholar] [CrossRef]
- Juárez, M.; Rabadán, M.P.; Martínez, L.D.; Tayahi, M.; Grande-Pérez, A.; Gómez, P. Natural hosts and genetic diversity of the emerging tomato leaf curl New Delhi virus in Spain. Front. Microbiol. 2019, 10, 140. [Google Scholar] [CrossRef]
- Perring, T.M.; Gruenhagen, N.M.; Farrar, C.A. Management of plant viral diseases through chemical control of insect vectors. Annu. Rev. Entomol. 1999, 44, 457–481. [Google Scholar] [CrossRef]
- Pappu, H.R.; Jones, R.A.; Jain, R.K. Global status of tospovirus epidemics in diverse cropping systems: Successes achieved and challenges ahead. Virus Res. 2009, 141, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Thieme, T.; Hoffmann, U.; Heimbach, U. Efficacy of plant protection substances against virus transmission by aphids infesting potato. J. Kult. 2009, 61, 21–30. [Google Scholar]
- Pilkington, L.J.; Messelink, G.; van Lenteren, J.C.; Le Mottee, K. Protected Biological Control–Biological pest management in the greenhouse industry. Biol. Control 2010, 52, 216–220. [Google Scholar] [CrossRef]
- Bragard, C.; Caciagli, P.; Lemaire, O.; López-Moya, J.J.; MacFarlane, S.; Peters, D.; Susi, P.; Torrance, L. Status and prospects of plant virus control through interference with vector transmission. Annu. Rev. Phytopathol. 2013, 51, 177–201. [Google Scholar] [CrossRef]
| Dates | Biological | Chemical 1 | Integrated 1 |
|---|---|---|---|
| 3 days before transplanting | Amblyseius swirskii (55 individuals/plant) | Amblyseius swirskii (55 individuals/plant) | |
| Weeks 1, 5 | acetamiprid + phosphoric soap | pymetrozine | |
| Weeks 2, 6 | pymetrozine + phosphoric soap | phosphoric soap | |
| Weeks 3, 7 | acetamiprid + phosphoric soap | phosphoric soap | |
| Week 4 | spirotetramat + azadirachtin | spirotetramat |
| Campaign | Greenhouses (n) | B. tabaci | A. swirskii | E. mundus | N. tenuis |
|---|---|---|---|---|---|
| 2015–2016 | 13 | 12 (92.3%) | 9 (69.2%) | 0 (0.0%) | 0 (0.0%) |
| 2016–2017 | 22 | 19 (86.4%) | 8 (36.4%) | 0 (0.0%) | 3 (13.6%) |
| 2017–2018 | 23 | 22 (95.6%) | 8 (34.8%) | 0 (0.0%) | 2 (8.7%) |
| Campaign | Greenhouses (n) | Pest Management (n (%)) | |
|---|---|---|---|
| Chemical | Integrated | ||
| 2015–2016 | 13 | 4 (30.8%) | 9 (69.2%) |
| 2016–2017 | 22 | 14 (63.6%) | 8 (36.4%) |
| 2017–2018 | 23 | 14 (60.9%) | 9 (39.1%) |
| Strategy | Mean | SE | 95% Confidence Interval (Wald) |
|---|---|---|---|
| Biological | 12.27a | 1.43 | 9.75–15.44 |
| Integrated | 24.46b | 2.86 | 19.45–30.78 |
| Chemical | 41.97c | 4.92 | 33.36–52.80 |
| Difference AUDPC Means | M1–M2 | 95% Confidence Interval (Wald) | t-Difference | p Value |
|---|---|---|---|---|
| Biological vs. Integrated | 3.36 | 1.02–7.77 | 1.607 | >0.0500 |
| Chemical vs. Integrated | 5.06 | 0.57–9.54 | 2.369 | 0.0292 |
| Chemical vs. Biological | 2.06 | −2.34–6.41 | 0.980 | >0.0500 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez, E.; Téllez, M.M.; Janssen, D. Whitefly Control Strategies against Tomato Leaf Curl New Delhi Virus in Greenhouse Zucchini. Int. J. Environ. Res. Public Health 2019, 16, 2673. https://doi.org/10.3390/ijerph16152673
Rodríguez E, Téllez MM, Janssen D. Whitefly Control Strategies against Tomato Leaf Curl New Delhi Virus in Greenhouse Zucchini. International Journal of Environmental Research and Public Health. 2019; 16(15):2673. https://doi.org/10.3390/ijerph16152673
Chicago/Turabian StyleRodríguez, Estefanía, Mª Mar Téllez, and Dirk Janssen. 2019. "Whitefly Control Strategies against Tomato Leaf Curl New Delhi Virus in Greenhouse Zucchini" International Journal of Environmental Research and Public Health 16, no. 15: 2673. https://doi.org/10.3390/ijerph16152673
APA StyleRodríguez, E., Téllez, M. M., & Janssen, D. (2019). Whitefly Control Strategies against Tomato Leaf Curl New Delhi Virus in Greenhouse Zucchini. International Journal of Environmental Research and Public Health, 16(15), 2673. https://doi.org/10.3390/ijerph16152673






