Relationship of Time-Activity-Adjusted Particle Number Concentration with Blood Pressure
Abstract
1. Introduction
2. Materials and Methods
2.1. Demographics and Health Data
2.2. Exposure Assessment
2.3. Statistical Analysis
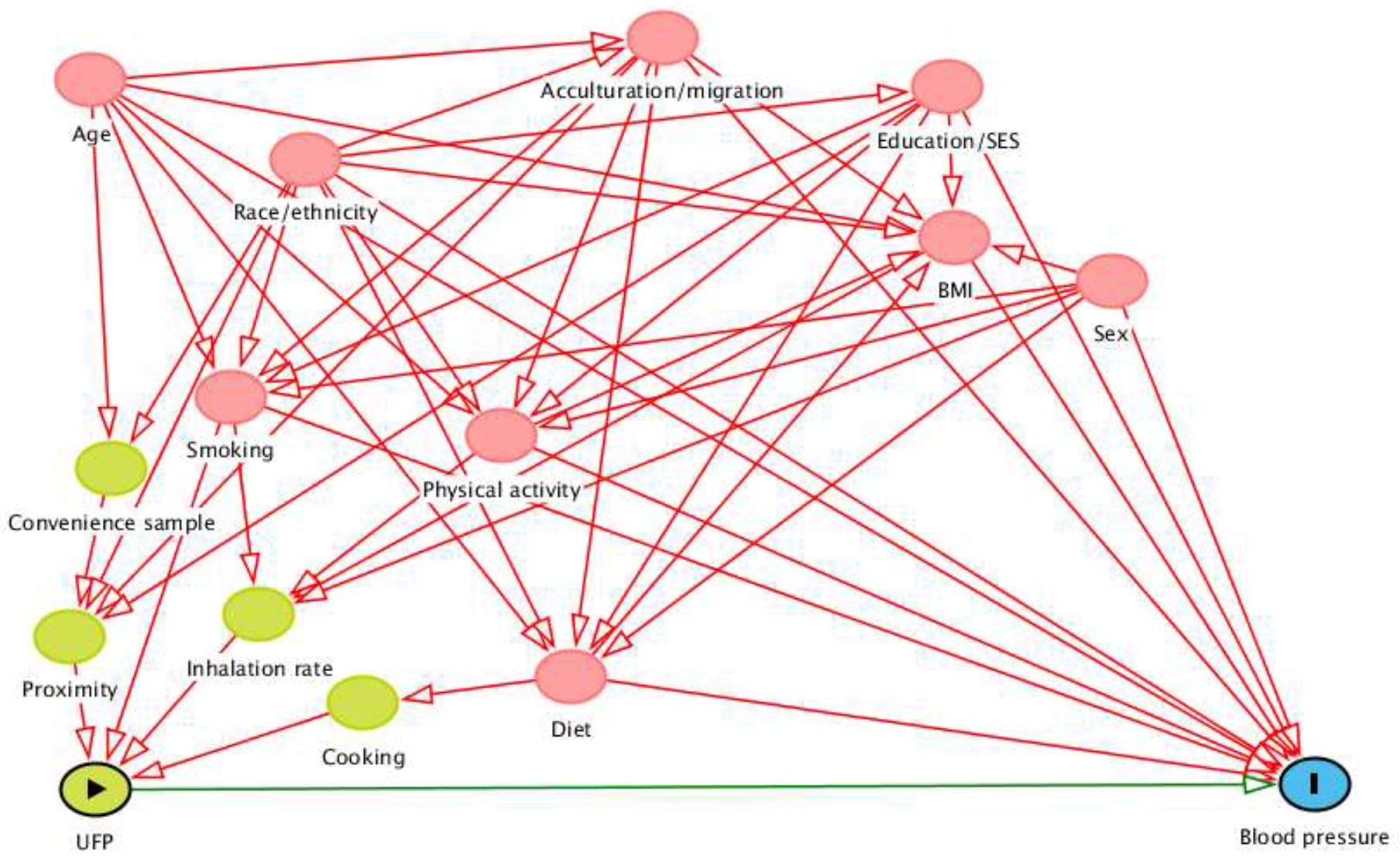
2.3.1. Conceptual Model
2.3.2. Primary Analysis and Sensitivity Analyses
2.3.3. Effect Modification
2.3.4. Consistency of Associations over Time
3. Results
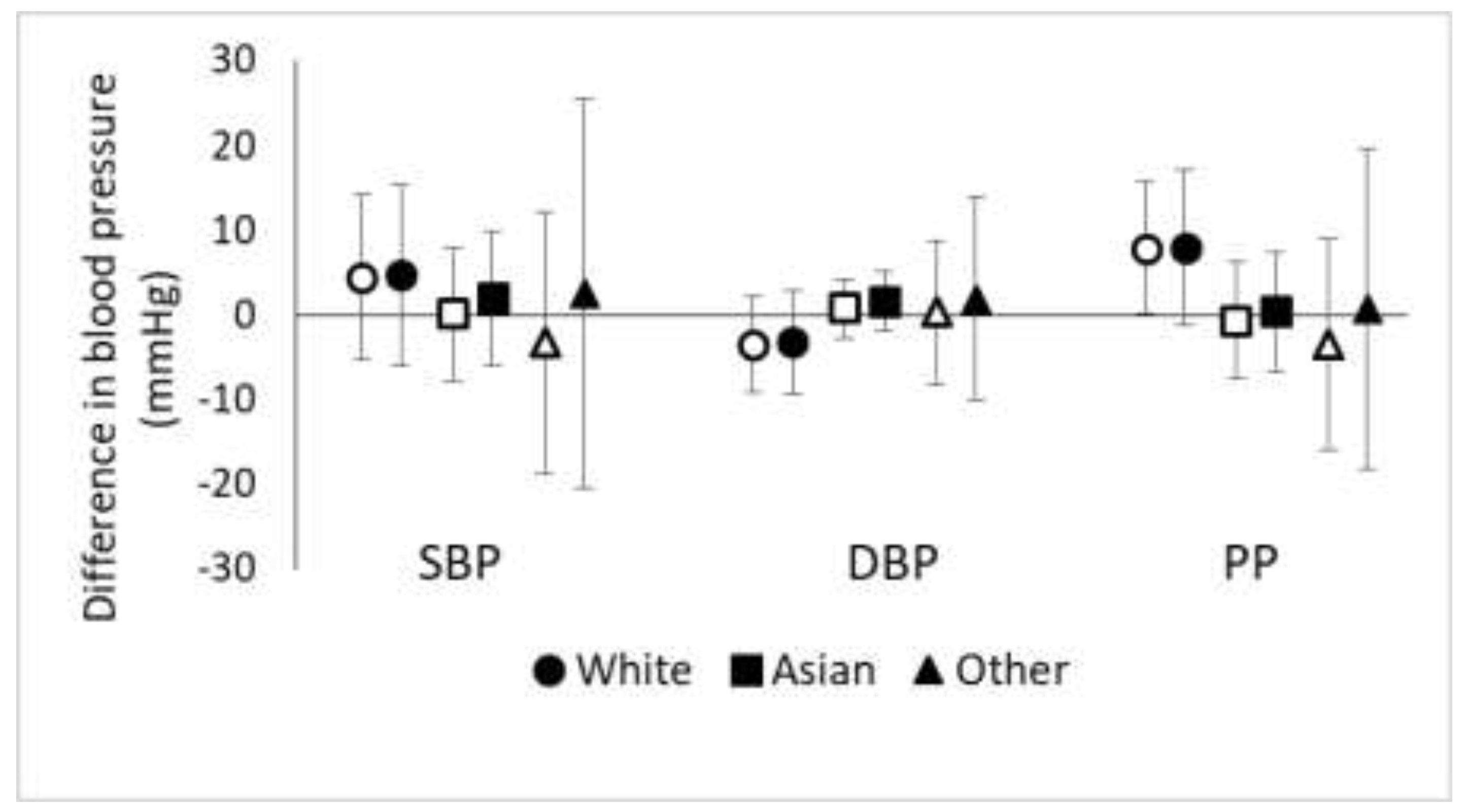
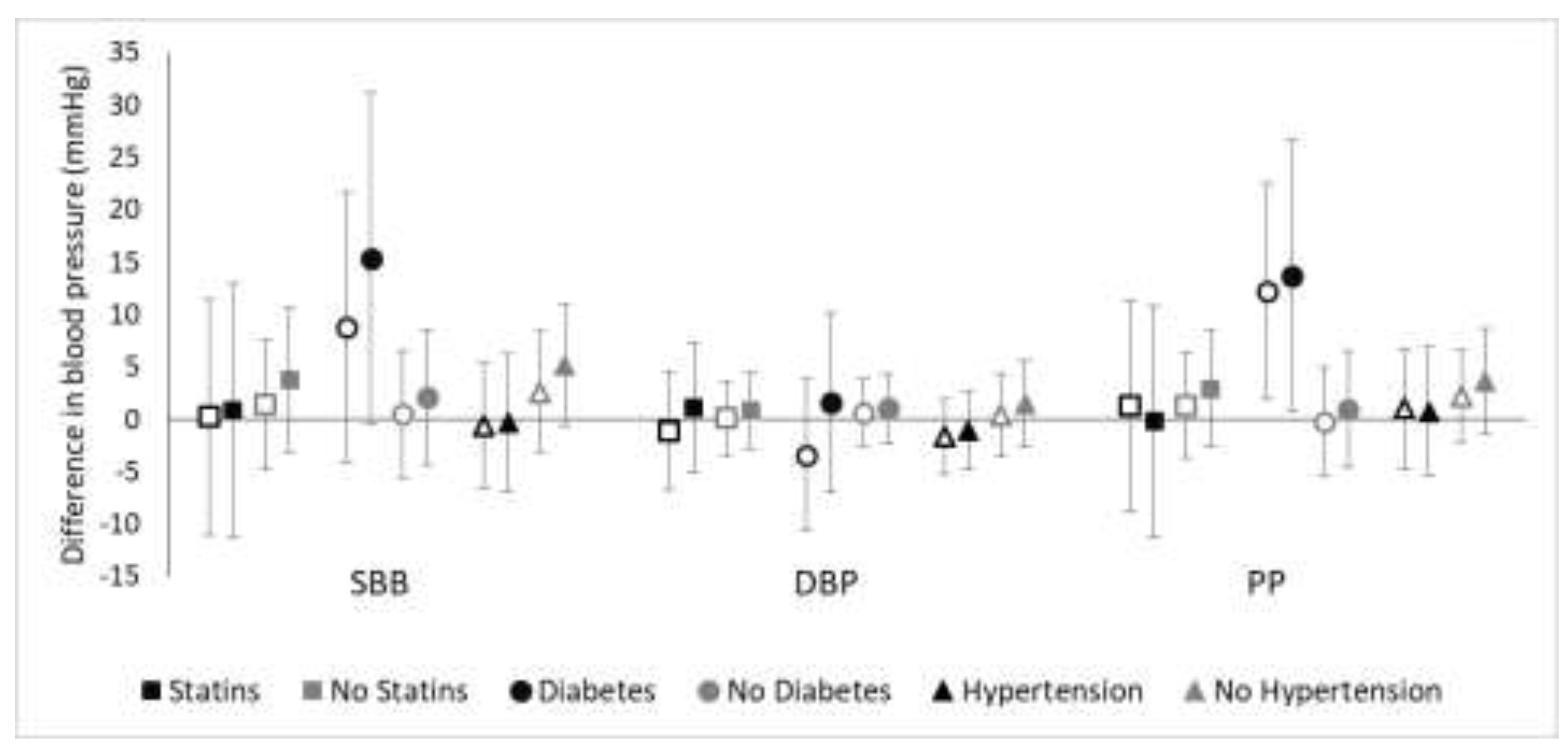
3.1. Effect Modification
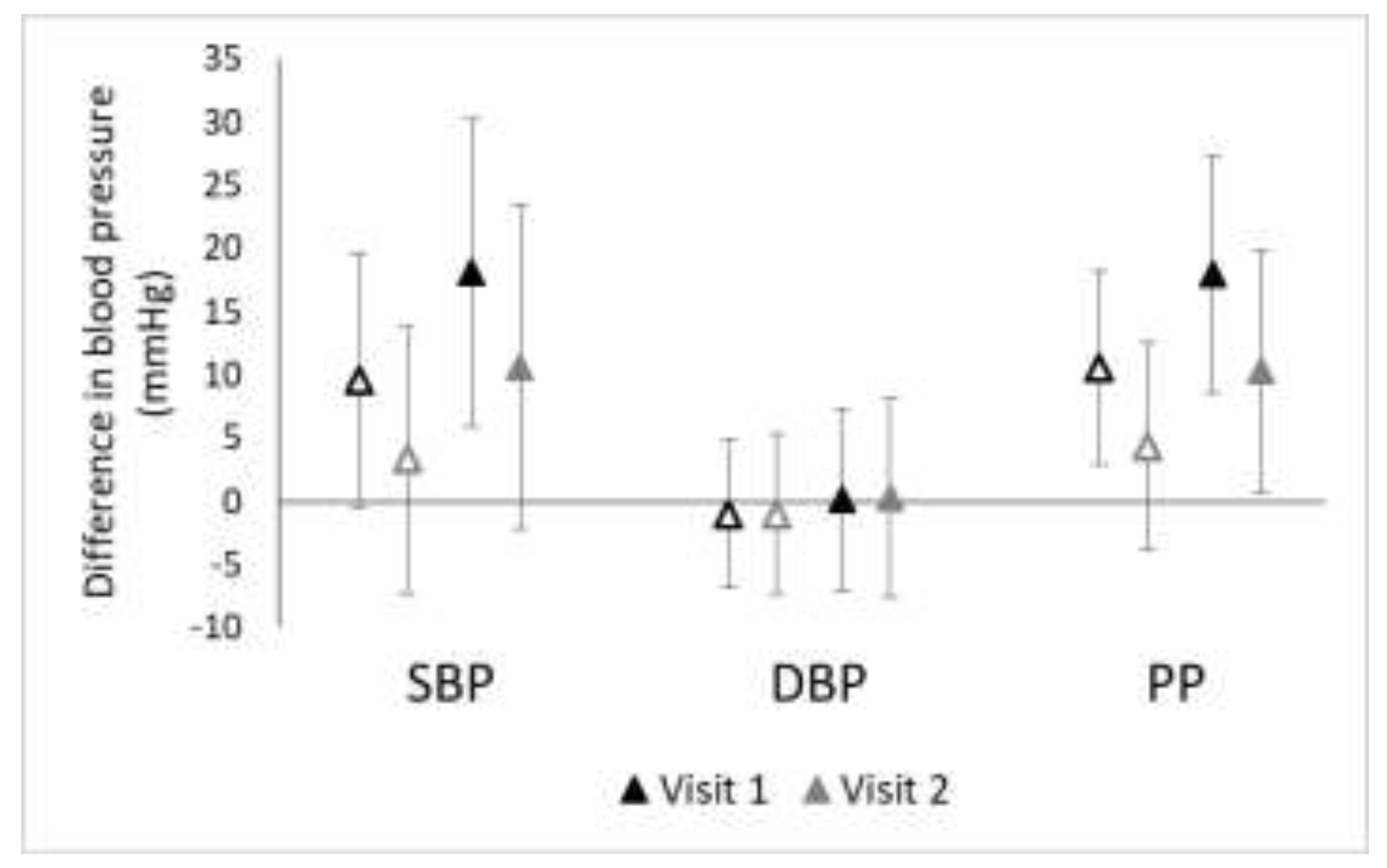
3.2. Consistency of Associations over Time
4. Discussion
5. Conclusions
Authors contributions
Funding
Acknowledgements
Conflicts of interest
Appendix A: Exposure Assessment
Appendix B: Directed Acyclic Graph Justification
References
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: An analysis of data from the Global Burden of Diseases Study 2015. The Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A.; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. American Heart Association Council on Epidemiology and Prevention, Council on the Kidney in Cardiovascular Disease, and Council on Nutrition, Physical Activity and Metabolism Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010, 121, 2331–2378. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.A.; Siscovick, D.S.; Sheppard, L.; Shepherd, K.; Sullivan, J.H.; Anderson, G.L.; Kaufman, J.D. Long-Term Exposure to Air Pollution and Incidence of Cardiovascular Events in Women. N. Engl. J. Med. 2007, 356, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Crouse, D.L.; Peters, P.A.; van Donkelaar, A.; Goldberg, M.S.; Villeneuve, P.J.; Brion, O.; Khan, S.; Atari, D.O.; Jerrett, M.; Pope, C.A.; et al. Risk of nonaccidental and cardiovascular mortality in relation to long-term exposure to low concentrations of fine particulate matter: A Canadian national-level cohort study. Environ. Health Perspect. 2012, 120, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Fuks, K.; Moebus, S.; Hertel, S.; Viehmann, A.; Nonnemacher, M.; Dragano, N.; Möhlenkamp, S.; Jakobs, H.; Kessler, C.; Erbel, R.; et al. Long-term urban particulate air pollution, traffic noise, and arterial blood pressure. Environ. Health Perspect. 2011, 119, 1706–1711. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Laden, F.; Forman, J.P.; Hart, J.E. Long-Term Exposure to Particulate Matter and Self-Reported Hypertension: A Prospective Analysis in the Nurses’ Health Study. Environ. Health Perspect. 2016, 124, 1414–1420. [Google Scholar] [CrossRef] [PubMed]
- Nemmar, A.; Hoet, P.H.M.; Vanquickenborne, B.; Dinsdale, D.; Thomeer, M.; Hoylaerts, M.F.; Vanbilloen, H.; Mortelmans, L.; Nemery, B. Passage of Inhaled Particles into the Blood Circulation in Humans. Circulation 2002, 105, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Kreyling, W.G.; Semmler-Behnke, M.; Möller, W. Ultrafine particle-lung interactions: Does size matter? J. Aerosol Med. Off. J. Int. Soc. Aerosols Med. 2006, 19, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G. Pulmonary effects of inhaled ultrafine particles. Int. Arch. Occup. Environ. Health 2000, 74, 1–8. [Google Scholar] [CrossRef]
- Delfino, R.J.; Sioutas, C.; Malik, S. Potential Role of Ultrafine Particles in Associations between Airborne Particle Mass and Cardiovascular Health. Environ. Health Perspect. 2005, 113, 934–946. [Google Scholar] [CrossRef] [PubMed]
- Araujo, J.A.; Barajas, B.; Kleinman, M.; Wang, X.; Bennett, B.J.; Gong, K.W.; Navab, M.; Harkema, J.; Sioutas, C.; Lusis, A.J.; et al. Ambient particulate pollutants in the ultrafine range promote early atherosclerosis and systemic oxidative stress. Circ. Res. 2008, 102, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Lawal, A.O. Air particulate matter induced oxidative stress and inflammation in cardiovascular disease and atherosclerosis: The role of Nrf2 and AhR-mediated pathways. Toxicol. Lett. 2017, 270, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Hu, J.; Goldberg, D.; Reynolds, P.; Hertz, A.; Bernstein, L.; Kleeman, M.J. Associations of mortality with long-term exposures to fine and ultrafine particles, species and sources: Results from the California Teachers Study Cohort. Environ. Health Perspect. 2015, 123, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, I.; Dratva, J.; Caviezel, S.; Burdet, L.; de Groot, E.; Ducret-Stich, R.E.; Eeftens, M.; Keidel, D.; Meier, R.; Perez, L.; et al. Particulate Matter and Subclinical Atherosclerosis: Associations between Different Particle Sizes and Sources with Carotid Intima-Media Thickness in the SAPALDIA Study. Environ. Health Perspect. 2016. [Google Scholar] [CrossRef] [PubMed]
- Viehmann, A.; Hertel, S.; Fuks, K.; Eisele, L.; Moebus, S.; Möhlenkamp, S.; Nonnemacher, M.; Jakobs, H.; Erbel, R.; Jöckel, K.-H.; et al. Long-term residential exposure to urban air pollution, and repeated measures of systemic blood markers of inflammation and coagulation. Occup. Environ. Med. 2015, 72, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Corlin, L.; Woodin, M.; Hart, J.; Simon, M.; Gute, D.; Tucker, K.; Durant, J.; Brugge, D. Longitudinal Associations of Long-term Exposure to Ultrafine Particles with Blood Pressure and Systemic Inflammation in Puerto Rican Adults. Environ. Health 2018, 17. [Google Scholar] [CrossRef] [PubMed]
- Pilz, V.; Wolf, K.; Breitner, S.; Rückerl, R.; Koenig, W.; Rathmann, W.; Cyrys, J.; Peters, A.; Schneider, A. C-reactive protein (CRP) and long-term air pollution with a focus on ultrafine particles. Int. J. Hyg. Environ. Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Chen, H.; Hatzopoulou, M.; Jerrett, M.; Kwong, J.C.; Burnett, R.T.; van Donkelaar, A.; Copes, R.; Martin, R.V.; Van Ryswyk, K.; et al. Exposure to Ambient Ultrafine Particles and Nitrogen Dioxide and Incident Hypertension and Diabetes. Epidemiology 2018, 29, 323. [Google Scholar] [CrossRef] [PubMed]
- Karner, A.A.; Eisinger, D.S.; Niemeier, D.A. Near-roadway air quality: Synthesizing the findings from real-world data. Environ. Sci. Technol. 2010, 44, 5334–5344. [Google Scholar] [CrossRef] [PubMed]
- Lane, K.J.; Levy, J.I.; Scammell, M.K.; Patton, A.P.; Durant, J.L.; Mwamburi, M.; Zamore, W.; Brugge, D. Effect of time-activity adjustment on exposure assessment for traffic-related ultrafine particles. J. Expos. Sci. Environ. Epid. 2015, 25, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Baldauf, R.W.; Devlin, R.B.; Gehr, P.; Giannelli, R.; Hassett-Sipple, B.; Jung, H.; Martini, G.; McDonald, J.; Sacks, J.D.; Walker, K. Ultrafine Particle Metrics and Research Considerations: Review of the 2015 UFP Workshop. Int. J. Environ. Res. Public. Health 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- HEI Understanding the Health Effects of Ambient UFP: HEI Review Panel on UFP. 2013. Available online: https://www.healtheffects.org/system/files/Perspectives3.pdf (accessed on 14 September 2018).
- Patton, A.P.; Zamore, W.; Naumova, E.N.; Levy, J.I.; Brugge, D.; Durant, J.L. Transferability and generalizability of regression models of ultrafine particles in urban neighborhoods in the Boston area. Environ. Sci. Technol. 2015, 49, 6051–6060. [Google Scholar] [CrossRef] [PubMed]
- Lane, K.J.; Levy, J.I.; Scammell, M.K.; Peters, J.L.; Patton, A.P.; Reisner, E.; Lowe, L.; Zamore, W.; Durant, J.L.; Brugge, D. Association of modeled long-term personal exposure to ultrafine particles with inflammatory and coagulation biomarkers. Environ. Int. 2016, 92–93, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lane, K.J.; Corlin, L.; Patton, A.P.; Durant, J.L.; Thanikachalam, M.; Woodin, M.; Wang, M.; Brugge, D. Association of Long-Term Near-Highway Exposure to Ultrafine Particles with Cardiovascular Diseases, Diabetes and Hypertension. Int. J. Environ. Res. Public. Health 2017, 14, 461. [Google Scholar] [CrossRef] [PubMed]
- Corlin, L.; Woodin, M.; Thanikachalam, M.; Lowe, L.; Brugge, D. Evidence for the healthy immigrant effect in older Chinese immigrants: A cross-sectional study. BMC Public Health 2014, 14, 603. [Google Scholar] [CrossRef] [PubMed]
- Miyata, R.; Hiraiwa, K.; Cheng, J.C.; Bai, N.; Vincent, R.; Francis, G.A.; Sin, D.D.; Van Eeden, S.F. Statins attenuate the development of atherosclerosis and endothelial dysfunction induced by exposure to urban particulate matter (PM10). Toxicol. Appl. Pharmacol. 2013, 272, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J.; Park, S.K.; O’Neill, M.S.; Vokonas, P.S.; Sparrow, D.; Weiss, S.; Kelsey, K. Glutathione-S-transferase M1, obesity, statins, and autonomic effects of particles: Gene-by-drug-by-environment interaction. Am. J. Respir. Crit. Care Med. 2005, 172, 1529–1533. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.H.; Patton, A.P.; Lane, K.; Laws, M.B.; Marden, A.; Carrasco, E.; Spengler, J.; Mwamburi, M.; Zamore, W.; Durant, J.L.; et al. A community participatory study of cardiovascular health and exposure to near-highway air pollution: Study design and methods. Rev. Environ. Health 2013, 28, 21–35. [Google Scholar] [CrossRef] [PubMed]
- NHIS Data, Questionnaires and Related Documentation. Available online: http://www.cdc.gov/nchs/nhis/data-questionnaires-documentation.htm (accessed on 30 November 2016).
- Padró-Martínez, L.T.; Patton, A.P.; Trull, J.B.; Zamore, W.; Brugge, D.; Durant, J.L. Mobile monitoring of particle number concentration and other traffic-related air pollutants in a near-highway neighborhood over the course of a year. Atmos. Environ. 2012, 61, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Patton, A.P.; Collins, C.; Naumova, E.N.; Zamore, W.; Brugge, D.; Durant, J.L. An hourly regression model for ultrafine particles in a near-highway urban area. Environ. Sci. Technol. 2014, 48, 3272–3280. [Google Scholar] [CrossRef] [PubMed]
- Lane, K.J.; Kangsen Scammell, M.; Levy, J.I.; Fuller, C.H.; Parambi, R.; Zamore, W.; Mwamburi, M.; Brugge, D. Positional error and time-activity patterns in near-highway proximity studies: An exposure misclassification analysis. Environ. Health 2013, 12, 75. [Google Scholar] [CrossRef] [PubMed]
- Greenland, S.; Pearl, J.; Robins, J.M. Causal diagrams for epidemiologic research. Epidemiology 1999, 10, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Franklin, S.S.; Gustin, W.; Wong, N.D.; Larson, M.G.; Weber, M.A.; Kannel, W.B.; Levy, D. Hemodynamic Patterns of Age-Related Changes in Blood Pressure. Circulation 1997, 96, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Daher, N.; Hasheminassab, S.; Shafer, M.; Schauer, J.; Sioutas, C. Seasonal and spatial variability in chemical composition and mass closure of ambient ultrafine particles in the megacity of Los Angeles. Environ. Sci. Process. Impacts 2013, 15, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Saffari, A.; Daher, N.; Shafer, M.M.; Schauer, J.J.; Sioutas, C. Seasonal and spatial variation of trace elements and metals in quasi-ultrafine (PM0.25) particles in the Los Angeles metropolitan area and characterization of their sources. Environ. Pollut. 2013, 181, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Woodhouse, P.R.; Khaw, K.T.; Plummer, M. Seasonal variation of blood pressure and its relationship to ambient temperature in an elderly population. J. Hypertens. 1993, 11, 1267–1274. [Google Scholar] [CrossRef] [PubMed]
- Modesti, P.A. Season, temperature and blood pressure: A complex interaction. Eur. J. Intern. Med. 2013, 24, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Mujahid, M.S.; Diez Roux, A.V.; Morenoff, J.D.; Raghunathan, T.E.; Cooper, R.S.; Ni, H.; Shea, S. Neighborhood characteristics and hypertension. Epidemiol. Camb. Mass 2008, 19, 590–598. [Google Scholar] [CrossRef] [PubMed]
- Dubowitz, T.; Ghosh-Dastidar, M.; Eibner, C.; Slaughter, M.E.; Fernandes, M.; Whitsel, E.A.; Bird, C.E.; Jewell, A.; Margolis, K.L.; Li, W.; et al. The Women’s Health Initiative: The Food Environment, Neighborhood Socioeconomic Status, BMI, and Blood Pressure. Obesity 2012, 20, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Harmer, P.; Cardinal, B.J.; Vongjaturapat, N. Built environment and changes in blood pressure in middle aged and older adults. Prev. Med. 2009, 48, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.; Wang, D.D.; Rizzo, A.M.; Gachette, D.; Delnord, M.; Parambi, R.; Kang, C.-M.; Brugge, D. Association of PNC, BC, and PM2.5 measured at a central monitoring site with blood pressure in a predominantly near highway population. Int. J. Environ. Res. Public. Health 2015, 12, 2765–2780. [Google Scholar] [CrossRef] [PubMed]
- Patton, A.P.; Perkins, J.; Zamore, W.; Levy, J.I.; Brugge, D.; Durant, J.L. Spatial and temporal differences in traffic-related air pollution in three urban neighborhoods near an interstate highway. Atmos. Environ. 2014, 99, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Aronow, W.S.; Fleg, J.L.; Pepine, C.J.; Artinian, N.T.; Bakris, G.; Brown, A.S.; Ferdinand, K.C.; Ann Forciea, M.; Frishman, W.H.; Jaigobin, C.; et al. ACCF/AHA 2011 expert consensus document on hypertension in the elderly: A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents developed in collaboration with the American Academy of Neurology, American Geriatrics Society, American Society for Preventive Cardiology, American Society of Hypertension, American Society of Nephrology, Association of Black Cardiologists, and European Society of Hypertension. J. Am. Soc. Hypertens. JASH 2011, 5, 259–352. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Brands, M.W.; Daniels, S.R.; Karanja, N.; Elmer, P.J.; Sacks, F.M. American Heart Association Dietary approaches to prevent and treat hypertension: A scientific statement from the American Heart Association. Hypertens. Dallas Tex 1979 2006, 47, 296–308. [Google Scholar] [CrossRef]
- Havranek, E.P.; Mujahid, M.S.; Barr, D.A.; Blair, I.V.; Cohen, M.S.; Cruz-Flores, S.; Davey-Smith, G.; Dennison-Himmelfarb, C.R.; Lauer, M.S.; Lockwood, D.W.; et al. Social Determinants of Risk and Outcomes for Cardiovascular Disease: A Scientific Statement from the American Heart Association. Circulation 2015, 132, 873–898. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; He, J.; Appel, L.J.; Cutler, J.A.; Havas, S.; Kotchen, T.A.; Roccella, E.J.; Stout, R.; Vallbona, C.; Winston, M.C.; et al. National High Blood Pressure Education Program Coordinating Committee Primary prevention of hypertension: Clinical and public health advisory from The National High Blood Pressure Education Program. JAMA 2002, 288, 1882–1888. [Google Scholar] [CrossRef] [PubMed]
- Guh, D.P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C.L.; Anis, A.H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 2009, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Strazzullo, P.; Kerry, S.M.; Barbato, A.; Versiero, M.; D’Elia, L.; Cappuccio, F.P. Do statins reduce blood pressure? A meta-analysis of randomized, controlled trials. Hypertens. Dallas Tex 1979 2007, 49, 792–798. [Google Scholar] [CrossRef] [PubMed]
- Linneberg, A.; Jacobsen, R.K.; Skaaby, T.; Taylor, A.E.; Fluharty, M.E.; Jeppesen, J.L.; Bjorngaard, J.H.; Åsvold, B.O.; Gabrielsen, M.E.; Campbell, A.; et al. Effect of Smoking on Blood Pressure and Resting Heart Rate: A Mendelian Randomization Meta-Analysis in the CARTA Consortium. Circ. Cardiovasc. Genet. 2015, 8, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Dolezsar, C.M.; McGrath, J.J.; Herzig, A.J.M.; Miller, S.B. Perceived racial discrimination and hypertension: A comprehensive systematic review. Health Psychol. Off. J. Div. Health Psychol. Am. Psychol. Assoc. 2014, 33, 20–34. [Google Scholar] [CrossRef] [PubMed]
- Kurian, A.K.; Cardarelli, K.M. Racial and ethnic differences in cardiovascular disease risk factors: A systematic review. Ethn. Dis. 2007, 17, 143–152. [Google Scholar] [PubMed]
- Freis, E.D. Age, race, sex and other indices of risk in hypertension. Am. J. Med. 1973, 55, 275–280. [Google Scholar] [CrossRef]
- Gillum, R.F. Pathophysiology of hypertension in blacks and whites. A review of the basis of racial blood pressure differences. Hypertension 1979, 1, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Winkleby, M.A.; Jatulis, D.E.; Frank, E.; Fortmann, S.P. Socioeconomic status and health: How education, income, and occupation contribute to risk factors for cardiovascular disease. Am. J. Public Health 1992, 82, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, G.A.; Keil, J.E. Socioeconomic factors and cardiovascular disease: A review of the literature. Circulation 1993, 88, 1973–1998. [Google Scholar] [CrossRef] [PubMed]
- Sowers, J.R.; Epstein, M. Diabetes mellitus and associated hypertension, vascular disease, and nephropathy. An update. Hypertens. Dallas Tex 1979 1995, 26, 869–879. [Google Scholar]
- Virdis, A.; Giannarelli, C.; Neves, M.F.; Taddei, S.; Ghiadoni, L. Cigarette smoking and hypertension. Curr. Pharm. Des. 2010, 16, 2518–2525. [Google Scholar] [CrossRef] [PubMed]
- Narkiewicz, K.; Kjeldsen, S.E.; Hedner, T. Is smoking a causative factor of hypertension? Blood Press. 2005, 14, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, T. The effect of migration on hypertension and other cardiovascular risk factors: A review. J. Am. Soc. Hypertens. JASH 2014, 8, 171–191. [Google Scholar] [CrossRef] [PubMed]
- Vasan, R.S.; Larson, M.G.; Leip, E.P.; Kannel, W.B.; Levy, D. Assessment of frequency of progression to hypertension in non-hypertensive participants in the Framingham Heart Study: A cohort study. The Lancet 2001, 358, 1682–1686. [Google Scholar] [CrossRef]
- Forman, J.P.; Stampfer, M.J.; Curhan, G.C. Diet and lifestyle risk factors associated with incident hypertension in women. JAMA 2009, 302, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Conen, D.; Glynn, R.J.; Ridker, P.M.; Buring, J.E.; Albert, M.A. Socioeconomic status, blood pressure progression, and incident hypertension in a prospective cohort of female health professionals. Eur. Heart J. 2009, 30, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Bowman, T.S.; Gaziano, J.M.; Buring, J.E.; Sesso, H.D. A prospective study of cigarette smoking and risk of incident hypertension in women. J. Am. Coll. Cardiol. 2007, 50, 2085–2092. [Google Scholar] [CrossRef] [PubMed]
- Niskanen, L.; Laaksonen, D.E.; Nyyssönen, K.; Punnonen, K.; Valkonen, V.-P.; Fuentes, R.; Tuomainen, T.-P.; Salonen, R.; Salonen, J.T. Inflammation, abdominal obesity, and smoking as predictors of hypertension. Hypertens. Dallas Tex 1979 2004, 44, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Crump, C.; Sundquist, J.; Winkleby, M.A.; Sundquist, K. Interactive Effects of Physical Fitness and Body Mass Index on the Risk of Hypertension. JAMA Intern. Med. 2016, 176, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Wallace, B.C.; Small, K.; Brodley, C.E.; Lau, J.; Trikalinos, T.A. Deploying an interactive machine learning system in an evidence-based practice center: Abstrackr. In Proceedings of the 2nd ACM SIGHIT International Health Informatics Symposium (IHI), Miami, FL, USA, 28–30 January 2012. [Google Scholar]
- Abrams, J.; Vela, B.S.; Coultas, D.B.; Samaan, S.A.; Malhotra, D.; Roche, R.J. Coronary risk factors and their modification: Lipids, smoking, hypertension, estrogen, and the elderly. Curr. Probl. Cardiol. 1995, 20, 533–610. [Google Scholar] [CrossRef]
- Alam, S.; Johnson, A.G. A meta-analysis of randomised controlled trials (RCT) among healthy normotensive and essential hypertensive elderly patients to determine the effect of high salt (NaCl) diet of blood pressure. J. Hum. Hypertens. 1999, 13, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Chang, J.; Chen, C.; Li, P.; Yang, K.; Chi, I. Investigating the effect of transcendental meditation on blood pressure: A systematic review and meta-analysis. J. Hum. Hypertens. 2015, 29, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Bønaa, K. Epidemiological and intervention studies on the effect of marine polyunsaturated fatty acids on blood pressure. J. Intern. Med. Suppl. 1989, 731, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, V.A.; Fagard, R.H. Effect of resistance training on resting blood pressure: A meta-analysis of randomized controlled trials. J. Hypertens. 2005, 23, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, V.A.; Fagard, R.H.; Coeckelberghs, E.; Vanhees, L. Impact of resistance training on blood pressure and other cardiovascular risk factors: A meta-analysis of randomized, controlled trials. Hypertens. Dallas Tex 1979 2011, 58, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Gasperin, D.; Netuveli, G.; Dias-da-Costa, J.S.; Pattussi, M.P. Effect of psychological stress on blood pressure increase: A meta-analysis of cohort studies. Cad. Saude Publica 2009, 25, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Ha, V.; Sievenpiper, J.L.; de Souza, R.J.; Chiavaroli, L.; Wang, D.D.; Cozma, A.I.; Mirrahimi, A.; Yu, M.E.; Carleton, A.J.; Dibuono, M.; et al. Effect of fructose on blood pressure: A systematic review and meta-analysis of controlled feeding trials. Hypertens. Dallas Tex 1979 2012, 59, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Hay, M. Sex, the brain and hypertension: Brain oestrogen receptors and high blood pressure risk factors. Clin. Sci. Lond. Engl. 1979 2016, 130, 9–18. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Whelton, P.K. Effect of dietary fiber and protein intake on blood pressure: A review of epidemiologic evidence. Clin. Exp. Hypertens. 1999, 21, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Hertz-Picciotto, I.; Croft, J. Review of the Relation between Blood Lead and Blood Pressure. Epidemiol. Rev. 1993, 15, 352–373. [Google Scholar] [CrossRef] [PubMed]
- Jee, S.H.; He, J.; Whelton, P.K.; Suh, I.; Klag, M.J. The effect of chronic coffee drinking on blood pressure: A meta-analysis of controlled clinical trials. Hypertens. Dallas Tex 1979 1999, 33, 647–652. [Google Scholar] [CrossRef]
- Jee, S.H.; Miller, E.R.; Guallar, E.; Singh, V.K.; Appel, L.J.; Klag, M.J. The effect of magnesium supplementation on blood pressure: A meta-analysis of randomized clinical trials. Am. J. Hypertens. 2002, 15, 691–696. [Google Scholar] [CrossRef]
- Johansen, H.L. Hypertension in Canada: Risk factor review and recommendations for further work. Can. J. Public Health Rev. Can. Sante Publique 1983, 74, 123–128. [Google Scholar]
- Kaufman, J.; Barkey, N. Hypertension in Africa: An overview of prevalence rates and causal risk factors. Ethn. Dis. 1993, 3 Suppl, S83–S101. [Google Scholar]
- Li, G.; Zhang, Y.; Thabane, L.; Mbuagbaw, L.; Liu, A.; Levine, M.A.H.; Holbrook, A. Effect of green tea supplementation on blood pressure among overweight and obese adults: A systematic review and meta-analysis. J. Hypertens. 2015, 33, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Luft, F.C.; Miller, J.Z.; Lyle, R.M.; Melby, C.L.; Fineberg, N.S.; McCarron, D.A.; Weinberger, M.H.; Morris, C.D. The effect of dietary interventions to reduce blood pressure in normal humans. J. Am. Coll. Nutr. 1989, 8, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Mesas, A.E.; Leon-Muñoz, L.M.; Rodriguez-Artalejo, F.; Lopez-Garcia, E. The effect of coffee on blood pressure and cardiovascular disease in hypertensive individuals: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2011, 94, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Millard, P.S.; Triplekorn, C.M. Effect of reduced dietary sodium on blood pressure. J. Fam. Pract. 1996, 43, 123–124. [Google Scholar] [PubMed]
- Murphy, M.H.; Nevill, A.M.; Murtagh, E.M.; Holder, R.L. The effect of walking on fitness, fatness and resting blood pressure: A meta-analysis of randomised, controlled trials. Prev. Med. 2007, 44, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Naing, C.; Aung, K. Prevalence and risk factors of hypertension in myanmar: A systematic review and meta-analysis. Medicine (Baltimore) 2014, 93, e100. [Google Scholar] [CrossRef] [PubMed]
- Nissensohn, M.; Román-Viñas, B.; Sánchez-Villegas, A.; Piscopo, S.; Serra-Majem, L. The Effect of the Mediterranean Diet on Hypertension: A Systematic Review and Meta-Analysis. J. Nutr. Educ. Behav. 2016, 48, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Sullivan, T.R.; Fakler, P.; Frank, O.R.; Stocks, N.P. Effect of cocoa on blood pressure. Cochrane Database Syst. Rev. 2012, CD008893. [Google Scholar] [CrossRef]
- Roberts, C.; Banning, M. Risk factors for hypertension and cardiovascular disease. Nurs. Stand. 1998, 39–42. [Google Scholar]
- Steffen, P.R.; Smith, T.B.; Larson, M.; Butler, L. Acculturation to Western society as a risk factor for high blood pressure: A meta-analytic review. Psychosom. Med. 2006, 68, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Steffen, M.; Kuhle, C.; Hensrud, D.; Erwin, P.J.; Murad, M.H. The effect of coffee consumption on blood pressure and the development of hypertension: A systematic review and meta-analysis. J. Hypertens. 2012, 30, 2245–2254. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Moran, A.E.; Liu, J.; Qi, Y.; Xie, W.; Tzong, K.; Zhao, D. A Meta-Analysis of Effect of Dietary Salt Restriction on Blood Pressure in Chinese Adults. Glob. Heart 2015, 10, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Whelton, S.P.; Chin, A.; Xin, X.; He, J. Effect of aerobic exercise on blood pressure: A meta-analysis of randomized, controlled trials. Ann. Intern. Med. 2002, 136, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.-Y.; Qin, L.-Q.; Wang, P.-Y.; Li, W.; Chang, C. Effect of milk tripeptides on blood pressure: A meta-analysis of randomized controlled trials. Nutr. Burbank Los Angel. Cty. Calif 2008, 24, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Residential Proximity to Major Highways—United States, 2010. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/su6203a8.htm (accessed on 20 July 2018).
- US EPA National Center for Environmental Assessment, W.D.; Moya, J. Exposure Factors Handbook 2011 Edition (Final Report). Available online: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=236252 (accessed on 20 July 2018).
- Buonanno, G.; Stabile, L.; Morawska, L. Personal exposure to ultrafine particles: The influence of time-activity patterns. Sci. Total Environ. 2014, 468–469, 903–907. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Marini, S.; Morawska, L.; Fuoco, F.C. Individual dose and exposure of Italian children to ultrafine particles. Sci. Total Environ. 2012, 438, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Morawska, L.; Afshari, A.; Bae, G.N.; Buonanno, G.; Chao, C.Y.H.; Hänninen, O.; Hofmann, W.; Isaxon, C.; Jayaratne, E.R.; Pasanen, P.; et al. Indoor aerosols: From personal exposure to risk assessment. Indoor Air 2013, 23, 462–487. [Google Scholar] [CrossRef] [PubMed]
- Wallace, L.; Ott, W. Personal exposure to ultrafine particles. J. Expo. Sci. Environ. Epidemiol. 2011, 21, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Int Panis, L.; de Geus, B.; Vandenbulcke, G.; Willems, H.; Degraeuwe, B.; Bleux, N.; Mishra, V.; Thomas, I.; Meeusen, R. Exposure to particulate matter in traffic: A comparison of cyclists and car passengers. Atmos. Environ. 2010, 44, 2263–2270. [Google Scholar] [CrossRef]
- Kelly, F. Oxidative stress: Its role in air pollution and adverse health effects. Occup. Environ. Med. 2003, 60, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Braniš, M.; Šafránek, J.; Hytychová, A. Exposure of children to airborne particulate matter of different size fractions during indoor physical education at school. Build. Environ. 2009, 44, 1246–1252. [Google Scholar] [CrossRef]
- Dennekamp, M.; Howarth, S.; Dick, C. a. J.; Cherrie, J.W.; Donaldson, K.; Seaton, A. Ultrafine particles and nitrogen oxides generated by gas and electric cooking. Occup. Environ. Med. 2001, 58, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Salant, T.; Lauderdale, D.S. Measuring culture: A critical review of acculturation and health in Asian immigrant populations. Soc. Sci. Med. 2003, 57, 71–90. [Google Scholar] [CrossRef]
- Oza-Frank, R.; Cunningham, S.A. The weight of US residence among immigrants: A systematic review. Obes. Rev. 2010, 11, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Ayala; Guadalupe, X.; Baquero, B.; Klinger, S. A Systematic Review of the Relationship between Acculturation and Diet among Latinos in the United States: Implications for Future Research. J. Am. Diet. Assoc. 2008, 108, 1330–1344. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Rankin, S.; Stewart, A.; Oka, R. Effects of acculturation on smoking behavior in Asian Americans: A meta-analysis. J. Cardiovasc. Nurs. 2008, 23, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Serafica, R.C. Dietary acculturation in Asian Americans. J. Cult. Divers. 2014, 21, 145–151. [Google Scholar] [PubMed]
- Afable-Munsuz, A.; Ponce, N.A.; Rodriguez, M.; Perez-Stable, E.J. Immigrant generation and physical activity among Mexican, Chinese & Filipino adults in the U.S. Soc. Sci. Med. 2010, 70, 1997–2005. [Google Scholar] [CrossRef] [PubMed]
- Gerber, M.; Barker, D.; Pühse, U. Acculturation and physical activity among immigrants: A systematic review. J. Public Health 2012, 20, 313–341. [Google Scholar] [CrossRef]
- Gorman, B.K.; Lariscy, J.T.; Kaushik, C. Gender, acculturation, and smoking behavior among U.S. Asian and Latino immigrants. Soc. Sci. Med. 2014, 106, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Jamal, A. Current Cigarette Smoking Among Adults—United States, 2005–2015. MMWR Morb. Mortal. Wkly. Rep. 2016, 65. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vaznaugh, E.V.; Kawachi, I.; Subramanian, S.V.; Sánchez, B.N.; Acevedo-Garcia, D. Differential effect of birthplace and length of residence on body mass index (BMI) by education, gender and race/ethnicity. Soc. Sci. Med. 2008, 67, 1300–1310. [Google Scholar] [CrossRef] [PubMed]
- Flegal, K.M.; Carroll, M.D.; Kit, B.K.; Ogden, C.L. Prevalence of Obesity and Trends in the Distribution of Body Mass Index Among US Adults, 1999-2010. JAMA 2012, 307, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Chastin, S.F.M.; Buck, C.; Freiberger, E.; Murphy, M.; Brug, J.; Cardon, G.; O’Donoghue, G.; Pigeot, I.; Oppert, J.-M. Systematic literature review of determinants of sedentary behaviour in older adults: A DEDIPAC study. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 127. [Google Scholar] [CrossRef] [PubMed]
- Trost, S.G.; Owen, N.; Bauman, A.E.; Sallis, J.F.; Brown, W. Correlates of adults’ participation in physical activity: Review and update. Med. Sci. Sports Exerc. 2002, 34, 1996–2001. [Google Scholar] [CrossRef] [PubMed]
- Wareham, N.J.; van Sluijs, E.M.F.; Ekelund, U. Physical activity and obesity prevention: A review of the current evidence. Proc. Nutr. Soc. 2005, 64, 229–247. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. CMAJ 2006, 174, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Freisling, H.; Knaze, V.; Slimani, N. A Systematic Review of Peer-Reviewed Studies on Diet Quality Indexes Applied to Old Age: A Multitude of Predictors of Diet Quality. In Diet Quality; Nutrition and Health; Humana Press: New York, NY, USA, 2013; pp. 365–381. ISBN 978-1-4614-7314-5. [Google Scholar]
- Satia-Abouta, J.; Patterson, R.E.; Kristal, A.R.; Teh, C.; Tu, S.-P. Psychosocial Predictors of Diet and Acculturation in Chinese American and Chinese Canadian Women. Ethn. Health 2002, 7, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Paeratakul, S.; Popkin, B.M.; Keyou, G.; Adair, L.S.; Stevens, J. Changes in diet and physical activity affect the body mass index of Chinese adults. Int. J. Obes. 1998, 22, 424–431. [Google Scholar] [CrossRef]
- Weiss, D.R.; O’Loughlin, J.L.; Platt, R.W.; Paradis, G. Five-year predictors of physical activity decline among adults in low-income communities: A prospective study. Int. J. Behav. Nutr. Phys. Act. 2007, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Roman, B.; Estruch, R. Scientific Evidence of Interventions Using the Mediterranean Diet: A Systematic Review. Nutr. Rev. 2006, 64, S27–S47. [Google Scholar] [CrossRef] [PubMed]
- Villareal, D.T.; Apovian, C.M.; Kushner, R.F.; Klein, S. Obesity in Older Adults: Technical Review and Position Statement of the American Society for Nutrition and NAASO, The Obesity Society. Obes. Res. 13, 1849–1863. [CrossRef] [PubMed]
- Morenga, L.T.; Mallard, S.; Mann, J. Dietary sugars and body weight: Systematic review and meta-analyses of randomised controlled trials and cohort studies. BMJ 2013, 346, e7492. [Google Scholar] [CrossRef] [PubMed]
- Soltani, S.; Shirani, F.; Chitsazi, M.J.; Salehi-Abargouei, A. The effect of dietary approaches to stop hypertension (DASH) diet on weight and body composition in adults: A systematic review and meta-analysis of randomized controlled clinical trials. Obes. Rev. 17, 442–454. [CrossRef] [PubMed]
- Sodowsky, G.R.; Lai, E.W.M.; Plake, B.S. Moderating Effects of Sociocultural Variables on Acculturation Attitudes of Hispanics and Asian Americans. J. Couns. Dev. 70, 194–204. [CrossRef]
- Textor, J.; Hardt, J.; Knüpppel, S. DAGitty: A Graphical Tool for Analyzing Causal Diagrams. Epidemiology 2011, 5, 745. [Google Scholar] [CrossRef] [PubMed]
| Total | White | Asian | Other | |||||
|---|---|---|---|---|---|---|---|---|
| Characteristic | n | % (n) or mean (sd) | n | % (n) or mean (sd) | N | % (n) or mean (sd) | N | % (n) or mean (sd) |
| TAA-PNC * (particles/cm3) | 409 | 22,000 (6500) | 178 | 20,000 (4900) | 149 | 24,000 (7900) | 82 | 21,000 (5000) |
| ln[(TAA-PNC) (particles/cm3)] | 409 | 9.9 (0.35) | 178 | 9.8 (0.28) | 149 | 10.0 (0.43) | 82 | 9.9 (0.27) |
| SBP (mmHg) | 409 | 137.5 (19.5) | 178 | 133.9 (18.3) | 149 | 141.2 (20.6) | 82 | 138.9 (18.8) |
| DBP (mmHg) | 409 | 77.7 (10.3) | 178 | 76.2 (10.7) | 149 | 77.3 (9.3) | 82 | 81.9 (10.4) |
| PP (mmHg) | 409 | 59.8 (16.5) | 178 | 57.6 (14.9) | 149 | 63.9 (18.4) | 82 | 57.1 (15.1) |
| Hypertension | 409 | 63.8 (261) | 178 | 55.6 (99) | 149 | 69.8 (104) | 82 | 70.7 (58) |
| Age (years) | 409 | 61.5 (12.8) | 178 | 59.8 (11.3) | 149 | 66.6 (13.4) | 82 | 56.0 (11.3) |
| BMI (kg/m2) | 393 | 27.7 (6.8) | 168 | 29.5 (6.9) | 149 | 24.1 (4.1) | 76 | 30.6 (7.7) |
| ln[light/moderate physical activity (min/week)] | 374 | 4.3 (2.2) | 164 | 3.8 (2.3) | 147 | 5.1 (1.6) | 63 | 3.5 (2.3) |
| Female | 409 | 59.2 (242) | 178 | 59.6 (106) | 149 | 56.4 (84) | 82 | 63.4 (52) |
| Smoker status | 398 | 176 | 145 | 77 | ||||
| Current | 21.1 (84) | 22.2 (39) | 14.5 (21) | 31.2 (24) | ||||
| Former | 30.7 (122) | 43.2 (76) | 15.9 (23) | 29.9 (23) | ||||
| Never | 48.2 (192) | 34.7 (61) | 69.7 (101) | 39.0 (30) | ||||
| Fruit and vegetable consumption ≥ 7x/week | 275 | 38.2 (105) | 125 | 48.0 (60) | 97 | 30.9 (30) | 53 | 28.3 (15) |
| Fried food consumption ≥ 1x/week | 405 | 33.8 (137) | 176 | 45.5 (80) | 149 | 14.8 (22) | 80 | 43.8 (35) |
| Educational Attainment | 409 | 178 | 149 | 82 | ||||
| < HS | 34.2 (140) | 11.2 (20) | 61.7 (92) | 34.2 (28) | ||||
| HS | 31.8 (130) | 36.5 (65) | 24.2 (36) | 35.4 (29) | ||||
| >HS | 34.0 (139) | 52.3 (93) | 14.1 (21) | 30.5 (25) | ||||
| Born in the USA | 404 | 45.5 (184) | 174 | 86.8 (151) | 149 | 0.0 (0) | 81 | 40.7 (33) |
| Statin Medications | 400 | 29.0 (116) | 176 | 31.3 (55) | 144 | 29.2 (42) | 80 | 23.8 (19) |
| Hypertension medications | 400 | 45.0 (180) | 176 | 35.8 (63) | 144 | 54.2 (78) | 80 | 48.8 (39) |
| Diabetes | 399 | 20.3 (81) | 175 | 17.7 (31) | 144 | 18.8 (27) | 80 | 28.8 (23) |
| Model | SBP (mmHg) | DBP (mmHg) | PP (mmHg) | Hypertension |
|---|---|---|---|---|
| β (95% CI) | β (95% CI) | β (95% CI) | OR (95% CI) | |
| Model A | 2.87 (−2.60, 8.33) | −0.18 (−3.09, 2.72) | 3.05 (−1.58, 7.68) | 1.53 (0.86, 2.72) |
| Model B | 5.23 (−0.68, 11.14) | 0.96 (−2.08, 4.00) | 4.27 (−0.79, 9.32) | 1.81 (0.94, 3.48) |
| Model C | 5.84 (−1.94, 13.61) | 1.79 (−1.95, 5.52) | 4.05 (−2.80, 10.90) | 1.53 (0.68, 3.43) |
| Model D | 2.41 (−3.51, 8.32) | 0.15 (−2.92, 3.21) | 2.26 (−2.83, 7.35) | 1.27 (0.64, 2.52) |
| Model E | 3.60 (−1.75, 8.95) | 1.10 (−1.95, 4.15) | 2.50 (−1.74, 6.74) | 1.72 (0.84, 3.55) |
| Model F | 1.67 (−3.87, 7.22) | 0.01 (−3.15, 3.18) | 1.66 (−2.78, 6.11) | 1.25 (0.57, 2.75) |
| Model G | 1.68 (−3.89, 7.24) | −0.19 (−3.35, 2.97) | 1.86 (−2.58, 6.31) | 1.31 (0.59, 2.91) |
| Model H | 5.67 (−0.40, 11.75) | 1.35 (−1.76, 4.45) | 4.33 (−0.88, 9.53) | 1.86 (0.95, 3.66) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corlin, L.; Ball, S.; Woodin, M.; Patton, A.P.; Lane, K.; Durant, J.L.; Brugge, D. Relationship of Time-Activity-Adjusted Particle Number Concentration with Blood Pressure. Int. J. Environ. Res. Public Health 2018, 15, 2036. https://doi.org/10.3390/ijerph15092036
Corlin L, Ball S, Woodin M, Patton AP, Lane K, Durant JL, Brugge D. Relationship of Time-Activity-Adjusted Particle Number Concentration with Blood Pressure. International Journal of Environmental Research and Public Health. 2018; 15(9):2036. https://doi.org/10.3390/ijerph15092036
Chicago/Turabian StyleCorlin, Laura, Shannon Ball, Mark Woodin, Allison P. Patton, Kevin Lane, John L. Durant, and Doug Brugge. 2018. "Relationship of Time-Activity-Adjusted Particle Number Concentration with Blood Pressure" International Journal of Environmental Research and Public Health 15, no. 9: 2036. https://doi.org/10.3390/ijerph15092036
APA StyleCorlin, L., Ball, S., Woodin, M., Patton, A. P., Lane, K., Durant, J. L., & Brugge, D. (2018). Relationship of Time-Activity-Adjusted Particle Number Concentration with Blood Pressure. International Journal of Environmental Research and Public Health, 15(9), 2036. https://doi.org/10.3390/ijerph15092036






