Seasonal Patterns in the Prevalence and Diversity of Tick-Borne Borrelia burgdorferi Sensu Lato, Anaplasma phagocytophilum and Rickettsia spp. in an Urban Temperate Forest in South Western Slovakia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area, Tick Sampling and Host Trapping
2.2. DNA Extraction and PCR Detection of Bacteria
2.3. Detection of Borrelia burgdorferi s.l.
2.4. Detection of A. phagocytophilum
2.5. Detection of R. helvetica and R. monacensis
2.6. Statistical Analysis
2.7. Nucleotide Sequence Accession Numbers
3. Results
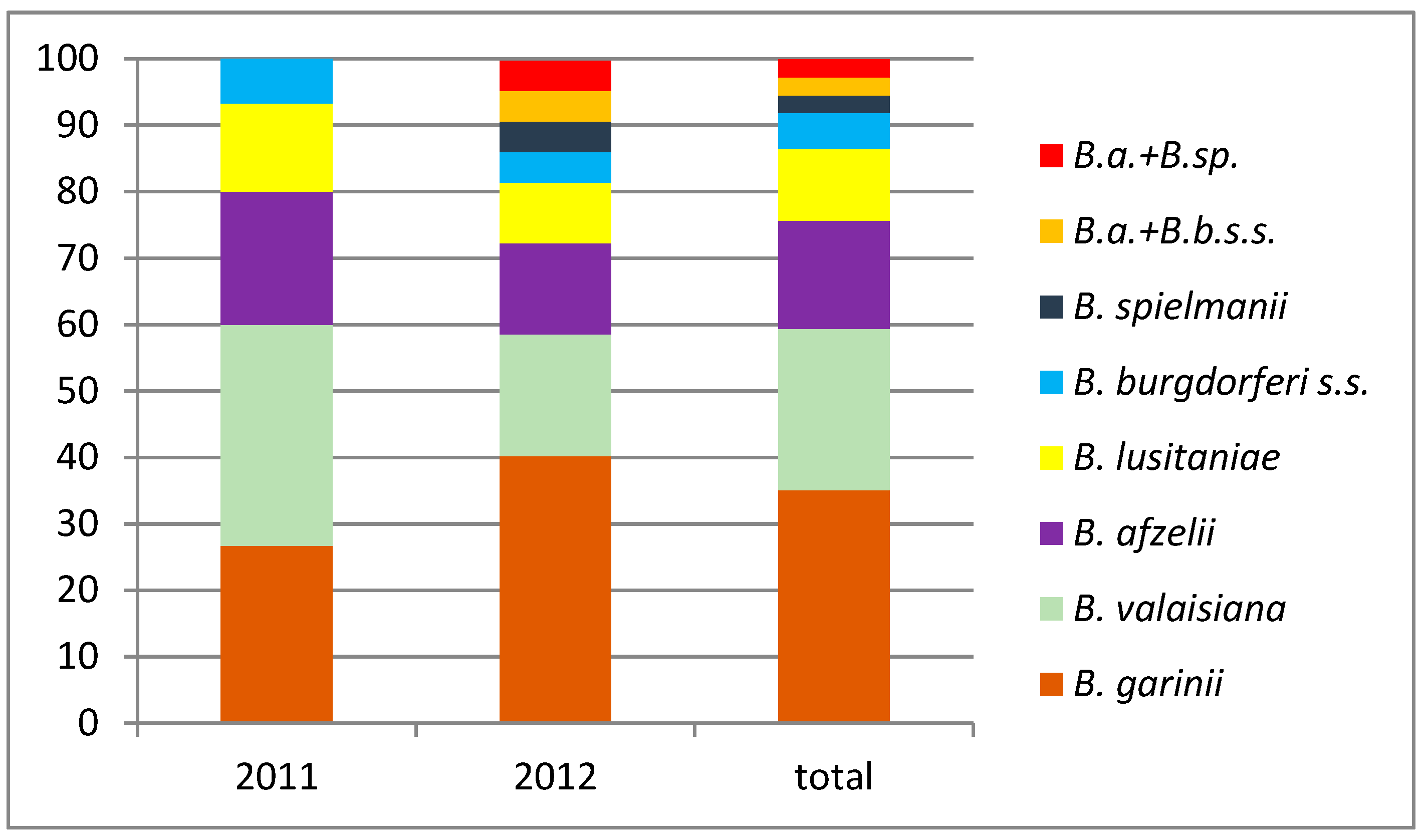
3.1. Pathogens in Questing I. ricinus
3.2. Pathogens in Bird-Feeding I. ricinus
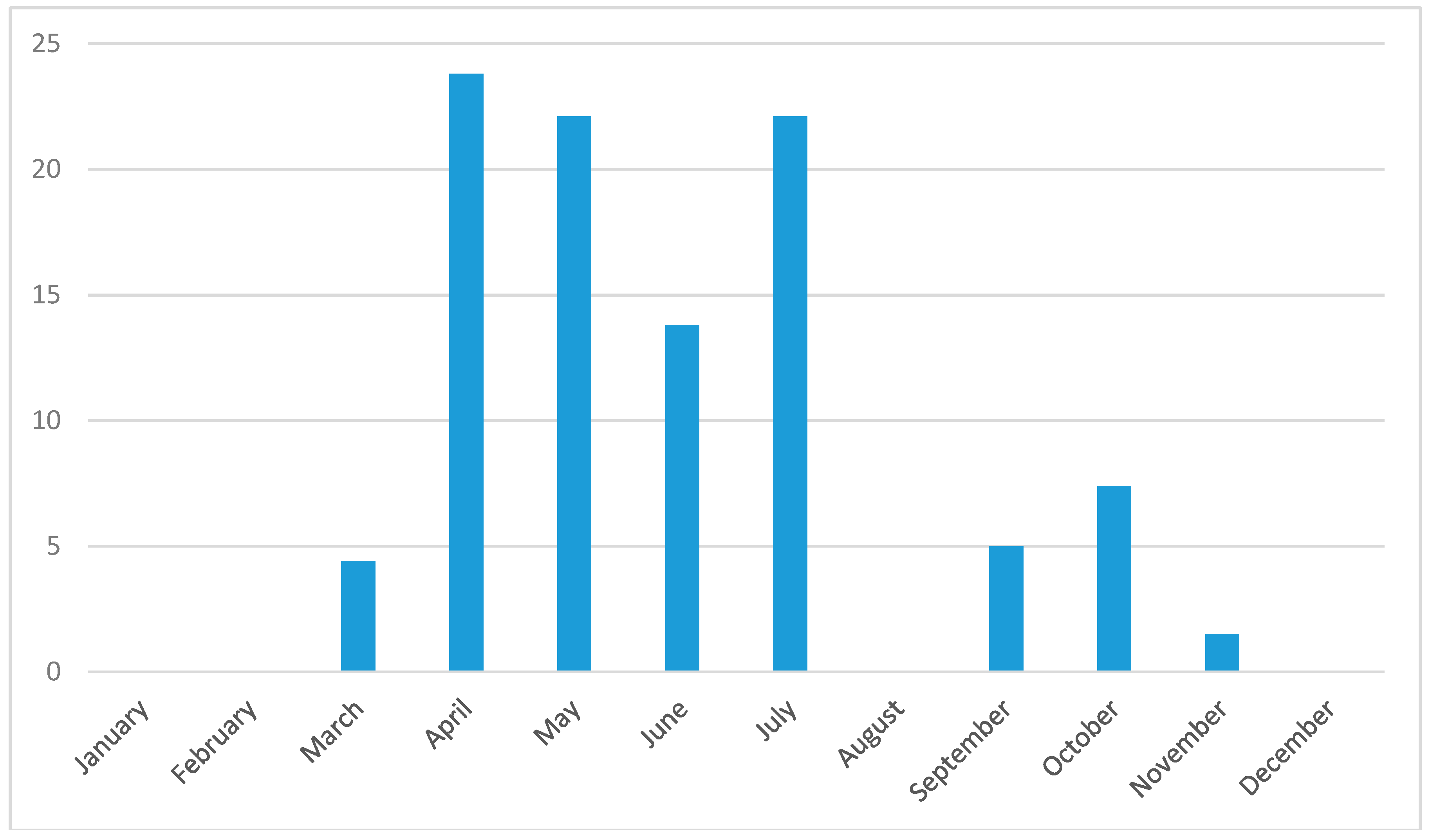
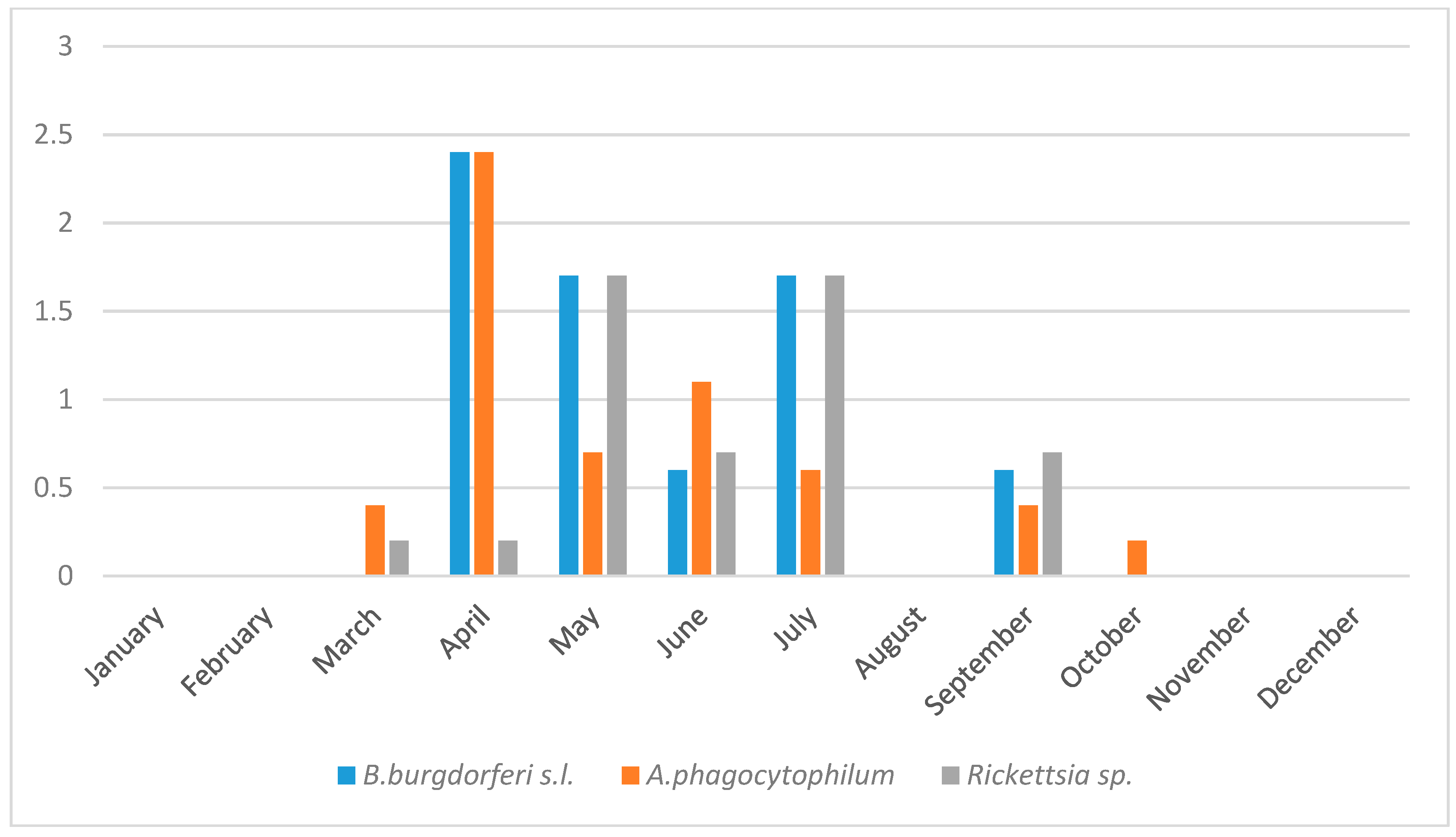
3.3. Seasonality in Tick Abundance and Pathogen Occurrence
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Rizzoli, A.; Silaghi, C.; Obiegala, A.; Rudolf, I.; Hubalek, Z.; Foldvari, G.; Plantard, O.; Vayssier-Taussat, M.; Bonnet, S.; Spitalska, E.; et al. Ixodes ricinus and Its Transmitted Pathogens in Urban and Peri-Urban Areas in Europe: New Hazards and Relevance for Public Health. Front. Public Health 2014, 2, 251. [Google Scholar] [CrossRef] [PubMed]
- Foldvari, G.; Rigo, K.; Jablonszky, M.; Biro, N.; Majoros, G.; Molnar, V.; Toth, M. Ticks and the city: Ectoparasites of the Northern white-breasted hedgehog (Erinaceus roumanicus) in an urban park. Ticks Tick Borne Dis. 2011, 2, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Foldvari, G.; Jahfari, S.; Rigo, K.; Jablonszky, M.; Szekeres, S.; Majoros, G.; Toth, M.; Molnar, V.; Coipan, E.C.; Sprong, H. Candidatus Neoehrlichia mikurensis and Anaplasma phagocytophilum in urban hedgehogs. Emerg. Infect. Dis. 2014, 20, 496–498. [Google Scholar] [CrossRef] [PubMed]
- Keesing, F.; Belden, L.K.; Daszak, P.; Dobson, A.; Harvell, C.D.; Holt, R.D.; Hudson, P.; Jolles, A.; Jones, K.E.; Mitchell, C.E.; et al. Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 2010, 468, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.R.; Fraser-Liggett, C.M.; Mongodin, E.F.; Qiu, W.G.; Dunn, J.J.; Luft, B.J.; Schutzer, S.E. Whole genome sequence of an unusual Borrelia burgdorferi sensu lato isolate. J. Bacteriol. 2011, 193, 1489–1490. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, L.B.; Tomova, A.; Gonzalez-Acuna, D.; Murua, R.; Moreno, C.X.; Hernandez, C.; Cabello, J.; Cabello, C.; Daniels, T.J.; Godfrey, H.P.; et al. Borrelia chilensis, a new member of the Borrelia burgdorferi sensu lato complex that extends the range of this genospecies in the Southern Hemisphere. Environ. Microbiol. 2014, 16, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Vollmer, S.A.; Cornet, M.; Garnier, M.; Fingerle, V.; Wilske, B.; Bormane, A.; Vitorino, L.; Collares-Pereira, M.; Drancourt, M.; et al. A new Borrelia species defined by multilocus sequence analysis of housekeeping genes. Appl. Environ. Microbiol. 2009, 75, 5410–5416. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Hojgaard, A.; Lane, R.S.; Cornet, M.; Fingerle, V.; Rudenko, N.; Ogden, N.; Aanensen, D.M.; Fish, D.; Piesman, J. Multilocus sequence analysis of Borrelia bissettii strains from North America reveals a new Borrelia species, Borrelia kurtenbachii. Ticks Tick Borne Dis. 2010, 1, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Postic, D.; Garnier, M.; Baranton, G. Multilocus sequence analysis of atypical Borrelia burgdorferi sensu lato isolates—Description of Borrelia californiensis sp. nov., and genomospecies 1 and 2. Int. J. Med. Microbiol. 2007, 297, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Postic, D.; Sertour, N.; Livey, I.; Matuschka, F.R.; Baranton, G. Delineation of Borrelia burgdorferi sensu lato species by multilocus sequence analysis and confirmation of the delineation of Borrelia spielmanii sp. nov. Int. J. Syst. Evol. Microbiol. 2006, 56, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Borrelia carolinensis sp. nov., a new (14th) member of the Borrelia burgdorferi Sensu Lato complex from the southeastern region of the United States. J. Clin. Microbiol. 2009, 47, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Lin, T.; Gao, L.; Grubhoffer, L.; Oliver, J.H., Jr. Delineation of a new species of the Borrelia burgdorferi Sensu Lato Complex, Borrelia americana sp. nov. J. Clin. Microbiol. 2009, 47, 3875–3880. [Google Scholar] [CrossRef] [PubMed]
- Rauter, C.; Hartung, T. Prevalence of Borrelia burgdorferi sensu lato genospecies in Ixodes ricinus ticks in Europe: A metaanalysis. Appl. Environ. Microbiol. 2005, 71, 7203–7216. [Google Scholar] [CrossRef] [PubMed]
- Strnad, M.; Honig, V.; Ruzek, D.; Grubhoffer, L.; Rego, R.O.M. Europe-Wide Meta-Analysis of Borrelia burgdorferi Sensu lato Prevalence in Questing Ixodes ricinus Ticks. Appl. Environ. Microbiol. 2017, 83, e00609-17. [Google Scholar] [CrossRef] [PubMed]
- Pangracova, L.; Derdakova, M.; Pekarik, L.; Hviscova, I.; Vichova, B.; Stanko, M.; Hlavata, H.; Petko, B. Ixodes ricinus abundance and its infection with the tick-borne pathogens in urban and suburban areas of Eastern Slovakia. Parasit. Vectors 2013, 6, 238. [Google Scholar] [CrossRef] [PubMed]
- Taragelova, V.R.; Mahrikova, L.; Selyemova, D.; Vaclav, R.; Derdakova, M. Natural foci of Borrelia lusitaniae in a mountain region of Central Europe. Ticks Tick Borne Dis. 2016, 7, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Bazovska, S.; Durovska, J.; Derdakova, M.; Taragelova, V.; Pancak, J.; Zaborska, M.; Traubner, P. The genospecies B. burgdorferi s.l., isolated from ticks and from neurological patients with suspected Lyme borreliosis. Neuro Endocrinol. Lett. 2011, 32, 491–495. [Google Scholar] [PubMed]
- Lencakova, D.; Hizo-Teufel, C.; Petko, B.; Schulte-Spechtel, U.; Stanko, M.; Wilske, B.; Fingerle, V. Prevalence of Borrelia burgdorferi s.l. OspA types in Ixodes ricinus ticks from selected localities in Slovakia and Poland. Int. J. Med. Microbiol. 2006, 296 (Suppl. 40), 108–118. [Google Scholar] [CrossRef] [PubMed]
- Hanincova, K.; Schafer, S.M.; Etti, S.; Sewell, H.S.; Taragelova, V.; Ziak, D.; Labuda, M.; Kurtenbach, K. Association of Borrelia afzelii with rodents in Europe. Parasitology 2003, 126, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Humair, P.F.; Gern, L. Relationship between Borrelia burgdorferi sensu lato species, red squirrels (Sciurus vulgaris) and Ixodes ricinus in enzootic areas in Switzerland. Acta Trop. 1998, 69, 213–227. [Google Scholar] [CrossRef]
- Humair, P.F.; Rais, O.; Gern, L. Transmission of Borrelia afzelii from Apodemus mice and Clethrionomys voles to Ixodes ricinus ticks: Differential transmission pattern and overwintering maintenance. Parasitology 1999, 118, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Huegli, D.; Hu, C.M.; Humair, P.F.; Wilske, B.; Gern, L. Apodemus species mice are reservoir hosts of Borrelia garinii OspA serotype 4 in Switzerland. J. Clin. Microbiol. 2002, 40, 4735–4737. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Schlee, D.B.; Allgower, R.; Matuschka, F.R. Relationships of a novel Lyme disease spirochete, Borrelia spielmani sp. nov., with its hosts in Central Europe. Appl. Environ. Microbiol. 2004, 70, 6414–6419. [Google Scholar] [CrossRef] [PubMed]
- Skuballa, J.; Petney, T.; Pfaffle, M.; Oehme, R.; Hartelt, K.; Fingerle, V.; Kimmig, P.; Taraschewski, H. Occurrence of different Borrelia burgdorferi sensu lato genospecies including B. afzelii, B. bavariensis, and B. spielmanii in hedgehogs (Erinaceus spp.) in Europe. Ticks Tick Borne Dis. 2012, 3, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Richter, D.; Schlee, D.B.; Matuschka, F.R. Reservoir competence of various rodents for the lyme disease Spirochete Borrelia spielmanii. Appl. Environ. Microbiol. 2011, 77, 3565–3570. [Google Scholar] [CrossRef] [PubMed]
- Taragelova, V.; Koci, J.; Hanincova, K.; Kurtenbach, K.; Derdakova, M.; Ogden, N.; Literak, I.; Kocianova, E.; Labuda, M. Blackbirds and song thrushes constitute a key reservoir of B. garinii, the causative agent of borreliosis in central Europe. Appl. Environ. Microbiol. 2008, 74, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Poupon, M.A.; Lommano, E.; Humair, P.F.; Douet, V.; Rais, O.; Schaad, M.; Jenni, L.; Gern, L. Prevalence of Borrelia burgdorferi sensu lato in ticks collected from migratory birds in Switzerland. Appl. Environ. Microbiol. 2006, 72, 976–979. [Google Scholar] [CrossRef] [PubMed]
- Majlathova, V.; Majlath, I.; Derdakova, M.; Vichova, B.; Pet’ko, B. Borrelia lusitaniae and green lizards (Lacerta viridis), Karst Region, Slovakia. Emerg. Infect. Dis. 2006, 12, 1895–1901. [Google Scholar] [CrossRef] [PubMed]
- Derdakova, M.; Stefancikova, A.; Spitalska, E.; Taragelova, V.; Kostalova, T.; Hrklova, G.; Kybicova, K.; Schanilec, P.; Majlathova, V.; Varady, M.; et al. Emergence and genetic variability of Anaplasma species in small ruminants and ticks from Central Europe. Vet. Microbiol. 2011, 153, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Derdakova, M.; Vaclav, R.; Pangracova-Blanarova, L.; Selyemova, D.; Koci, J.; Walder, G.; Spitalska, E. Candidatus Neoehrlichia mikurensis and its co-circulation with Anaplasma phagocytophilum in Ixodes ricinus ticks across ecologically different habitats of Central Europe. Parasit. Vectors 2014, 7, 160. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, G.; Sekeyova, Z.; Raoult, D.; Mediannikov, O. Multiple tick-associated bacteria in Ixodes ricinus from Slovakia. Ticks Tick Borne Dis. 2012, 3, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Vichova, B.; Majlathova, V.; Novakova, M.; Stanko, M.; Hviscova, I.; Pangracova, L.; Chrudimsky, T.; Curlik, J.; Petko, B. Anaplasma infections in ticks and reservoir host from Slovakia. Infect. Genet. Evol. 2014, 22, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Massung, R.F.; Levin, M.L.; Miller, N.J.; Mather, T.N. Reservoir competency of goats for Anaplasma phagocytophilum. Ann. N. Y. Acad. Sci. 2006, 1078, 476–478. [Google Scholar] [CrossRef] [PubMed]
- Stuen, S. Anaplasma phagocytophilum—The most widespread tick-borne infection in animals in Europe. Vet. Res. Commun. 2007, 31 (Suppl. 1), 79–84. [Google Scholar] [CrossRef] [PubMed]
- Stuen, S.; Pettersen, K.S.; Granquist, E.G.; Bergstrom, K.; Bown, K.J.; Birtles, R.J. Anaplasma phagocytophilum variants in sympatric red deer (Cervus elaphus) and sheep in southern Norway. Ticks Tick Borne Dis. 2013, 4, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Stuen, S.; Granquist, E.G.; Silaghi, C. Anaplasma phagocytophilum—A widespread multi-host pathogen with highly adaptive strategies. Front. Cell. Infect. Microbiol. 2013, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Blanarova, L.; Stanko, M.; Carpi, G.; Miklisova, D.; Vichova, B.; Mosansky, L.; Bona, M.; Derdakova, M. Distinct Anaplasma phagocytophilum genotypes associated with Ixodes trianguliceps ticks and rodents in Central Europe. Ticks Tick Borne Dis. 2014, 5, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Bown, K.J.; Begon, M.; Bennett, M.; Woldehiwet, Z.; Ogden, N.H. Seasonal dynamics of Anaplasma phagocytophila in a rodent-tick (Ixodes trianguliceps) system, United Kingdom. Emerg. Infect. Dis. 2003, 9, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Rosef, O.; Paulauskas, A.; Radzijevskaja, J. Prevalence of Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in questing Ixodes ricinus ticks in relation to the density of wild cervids. Acta Vet. Scand. 2009, 51, 47. [Google Scholar] [CrossRef] [PubMed]
- Jado, I.; Oteo, J.A.; Aldamiz, M.; Gil, H.; Escudero, R.; Ibarra, V.; Portu, J.; Portillo, A.; Lezaun, M.J.; Garcia-Amil, C.; et al. Rickettsia monacensis and human disease, Spain. Emerg. Infect. Dis. 2007, 13, 1405–1407. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, K.; Lindquist, O.; Pahlson, C. Association of Rickettsia helvetica with chronic perimyocarditis in sudden cardiac death. Lancet 1999, 354, 1169–1173. [Google Scholar] [CrossRef]
- Oteo, J.A.; Portillo, A. Tick-borne rickettsioses in Europe. Ticks Tick Borne Dis. 2012, 3, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Spitalska, E.; Boldis, V.; Derdakova, M.; Selyemova, D.; Rusnakova Taragelova, V. Rickettsial infection in Ixodes ricinus ticks in urban and natural habitats of Slovakia. Ticks Tick Borne Dis. 2014, 5, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Spitalska, E.; Stanko, M.; Mosansky, L.; Kraljik, J.; Miklisova, D.; Mahrikova, L.; Bona, M.; Kazimirova, M. Seasonal analysis of Rickettsia species in ticks in an agricultural site of Slovakia. Exp. Appl. Acarol. 2016, 68, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Spitalska, E.; Boldis, V.; Kostanova, Z.; Kocianova, E.; Stefanidesova, K. Incidence of various tick-borne microorganisms in rodents and ticks of central Slovakia. Acta Virol. 2008, 52, 175–179. [Google Scholar] [PubMed]
- Stefanidesova, K.; Kocianova, E.; Boldis, V.; Kostanova, Z.; Kanka, P.; Spitalska, E. Evidence of Anaplasma phagocytophilum and Rickettsia helvetica infection in free-ranging ungulates in central Slovakia. Eur. J. Wildl. Res. 2008, 54, 519–524. [Google Scholar] [CrossRef]
- Matuschka, F.R.; Fischer, P.; Musgrave, K.; Richter, D.; Spielman, A. Hosts on which nymphal Ixodes ricinus most abundantly feed. Am. J. Trop. Med. Hyg. 1991, 44, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Mejlon, H.A.; Jaenson, T.G.T. Questing behaviour of Ixodes ricinus ticks. Exp. Appl. Acarol. 1997, 21, 747–754. [Google Scholar] [CrossRef]
- Norman, R.; Bowers, R.G.; Begon, M.; Hudson, P.J. Persistence of tick-borne virus in the presence of multiple host species: Tick reservoirs and parasite mediated competition. J. Theor. Biol. 1999, 200, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Begon, M. Effects of Host Diversity on Disease Dynamics; Princeton University Press: Princeton, NJ, USA, 2008. [Google Scholar]
- Ogden, N.H.; Tsao, J.I. Biodiversity and Lyme disease: Dilution or amplification? Epidemics 2009, 1, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Jaenson, T.G.T.; Talleklint, L. Incompetence of Roe Deer as Reservoirs of the Lyme Borreliosis Spirochete. J. Med. Entomol. 1992, 29, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Kurtenbach, K.; De Michelis, S.; Etti, S.; Schafer, S.M.; Sewell, H.S.; Brade, V.; Kraiczy, P. Host association of Borrelia burgdorferi sensu lato—The key role of host complement. Trends Microbiol. 2002, 10, 74–79. [Google Scholar] [CrossRef]
- Wood, C.L.; Lafferty, K.D. Biodiversity and disease: A synthesis of ecological perspectives on Lyme disease transmission. Trends Ecol. Evol. 2013, 28, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Ostfeld, R.S.; Keesing, F. Biodiversity and disease risk: The case of Lyme disease. Conserv. Biol. 2000, 14, 722–728. [Google Scholar] [CrossRef]
- Randolph, S.E.; Dobson, A.D. Pangloss revisited: A critique of the dilution effect and the biodiversity-buffers-disease paradigm. Parasitology 2012, 139, 847–863. [Google Scholar] [CrossRef] [PubMed]
- Mysterud, A.; Easterday, W.R.; Qviller, L.; Viljugrein, H.; Ytrehus, B. Spatial and seasonal variation in the prevalence of Anaplasma phagocytophilum and Borrelia burgdorferi sensu lato in questing Ixodes ricinus ticks in Norway. Parasites Vectors 2013, 6, 187. [Google Scholar] [CrossRef] [PubMed]
- Gill, F.B. Ornithology, 3rd ed.; W. H. Freeman and Company: New York, NY, USA, 2007. [Google Scholar]
- Svitalkova, Z.; Harustiakova, D.; Mahrikova, L.; Berthova, L.; Slovak, M.; Kocianova, E.; Kazimirova, M. Anaplasma phagocytophilum prevalence in ticks and rodents in an urban and natural habitat in South-Western Slovakia. Parasites Vectors 2015, 8, 276. [Google Scholar] [CrossRef] [PubMed]
- Taragelova, V.; Koci, J.; Hanincova, K.; Oleksak, M.; Labuda, M. Songbirds as hosts for ticks (Acari, Ixodidae) in Slovakia. Biologia 2005, 60, 529–537. [Google Scholar]
- Cepák, J. Atlas Migrace Ptáků: České a Slovenské Republiky; Aventinum: Prague, Czech Republic, 2008. [Google Scholar]
- Falco, R.C.; Fish, D. A comparison of methods for sampling the deer tick, Ixodes dammini, in a Lyme disease endemic area. Exp. Appl. Acarol. 1992, 14, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Stanko, M. Small mammals communities of windbreaks and adjacent fields in the eastern Slovakian lowlands. Folia Zool. 1994, 43, 135–143. [Google Scholar]
- Slovak, M. Pictorial key to the adults of ticks (Acari: Ixodida) of the Slovakia fauna. Entomofauna Carpath. 2010, 22, 8–13. [Google Scholar]
- Guy, E.C.; Stanek, G. Detection of Borrelia burgdorferi in patients with Lyme disease by the polymerase chain reaction. J. Clin. Pathol. 1991, 44, 610–611. [Google Scholar] [CrossRef] [PubMed]
- Black, W.C.; Roehrdanz, R.L. Mitochondrial gene order is not conserved in arthropods: Prostriate and metastriate tick mitochondrial genomes. Mol. Biol. Evol. 1998, 15, 1772–1785. [Google Scholar] [CrossRef] [PubMed]
- Derdakova, M.; Beati, L.; Pet’ko, B.; Stanko, M.; Fish, D. Genetic variability within Borrelia burgdorferi sensu lato genospecies established by PCR-single-strand conformation polymorphism analysis of the rrfA-rrlB intergenic spacer in Ixodes ricinus ticks from the Czech Republic. Appl. Environ. Microbiol. 2003, 69, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Courtney, J.W.; Kostelnik, L.M.; Zeidner, N.S.; Massung, R.F. Multiplex real-time PCR for detection of Anaplasma phagocytophilum and Borrelia burgdorferi. J. Clin. Microbiol. 2004, 42, 3164–3168. [Google Scholar] [CrossRef] [PubMed]
- Regnery, R.L.; Spruill, C.L.; Plikaytis, B.D. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J. Bacteriol. 1991, 173, 1576–1589. [Google Scholar] [CrossRef] [PubMed]
- Boretti, F.S.; Perreten, A.; Meli, M.L.; Cattori, V.; Willi, B.; Wengi, N.; Hornok, S.; Honegger, H.; Hegglin, D.; Woelfel, R.; et al. Molecular Investigations of Rickettsia helvetica infection in dogs, foxes, humans, and Ixodes ticks. Appl. Environ. Microbiol. 2009, 75, 3230–3237. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Legendre, L.F. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 2012; Volume 24, p. 1006. [Google Scholar]
- Oksanen, J.; Guillaume, B.F.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Henry, M.; et al. Vegan: Community Ecology Package. R Package Version 2.4-1. Available online: https://CRAN.R-project.org/package=vegan (accessed on 15 April 2018).
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 15 April 2018).
- Hervé, M. RVAideMemoire: Diverse Basic Statistical and Graphical Functions. R Package Version 0.9-62. Available online: https://CRAN.R-project.org/package=RVAideMemoire (accessed on 15 April 2018).
- Estrada-Peña, A.; Ortega, C.; Sánchez, N.; DeSimone, L.; Sudre, B.; Suk, J.E.; Semenza, J.C. Correlation of Borrelia burgdorferi sensu lato prevalence in questing Ixodes ricinus ticks with specific abiotic traits in the western Palearctic. Appl. Environ. Microbiol. 2011, 77, 3838–3845. [Google Scholar] [CrossRef] [PubMed]
- Kurtenbach, K.; Hanincova, K.; Tsao, J.I.; Margos, G.; Fish, D.; Ogden, N.H. Fundamental processes in the evolutionary ecology of Lyme borreliosis. Nat. Rev. Microbiol. 2006, 4, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Tagliapietra, V.; Rosa, R.; Arnoldi, D.; Cagnacci, F.; Capelli, G.; Montarsi, F.; Hauffe, H.C.; Rizzoli, A. Saturation deficit and deer density affect questing activity and local abundance of Ixodes ricinus (Acari, Ixodidae) in Italy. Vet. Parasitol. 2011, 183, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Heyman, P.; Cochez, C.; Simons, L.; Vanwambeke, S.O. A multi-level analysis of the relationship between environmental factors and questing Ixodes ricinus dynamics in Belgium. Parasites Vectors 2012, 5, 149. [Google Scholar] [CrossRef] [PubMed]
- Cayol, C.; Koskela, E.; Mappes, T.; Siukkola, A.; Kallio, E.R. Temporal dynamics of the tick Ixodes ricinus in northern Europe: Epidemiological implications. Parasites Vectors 2017, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Tomasiewicz, K.; Modrzewska, R.; Buczek, A.; Stanczak, J.; Maciukajc, J. The risk of exposure to Anaplasma phagocytophilum infection in Mid-Eastern Poland. Ann. Agric. Environ. Med. 2004, 11, 261–264. [Google Scholar] [PubMed]
- Skotarczak, B.; Rymaszewska, A.; Wodecka, B.; Sawczuk, M.; Adamska, M.; Maciejewska, A. PCR detection of granulocytic Anaplasma and Babesia in Ixodes ricinus ticks and birds in west-central Poland. Ann. Agric. Environ. Med. 2006, 13, 21–23. [Google Scholar] [PubMed]
- Rigo, K.; Gyuranecz, M.; Toth, A.G.; Foldvari, G. Detection of Borrelia burgdorferi Sensu Lato and Anaplasma phagocytophilum in small mammals and ectoparasites in Hungary. Vector Borne Zoon. Dis. 2011, 11, 1499–1501. [Google Scholar] [CrossRef] [PubMed]
- Massung, R.F.; Mauel, M.J.; Owens, J.H.; Allan, N.; Courtney, J.W.; Stafford, K.C., 3rd; Mather, T.N. Genetic variants of Ehrlichia phagocytophila, Rhode Island and Connecticut. Emerg. Infect. Dis. 2002, 8, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Bown, K.J.; Lambin, X.; Ogden, N.H.; Begon, M.; Telford, G.; Woldehiwet, Z.; Birtles, R.J. Delineating Anaplasma phagocytophilum ecotypes in coexisting, discrete enzootic cycles. Emerg. Infect. Dis. 2009, 15, 1948–1954. [Google Scholar] [CrossRef] [PubMed]
- Silaghi, C.; Skuballa, J.; Thiel, C.; Pfister, K.; Petney, T.; Pfaffle, M.; Taraschewski, H.; Passos, L.M. The European hedgehog (Erinaceus europaeus)—A suitable reservoir for variants of Anaplasma phagocytophilum? Ticks Tick Borne Dis. 2012, 3, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Barakova, I.; Derdakova, M.; Carpi, G.; Rosso, F.; Collini, M.; Tagliapietra, V.; Ramponi, C.; Hauffe, H.C.; Rizzoli, A. Genetic and ecologic variability among Anaplasma phagocytophilum strains, northern Italy. Emerg. Infect. Dis. 2014, 20, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Hanincova, K.; Taragelova, V.; Koci, J.; Schafer, S.M.; Hails, R.; Ullmann, A.J.; Piesman, J.; Labuda, M.; Kurtenbach, K. Association of Borrelia garinii and B. valaisiana with songbirds in Slovakia. Appl. Environ. Microbiol. 2003, 69, 2825–2830. [Google Scholar] [CrossRef] [PubMed]
- Didyk, Y.M.; Blanarova, L.; Pogrebnyak, S.; Akimov, I.; Petko, B.; Vichova, B. Emergence of tick-borne pathogens (Borrelia burgdorferi sensu lato, Anaplasma phagocytophilum, Ricketsia raoultii and Babesia microti) in the Kyiv urban parks, Ukraine. Ticks Tick Borne Dis. 2017, 8, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Glatz, M.; Mullegger, R.R.; Maurer, F.; Fingerle, V.; Achermann, Y.; Wilske, B.; Bloemberg, G.V. Detection of Candidatus Neoehrlichia mikurensis, Borrelia burgdorferi sensu lato genospecies and Anaplasma phagocytophilum in a tick population from Austria. Ticks Tick Borne Dis. 2014, 5, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Skotarczak, B.; Rymaszewska, A.; Wodecka, B.; Sawczuk, M. Molecular evidence of coinfection of Borrelia burgdorferi sensu lato, human granulocytic ehrlichiosis agent, and Babesia microti in ticks from northwestern Poland. J. Parasitol. 2003, 89, 194–196. [Google Scholar] [CrossRef]
- Stanczak, J.; Kubica-Biernat, B.; Racewicz, M.; Kruminis-Lozowska, W.; Kur, J. Detection of three genospecies of Borrelia burgdorferi sensu lato in Ixodes ricinus ticks collected in different regions of Poland. Int. J. Med. Microbiol. 2000, 290, 559–566. [Google Scholar] [CrossRef]
- Stunzner, D.; Hubalek, Z.; Halouzka, J.; Postic, D.; Pierer, K.; Marth, E. Prevalence of Borrelia burgdorferi s.I. in Ixodes ricinus ticks from Styria (Austria) and species identification by PCR-RFLP analysis. Zent. Bakteriol. 1998, 288, 471–478. [Google Scholar] [CrossRef]
- Mihalca, A.D.; Sandor, A.D. The role of rodents in the ecology of Ixodes ricinus and associated pathogens in Central and Eastern Europe. Front. Cell. Infect. Microbiol. 2013, 3, 56. [Google Scholar] [CrossRef] [PubMed]
- Perkins, S.E.; Cattadori, I.M.; Tagliapietra, V.; Rizzoli, A.P.; Hudson, P.J. Localized deer absence leads to tick amplification. Ecology 2006, 87, 1981–1986. [Google Scholar] [CrossRef]
- Berthova, L.; Slobodnik, V.; Slobodnik, R.; Oleksak, M.; Sekeyova, Z.; Svitalkova, Z.; Kazimirova, M.; Spitalska, E. The natural infection of birds and ticks feeding on birds with Rickettsia spp. and Coxiella burnetii in Slovakia. Exp. Appl. Acarol. 2016, 68, 299–314. [Google Scholar] [CrossRef] [PubMed]
- Biernat, B.; Stanczak, J.; Michalik, J.; Sikora, B.; Wierzbicka, A. Prevalence of infection with Rickettsia helvetica in Ixodes ricinus ticks feeding on non-rickettsiemic rodent hosts in sylvatic habitats of west-central Poland. Ticks Tick Borne Dis. 2016, 7, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Burri, C.; Schumann, O.; Schumann, C.; Gern, L. Are Apodemus spp. mice and Myodes glareolus reservoirs for Borrelia miyamotoi, Candidatus Neoehrlichia mikurensis, Rickettsia helvetica, R. monacensis and Anaplasma phagocytophilum? Ticks Tick Borne Dis. 2014, 5, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Franke, J.; Fritzsch, J.; Tomaso, H.; Straube, E.; Dorn, W.; Hildebrandt, A. Coexistence of pathogens in host-seeking and feeding ticks within a single natural habitat in Central Germany. Appl. Environ. Microbiol. 2010, 76, 6829–6836. [Google Scholar] [CrossRef] [PubMed]
- Inokuma, H.; Seino, N.; Suzuki, M.; Kaji, K.; Takahashi, H.; Igota, H.; Inoue, S. Detection of Rickettsia helvetica DNA from peripheral blood of Sika deer (Cervus nippon yesoensis) in Japan. J. Wildl. Dis. 2008, 44, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Sprong, H.; Wielinga, P.R.; Fonville, M.; Reusken, C.; Brandenburg, A.H.; Borgsteede, F.; Gaasenbeek, C.; van der Giessen, J.W. Ixodes ricinus ticks are reservoir hosts for Rickettsia helvetica and potentially carry flea-borne Rickettsia species. Parasites Vectors 2009, 2, 41. [Google Scholar] [CrossRef] [PubMed]
- Vaclav, R.; Ficova, M.; Prokop, P.; Betakova, T. Associations between coinfection prevalence of Borrelia lusitaniae, Anaplasma sp., and Rickettsia sp. in hard ticks feeding on reptile hosts. Microb. Ecol. 2011, 61, 245–253. [Google Scholar] [CrossRef] [PubMed]

| Bird Species | Number of Birds | B. burgdorferi s.l.-Positive/Examined (%) | R. helvetica-Positive/Examined (%) |
|---|---|---|---|
| Parus major | 18 | 28/255 (11%) | 13/255 (5.1%) |
| Sitta europaea | 5 | 3/9 (33.3%) | 0/9 (0%) |
| Turdus merula | 2 | 5/9 (55.6%) | 0/9 (0%) |
| Erithacus rubecula | 2 | 1/15 (6.7%) | 0/15 (0%) |
| Dendrocopos major | 2 | 0 | 0 |
| Parus montanus | 2 | 0/5 (0%) | 0/5 (0%) |
| Fringilla coelebs | 1 | 0 | 0 |
| Parus caeruleus | 1 | 0 | 0 |
| Muscicapa striata | 1 | 0/2 (0%) | 0/2 (0%) |
| Total | 34 | 37/295 (12.5%) | 13/295 (4.4%) |
| B. burgdorferi s.l. Positive/Examined (%) | R. helvetica Positive/Examined (%) | |
|---|---|---|
| larvae | 34/275 (12.4%) | 12/275 (4.4%) |
| nymphs | 3/20 (15%) | 1/20 (5%) |
| Total | 37/295 (12.5%) | 13/295 (4.4%) |
| Total Examined Ticks | B. burgdorferi s.l. Positive (%) | Rickettsia sp. Positive (%) | A.P. Positive (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | B.G. | B.V. | B.A. | B.L. | B.B.S.S. | B.S. | Mixed | R.H. | R.M. | |||
| Nymphs | 485 | 35 (7.21%) | 12 (2.47%) | 8 (1.65%) | 6 (1.24%) | 4 (0.83%) | 2 (0.41%) | 1 (0.21%) | 2 (0.41%) | 23 (4.75%) | 0 | 23 (6%) |
| Adults | 58 | 2 (3.38%) | 1 (1.69%) | 1 (1.69%) | 0 | 0 | 0 | 0 | 0 | 4 (6.78%) | 1 (1.69%) | 3 (5.08%) |
| Total | 543 | 37 (6.81%) | 13 (2.39%) | 9 (1.66%) | 6 (1.1%) | 4 (0.74%) | 2 (0.37%) | 1 (0.18%) | 2 (0.37%) | 27 (4.97%) | 1 (0.18%) | 32 (5.89%) |
| Study Site (Type) | Year of Tick Collection | A. phagocytophilum in Questing I. ricinus Ticks | Author |
|---|---|---|---|
| Bratislava (urban forest) | 2011–2012 | 5.9% (32/543) | This study |
| Fugelka (natural forest) | 2011–2013 | 3.1% (71/2257) n.s. | [59] |
| Bratislava (urban forest) | 2011–2013 | 7.2% (153/2117) n.s. | [59] |
| Košice (urban forest) | 2010 | 4.5% (10/224) n.s. | [30] |
| Bardejov (suburban forest) | 2008 | 1.7% (3/179) n.s. | [30] |
| Bratislava (suburban/urban) | 2009–2012 | 4% (10/248) n.s. | [30] |
| Čermeľ (natural forest) | 2011–2012 | 0.9% (2/220) * | [37] |
| Hýľov (natural forest) | 2011–2012 | 0.8% (2/266) ** | [37] |
| Košice (suburban forest) | 2011–2012 | 1.1% (2/176) n.s. | [37] |
| Rozhanovce (natural forest) | 2011–2012 | 0.6% (4/714) ** | [37] |
| Study Site (Type) | Year of Ticks Collection | B.b.s.l. Prevalence (%) in Questing I. ricinus Nymphs | Author |
|---|---|---|---|
| Bratislava (urban forest) | 2011–2012 | 7.2% (35/485) | This study |
| Šúr (natural forest) | 2001–2002 | 27.8% (57/205) ** | [26] |
| Malacky (urban park) | 2001–2002 | 14.4% (31/215) n.s. | [26] |
| Brzotín (natural forest) | 2001–2002 | 20% (14/70) * | [26] |
| Biskupice (natural forest) | 1999 | 21.5% (28/130) ** | [87] |
| Malacky (urban park) | 1999 | 18.7% (37/198) ** | [87] |
| Záhorská Ves (natural forest) | 1999 | 27.8% (15/54) ** | [87] |
| Záhorská Ves (natural forest) | 2000 | 19% (33/174) ** | [87] |
| Study Site (Type) | Year of Tick Collection | B. garinii Prevalence in Bird-Feeding I. ricinus Ticks | Author |
|---|---|---|---|
| Bratislava (urban forest) | 2011–2012 | 97% (36/37) | This study |
| All localities (natural forest) | 2001–2002 | 44% (155/352) ** | [26] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chvostáč, M.; Špitalská, E.; Václav, R.; Vaculová, T.; Minichová, L.; Derdáková, M. Seasonal Patterns in the Prevalence and Diversity of Tick-Borne Borrelia burgdorferi Sensu Lato, Anaplasma phagocytophilum and Rickettsia spp. in an Urban Temperate Forest in South Western Slovakia. Int. J. Environ. Res. Public Health 2018, 15, 994. https://doi.org/10.3390/ijerph15050994
Chvostáč M, Špitalská E, Václav R, Vaculová T, Minichová L, Derdáková M. Seasonal Patterns in the Prevalence and Diversity of Tick-Borne Borrelia burgdorferi Sensu Lato, Anaplasma phagocytophilum and Rickettsia spp. in an Urban Temperate Forest in South Western Slovakia. International Journal of Environmental Research and Public Health. 2018; 15(5):994. https://doi.org/10.3390/ijerph15050994
Chicago/Turabian StyleChvostáč, Michal, Eva Špitalská, Radovan Václav, Tatiana Vaculová, Lenka Minichová, and Markéta Derdáková. 2018. "Seasonal Patterns in the Prevalence and Diversity of Tick-Borne Borrelia burgdorferi Sensu Lato, Anaplasma phagocytophilum and Rickettsia spp. in an Urban Temperate Forest in South Western Slovakia" International Journal of Environmental Research and Public Health 15, no. 5: 994. https://doi.org/10.3390/ijerph15050994
APA StyleChvostáč, M., Špitalská, E., Václav, R., Vaculová, T., Minichová, L., & Derdáková, M. (2018). Seasonal Patterns in the Prevalence and Diversity of Tick-Borne Borrelia burgdorferi Sensu Lato, Anaplasma phagocytophilum and Rickettsia spp. in an Urban Temperate Forest in South Western Slovakia. International Journal of Environmental Research and Public Health, 15(5), 994. https://doi.org/10.3390/ijerph15050994





