Arsenic and Other Elemental Concentrations in Mushrooms from Bangladesh: Health Risks
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mushroom Cultivation and Consumption in Bangladesh
2.2. Sampling, Sample Processing, Digestion and Analysis
2.3. Sample Preparation
2.4. Sample Analysis
2.5. Quality Control: Analysis of Standard Reference Material (SRM)
2.6. Health Risk Index (HRI)
2.7. Statistical Analysis
3. Results and Discussion
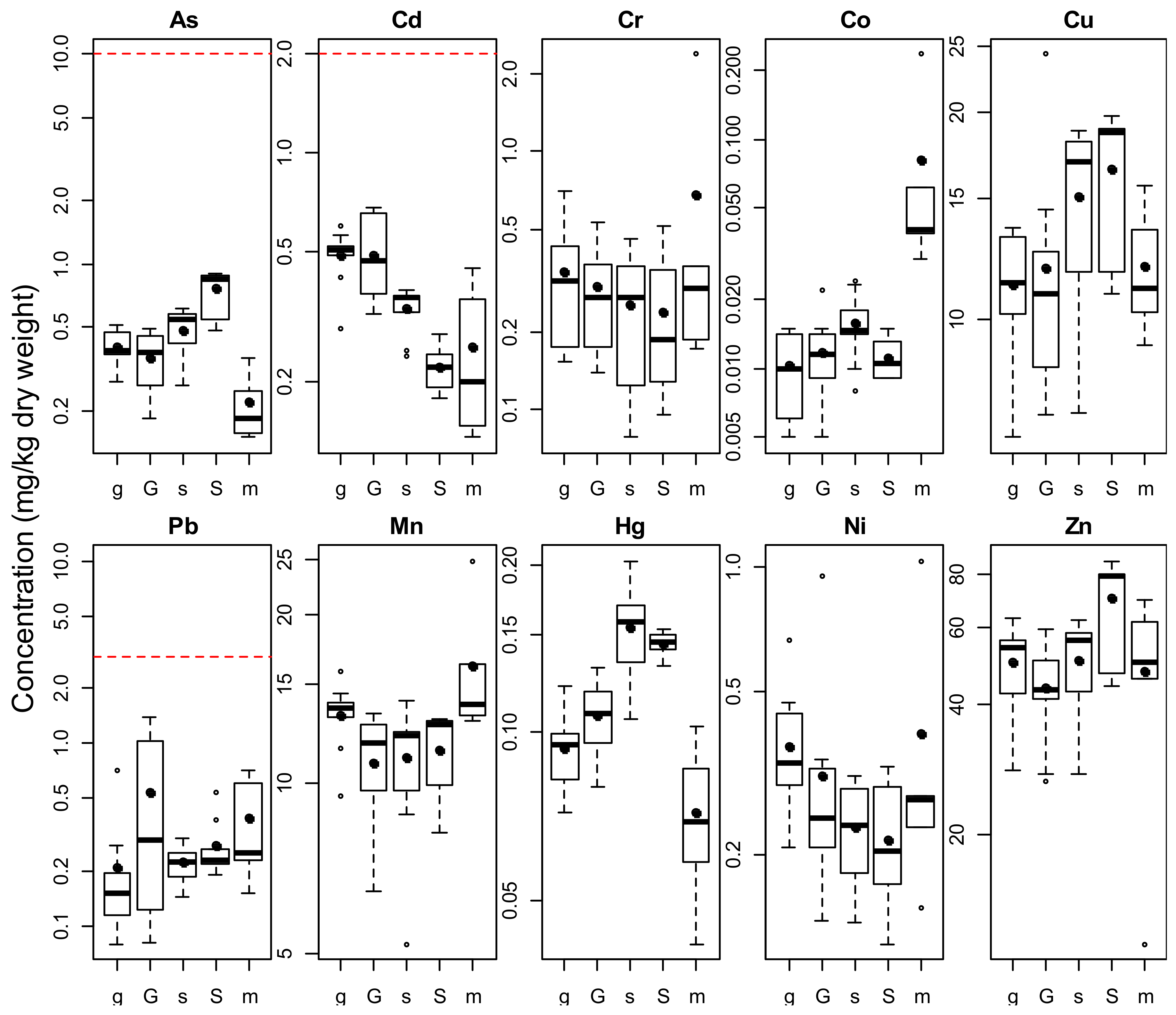
3.1. Arsenic
3.2. Cadmium
3.3. Chromium
3.4. Cobalt
3.5. Copper
3.6. Lead
3.7. Manganese
3.8. Mercury
3.9. Nickel
3.10. Zinc
3.11. Concentrations of Elements in Powdered Mushrooms for Sale
3.12. Elemental Accumulation
3.13. Correlations between Elements in Mushrooms
3.14. Correlations between Elemental Concentrations in the Medium and Mushroom
3.15. Market Basket Study
3.16. Daily Consumption of As and Other Elements from Mushrooms: Health Risk Index
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chakrabarti, D.; Singh, S.K.; Rashid, M.H.; Rahman, M.M. Arsenic: Occurrence in groundwater. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netharland, 2013. [Google Scholar]
- Rahman, M.M.; Owens, G.; Naidu, R. Arsenic levels in rice grain and assessment of daily dietary intake of arsenic from rice in arsenic-contaminated regions of Bangladesh—Implications to groundwater irrigation. Environ. Geochem. Health 2009, 31, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Biswas, M.; Mandal, M. Irrigation Management for Crop Diversification in Bangladesh; University Press: Dhaka, Bangladesh, 1993. [Google Scholar]
- Chakraborti, D.; Rahman, M.M.; Das, B.; Murrill, M.; Dey, S.; Mukherjee, S.C.; Dhar, R.K.; Biswas, B.K.; Chowdhury, U.K.; Roy, S.; et al. Status of groundwater arsenic contamination in Bangladesh: A 14-year study report. Water Res. 2010, 44, 5789–5802. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.; Badruzzaman, A.; Jalil, M.; Hossain, M.D.; Ahmed, M.F.; Masud, A.; Kamruzzaman, M.; Rahman, M.A. Fate of arsenic extracted with groundwater. In Fate of Arsenic in the Environment; United Nations University: Tokyo, Japan, 2003; pp. 7–20. ISBN 984-32-0507-3. [Google Scholar]
- Abedin, M.J.; Cresser, M.S.; Meharg, A.A.; Feldmann, J.; Cotter-Howells, J. Arsenic accumulation and metabolism in rice (Oryza sativa L.). Environ. Sci. Technol. 2002, 36, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Frisbie, S.H.; Mitchell, E.J.; Mastera, L.J.; Maynard, D.M.; Yusuf, A.Z.; Siddiq, M.Y.; Ortega, R.; Dunn, R.K.; Westerman, D.S.; Bacquart, T. Public health strategies for western Bangladesh that address arsenic, manganese, uranium, and other toxic elements in drinking water. Environ. Health Perspect. 2009, 117, 410. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, M. The Fungi: 1, 2, 3… 5.1 million species? Am. J. Bot. 2011, 98, 426–438. [Google Scholar] [CrossRef] [PubMed]
- Senn-Irlet, B.; Heilmann-Clausen, J.; Genney, D.; Dahlberg, A.; Guidance for Conservation of Macrofungi in Europe. ECCF Strasbg. 2007. Available online: https://www.wsl.ch/eccf/Guidance_Fungi.pdf (accessed on 4 May 2018).
- Manzi, P.; Aguzzi, A.; Pizzoferrato, L. Nutritional value of mushrooms widely consumed in Italy. Food Chem. 2001, 73, 321–325. [Google Scholar] [CrossRef]
- Valverde, M.E.; Hernández-Pérez, T.; Paredes-López, O. Edible mushrooms: Improving human health and promoting quality life. Int. J. Microbiol. 2015, 2015, 376387. [Google Scholar] [CrossRef] [PubMed]
- Dıez, V.; Alvarez, A. Compositional and nutritional studies on two wild edible mushrooms from northwest Spain. Food Chem. 2001, 75, 417–422. [Google Scholar] [CrossRef]
- Li, J.; Zhang, J.; Chen, H.; Chen, X.; Lan, J.; Liu, C. Complete mitochondrial genome of the medicinal mushroom Ganoderma lucidum. PLoS ONE 2013, 8, e72038. [Google Scholar] [CrossRef] [PubMed]
- Alom, M.M.; Bari, M.W. Investment in mushroom cultivation at Savar Upazilla: A prospective sector for Bangladesh. ASA Univ. Rev. 2010, 4, 182–185. [Google Scholar]
- Sánchez, C. Cultivation of Pleurotus ostreatus and other edible mushrooms. Appl. Microbiol. Biotechnol. 2010, 85, 1321–1337. [Google Scholar] [CrossRef] [PubMed]
- Niedzielski, P.; Mleczek, M.; Siwulski, M.; Gąsecka, M.; Kozak, L.; Rissmann, I.; Mikołajczak, P. Efficacy of supplementation of selected medicinal mushrooms with inorganic selenium salts. J. Environ. Sci. Health Part B 2014, 49, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Mleczek, M.; Niedzielski, P.; Siwulski, M.; Rzymski, P.; Gąsecka, M.; Goliński, P.; Kozak, L.; Kozubik, T. Importance of low substrate arsenic content in mushroom cultivation and safety of final food product. Eur. Food Res. Technol. 2016, 242, 355–362. [Google Scholar] [CrossRef]
- Rzymski, P.; Mleczek, M.; Siwulski, M.; Gąsecka, M.; Niedzielski, P. The risk of high mercury accumulation in edible mushrooms cultivated on contaminated substrates. J. Food Compos. Anal. 2016, 51, 55–60. [Google Scholar] [CrossRef]
- Baloch, U.K. WHEAT: Post-Harvest Operations; FAO: Rome, Italy, 1999. [Google Scholar]
- Chakma, S.; Rahman, M.; Islam, P.; Awal, M.; Roy, U.; Haq, M. Arsenic in rice and rice straw. Bangladesh Vet. 2012, 29, 1–6. [Google Scholar] [CrossRef]
- Kalač, P.; Svoboda, L.R. A review of trace element concentrations in edible mushrooms. Food Chem. 2000, 69, 273–281. [Google Scholar] [CrossRef]
- Barmon, B.K.; Sharmin, I.; Abbasi, P.K.; Mamun, A. Economics of mushroom (Agaricus bisporus) production in a selected Upazila of Bangladesh. Agriculturists 2012, 10, 77–89. [Google Scholar] [CrossRef]
- Kamal, A.; Fauzia, B.; Abul, K. Mushroom production in Bangladesh: Present scenario and potentialities. SAARC J. Agric. 2009, 7, 91–105. [Google Scholar]
- Water Content of Mushrooms. Available online: http://wholefoodcatalog.info/nutrient/water/mushrooms/ (accessed on 4 May 2018).
- Cui, Y.-J.; Zhu, Y.-G.; Zhai, R.-H.; Chen, D.-Y.; Huang, Y.-Z.; Qiu, Y.; Liang, J.-Z. Transfer of metals from soil to vegetables in an area near a smelter in Nanning, China. Environ. Int. 2004, 30, 785–791. [Google Scholar] [CrossRef] [PubMed]
- USEPA, R.-B.C.T.; Manual, E.P.S.G. Stressor Identification Guidance Document; EPA 822-B-00-025; Office of Water and Office of Research and Development, Environmental Protection Agency: Washington, DC, USA, 2000.
- Rahman, M.M.; Asaduzzaman, M.; Naidu, R. Consumption of arsenic and other elements from vegetables and drinking water from an arsenic-contaminated area of Bangladesh. J. Hazard. Mater. 2013, 262, 1056–1063. [Google Scholar] [CrossRef] [PubMed]
- Team, R.C. R: A Language and Environment for Statistical Computing. 2013. Available online: https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006 (accessed on 4 May 2018).
- Kula, I.; Solak, M.H.; Uğurlu, M.; Işıloğlu, M.; Arslan, Y. Determination of mercury, cadmium, lead, zinc, selenium and iron by ICP-OES in mushroom samples from around thermal power plant in Muğla, Turkey. Bull. Environ. Contam. Toxicol. 2011, 87, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Falandysz, J.; Borovička, J. Macro and trace mineral constituents and radionuclides in mushrooms: Health benefits and risks. Appl. Microbiol. Biotechnol. 2013, 97, 477–501. [Google Scholar] [CrossRef] [PubMed]
- Mohiuddin, K.; Alam, M.M.; Arefin, M.T.; Ahmed, I. Assessment of nutritional composition and heavy metal content in some edible mushroom varieties collected from different areas of Bangladesh. Asian J. Med. Biol. Res. 2016, 1, 495–501. [Google Scholar] [CrossRef]
- Llorente-Mirandes, T.; Llorens-Munoz, M.; Funes-Collado, V.; Sahuquillo, A.; Lopez-Sanchez, J.F. Assessment of arsenic bioaccessibility in raw and cooked edible mushrooms by a PBET method. Food Chem. 2016, 194, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Braeuer, S.; Goessler, W.; Kamenik, J.; Konvalinkova, T.; Zigova, A.; Borovicka, J. Arsenic hyperaccumulation and speciation in the edible ink stain bolete (Cyanoboletus pulverulentus). Food Chem. 2018, 242, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Smitht, P.G.; Koch, I.; Reimer, K.J. Arsenic speciation analysis of cultivated white button mushrooms (Agaricus bisporus) using high-performance liquid chromatography—Inductively coupled plasma mass spectrometry, and X-ray absorption Spectroscopy. Environ. Sci. Technol. 2007, 41, 6947–6954. [Google Scholar] [CrossRef]
- Rzymski, P.; Mleczek, M.; Siwulski, M.; Jasinska, A.; Budka, A.; Niedzielski, P.; Kalac, P.; Gasecka, M.; Budzynska, S. Multielemental analysis of fruit bodies of three cultivated commercial Agaricus species. J. Food Compos. Anal. 2017, 59, 170–178. [Google Scholar] [CrossRef]
- Maihara, V.; Moura, P.; Catharino, M.; Castro, L.; Figueira, R. Arsenic and cadmium content in edible mushrooms from São Paulo, Brazil determined by INAA and GF AAS. J. Radioanal. Nucl. Chem. 2008, 278, 395–397. [Google Scholar] [CrossRef]
- Quarcoo, A.; Adotey, G. Determination of heavy metals in Pleurotus ostreatus (Oyster mushroom) and Termitomyces clypeatus (Termite mushroom) sold on selected markets in Accra, Ghana. Mycosphere 2013, 4, 960–967. [Google Scholar] [CrossRef]
- Reddy, N.M.; Reddi, U.B.E.; Reddy, B.T. Assessment of heavy metal threat in agaricus bisporus mushrooms cultivated from water hyacinth weed of Kolleru lake, Andhra pradesh-India. Int. J. Environ. Sci. 2012, 3, 28. [Google Scholar]
- Muñoz, A.H.S.; Corona, F.G.; Wrobel, K.; Soto, G.M.; Wrobel, K. Subcellular distribution of aluminum, bismuth, cadmium, chromium, copper, iron, manganese, nickel, and lead in cultivated mushrooms (Agaricus bisporus and Pleurotus ostreatus). Biol. Trace Elem. Res. 2005, 106, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Koyyalamudi, S.R.; Jeong, S.-C.; Manavalan, S.; Vysetti, B.; Pang, G. Micronutrient mineral content of the fruiting bodies of Australian cultivated Agaricus bisporus white button mushrooms. J. Food Compos. Anal. 2013, 31, 109–114. [Google Scholar] [CrossRef]
- Sivrikaya, H.; Bacak, L.; Saraçbaşı, A.; Eroğlu, H. Trace elements in Pleurotus sajor-caju cultivated on chemithermomechanical pulp for bio-bleaching. Food Chem. 2002, 79, 173–176. [Google Scholar] [CrossRef]
- Vetter, J. Chemical composition of fresh and conserved Agaricus bisporus mushroom. Eur. Food Res. Technol. 2003, 217, 10–12. [Google Scholar] [CrossRef]
- Michael, H.W.; Bultosa, G.; Pant, L.M. Nutritional contents of three edible oyster mushrooms grown on two substrates at Haramaya, Ethiopia, and sensory properties of boiled mushroom and mushroom sauce. Int. J. Food Sci. Technol. 2011, 46, 732–738. [Google Scholar] [CrossRef]
- Kalač, P. Chemical composition and nutritional value of European species of wild growing mushrooms: A review. Food Chem. 2009, 113, 9–16. [Google Scholar] [CrossRef]
- Kalač, P. Trace element contents in European species of wild growing edible mushrooms: A review for the period 2000–2009. Food Chem. 2010, 122, 2–15. [Google Scholar] [CrossRef]
- Survey of Metals in a Variety of Foods. Food Survey Information Sheet. 2007. Available online: http://www.food.gov.UK/Science/Surveillance (accessed on 4 May 2018).
- ANZFA (Australia New Zealand Food Authority) Food Standards Code. Standard 1.4.1. Contaminants and Natural Toxicants. 2011. Available online: https://www.legislation.gov.au/Details/F2015C00052 (accessed on 4 May 2018).
- China, E. Maximum Levels of Contaminants in Foods GB2762-2005. China State Environmental Protection Administration: Beijing, China, 2005. Available online: http://www.speciation.net/Database/Links/USDA-FAIRS-Product-Specific-Maximum-Levels-of--Contaminants-in-Foods-2006-;i3429Contaminants-in-Foods-2006-;i3429 (accessed on 4 May 2018).
- Roy, P.; Saha, A. Metabolism and toxicity of arsenic: A human carcinogen. Curr. Sci. 2002, 81, 38–45. [Google Scholar]
- Gonzálvez, A.; Llorens, A.; Cervera, M.; Armenta, S.; de la Guardia, M. Non-chromatographic speciation of inorganic arsenic in mushrooms by hydride generation atomic fluorescence spectrometry. Food Chem. 2009, 115, 360–364. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. A review of human carcinogens: Arsenic, metals, fibres, and dusts. IARC Monogr. Eval. Carcinog. Risks Hum. C 2012, 100, 169–211. [Google Scholar]
- US EPA. Office of Pesticide Programs Chemicals Evaluated for Carcinogenic Potential—Annual Cancer Report; EPA: Washington DC, USA, 2016. [Google Scholar]
- European Commission. Commission Regulation (EC) No. 466/2001; Directive 2001/22/EC; European Commission EU: Brussels, Belgium, 2001. [Google Scholar]
- Bernhoft, R.A. Mercury toxicity and treatment: A review of the literature. J. Environ. Public Health 2012, 2012, 460508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Cai, Y.; Tu, C.; Ma, L.Q. Arsenic speciation and distribution in an arsenic hyperaccumulating plant. Sci. Total Environ. 2002, 300, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Rácz, L.; Oldal, V. Investigation of uptake processes in a soil/mushroom system by AES and AAS methods. Microchem. J. 2000, 67, 115–118. [Google Scholar] [CrossRef]
- Kalac, P.; Niznanska, M.; Bevilaqua, D.; Staskova, I. Concentrations of mercury, copper, cadmium and lead in fruiting bodies of edible mushrooms in the vicinity of a mercury smelter and a copper smelter. Sci. Total Environ. 1996, 177, 251–258. [Google Scholar] [CrossRef]
- Cibulka, J.; Miholova, D.; Szakova, J.; Fucikova, A.; Slamova, A.; Stehulova, I.; Barlakova, S.; Sisak, L.; Pulkrab, K. Cadmium, Lead, Mercury and Caesium Levels in Wild Mushrooms and Forest Berries from Different Localities of the Czech Republic. Sci. Agric. Bohem. 1996. Available online: http://agris.fao.org/agris-search/search.do?recordID=CZ9601339 (accessed on 4 May 2018).
- Säumel, I.; Kotsyuk, I.; Hölscher, M.; Lenkereit, C.; Weber, F.; Kowarik, I. How healthy is urban horticulture in high traffic areas? Trace metal concentrations in vegetable crops from plantings within inner city neighbourhoods in Berlin, Germany. Environ. Pollut. 2012, 165, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Morton-Bermea, O.; Hernández-Álvarez, E.; González-Hernández, G.; Romero, F.; Lozano, R.; Beramendi-Orosco, L. Assessment of heavy metal pollution in urban topsoils from the metropolitan area of Mexico City. J. Geochem. Explor. 2009, 101, 218–224. [Google Scholar] [CrossRef]
- Duong, T.T.; Lee, B.-K. Determining contamination level of heavy metals in road dust from busy traffic areas with different characteristics. J. Environ. Manag. 2011, 92, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Lepšová, A.; Mejstřík, V. Accumulation of trace elements in the fruiting bodies of macrofungi in the Krušné hory mountains, Czechoslovakia. Sci. Total Environ. 1988, 76, 117–128. [Google Scholar] [CrossRef]
- Aktaruzzaman, M.; Fakhruddin, A.; Chowdhury, M.; Fardous, Z.; Alam, M. Accumulation of heavy metals in soil and their transfer to leafy vegetables in the region of Dhaka Aricha Highway, Savar, Bangladesh. Pak. J. Biol. Sci. 2013, 16, 332–338. [Google Scholar] [PubMed]
- Melgar, M.J.; Alonso, J.; Garcia, M.A. Total contents of arsenic and associated health risks in edible mushrooms, mushroom supplements and growth substrates from Galicia (NW Spain). Food Chem. Toxicol. 2014, 73, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Johanson, K.J.; Nikolova, I.; Taylor, A.F.; Vinichuk, M.M. Uptake of Elements by Fungi in the Forsmark Area; SKB: Orange, CA, USA, 2004; Volume 4. [Google Scholar]
- Joint FAO/WHO Expert Committee on Food Additives (JECFA); World Health Organization. Compendium of Food Additive Specifications: Addendum 8; Food & Agriculture Organization, 2000; Volume 52. [Google Scholar]
- USEPA. Integrated Risk Information System; USEPA: Washington, DC, USA, 2002. [Google Scholar]
- Fang, Y.; Sun, X.Y.; Yang, W.J.; Ma, N.; Xin, Z.H.; Fu, J.; Liu, X.C.; Liu, M.; Mariga, A.M.; Zhu, X.F.; et al. Concentrations and health risks of lead, cadmium, arsenic, and mercury in rice and edible mushrooms in China (vol 147, pg 147, 2013). Food Chem. 2014, 151, 379. [Google Scholar] [CrossRef]
- Yu, T.H.; Peng, Y.Y.; Lin, C.X.; Qin, J.H.; Li, H.S. Application of iron and silicon fertilizers reduces arsenic accumulation by two Ipomoea aquatica varities. J. Integr. Agric. 2016, 15, 2613–2619. [Google Scholar] [CrossRef]
| Element | Mushroom Species | Mean ± SD | Median | Range | NIST SRM 1573a (Tomato Leaves) | ||
|---|---|---|---|---|---|---|---|
| Certified Values | Observed Values | Recovery (%) | |||||
| As | P. high-king | 0.56 ± 0.25 | 0.489 | 0.18–0.91 | 0.112 ± 0.004 | 0.122 ± 0.003 | 109 |
| P. ostreatus | 0.45 ± 0.11 | 0.45 | 0.263–0.62 | ||||
| Cd | P. high-king | 0.35 ± 0.17 | 0.301 | 0.18–0.68 | 1.52 ± 0.04 | 1.46 ± 0.060 | 96.1 |
| P. ostreatus | 0.41 ± 0.10 | 0.38 | 0.24–0.599 | ||||
| Cr | P. high-king | 0.27 ± 0.14 | 0.223 | 0.096–0.533 | 1.99 ± 0.06 | 1.76 ± 0.050 | 88.4 |
| P. ostreatus | 0.3 ± 0.16 | 0.313 | 0.078–0.697 | ||||
| Co | P. high-king | 0.011 ± 0.004 | 0.011 | 0.005–0.022 | 0.57 ± 0.02 | 0.61 ± 0.040 | 107 |
| P. ostreatus | 0.013 ± 0.005 | 0.014 | 0.005–0.024 | ||||
| Cu | P. high-king | 14.2 ± 4.9 | 12.5 | 7.30–24.3 | 4.7 ± 0.14 | 5.3 ± 0.19 | 113 |
| P. ostreatus | 13.2 ± 3.7 | 12.6 | 6.70–18.8 | ||||
| Pb | P. high-king | 0.40 ± 0.39 | 0.23 | 0.08–1.37 | |||
| P. ostreatus | 0.22 ± 0.13 | 0.194 | 0.079–0.72 | ||||
| Mn | P. high-king | 11.2 ± 2.17 | 12.2 | 6.45–13.4 | 246 ± 8 | 232 ± 12 | 94.3 |
| P. ostreatus | 12.2 ± 2.4 | 12.7 | 5.1–15.8 | ||||
| Hg | P. high-king | 0.126 ± 0.03 | 0.12 | 0.11–0.144 | 0.034 ± 0.004 | 0.032 ± 0.006 | 94.1 |
| P. ostreatus | 0.124 ± 0.03 | 0.121 | 0.094–0.154 | ||||
| Ni | P. high-king | 0.263 ± 0.18 | 0.22 | 0.12–0.95 | 1.59 ± 0.07 | 1.50 ± 0.08 | 94.3 |
| P. ostreatus | 0.3 ± 0.12 | 0.292 | 0.136–0.665 | ||||
| Zn | P. high-king | 56.9 ± 19.5 | 50.4 | 26.3–85.8 | 30.9 ± 0.7 | 27.6 ± 0.6 | 89.3 |
| P. ostreatus | 50.2 ± 11.3 | 54.8 | 27.5–63.2 | ||||
| Element | Study Location | Species | Concentration | Reference | |
|---|---|---|---|---|---|
| Mean | Range | ||||
| As | Bangladesh | P. high-king | 0.56 ± 0.25 | 0.18–0.91 | This study |
| P. ostreatus | 0.45 ± 0.11 | 0.263–0.62 | |||
| Bangladesh | P. ostreatus | BDL | [31] | ||
| A. bisporus | |||||
| V. volvacea | |||||
| G. lucidum | |||||
| Spain | L. edodes | 1.393 | [32,33] | ||
| A. bisporus | 0.185 | ||||
| P. ostreatus | 0.335 | ||||
| Canada | A. bisporus | 0.14 ± 0.04 | [34] | ||
| Poland | A. arvensis | BDL | [35] | ||
| A. bisporus | 0.08–0.71 | ||||
| A. Bisporus | 0.15–1.4 | ||||
| Brazil | P. ostreatus | 0.056 ± 0.004 | [36] | ||
| P. florida | 0.073 ± 0.018 | ||||
| P. eryngui | 0.009 ± 0.003 | ||||
| P. salmoneostramineus | 0.043 ± 0.004 | ||||
| A. sp. | 0.125 ± 0.014 | ||||
| A. bisporus | 0.097 ± 0.024 | ||||
| L. edodes | 0.210 ± 0.009 | ||||
| Ghana | P. ostreatus | 0.04 | [37] | ||
| T. clypeatus | 0.1 | ||||
| India | A. bisporus | 0.64 ± 0.16 | [38] | ||
| Cd | Bangladesh | P. high-king | 0.35 ± 0.17 | 0.18–0.68 | This study |
| P. ostreatus | 0.41 ± 0.10 | 0.24–0.60 | |||
| Bangladesh | P. ostreatus | BDL | [31] | ||
| A. bisporus | |||||
| V. volvacea | |||||
| G. lucidum | |||||
| Mexico | P. ostreatus | 5.39 | [39] | ||
| A. bisporus (caps) | 0.54 | ||||
| Brazil | P. ostreatus | 0.074 ± 0.002 | [36] | ||
| P. florida | 0.220 ± 0.013 | ||||
| P. eryngui | 0.011 ± 0.003 | ||||
| P. salmoneostramineus | 0.229 ± 0.004 | ||||
| A. sp. | <LD | ||||
| A. bisporus | <LD | ||||
| L. edodes | 0.190 ± 0.011 | ||||
| Australia | A. bisporus | 0.18 ± 0.02 | [40] | ||
| Cr | Bangladesh | P. high-king | 0.27 ± 0.14 | 0.096–0.53 | This study |
| P. ostreatus | 0.30 ± 0.16 | 0.078–0.70 | |||
| Bangladesh | P. ostreatus | 0.21 ± 0.00–0.30 ± 0.01 | [31] | ||
| A. bisporus | 0.23 ± 0.01–0.30 ± 0.01 | ||||
| V. volvacea | 0.24 ± 0.01 | ||||
| G. lucidum | 0.21 ± 0.01 | ||||
| Mexico | P. ostreatus | 63.0 | [39] | ||
| A. bisporus (caps) | 4.06 | ||||
| Turkey | P. sajor-caju | 8.50 | [41] | ||
| Co | Bangladesh | P. high-king | 0.011 ± 0.004 | 0.005–0.022 | This study |
| P. ostreatus | 0.013 ± 0.005 | 0.005–0.024 | |||
| Turkey | P. sajor-caju | 12.5 | [41] | ||
| Australia | A. bisporus | 0.126 ± 0.001 | [40] | ||
| Cu | Bangladesh | P. high-king | 14.2 ± 4.9 | 7.3–24.3 | This study |
| P. ostreatus | 13.2 ± 3.7 | 6.7–18.8 | |||
| Bangladesh | P. ostreatus | 39.2 ± 0.88–102.1 ± 2.6 | [31] | ||
| A. bisporus | 54.6 ± 0.86–163.4 ± 3.9 | ||||
| V. volvacea | 101.8 ± 2.3 | ||||
| G. lucidum | 72.5 ± 1.22 | ||||
| Turkey | P. sajor-caju | 10.5 | [41] | ||
| Mexico | P. ostreatus | 732 | [39] | ||
| A. bisporus (caps) | 352 | ||||
| Australia | A. bisporus | 53.4 ± 0.251 | [40] | ||
| Hungary | A. bisporus | 58 ± 2–65 ± 1 | [42] | ||
| Pb | Bangladesh | P. high-king | 0.40 ± 0.39 | 0.08–1.37 | This study |
| P. ostreatus | 0.22 ± 0.13 | 0.079–0.72 | |||
| Bangladesh | P. ostreatus | 0.14 ± 0.02–0.59 ± 0.03 | [31] | ||
| A. bisporus | 0.15 ± 0.01–0.22 ± 0.02 | ||||
| V. volvacea | 0.25 ± 0.02 | ||||
| G. lucidum | 0.13 ± 0.01 | ||||
| Turkey | P. sajor-caju | 27.5 | [41] | ||
| Mexico | P. ostreatus | 0.91 | [39] | ||
| A. bisporus (caps) | 0.41 | ||||
| Australia | A bisporus | 3.9 ± 0.49 | [40] | ||
| Mn | Bangladesh | P. high-king | 11.2 ± 2.17 | 6.45–13.4 | This study |
| P. ostreatus | 12.2 ± 2.4 | 5.1–15.8 | |||
| Bangladesh | P. ostreatus | 52.9 ± 1.04–104.5 ± 1.8 | [31] | ||
| A. bisporus | 56.2 ± 1.34–91.1 ± 1.38 | ||||
| V. volvacea | 78.5 ± 0.97 | ||||
| G. lucidum | 64.0 ± 0.92 | ||||
| Turkey | P. sajor-caju | 17.5 | [41] | ||
| Mexico | P. ostreatus | 18.3 | [39] | ||
| A. bisporus (caps) | 9.42 | ||||
| Australia | A. bisporus | 10.6 ± 0.01 | [40] | ||
| Hg | Bangladesh | P. high-king | 0.126 ± 0.03 | 0.11–0.14 | This study |
| P. ostreatus | 0.124 ± 0.03 | 0.094–0.15 | |||
| Poland | P. ostreatus | 0.10 ± 0.01 (cap), 0.02 ± 0.01 (stipe) | [18] | ||
| A. bisporus | 0.08 ± 0.01 (cap), 0.05 ± 0.01 (stipe) | ||||
| H. erinaceus | 0.07 ± 0.02 (cap), 0.03 ± 0.01 (stipe) | ||||
| Ni | Bangladesh | P. high-king | 0.263 ± 0.18 | 0.12–0.95 | This study |
| P. ostreatus | 0.3 ± 0.12 | 0.136–0.665 | |||
| Turkey | P. sajor-caju | 17.5 | [41] | ||
| Mexico | P. ostreatus | 31.5 | [39] | ||
| A. bisporus (caps) | 9.02 | ||||
| Zn | Bangladesh | P. high-king | 56.9 ± 19.5 | 26.3–85.8 | This study |
| P. ostreatus | 50.2 ± 11.3 | 27.5–63.2 | |||
| Bangladesh | P. ostreatus | 30.1 ± 0.19–75.5 ± 0.54 | [31] | ||
| A. bisporus | 36.3 ± 0.23–47.6 ± 0.46 | ||||
| V. volvacea | 36.5 ± 0.43 | ||||
| G. lucidum | 52.2 ± 0.47 | ||||
| Turkey | P. sajor-caju | 110 | [41] | ||
| Ethiopia | P. ostreatus | 59.2 ± 0.02 | [43] | ||
| P. sajor-caju | 59.2 ± 0.03 | ||||
| Australia | A. bisporus | 43.8 ± 0.20 | [40] | ||
| Hungary | A. bisporus | 60 ± 0–62 ± 0 | [42] | ||
| Location | Mushroom | As | Cd | Cr | Co | Cu | Pb | Mn | Hg | Ni | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Savar | P. high-king | 1.35 | 1.30 | 0.132 | 0.009 | 3.28 | 0.073 | 0.008 | 1.63 | 0.058 | 1.18 |
| P. ostreatus | 1.54 | 3.12 | 0.249 | 0.038 | 2.03 | 0.090 | 0.016 | 0.82 | 0.047 | 1.01 | |
| Gazipur | P. high-king | 0.79 | 2.11 | 0.293 | 0.032 | 1.61 | 0.271 | 0.012 | 0.79 | 0.042 | 0.76 |
| P. ostreatus | 0.98 | 2.19 | 0.378 | 0.032 | 1.42 | 0.121 | 0.013 | 0.58 | 0.05 | 0.92 | |
| Mean | 1.07 | 2.19 | 0.263 | 0.028 | 2.09 | 0.139 | 0.012 | 0.956 | 0.049 | 0.97 | |
| SD | 0.51 | 1.19 | 0.189 | 0.020 | 1.13 | 0.169 | 0.006 | 0.754 | 0.020 | 0.43 | |
| As | Cd | Cr | Co | Cu | Pb | Mn | Hg | Ni | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|
| As | 1.000 | |||||||||
| Cd | −0.508 ** | 1.000 | ||||||||
| Cr | −0.016 ns | 0.343 ns | 1.000 | |||||||
| Co | −0.119 ns | 0.132 ns | 0.504 ** | 1.000 | ||||||
| Cu | 0.778 ** | −0.056 ns | 0.170 ns | 0.130 ns | 1.000 | |||||
| Pb | −0.312 ns | 0.351 ns | 0.679 ** | 0.522 ** | −0.288 ns | 1.000 | ||||
| Mn | 0.552 ** | 0.177 ns | 0.312 ns | 0.234 ns | 0.645 ** | −0.004 ns | 1.000 | |||
| Hg | 0.891 ** | −0.470 * | −0.158 ns | −0.259 ns | 0.666 ** | −0.383 ns | 0.352 ns | 1.000 | ||
| Ni | −0.055 ns | 0.522 ** | 0.215 ns | 0.259 ns | 0.117 ns | 0.290 ns | 0.154 ns | 0.027 ns | 1.000 | |
| Zn | 0.956 ** | −0.322 ns | 0.121 ns | −0.044 ns | 0.806 ** | −0.168 ns | 0.699 ** | 0.826 ** | 0.075 ns | 1.000 |
| Element | Weekly Intake Rate (Fresh wt, g) | Median Concentration wt (mg/kg) | Weekly Intake (mg) of Elements | Weekly Intake of Element mg/kg/bw | Oral Reference Dose mg/kg/day | Oral Reference Dose mg/kg/week | HRI | PTWI µg/kg | PTWI mg/45 kg | Maximum Consumption per Week * (g) |
|---|---|---|---|---|---|---|---|---|---|---|
| As | 0.09 | 0.047 | 0.0042 | 0.00009 | 0.0003 | 0.0021 | 0.0448 | 15 | 0.675 | 14.4 |
| Cd | 0.034 | 0.0030 | 0.00007 | 0.001 | 0.007 | 0.0097 | 7 | 0.315 | 9.3 | |
| Cr | 0.027 | 0.0024 | 0.00005 | 0.003 | 0.021 | 0.0026 | ||||
| Co | 0.001 | 0.0001 | 0.00000 | |||||||
| Cu | 1.250 | 0.1125 | 0.00250 | 0.04 | 0.28 | 0.0089 | ||||
| Pb | 0.021 | 0.0019 | 0.00004 | 0.004 | 0.028 | 0.0015 | 25 | 1.125 | 53.6 | |
| Mn | 1.240 | 0.1116 | 0.00248 | 0.14 | 0.98 | 0.0025 | ||||
| Hg | 0.012 | 0.0011 | 0.00002 | 5 | 0.225 | 18.8 | ||||
| Ni | 0.026 | 0.0023 | 0.00005 | 0.02 | 0.14 | 0.0004 | ||||
| Zn | 5.260 | 0.4734 | 0.01052 | 0.3 | 2.1 | 0.0050 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rashid, M.H.; Rahman, M.M.; Correll, R.; Naidu, R. Arsenic and Other Elemental Concentrations in Mushrooms from Bangladesh: Health Risks. Int. J. Environ. Res. Public Health 2018, 15, 919. https://doi.org/10.3390/ijerph15050919
Rashid MH, Rahman MM, Correll R, Naidu R. Arsenic and Other Elemental Concentrations in Mushrooms from Bangladesh: Health Risks. International Journal of Environmental Research and Public Health. 2018; 15(5):919. https://doi.org/10.3390/ijerph15050919
Chicago/Turabian StyleRashid, Md Harunur, Mohammad Mahmudur Rahman, Ray Correll, and Ravi Naidu. 2018. "Arsenic and Other Elemental Concentrations in Mushrooms from Bangladesh: Health Risks" International Journal of Environmental Research and Public Health 15, no. 5: 919. https://doi.org/10.3390/ijerph15050919
APA StyleRashid, M. H., Rahman, M. M., Correll, R., & Naidu, R. (2018). Arsenic and Other Elemental Concentrations in Mushrooms from Bangladesh: Health Risks. International Journal of Environmental Research and Public Health, 15(5), 919. https://doi.org/10.3390/ijerph15050919








