Adult Premenopausal Bone Health Related to Reproductive Characteristics—Population-Based Data from the Canadian Multicentre Osteoporosis Study (CaMos)
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
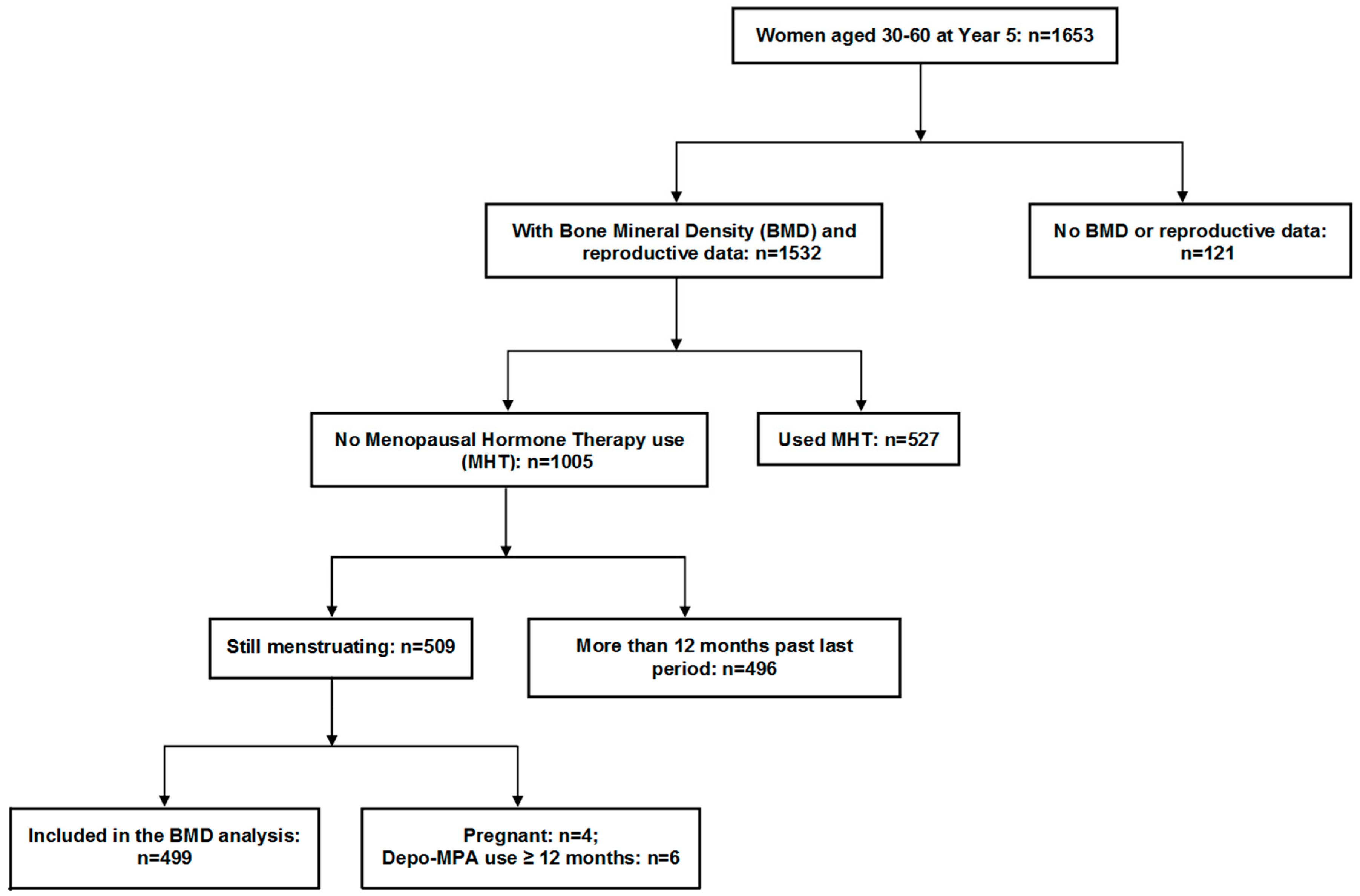
2.2. Flow of Participants through the Study
2.3. Methods
2.4. Statistical Analysis
3. Results
3.1. Reproductive Characteristics
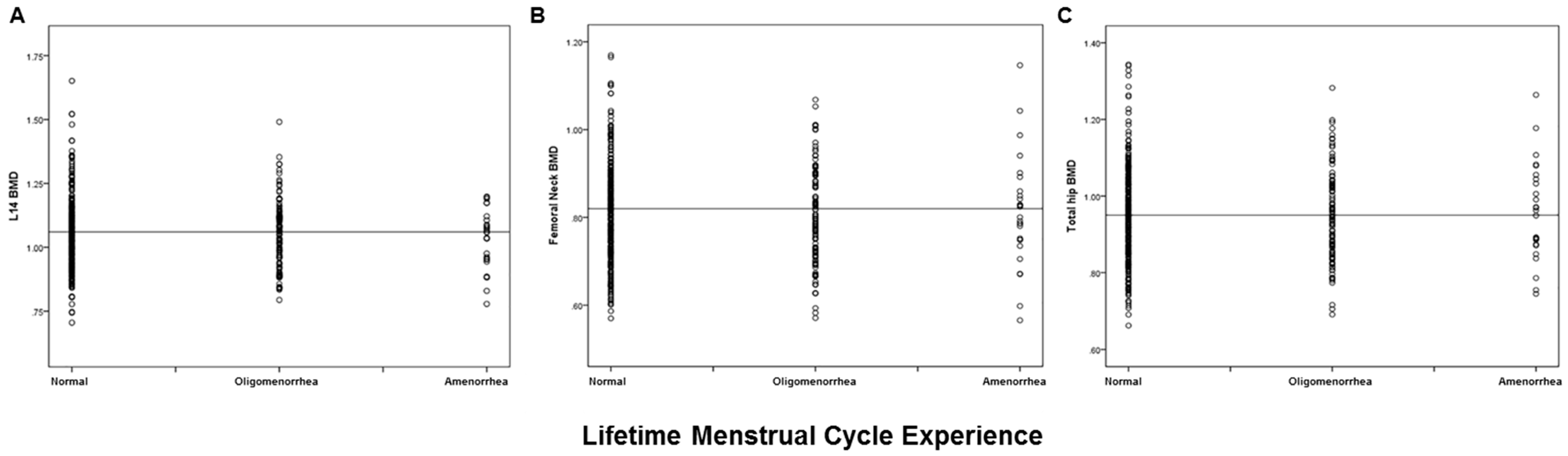
3.2. Relationship of Reproductive Variables with Bone Health (BMD, Prevalent Fracture)
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| AAE | Anovulatory Androgen Excess |
| ANOVA | Analysis of variance |
| BMD | areal Bone Mineral Density |
| BMI | Body Mass Index |
| CaMos | Canadian Multicentre Osteoporosis Study |
| CeMCOR | Centre for Menstrual Cycle and Ovulation Research |
| CHC | Combined Hormonal Contraceptives |
| FN | Femoral Neck |
| LS | Lumbar Spine |
| PCOS | Polycystic Ovary Syndrome |
| TH | Total Hip |
References
- Van Hooff, M.H.; Voorhorst, F.J.; Kaptein, M.B.; Hirasing, R.A.; Koppenaal, C.; Schoemaker, J. Relationship of the menstrual cycle pattern in 14–17 year old old adolescents with gynaecological age, body mass index and historical parameters. Hum. Reprod. 1998, 13, 2252–2260. [Google Scholar] [CrossRef] [PubMed]
- Munster, K.; Schmidt, L.; Helm, P. Length and variation in the menstrual cycle—A cross-sectional study from a Danish county. Br. J. Obstet. Gynaecol. 1992, 99, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Naess, M.; Langhammer, A.; Forsmo, S. Ovulation Prevalence in Women with Spontaneous Normal-Length Menstrual Cycles—A Population-Based Cohort from HUNT3, Norway. PLoS ONE 2015, 10, e0134473. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.A.; Prior, J.C.; Koehoorn, M. Age at Menarche in the Canadian Population: Secular Trends and Relationship to Adulthood BMI. J. Adolesc. Health 2008, 43, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, B.K.; Heuch, I.; Kvale, G. Association of low age at menarche with increased all-cause mortality: A 37-year follow-up of 61,319 Norwegian women. Am. J. Epidemiol. 2007, 166, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- De Laet, C.; Kanis, J.A.; Oden, A.; Johanson, H.; Johnell, O.; Delmas, P.; Eisman, J.A.; Kroger, H.; Fujiwara, S.; Garnero, P.; et al. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporos. Int. 2005, 16, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.; Kanis, J.A.; Oden, A.; McCloskey, E.; Chapurlat, R.D.; Christiansen, C.; Cummings, S.R.; Diez-Perez, A.; Eisman, J.A.; Fujiwara, S.; et al. A meta-analysis of the association of fracture risk and body mass index in women. J. Bone Miner. Res. 2014, 29, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, K.K.; Folsom, A.R.; Anderson, K.E. Menstrual history and risk of hip fractures in postmenopausal women. The Iowa Women’s Health Study. Am. J. Epidemiol. 2001, 153, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Kreiger, N.; Tenenhouse, A.; Joseph, L.; Mackenzie, M.D.; Poliquin, S.; Brown, J.P.; Prior, J.C.; Rittmaster, R.S. The Canadian Multicentre Osteoporosis Study (CaMos): Background, rationale, methods. Can. J. Aging 1999, 18, 376–387. [Google Scholar] [CrossRef]
- Prior, J.C.; Kirkland, S.; Joseph, L.; Kreiger, N.; Murray, T.M.; Hanley, D.A.; Adachi, J.D.; Vigna, Y.M.; Berger, C.; Blondeau, L.; et al. Oral contraceptive agent use and bone mineral density in premenopausal women: Cross-sectional, population-based data from the Canadian Multicentre Osteoporosis Study. Can. Med. Assoc. J. 2001, 165, 1023–1029. [Google Scholar]
- Wikipedia. Available online: https://en.wikipedia.org/wiki/Demographics_of_Canada#Ethnic_origin (accessed on 21 February 2018).
- Kingwell, E.; Prior, J.C.; Ratner, P.A.; Kennedy, S.M. Direct-to-participant feedback and awareness of bone mineral density testing results in a population-based sample of mid-aged Canadians. Osteoporos. Int. 2010, 21, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Genant, H.K.; Grampp, S.; Gluer, C.C.; Faulkner, K.G.; Jergas, M.; Engelke, K.; Hagiwara, S.; Van Kuijk, C. Universal standardization for dual-x-ray absorptiometry: Patient and phantom cross-calibration results. J. Bone Miner. Res. 1994, 9, 1503–1514. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Goltzman, D.; Langsetmo, L.; Joseph, L.; Jackson, S.; Kreiger, N.; Tenenhouse, A.; Davison, K.S.; Josse, R.G.; Prior, J.; et al. Peak bone mass from longitudinal data: Implications for the prevalence, pathophysiology, and diagnosis of osteoporosis. J. Bone Miner. Res. 2010, 25, 1948–1957. [Google Scholar] [CrossRef] [PubMed]
- Tenenhouse, A.; Joseph, L.; Kreiger, N.; Poliquin, S.; Murray, T.M.; Prior, J.C.; Berger, C.; Hanley, D.A.; Prior, J.C.; CaMos Research Group, Canadian Multicentre Osteoporosis Study. Estimation of prevalence of low bone density in Canadian women and men using a population-specific DEXA reference standard: The Canadian Multicentre Osteoporosis Study (CaMos). Osteoporos. Int. 2000, 11, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Kalyan, S.; Seifert-Klauss, V. Re-naming pcos—Suggest anovulatory androgen excess. J. Clin. Endocrinol. Metab. 2013, 98, 4325–4328. [Google Scholar]
- Buchanan, J.R.; Hospodar, P.; Myers, C.A.; Leuenberger, P.; Demers, L.M. Effect of excess endogenous androgens on bone density in young women. J. Clin. Endocrinol. Metab. 1988, 67, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Simberg, N.; Tiitinen, A.; Silfvast, A.; Viinikka, L.; Ylikorkala, O. High bone density in hyperandrogenic women: Effect of gonadotropin-releasing hormone agonist alone or in conjunction with estrogen-progestin replacement. J. Clin. Endocrinol. Metab. 1996, 81, 646–651. [Google Scholar] [PubMed]
- Prezelj, J.; Kocijancic, A. Bone mineral density in hyperandrogenic amenorrhoea. Calcif. Tissue Int. 1993, 52, 422–424. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Vigna, Y.M.; Schechter, M.T.; Burgess, A.E. Spinal bone loss and ovulatory disturbances. N. Engl. J. Med. 1990, 323, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Hitchcock, C.L.; Barr, S.I.; Yu, T.; Prior, J.C. Negative Spinal Bone Mineral Density Changes and Subclinical Ovulatory Disturbances—Prospective Data in Healthy Premenopausal Women With Regular Menstrual Cycles. Epidemiol. Rev. 2014, 36, 147. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, F.; Wang, X.; Arguelles, L.; Rosul, L.L.; Venners, S.A.; Chen, C.; Hsu, Y.H.; Terwedow, H.; Wu, D.; Tang, G.; et al. Menstrual cycle lengths and bone mineral density: A cross-sectional, population-based study in rural Chinese women ages 30–49 years. Osteoporos. Int. 2007, 18, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Bainbridge, K.E.; Sowers, M.R.; Crutchfield, M.; Lin, X.; Jannausch, M.; Harlow, S.D. Natural history of bone loss over 6 years among premenopausal and early postmenopausal women. Am. J. Epidemiol. 2002, 156, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Lau, H.H.; Ng, M.Y.; Ho, A.Y.; Luk, K.D.; Kung, A.W. Genetic and environmental determinants of bone mineral density in Chinese women. Bone 2005, 36, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Hawker, G.A.; Forsmo, S.; Cadarette, S.M.; Schei, B.; Jaglal, S.B.; Forsen, L.; Langhammer, A. Correlates of forearm bone mineral density in young Norwegian women: The Nord-Trondelag Health Study. Am. J. Epidemiol. 2002, 156, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Winther, A.; Dennison, E.; Ahmed, L.A.; Furberg, A.S.; Grimnes, G.; Jorde, R.; Gjesdal, C.G.; Emaus, N. The Tromso Study: Fit Futures: A study of Norwegian adolescents’ lifestyle and bone health. Arch. Osteoporos. 2014, 9, 185. [Google Scholar] [CrossRef] [PubMed]
- Chevalley, T.; Bonjour, J.P.; Van Rietbergen, B.; Rizzoli, R.; Ferrari, S. Fractures in healthy females followed from childhood to early adulthood are associated with later menarcheal age and with impaired bone microstructure at peak bone mass. J. Clin. Endocrinol. Metab. 2012, 97, 4174–4181. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Jones, G.; Sambrook, P.N.; White, C.P.; Kelly, P.J.; Eisman, J.A. Effects of estrogen exposure and reproductive factors on bone mineral density and osteoporotic fractures. J. Clin. Endocrinol. Metab. 1995, 80, 2709–2713. [Google Scholar] [PubMed]
- Forsmo, S.; Schei, B.; Langhammer, A.; Forsen, L. How do reproductive and lifestyle factors influence bone density in distal and ultradistal radius of early postmenopausal women? The Nord-Trondelag Health Survey, Norway. Osteoporos. Int. 2001, 12, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, C.S. Maternal mineral and bone metabolism during pregnancy, lactation, and post-weaning recovery. Physiol. Rev. 2016, 96, 449–547. [Google Scholar] [CrossRef] [PubMed]
- Crandall, C.J.; Liu, J.; Cauley, J.; Newcomb, P.A.; Manson, J.E.; Vitolins, M.Z.; Jacobson, L.T.; Rykman, K.K.; Stefanick, M.L. Associations of Parity, Breastfeeding, and Fractures in the Women’s Health Observational Study. Obstet. Gynecol. 2017, 130, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Goulding, A.; Jones, I.E.; Taylor, R.W.; Manning, P.J.; Williams, S.M. More broken bones: A 4-year double cohort study of young girls with and without distal forearm fractures. J. Bone Miner. Res. 2000, 15, 2011–2018. [Google Scholar] [CrossRef] [PubMed]
- Chevalley, T.; Bonjour, J.P.; Ferrari, S.; Rizzoli, R. Deleterious effect of late menarche on distal tibia microstructure in healthy 20-year-old and premenopausal middle-aged women. J. Bone Miner. Res. 2009, 24, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Fogelholm, M.; Sievanen, H.; Heinonen, A.; Virtanen, M.; Uusi-Rasi, K.; Pasanen, M.; Vuori, I. Association between weight cycling history and bone mineral density in premenopausal women. Osteoporos. Int. 1997, 7, 354–358. [Google Scholar] [CrossRef] [PubMed]
- Meyer, H.E.; Tverdal, A.; Selmer, R. Weight variability, weight change and the incidence of hip fractures: A prospective study of 39000 middle-aged Norwegians. Osteoporos. Int. 1998, 8, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Bedford, J.L.; Prior, J.C.; Barr, S.I. A prospective exploration of cognitive dietary restraint, subclinical ovulatory disturbances, cortisol and change in bone density over two years in healthy young women. J. Clin. Endocrinol. Metab. 2010, 95, 3291–3299. [Google Scholar] [CrossRef] [PubMed]
- Hagemans, M.L.; Van Der Schouw, Y.T.; de Kleijn, M.J.; van Staveren, W.A.; Pop, V.J.; Leusink, G.L.; Grobbee, D.E. Indicators for the total duration of premenopausal endogenous estrogen exposure in relation to BMD. Hum. Reprod. 2004, 19, 2163–2169. [Google Scholar] [CrossRef] [PubMed]
- Brot, C.; Jensen, L.B.; Sorensen, O.H. Bone mass and risk factors for bone loss in perimenopausal Danish women. J. Intern. Med. 1997, 242, 505–511. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Mean (SD/%) | n |
|---|---|---|
| Age, years | 51.48 (7.43) | 1653 |
| Age at menarche, years | 12.75 (1.54) | 1646 |
| ≤10 years at menarche, n (%) | 97 (5.9) | |
| ≥16 years at menarche, n (%) | 84 (5.1) | |
| Regular menses after menarche, n (%) | ||
| Immediately | 1318 (79.8) | 1651 |
| Became regular spontaneously | 64 (19.5) | 329 |
| Became regular with medication | 151 (56) | 268 |
| Parity, n (%) | 1403 | |
| Nulliparous (0 births) | 62 (4.4) | |
| 1–2 births | 874 (62.3) | |
| 3 or more births | 467 (31.1) | |
| 1 Breast feeding, n (%) months | 1328 | |
| Never | 496 (37.3) | |
| ≤6 months | 367 (27.6) | |
| >6 months | 465 (35) | |
| 1 CHC use, n (%) duration | 1390 | |
| ≤3 months = “never” | 66 (4.7) | |
| 4–12 months | 215 (15.5) | |
| >12 months | 1109 (79.8) | |
| 2 Oligomenorrhea, yes, n (%) | 164 (10.1) | 1505 |
| 3 Amenorrhea, yes, n (%) | 137 (8.3) | 1516 |
| Infertility, n (%) | 145 (8.8) | 1653 |
| Hormonal | 45 (2.7) | |
| Anatomical, obstructive | 37 (2.2) | |
| Male factor | 15 (0.9) | |
| 4 Other | 48 (2.9) | |
| Androgen Excess, yes, n (%) | 199 (13.0) | 1653 |
| Hysterectomy, yes, n (%) | 393 (23.8) | 1653 |
| Ovariectomy, n (%) | 230 (13.9) | 1653 |
| Unilateral | 95 (5.7) | |
| Bilateral | 130 (7.9) | |
| Unsure | 5 (0.3) | |
| Natural menopause, yes, n (%) | 532 (43.7) | 1217 |
| Age at natural menopause, years | 50.6 (4.1) | |
| Reproductive lifespan, years | 38.7 (7.6) |
| Characteristic * | Overall Mean (SD) | n | Lumbar Spine (L1-4) BMD Quartiles | p ** | |||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||||
| L1-4 BMD mean (SD), g/cm2 | 1.053 (0.135) | 462 | 0.891 (0.050) n:115 | 1.005 (0.026) n:115 | 1.088 (0.024) n:116 | 1.228 (0.096) n:116 | – |
| Range of LS (L1-4) values g/cm2 | 0.71 to 1.65 | 0.71 to 0.95 | 0.96 to 1.04 | 1.05 to 1.12 | 1.13 to 1.65 | – | |
| Mean age (SD), years | 43.7 (6.9) | 462 | 44.1 (6.8) n:115 | 43.6 (7.2) n:115 | 44.2 (6.6) n:116 | 43.1 (7.0) n:116 | 0.59 |
| Mean current height (SD), cm | 162.4 (6.5) | 425 | 160.5 (6.5) n: 102 | 161.6 (6.7) n:105 | 163.2(6.3) n:106 | 164.3 (6.1) n:112 | 0.001 |
| Mean current weight (SD), kg | 69.5 (15.4) | 424 | 60.9 (11.8) n:102 | 67.4 (12.8) n:105 | 71.8 (14.3) n:106 | 77.2 (17.3) n:111 | 0.001 |
| Mean current BMI (SD), kg/m2 | 26.31 (5.52) | 424 | 23.65 (4.51) n:102 | 25.79 (4.55) n:105 | 26.97 (5.36) n:106 | 28.06 (6.25) n:111 | 0.001 |
| Mean BMI at age 18 (SD), kg/m2 | 21.06 (3.10) | 433 | 20.15 (2.69) N:110 | 20.56 (2.69) n:108 | 21.54 (3.37) n:106 | 21.99 (3.26) n:109 | 0.001 |
| Mean BMI change from age 18 (SD), kg/m2 | 5.25 (4.59) | 399 | 3.58 (4.20) n:99 | 5.20 (3.97) n:99 | 5.62 (4.31) n:97 | 6.54 (5.28) n:104 | 0.001 |
| Race (%), white | 94.2 | 499 | 92.2 | 91.3 | 94.8 | 98.3 | 0.10 |
| Education (% yes) ≥ professional certif. | 71.9 | 499 | 72.2 | 76.5 | 71.6 | 67.2 | 0.48 |
| * Ever smoked (% yes) | 45.0 | 499 | 40.9 | 38.3 | 45.7 | 55.2 | 0.051 |
| Mean alcohol use (SD), drink/week | 2.4 (3.6) | 462 | 1.9 (2.9) n:115 | 2.5 (4.2) n:115 | 2.8 (4.6) n:116 | 2.4 (3.7) n:116 | 0.61 |
| * Exercise mean (SD) expenditure Kjoules/week | 4627 (3582) | 451 | 4051 (3423) n:111 | 4482 (3616) n:112 | 4842 (3706) n:112 | 5111 (3536) n:116 | 0.13 |
| * Calcium—mean intake (SD), mg/d | 955 (572) | 446 | 905 (620) n:111 | 999 (587) n:111 | 984 (630) n:110 | 935 (435) N:114 | 0.58 |
| * Vitamin D mean intake (SD), µg/d | 4.7 (5.5) | 457 | 4.7 (6.3) n: 113 | 5.2 (5.5) n: 114 | 4.5 (4.7) n: 114 | 4.4 (5.5) n: 116 | 0.71 |
| * Weight cycling (%), yes | 30.3 | 26.1 | 23.5 | 29.3 | 42.2 | 0.005 | |
| Age at menarche mean (SD), years | 12.8 (1.4) | 459 | 13.1 (1.6) n:114 | 12.7 (1.5) n:113 | 12.7 (1.2) n:116 | 12.6 (1.5) n:116 | 0.07 |
| * Gynecological age mean (SD), years | 31.0 (7.1) | 459 | 31.1 (7.1) n:114 | 30.9 (7.5) n:113 | 31.5 (6.8) n:116 | 30.5 (6.9) n:116 | 0.40 |
| * Parous (%), yes | 94.3 | 395 | 94.5 | 97.8 | 95.9 | 89 | 0.16 |
| * Ever used CHC (%), yes | 85.3 | 443 | 85.2 | 86.1 | 86.2 | 83.6 | 0.94 |
| * Ever Breastfed (%), yes | 37.7 | 301 | 27.0 | 40.0 | 44.8 | 38.8 | 0.037 |
| * Androgen excess (%), yes | 16.7 | 499 | 22.6 | 14.8 | 11.2 | 18.2 | 0.11 |
| * Oligomenorrhea (%), yes | 24.9 | 499 | 23.5 | 23.5 | 28.4 | 24.1 | 0.78 |
| * Amenorrhea (%), yes | 7.6 | 499 | 11.3 | 5.2 | 7.8 | 6.1 | 0.31 |
| Femoral neck BMD (SD), g/cm2 | 0.810 (0.112) | 449 | 0.673 (0.042) n:112 | 0.769 (0.022) n: 112 | 0.842 (0.021) n:113 | 0.958 (0.066) n:113 | − |
| Total hip BMD mean (SD), g/cm2 | 0.951 (0.122) | 444 | 0.804 (0.048) n:111 | 0.902 (0.025) n:111 | 0.987 (0.027) n:111 | 1.110 (0.122) n:111 | − |
| Women with any Prevalent Fragility Fracture, n (%) | 62 (13.5) | 499 | 20 (17.5) | 14 (12.2) | 21(18.3) | 7 (6.1) | 0.03 |
| Estimate (95% CI) | Lumbar Spine (n = 423) g/cm2 | Femoral Neck (n = 409) g/cm2 | Total Hip (n = 407) g/cm2 |
|---|---|---|---|
| Current BMI (kg/m2) | 0.009 (0.006 to 0.012) | 0.008 (0.05 to 0.010) | 0.010 (0.008 to 0.013) |
| Current Height (cm) | 0.004 (0.001 to 0.006) | 0.004 (0.002 to 0.006) | 0.003 (0.001 to 0.005) |
| Menarche age (years) | −0.011 (−0.020 to −0.001) | ||
| Amenorrhea (yes) | −0.079 (−0.141 to −0.018) | ||
| Androgen excess (yes) | −0.050 (−0.092 to −0.008) | ||
| Total R2 | 0.176 | 0.209 | 0.232 |
| Model 1—BMI: R2 = 0.111 | Model 1—BMI: R2 = 0.153 | Model 1—BMI: R2 = 0.205 | |
| Model 2—height added to R2 0.025 Model 3—amenorrhea added to R2 0.021 | Model 2—height added to R2 0.039 Model 3—age at menarche added to R2 0.017 | Model 2—height added to R2 0.027 | |
| Model 4—androgen excess added to R2 0.019 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goshtasebi, A.; Berger, C.; Barr, S.I.; Kovacs, C.S.; Towheed, T.; Davison, K.S.; Prior, J.C. Adult Premenopausal Bone Health Related to Reproductive Characteristics—Population-Based Data from the Canadian Multicentre Osteoporosis Study (CaMos). Int. J. Environ. Res. Public Health 2018, 15, 1023. https://doi.org/10.3390/ijerph15051023
Goshtasebi A, Berger C, Barr SI, Kovacs CS, Towheed T, Davison KS, Prior JC. Adult Premenopausal Bone Health Related to Reproductive Characteristics—Population-Based Data from the Canadian Multicentre Osteoporosis Study (CaMos). International Journal of Environmental Research and Public Health. 2018; 15(5):1023. https://doi.org/10.3390/ijerph15051023
Chicago/Turabian StyleGoshtasebi, Azita, Claudie Berger, Susan I. Barr, Christopher S. Kovacs, Tanveer Towheed, K. Shawn Davison, and Jerilynn C. Prior. 2018. "Adult Premenopausal Bone Health Related to Reproductive Characteristics—Population-Based Data from the Canadian Multicentre Osteoporosis Study (CaMos)" International Journal of Environmental Research and Public Health 15, no. 5: 1023. https://doi.org/10.3390/ijerph15051023
APA StyleGoshtasebi, A., Berger, C., Barr, S. I., Kovacs, C. S., Towheed, T., Davison, K. S., & Prior, J. C. (2018). Adult Premenopausal Bone Health Related to Reproductive Characteristics—Population-Based Data from the Canadian Multicentre Osteoporosis Study (CaMos). International Journal of Environmental Research and Public Health, 15(5), 1023. https://doi.org/10.3390/ijerph15051023





