Stress and Alterations in the Pain Matrix: A Biopsychosocial Perspective on Back Pain and Its Prevention and Treatment
Abstract
1. Introduction
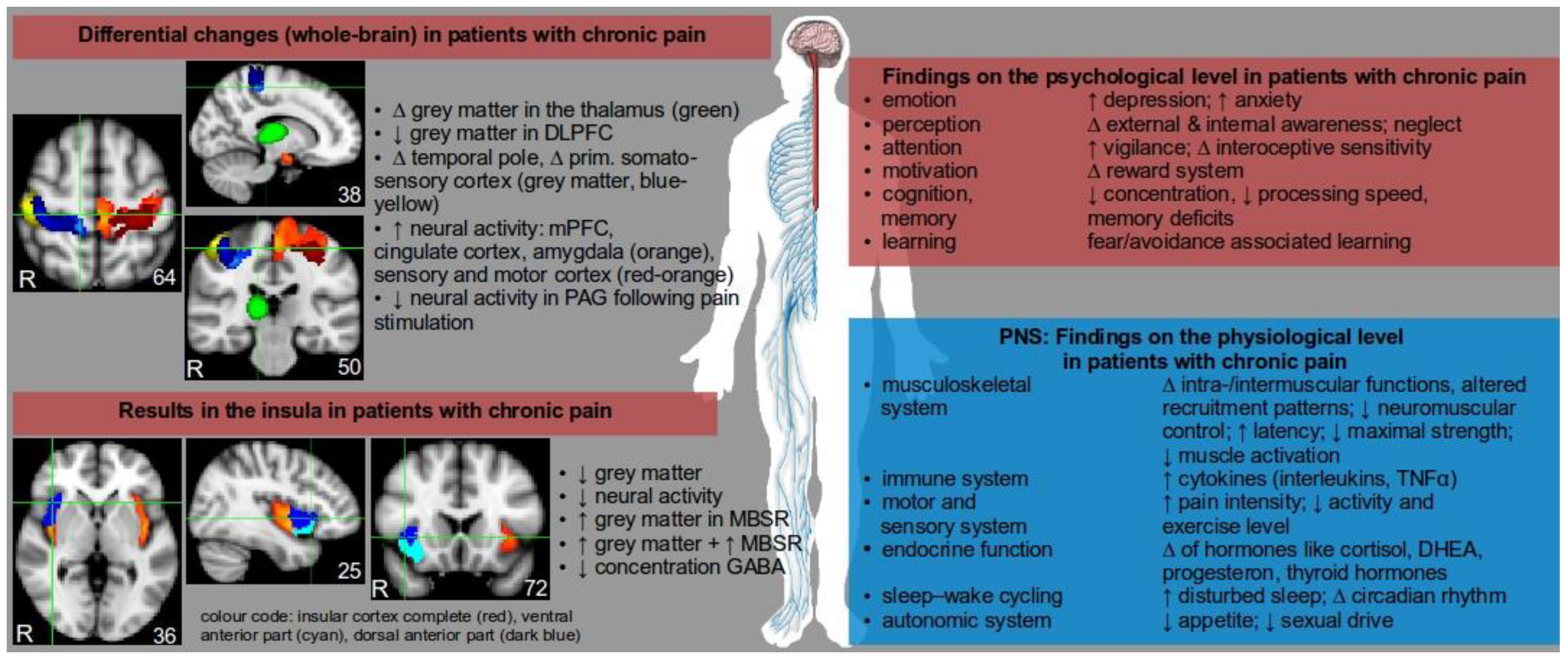
2. Neurobiological Consequences of Pain
3. Neurobiological Consequences of Physical Stress (PS) Such as Exercise
3.1. Adaptation to Exercise in the Prevention of Chronic Pain
3.2. Adaptation to Exercise in Therapy
4. Neurobiological Consequences of Mental Stress (MS)
4.1. Adaptation to Psychosocial Stress
4.2. Adaptation to Relaxation Treatments in Therapy
5. Interaction between Mental Stress and Physical Stress (MS and PS)
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ANS | autonomic nervous system |
| CNS | central nervous system |
| DLPFC | dorsolateral prefrontal cortex |
| EIH | Exercise-induces hypoalgesia |
| fMRI | functional magnetic resonance imaging |
| GABA | γ-aminobutyric acid |
| HPA | hypothalamic–pituitary–adrenal axis |
| HPG | hypothalamic–pituitary–gonadal axis |
| HPP | hypothalamic–pituitary–prolactin axis |
| HPS | hypothalamic–pituitary–somatotropic axis |
| HPT | hypo-thalamic–pituitary–thyroid axis |
| LBP | low back pain |
| CLBP | chronic low back pain |
| MBSR | mindfulness-based stress reduction program |
| MPFC | medial prefrontal cortex |
| MS | mental stress |
| PS | physical stress |
| rsfMRI | resting-state functional magnetic resonance imaging |
| SMN | sensory-motor neural network |
| sMRI | structural magnetic resonance imaging |
| SMT | sensory-motor training |
| SNRIs | selective noradrenaline reuptake inhibitors |
| SSRIs | selective serotonin reuptake inhibitors |
| NSAIDs | nonsteroidal anti-inflammatory drugs |
References
- Vos, T.; Barber, R.M.; Bell, B.; Bertozzi-Villa, A.; Biryukov, S.; Bolliger, I.; Charlson, F.; Davis, A.; Degenhardt, L.; Dicker, D.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 743–800. [Google Scholar] [CrossRef]
- Henschke, N.; Kamper, S.J.; Maher, C.G. The epidemiology and economic consequences of pain. Mayo Clin. Proc. 2015, 90, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Hoy, D.; Brooks, P.; Blyth, F.; Buchbinder, R. The epidemiology of low back pain. Best Pract. Res. Clin. Rheumatol. 2010, 24, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.B.; Goode, A.P.; George, S.Z.; Cook, C.E. Incidence and risk factors for first-time incident low back pain: A systematic review and meta-analysis. Spine J. 2014, 14, 2299–2319. [Google Scholar] [CrossRef] [PubMed]
- Diatchenko, L.; Nackley, A.G.; Slade, G.D.; Fillingim, R.B.; Maixner, W. Idiopathic pain disorders-pathways of vulnerability. Pain 2006, 123, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Henry, D.E.; Chiodo, A.E.; Wang, W. Central nervous system reorganization in a variety of chronic pain states: A review. PMR 2011, 3, 1116–1125. [Google Scholar]
- Fabbro, F.; Crescentini, C. Facing the experience of pain: A neuropsychological perspective. Phys. Life Rev. 2014, 11, 540–542. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Wilcke, T.; Leinisch, E.; Gänssbauer, S.; Draganski, B.; Bogdahn, U.; Altmeppen, J.; May, A. Affective components and intensity of pain correlate with structural differences in gray matter in chronic back pain patients. Pain 2006, 125, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Apkarian, A.V.; Sosa, Y.; Sonty, S.; Levy, R.M.; Harden, R.N.; Parrish, T.B.; Gitelman, D.R. Chronic back pain is associated with decreased prefrontal and thalamic gray matter density. J. Neurosci. 2004, 24, 10410–10415. [Google Scholar] [CrossRef] [PubMed]
- Kregel, J.; Meeus, M.; Malfliet, A.; Dolphens, M.; Danneels, L.; Nijs, J.; Cagnie, B. Structural and functional brain abnormalities in chronic low back pain: A systematic review. Semin. Arthritis Rheum. 2015, 45, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Baliki, M.N.; Petre, B.; Torbey, S.; Herrmann, K.M.; Huang, L.; Schnitzer, T.J.; Fields, H.L.; Apkarian, A.V. Corticostriatal functional connectivity predicts transition to chronic back pain. Nat. Neurosci. 2012, 15, 1117–1119. [Google Scholar] [CrossRef] [PubMed]
- Franklin, T.R.; Wetherill, R.R.; Jagannathan, K.; Hager, N.; O'Brien, C.P.; Childress, A.R. Limitations of the use of the MP-RAGE to identify neural changes in the brain: Recent cigarette smoking alters gray matter indices in the striatum. Front. Hum. Neurosci. 2014, 8, 1052. [Google Scholar] [CrossRef] [PubMed]
- Baliki, M.N.; Geha, P.Y.; Apkarian, A.V.; Chialvo, D.R. Beyond feeling: Chronic pain hurts the brain, disrupting the default-mode network dynamics. J. Neurosci. 2008, 28, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Giesecke, T.; Gracely, R.H.; Grant, M.A.; Nachemson, A.; Petzke, F.; Williams, D.A.; Clauw, D.J. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 2004, 50, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Paulus, M.P. Neural basis of mindfulness interventions that moderate the impact of stress on the brain. Neuropsychopharmacology 2016, 41, 373. [Google Scholar] [CrossRef] [PubMed]
- Harvey, V.; Dickenson, A. Neurobiology of pain. In Evidence-Based Chronic Pain Management; Stannard, C., Kalso, E., Ballantyne, J., Eds.; Wiley-Blackwell: Oxford, UK, 2010; pp. 42–51. [Google Scholar]
- Borghuis, J.A.; Hof, L.; Lemmink, K.A. The importance of sensory-motor control in providing core stability: Implications for measurement and training. Sports Med. 2008, 38, 893–916. [Google Scholar] [CrossRef] [PubMed]
- Burciu, R.G.; Fritsche, N.; Granert, O.; Schmitz, L.; Spönemann, N.; Konczak, J.; Theyson, N.; Gerwig, M.; van Eimeren, T.; Timmann, D. Brain changes associated with postural training in patients with cerebellar degeneration: A voxel-based morphometry study. J. Neurosci. 2013, 33, 4594–4604. [Google Scholar] [CrossRef] [PubMed]
- Vivar, C.; Potter, M.C.; van Praag, H. All about running: Synaptic plasticity, growth factors and adult hippocampal neurogenesis. Curr. Top. Behav. Neurosci. 2013, 15, 189–210. [Google Scholar] [PubMed]
- Erickson, K.I.; Gildengers, A.G.; Butters, M.A. Physical activity and brain plasticity in late adulthood. Dial. Clin. Neurosci. 2013, 15, 99–108. [Google Scholar]
- Hoeger-Bement, M.K. Exercise-induced hypoalgesia: An evidenced-based review. In Management and Mechanisms of Pain for the Physical Therapist; Bement, M.K.H., Sluka, K.A., Eds.; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2009. [Google Scholar]
- Naugle, K.M.; Fillingim, R.B.; Riley, J.L. A meta-analytic review of the hypoalgesic effects of exercise. J. Pain 2012, 13, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Tesarz, J.; Schuster, A.K.; Hartmann, M.; Gerhardt, A.; Eich, W. Pain perception in athletes compared to normally active controls: A systematic review with meta-analysis. Pain 2012, 153, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Naugle, K.M.; Naugle, K.E.; Fillingim, R.B.; Samuels, B.; Riley, J.L. Intensity thresholds for aerobic exercise–induced hypoalgesia. Med. Sci. Sports Exerc. 2014, 46, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, K.F. Exercise-induced hypoalgesia and intensity of exercise. Sports Med. 2012, 32, 477–487. [Google Scholar] [CrossRef]
- O’Leary, S.; Falla, D.; Hodges, P.W. Specific therapeutic exercise of the neck induces immediate local hypoalgesia. J. Pain 2007, 8, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Tesarz, J.; Gerhardt, A.; Schommer, K.; Treede, R.D.; Eich, W. Alterations in endogenous pain modulation in endurance athletes: An experimental study using quantitative sensory testing and the cold-pressor task. Pain 2013, 154, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Koltyn, K.F.; Brellenthin, A.G.; Cook, D.B.; Sehgal, N.; Hillard, C. Mechanisms of exercise-induced hypoalgesia. J. Pain 2014, 15, 1294–1304. [Google Scholar] [CrossRef] [PubMed]
- Radebold, A.; Cholewicki, J.; Panjabi, M.M.; Patel, T.C. Muscle response pattern to sudden trunk loading in healthy individuals and in patients with chronic low back pain. Spine (Phila Pa 1976) 2000, 25, 947–954. [Google Scholar] [CrossRef]
- Bayramoglu, M.; Akman, M.N.; Klnç, S.; Çetin, N.; Yavuz, N.; Özker, R. Isokinetic measurement of trunk muscle strength in women with chronic low-back pain. Am. J. Phys. Med. Rehabil. 2001, 80, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Standaert, C.J.; Weinstein, S.M.; Rumpeltes, J. Evidence-informed management of chronic low back pain with lumbar stabilization exercises. Spine J. 2008, 8, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Hayden, J.A.; Van Tulder, M.W.; Tomlinson, G. Systematic review: Strategies for using exercise therapy to improve outcomes in chronic low back pain. Ann. Intern. Med. 2005, 142, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Saragiotto, B.T.; Maher, C.G.; Yamato, T.P.; Costa, L.O.; Costa, L.C.; Ostelo, R.W.; Macedo, L.G. Motor control exercise for chronic non-specific low back pain. Cochrane Database Syst. Rev. 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Hayden, J.A.; van Tulder, M.W.; Malmivaara, A.; Koes, B.W. Exercise therapy for treatment of non-specific low back pain. Cochrane Database Syst. Rev. 2005, 1, CD000335. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Shekelle, P. Will this patient develop persistent disabling low back pain? JAMA 2010, 303, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.R. Individual differences in endogenous pain modulation as a risk factor for chronic pain. Neurology 2005, 65, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Riedl, V.; Valet, M.; Wöller, A.; Sorg, C.; Vogel, D.; Sprenger, T.; Boecker, H.; Wohlschläger, A.M.; Tölle, T. Repeated pain induces adaptations of intrinsic brain activity to reflect past and predict future pain. Neuroimage 2011, 57, 206–213. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Nasveld, P.; Palmer, M.; Anderson, R. Allostatic Load: A Review of the Literature; Department of Veterans’ Affairs: Canberra, Australia, 2012.
- Buhle, J.T.; Stevens, B.L.; Friedman, J.J.; Wager, T.D. Distraction and placebo two separate routes to pain control. Psychol. Sci. 2012, 23, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Villemure, C.; Bushnell, M.C. Mood influences supraspinal pain processing separately from attention. J. Neurosci. 2009, 29, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Valet, M.; Sprenger, T.; Boecker, H.; Willoch, F.; Rummeny, E.; Conrad, B.; Erhard, P.; Tolle, T.R. Distraction modulates connectivity of the cingulo-frontal cortex and the midbrain during pain—An fmri analysis. Pain 2004, 109, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Villarreal, E.A.G.; Brattico, E.; Vase, L.; Østergaard, L.; Vuust, P. Superior analgesic effect of an active distraction versus pleasant unfamiliar sounds and music: The influence of emotion and cognitive style. PLoS ONE 2012, 7, e29397. [Google Scholar] [CrossRef] [PubMed]
- Wippert, P.M.; de Witt Huberts, J.; Klipker, K.; Gantz, S.; Schiltenwolf, M.; Mayer, F. Beschreibung und empirische fundierung des verhaltenstherapeutischen moduls der MiSpEx-intervention. Der Schmerz 2015, 29, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Niederer, D.; Vogt, L.; Wippert, P.M.; Puschmann, A.K.; Pfeifer, A.C.; Schiltenwolf, M.; Banzer, W.; Mayer, F. Medicine in spine exercise (MiSpEx) for nonspecific low back pain patients: Study protocol for a multicentre, single-blind randomized controlled trial. Trials 2016, 17, 507. [Google Scholar] [CrossRef] [PubMed]
- Wippert, P.M.; Rector, M.; Kuhn, G.; Wuertz-Kozak, K. Stress and alterations in bones: An interdisciplinary perspective. Front. Endocrinol. 2017, 8, 96. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Horm. Behav. 2003, 43, 2–15. [Google Scholar] [CrossRef]
- McEwen, B.S. Physiology and neurobiology of stress and adaptation: Central role of the brain. Physiol. Rev. 2007, 87, 873–904. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Kalia, M. The role of corticosteroids and stress in chronic pain conditions. Metabolism 2010, 59, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Jiao, K.; Niu, L.; Xu, X.; Liu, Y.; Li, X.; Tay, F.R.; Wang, H. Norepinephrine regulates condylar bone loss via comorbid factors. J. Dent. Res. 2015, 94, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Generaal, E.; Vogelzangs, N.; Macfarlane, G.J.; Geenen, R.; Smit, J.H.; Penninx, B.W.; Dekker, J. Reduced hypothalamic-pituitary-adrenal axis activity in chronic multi-site musculoskeletal pain: Partly masked by depressive and anxiety disorders. BMC Musculoskelet. Disord. 2014, 15, 227. [Google Scholar] [CrossRef] [PubMed]
- Van Houdenhove, B.; Luyten, P. Beyond dualism: The role of life stress in chronic pain. Pain 2005, 113, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Verheijen, M.; Chrast, R.; Burrola, P.; Lemke, G. Local regulation of fat metabolism in peripheral nerves. Genes Dev. 2003, 17, 2450–2464. [Google Scholar] [CrossRef] [PubMed]
- Amsalem, M.; Poilbout, C.; Ferracci, G.; Delmas, P.; Padilla, F. Membrane cholesterol depletion as a trigger of Nav1.9 channel-mediated inflammatory pain. EMBO J. 2018, 37, e97349. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.; Singh, S.; Sibinga, E.M.S.; Gould, N.F.; Rowland-Seymour, A.; Sharma, R.; Berger, Z.; Sleicher, D.; Maron, D.D.; Shihab, H.M.; et al. Meditation programs for psychological stress and well-being—A systematic review and meta-analysis. JAMA Intern. Med. 2014, 174, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Marchand, W.R. Mindfulness-based stress reduction, mindfulness-based cognitive therapy, and zen meditation for depression, anxiety, pain, and psychological distress. J. Psychiatr. Pract. 2012, 18, 233–252. [Google Scholar] [CrossRef] [PubMed]
- Morone, N.E.; Greco, C.M.; Weiner, D.K. Mindfulness meditation for the treatment of chronic low back pain in older adults: A randomized controlled pilot study. Pain 2008, 134, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Veehof, M.M.; Oskam, M.J.; Schreurs, K.M.; Bohlmeijer, E.T. Acceptance-based interventions for the treatment of chronic pain: A systematic review and meta-analysis. Pain 2011, 152, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Critchley, H.D.; Wiens, S.; Rotshtein, P.; Öhman, A.; Dolan, R.J. Neural systems supporting interoceptive awareness. Neural Syst. Support. Interoceptive Awareness 2004, 7, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Wiebking, C.; Duncan, N.W.; Tiret, B.; Hayes, D.J.; Marjaǹska, M.; Doyon, J.; Bajbouj, M.; Northoff, G. Gaba in the insula—A predictor of the neural response to interoceptive awareness. Neuroimage 2014, 86, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, F.; Grant, J.A.; Brown, C.A.; McHaffie, J.G.; Coghill, R.C. Mindfulness meditation-related pain relief: Evidence for unique brain mechanisms in the regulation of pain. Neurosci. Lett. 2012, 520, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Villemure, C.; Čeko, M.; Cotton, V.A.; Bushnell, M.C. Insular cortex mediates increased pain tolerance in yoga practitioners. Cereb. Cortex 2013, 24, 2732–2740. [Google Scholar] [CrossRef] [PubMed]
- Henschke, N.; Ostelo, R.W.; van Tulder, M.W.; Vlaeyen, J.W.; Morley, S.; Assendelft, W.J.; Main, C.J. Behavioural treatment for chronic low-back pain. Cochrane Database Syst. Rev. 2010, 7, CD002014. [Google Scholar] [CrossRef] [PubMed]
- Engers, A.J.; Jellema, P.; Wensing, M.; van der Windt, D.A.; Grol, R.; van Tulder, M.W. Individual patient education for low back pain. Cochrane Database Syst. Rev. 2008, 1, CD004057. [Google Scholar] [CrossRef] [PubMed]
- Heymans, M.W.; Van Tulder, M.W.; Esmail, R.; Bombardier, C.; Koes, B.W. Back schools for nonspecific low back pain. Cochrane Database Syst. Rev. 2004, 8, CD011674. [Google Scholar] [CrossRef]
- Cook, D.B.; Stegner, A.J.; Ellingson, L.D. Exercise alters pain sensitivity in Gulf War veterans with chronic musculoskeletal pain. J. Pain 2010, 11, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Wippert, P.M.; Puschmann, A.K.; Drießlein, D.; Arampatzis, A.; Banzer, W.; Beck, H.; Schiltenwolf, M.; Schmidt, H.; Schneider, C.; Mayer, F. Development of a risk stratification and prevention index for stratified care in chronic low back pain. Focus: Yellow flags (MiSpEx network). Pain Rep. 2017, 2, e623. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wippert, P.-M.; Wiebking, C. Stress and Alterations in the Pain Matrix: A Biopsychosocial Perspective on Back Pain and Its Prevention and Treatment. Int. J. Environ. Res. Public Health 2018, 15, 785. https://doi.org/10.3390/ijerph15040785
Wippert P-M, Wiebking C. Stress and Alterations in the Pain Matrix: A Biopsychosocial Perspective on Back Pain and Its Prevention and Treatment. International Journal of Environmental Research and Public Health. 2018; 15(4):785. https://doi.org/10.3390/ijerph15040785
Chicago/Turabian StyleWippert, Pia-Maria, and Christine Wiebking. 2018. "Stress and Alterations in the Pain Matrix: A Biopsychosocial Perspective on Back Pain and Its Prevention and Treatment" International Journal of Environmental Research and Public Health 15, no. 4: 785. https://doi.org/10.3390/ijerph15040785
APA StyleWippert, P.-M., & Wiebking, C. (2018). Stress and Alterations in the Pain Matrix: A Biopsychosocial Perspective on Back Pain and Its Prevention and Treatment. International Journal of Environmental Research and Public Health, 15(4), 785. https://doi.org/10.3390/ijerph15040785



