A Novel Pb-Resistant Bacillus subtilis Bacterium Isolate for Co-Biosorption of Hazardous Sb(III) and Pb(II): Thermodynamics and Application Strategy
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Bacillus subtilis
2.2. Chemicals and Determination
2.3. Batch Experiments
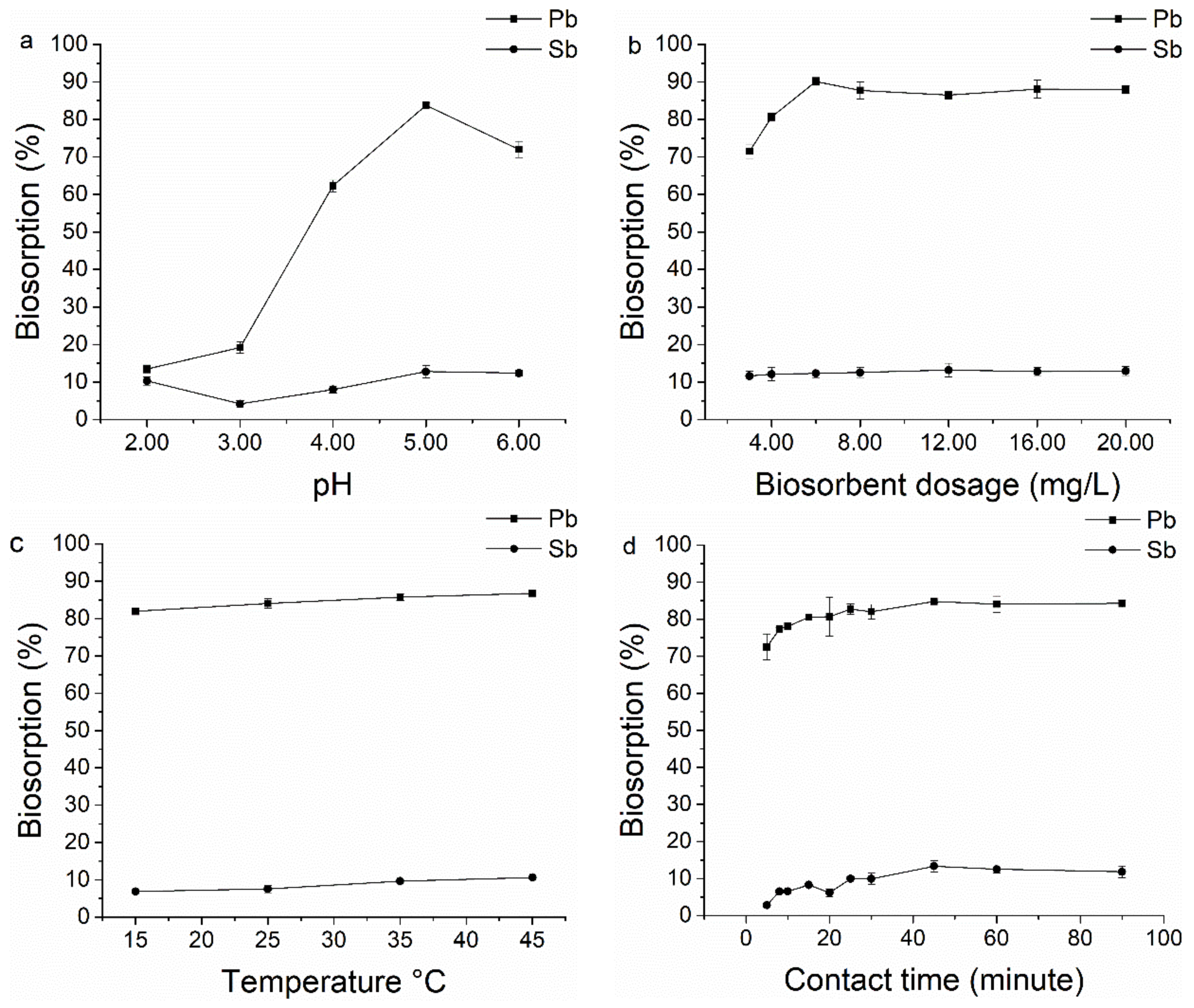
2.3.1. Effect of pH
2.3.2. Effect of Biomass Dosage
2.3.3. Effect of Temperature
2.3.4. Effect of Contact Time
2.4. Biosorption Isotherm Models
2.4.1. Langmuir Isotherm Model
2.4.2. Freundlich Isotherm Model
2.4.3. Temkin Isotherm Model
2.4.4. Dubinin-Radushkevich (D-R) Isotherm Model
2.5. Kinetic Models
2.5.1. Pseudo-First-Order Model
2.5.2. Pseudo-Second-Order Model
2.6. Microbial Bioleaching Experiment
3. Results
3.1. Optimum Conditions
3.1.1. pH
3.1.2. Biomass Dosage
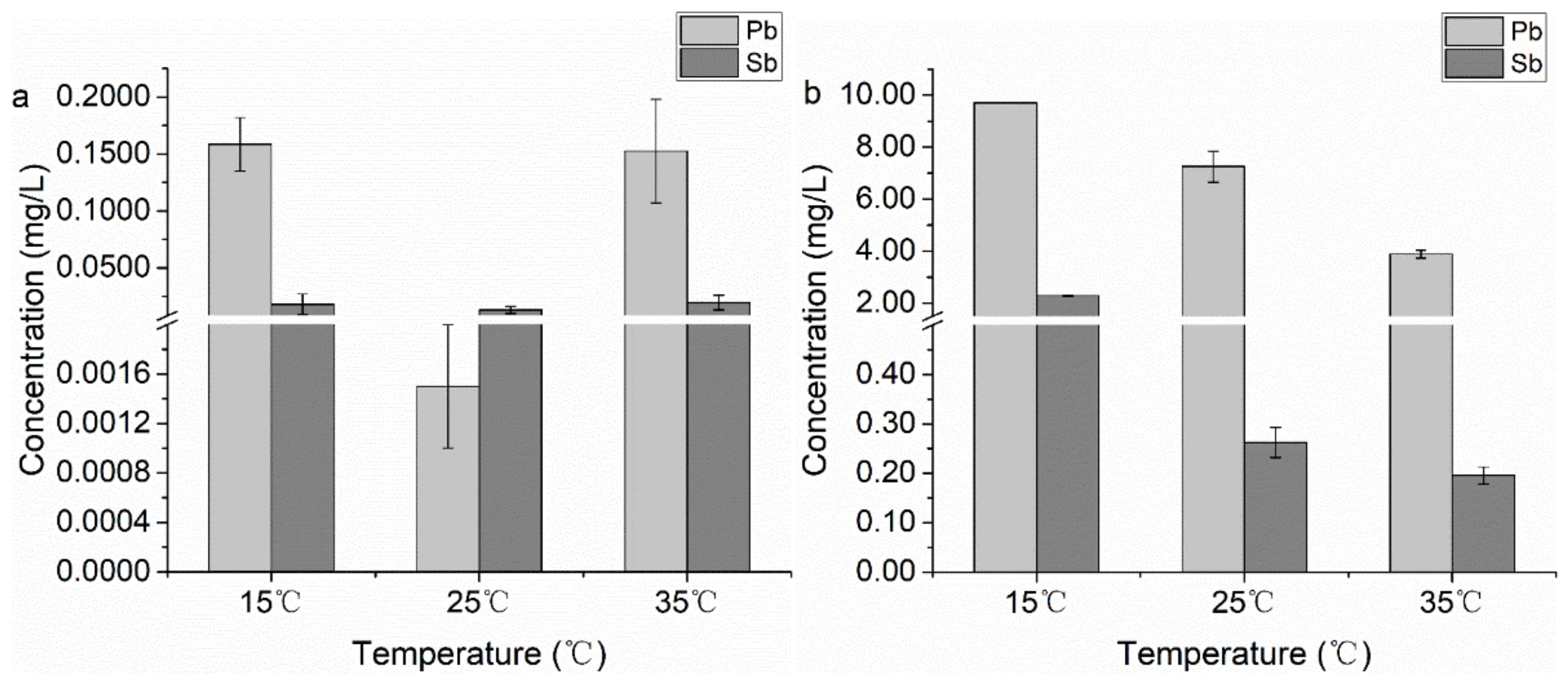
3.1.3. Temperature
3.1.4. Contact Time
3.2. FT-IR Analysis
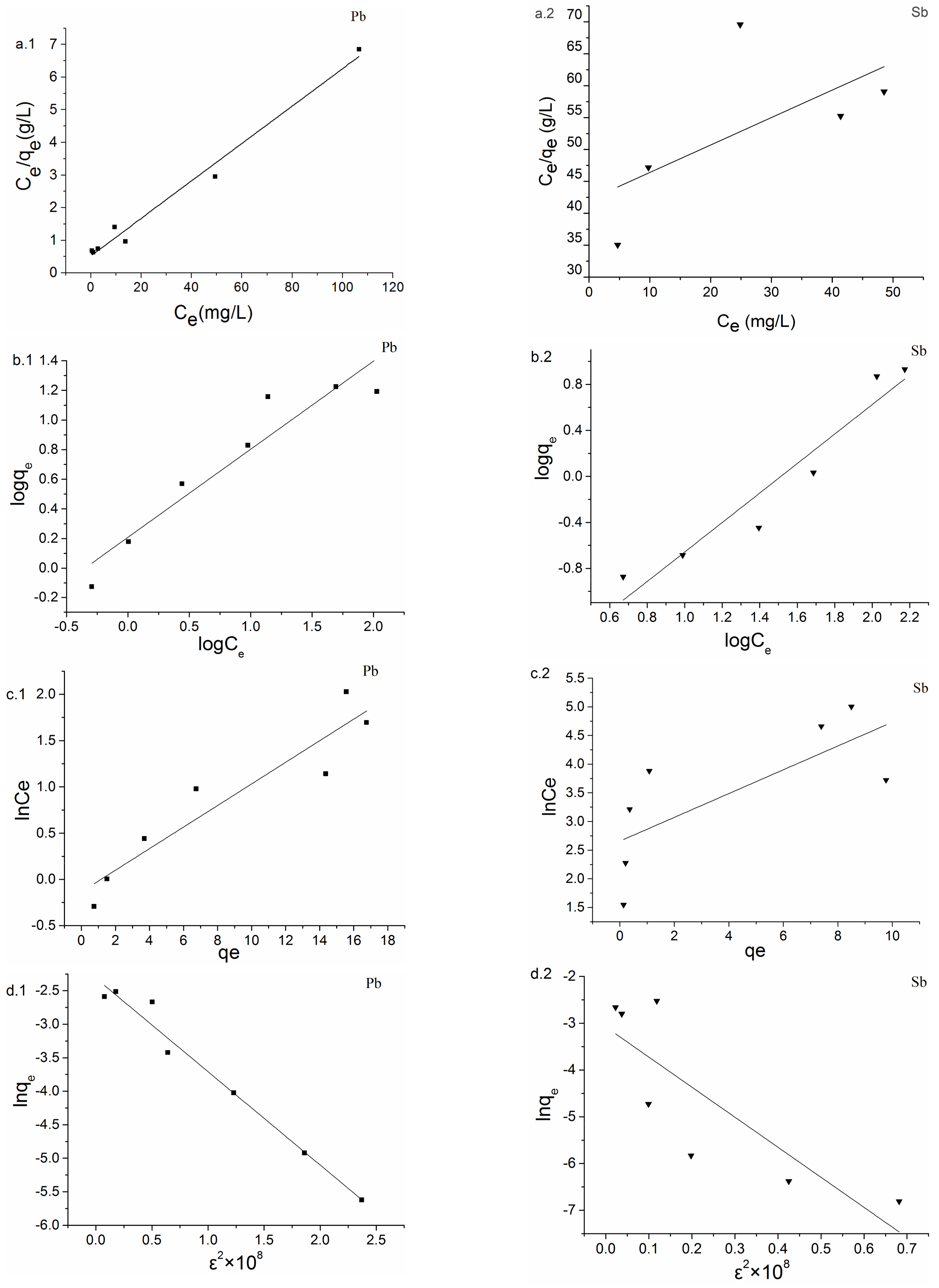
3.3. Biosorption Isotherm
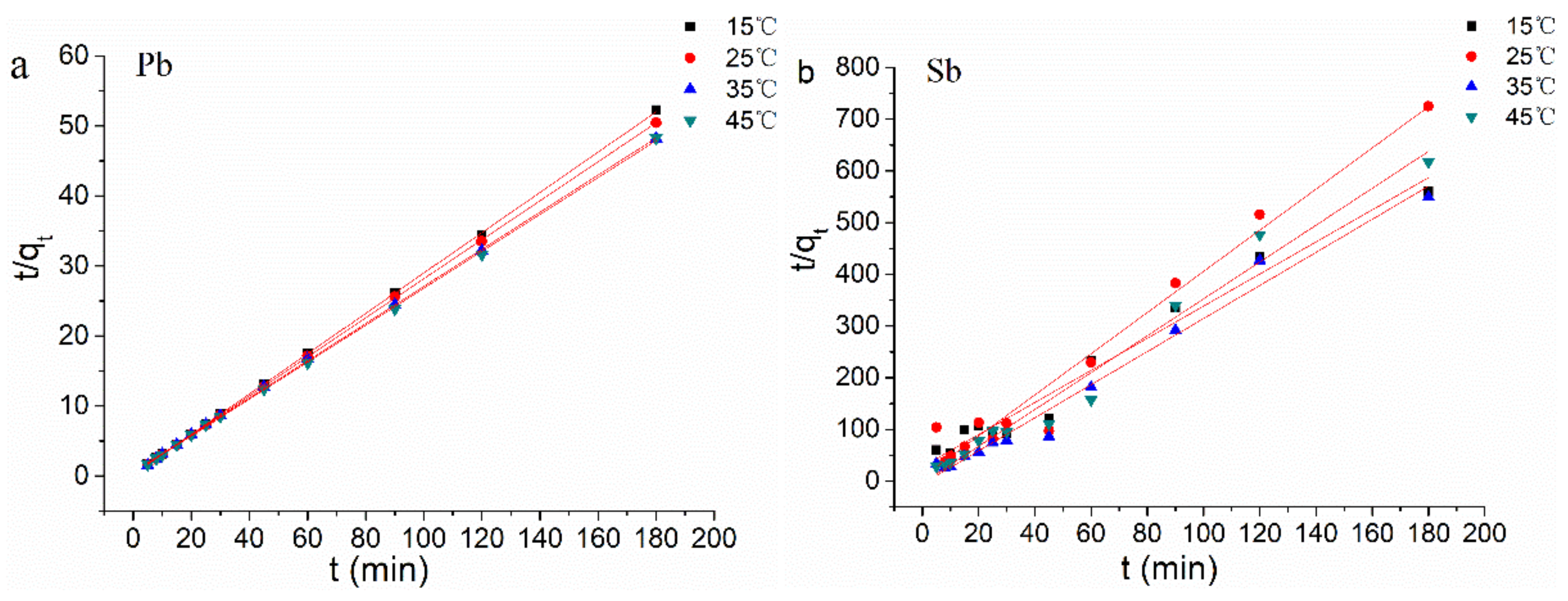
3.4. Biosorption Kinetics
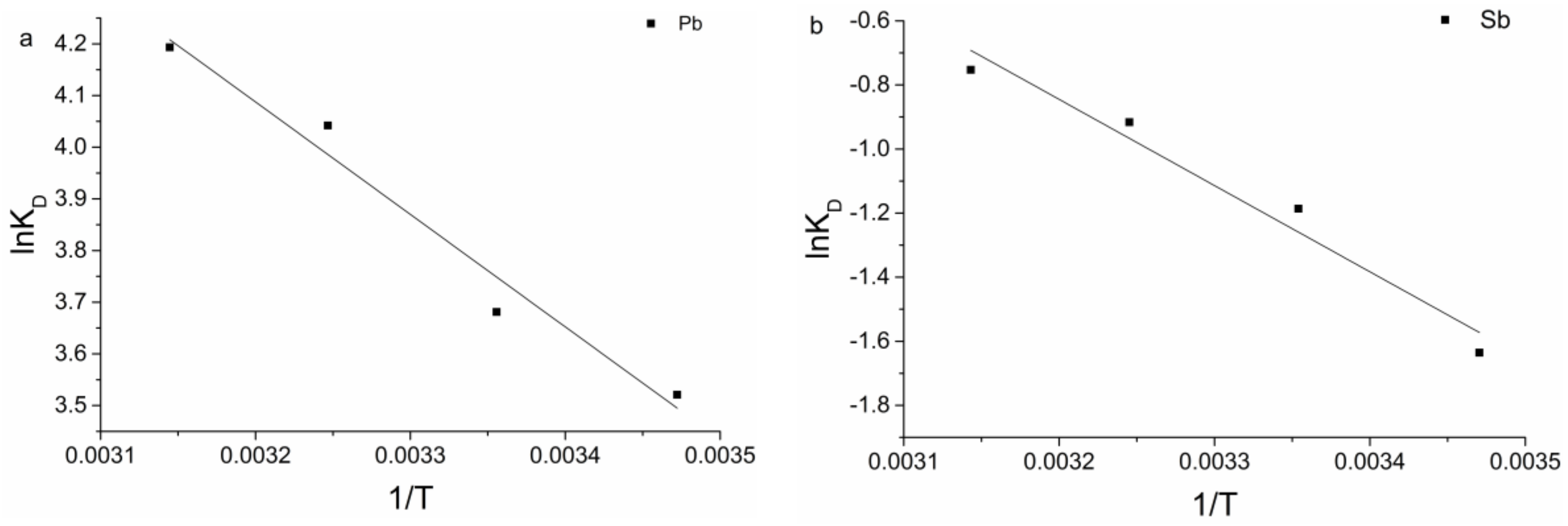
3.5. Biosorption Thermodynamics
4. Discussion
4.1. Strategy for Separation and Removal of Pb(II) and Sb(III) from Industrial Effluent and Wastewater
- Physicochemical characteristics of ‘real’ wastewater on the basis of thermodynamics and reaction kinetics.
- Screening biosorbents with high metal-binding capacity and selectivity.
- Optimization of parameters.
- Combination of biosorption with physicochemical treatment technologies for ‘complete’ wastewater treatment and recovery/reuse of metals.
4.2. Strategy for Possiblely Assisted Phytoremediation of Sb(III) and Pb(II) from Contaminated Soil
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mahmoud, M.E.; Nabil, G.M.; Mahmoud, S.M.E. High Performance Nano-Zirconium Silicate Adsorbent for Efficient Removal of Copper(II), Cadmium(II) and Lead(II). J. Environ. Chem. Eng. 2015, 3, 1320–1328. [Google Scholar] [CrossRef]
- Wu, S.C.; Peng, X.L.; Cheung, K.C.; Liu, S.L.; Wong, M.H. Adsorption kinetics of Pb and Cd by two plant growth promoting rhizobacteria. Bioresour. Technol. 2009, 100, 4559–4563. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Rastogi, A. Biosorption of lead(II) from aqueous solutions by non-living algal biomass Oedogonium sp. and Nostoc sp.—A comparative study. Colloids Surf. B Biointerfaces 2008, 64, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Tiwari, S.; Sharma, A.K.; Sanghi, R. Removal of lead from aqueous solutions using Cassia grandis seed gum-graft-poly(methylmethacrylate). J. Colloid Interface Sci. 2007, 316, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, N.; Hwang, G.; Sathishkumar, M.; Choi, T.K.; Lee, K.J.; Oh, B.T.; Lee, Y.S. Isolation, identification, Pb(II) biosorption isotherms and kinetics of a lead adsorbing Penicillium sp. MRF-1 from South Korean mine soil. J. Environ. Sci. 2010, 22, 1049–1056. [Google Scholar] [CrossRef]
- Hasan, S.H.; Srivastava, P.; Talat, M. Biosorption of Pb(II) from water using biomass of Aeromonas hydrophila: Central composite design for optimization of process variables. J. Hazard. Mater. 2009, 168, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Ungureanu, G.; Santos, S.; Boaventura, R.; Botelho, C. Biosorption of antimony by brown algae S. muticum and A. nodosum. Environ. Eng. Manag. J. 2015, 14, 455–463. [Google Scholar]
- Okkenhaug, G.; Zhu, Y.G.; Luo, L.; Lei, M.; Li, X.; Mulder, J. Distribution, speciation and availability of antimony (Sb) in soils and terrestrial plants from an active Sb mining area. Environ. Pollut. 2011, 159, 2427–2434. [Google Scholar] [CrossRef] [PubMed]
- Sigel, H.; Sigel, A.; Sigel, H. Handbook on Metals in Clinical and Analytical Chemistry; CRC Press: Boca Raton, FL, USA, 1994. [Google Scholar]
- Johnson, C.A.; Moench, H.; Wersin, P.; Kugler, P.; Wenger, C. Solubility of antimony and other elements in samples taken from shooting ranges. J. Environ. Qual. 2005, 34, 248. [Google Scholar] [PubMed]
- USEPA. Water Related Fate of the 129 Priority Pollutants; USEPA: Washington, DC, USA, 1979; Volume 1.
- USEPA. National Primary Drinking Water Standards; USEPA: Washington, DC, USA, 1999.
- USEPA. Lead in Drinking Water; USEPA: Washington, DC, USA, 2013; p. 1.
- Pan, X.; Wang, J.; Zhang, D. Biosorption of Pb(II) by Pleurotus Ostreatus Immobilized in Calcium Alginate Gel. Process Biochem. 2005, 40, 2799–2803. [Google Scholar]
- Naushad, M.; ALOthman, Z.A.; Awual, M.R.; Alam, M.M.; Eldesoky, G.E. Adsorption kinetics, isotherms, and thermodynamic studies for the adsorption of Pb2+ and Hg2+ metal ions from aqueous medium using Ti(IV) iodovanadate cation exchanger. Ionics 2015, 21, 2237–2245. [Google Scholar] [CrossRef]
- Ghasemi, M.; Naushad, M.; Ghasemi, N.; Khosravi-Fard, Y. Adsorption of Pb(II) from aqueous solution using new adsorbents prepared from agricultural waste: Adsorption isotherm and kinetic studies. J. Ind. Eng. Chem. 2014, 20, 2193–2199. [Google Scholar] [CrossRef]
- Al-Othman, Z.A.; Naushad, M.; Nilchi, A. Development, Characterization and Ion Exchange Thermodynamics for a New Crystalline Composite Cation Exchange Material: Application for the Removal of Pb2+ Ion from a Standard Sample (Rompin Hematite). J. Inorg. Organomet. Polym. Mater. 2011, 21, 547–559. [Google Scholar] [CrossRef]
- Bushra, R.; Naushad, M.; Adnan, R.; ALOthman, Z.A.; Rafatullah, M. Polyaniline supported nanocomposite cation exchanger: Synthesis, characterization and applications for the efficient removal of Pb2+ ion from aqueous medium. J. Ind. Eng. Chem. 2015, 21, 1112–1118. [Google Scholar] [CrossRef]
- Naushad, M.; ALOthman, Z.A.; Javadian, H. Removal of Pb(II) from aqueous solution using ethylene diamine tetra acetic acid-Zr(IV) iodate composite cation exchanger: Kinetics, isotherms and thermodynamic studies. J. Ind. Eng. Chem. 2015, 25, 35–41. [Google Scholar] [CrossRef]
- Naushad, M. Surfactant assisted nano-composite cation exchanger: Development, characterization and applications for the removal of toxic Pb2+ from aqueous medium. Chem. Eng. J. 2014, 235, 100–108. [Google Scholar] [CrossRef]
- Ali, I. New Generation Adsorbents for Water Treatment. Chem. Rev. 2012, 112, 5073–5091. [Google Scholar] [CrossRef] [PubMed]
- Congeevaram, S.; Dhanarani, S.; Park, J.; Dexilin, M.; Thamaraiselvi, K. Biosorption of chromium and nickel by heavy metal resistant fungal and bacterial isolates. J. Hazard. Mater. 2007, 146, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Shao, Z. Biosorption and bioaccumulation of lead by Penicillium sp. Psf-2 isolated from the deep sea sediment of the Pacific Ocean. Extremophiles 2007, 11, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Padmesh, T.V.N.; Palanivelu, K.; Velan, M. Biosorption of nickel(II) ions onto Sargassum wightii: Application of two-parameter and three-parameter isotherm models. J. Hazard. Mater. 2006, 133, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Yun, Y.S. Bacterial biosorbents and biosorption. Biotechnol. Adv. 2008, 26, 266–291. [Google Scholar] [CrossRef] [PubMed]
- Vieira, R.H.S.F.; Volesky, B. Biosorption: A Solution to pollution. Int. Mycrobiol. 2000, 3, 17–24. [Google Scholar]
- Remacle, J. The Cell Wall and Metal Binding. Biosorption of Heavy Metals; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Prasenjit, B.; Sumathi, S. Uptake of chromium by Aspergillus foetidus. J. Mater. Cycles Waste Manag. 2005, 7, 88–92. [Google Scholar] [CrossRef]
- Bautista-Hernández, D.A.; Ramírez-Burgos, L.I.; Duran-Páramo, E.; Fernández-Linares, L. Zinc and Lead Biosorption by Delftia tsuruhatensis: A Bacterial Strain Resistant to Metals Isolated from Mine Tailings. J. Water Resour. Protect. 2012, 4, 207–216. [Google Scholar] [CrossRef]
- Maldonado, J.; de los Rios, A.; Esteve, I.; Ascaso, C.; Puyen, Z.M.; Brambilla, C.; Solé, A. Sequestration and in vivo effect of lead on DE2009 microalga, using high-resolution microscopic techniques. J. Hazard. Mater. 2010, 183, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.-N.; Shim, J.; You, Y.; Myung, H.; Bang, K.S.; Cho, M.; Kamala-Kannan, S.; Oh, B.-T. Characterization of lead resistant endophytic Bacillus sp. MN3-4 and its potential for promoting lead accumulation in metal hyperaccumulator Alnus firma. J. Hazard. Mater. 2012, 199, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Yang, X.; Du, R.; Chen, Y.; Wang, S.; Qiu, R. Biosorption mechanisms involved in immobilization of soil Pb by Bacillus subtilis DBM in a multi-metal-contaminated soil. J. Environ. Sci. 2014, 26, 2056–2064. [Google Scholar] [CrossRef] [PubMed]
- Naik, M.M.; Dubey, S.K. Lead resistant bacteria: Lead resistance mechanisms, their applications in lead bioremediation and biomonitoring. Ecotoxicol. Environ. Saf. 2013, 98, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Pan, X.; Chen, H.; Guan, X.; Lin, Z. Biomineralization of Pb(II) into Pb-hydroxyapatite induced by Bacillus cereus 12-2 isolated from Lead-Zinc mine tailings. J. Hazard. Mater. 2016, 301, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Heng, S.; Munis, M.F.H.; Fahad, S.; Yang, X. Phytoremediation of heavy metals assisted by plant growth promoting (PGP) bacteria: A review. Environ. Exp. Bot. 2015, 117, 28–40. [Google Scholar] [CrossRef]
- Filella, M.; Belzile, N.; Lett, M.-C. Antimony in the environment: A review focused on natural waters. III. Microbiota relevant interactions. Earth Sci. Rev. 2007, 80, 195–217. [Google Scholar] [CrossRef]
- Li, D.; Xu, X.; Yu, H.; Han, X. Characterization of Pb2+ biosorption by psychrotrophic strain Pseudomonas sp. I3 isolated from permafrost soil of Mohe wetland in Northeast China. J. Environ. Manag. 2017, 196, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Çolak, F.; Atar, N.; Yazıcıoğlu, D.; Olgun, A. Biosorption of lead from aqueous solutions by Bacillus strains possessing heavy-metal resistance. Chem. Eng. J. 2011, 173, 422–428. [Google Scholar] [CrossRef]
- Matyara, F.; Kayab, A.; Dinçerb, S. Antibacterial agents and heavy metal resistance in gram-negative bacteria isolated from seawater, shrimp and sediment in iskenderun bay, turkey. Sci. Total Environ. 2008, 407, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, T.J. Role of Cellular Design in Bacterial Metal Accumulation and Mineralization. Ann. Rev. Microbiol. 1989, 43, 147–171. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, A.C.A. Chemical Interactions between Mercurial Species and Surface Biomolecules from Structural Components of Some Biological Systems. In Mercury Contaminated Sites: Characterization, Risk Assessment and Remediation; Ebinghaus, R., Ed.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 159–178. [Google Scholar]
- Dursun, A.Y.; Uslu, G.; Cuci, Y.; Aksu, Z. Bioaccumulation of copper(II), lead(II) and chromium(VI) by growing Aspergillus niger. Process Biochem. 2003, 38, 1647–1651. [Google Scholar] [CrossRef]
- Hemambika, B.; Rani, M.J.; Kannan, V.R. Biosorption of heavy metals by immobilized and dead fungal cells: A comparative assessmen. J. Ecol. Nat. Environ. 2011, 3, 168–175. [Google Scholar]
- Aksu, Z. Application of biosorption for the removal of organic pollutants: A review. Process Biochem. 2005, 40, 997–1026. [Google Scholar] [CrossRef]
- Turpeinen, R.; Kairesalo, T.; Häggblom, M.M. Microbial community structure and activity in arsenic-, chromium- and copper-contaminated soils. FEMS Microbiol. Ecol. 2004, 47, 39–50. [Google Scholar] [CrossRef]
- MIDI. MIDI Sherlock Microbial Identification System Operating Manual; MIDI: Newark, NJ, USA, 1995. [Google Scholar]
- Das, N. Recovery of precious metals through biosorption—A review. Hydrometallurgy 2010, 103, 180–189. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Chem. Phys. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Uber Die Adsorption in Lasungen. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar]
- Isik, M. Biosorption of Ni(II) from aqueous solutions by living and non-living ureolytic mixed culture. Colloids Surf. B Biointerfaces 2008, 62, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Günay, A.; Arslankaya, E.; Tosun, İ. Lead removal from aqueous solution by natural and pretreated clinoptilolite: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2007, 146, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Dubinin, M.M.; Zaverina, E.D.; Radushkevich, L.V. Sorption and Structure of Active Carbons I. Adsorption of Organic Vapors. Zhurnal Fizicheskoi Khimii 1947, 1947, 1351–1362. [Google Scholar]
- Lagergren, S.Y. ur theorie der sogenannten adsorption geloster stoffe, Kungliga Svenska Vetenskapsakademiens. Handlingar 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Lodeiro, P.; Barriada, J.L.; Herrero, R.; De Vicente, M.S. The marine macroalga Cystoseira baccata as biosorbent for cadmium(II) and lead(II) removal: Kinetic and equilibrium studies. Environ. Pollut. 2006, 142, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Woodburn, J.M.Q.Y.G. Biosorption of cadmium from aqueous solutions by pretreated biomass of marine alga Durvillaea potatorum. Water Res. 1999, 33, 335–342. [Google Scholar]
- Karthikeyan, S.; Balasubramanian, R.; Iyer, C.S.P. Evaluation of the marine algae Ulva fasciata and Sargassum sp. for the biosorption of Cu(II) from aqueous solutions. Bioresour. Technol. 2007, 98, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Panda, G.C.; Das, S.K.; Chatterjee, S.; Maity, P.B.; Bandopadhyay, T.S.; Guha, A.K. Adsorption of cadmium on husk of Lathyrus sativus: Physico-chemical study. Colloids Surf. B Biointerfaces 2006, 50, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Anayurt, R.A.; Sari, A.; Tuzen, M. Equilibrium, thermodynamic and kinetic studies on biosorption of Pb(II) and Cd(II) from aqueous solution by macrofungus (Lactarius scrobiculatus) biomass. Chem. Eng. J. 2009, 151, 255–261. [Google Scholar] [CrossRef]
- Uluozlu, O.D.; Sari, A.; Tuzen, M.; Soylak, M. Biosorption of Pb(II) and Cr(III) from aqueous solution by lichen (Parmelina tiliaceae) biomass. Bioresour. Technol. 2008, 99, 2972–2980. [Google Scholar] [CrossRef] [PubMed]
- Mckay, Y.H.G. The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res. 2000, 34, 735–742. [Google Scholar]
- Veglio, F.; Beolchini, F. Removal of metals by biosorption: A review. Hydrometallurgy 1997, 44, 301–316. [Google Scholar] [CrossRef]
- Martins, B.L.; Cruz, C.C.; Luna, A.S.; Henriques, C.A. Sorption and desorption of Pb2+ ions by dead Sargassum sp. biomass. Biochem. Eng. J. 2006, 27, 310–314. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Biosorption of heavy metals by Saccharomyces cerevisiae: A review. Biotechnol. Adv. 2006, 24, 427–451. [Google Scholar] [CrossRef] [PubMed]
- David Kratochvil, B.V. Advances in the biosorption of heavy metals. Trends Biotechnol. 1998, 16, 291–300. [Google Scholar] [CrossRef]
- Parvathi, K.; Nagendran, R.; Kumar, R.N. Lead biosorption onto waste beer yeast by-product, a means to decontaminate effluent generated from battery manufacturing industry. Electron. J. Biotechnol. 2007, 10, 92–105. [Google Scholar] [CrossRef][Green Version]
- Zhang, D.; Pan, X.; Zhao, L.; Mu, G. Biosorption of Antimony (Sb) by the Cyanobacterium Synechocystis sp. Pol. J. Environ. Stud. 2011, 20, 1353–1358. [Google Scholar]
- Pan, J.; Ge, X.; Liu, R.; Tang, H. Characteristic features of Bacillus cereus cell surfaces with biosorption of Pb(II) ions by AFM and FT-IR. Colloids Surf. B Biointerfaces 2006, 52, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Sun, F.; Wu, S.; Yan, Y.; Xing, B. Removal of antimony(III) from aqueous solution by freshwater cyanobacteria Microcystis biomass. Chem. Eng. J. 2012, 183, 172–179. [Google Scholar] [CrossRef]
- Tunali, S.; Çabuk, A.; Akar, T. Removal of lead and copper ions from aqueous solutions by bacterial strain isolated from soil. Chem. Eng. J. 2006, 115, 203–211. [Google Scholar] [CrossRef]
- Pan, J.-H.; Liu, R.-X.; Tang, H.-X. Surface reaction of Bacillus cereus biomass and its biosorption for lead and copper ions. J. Environ. Sci. 2007, 19, 403–408. [Google Scholar] [CrossRef]
- Filella, M.; Belzile, N.; Chen, Y.-W. Antimony in the environment: A review focused on natural waters: I. occurrence. Earth Sci. Rev. 2002, 57, 125–176. [Google Scholar] [CrossRef]
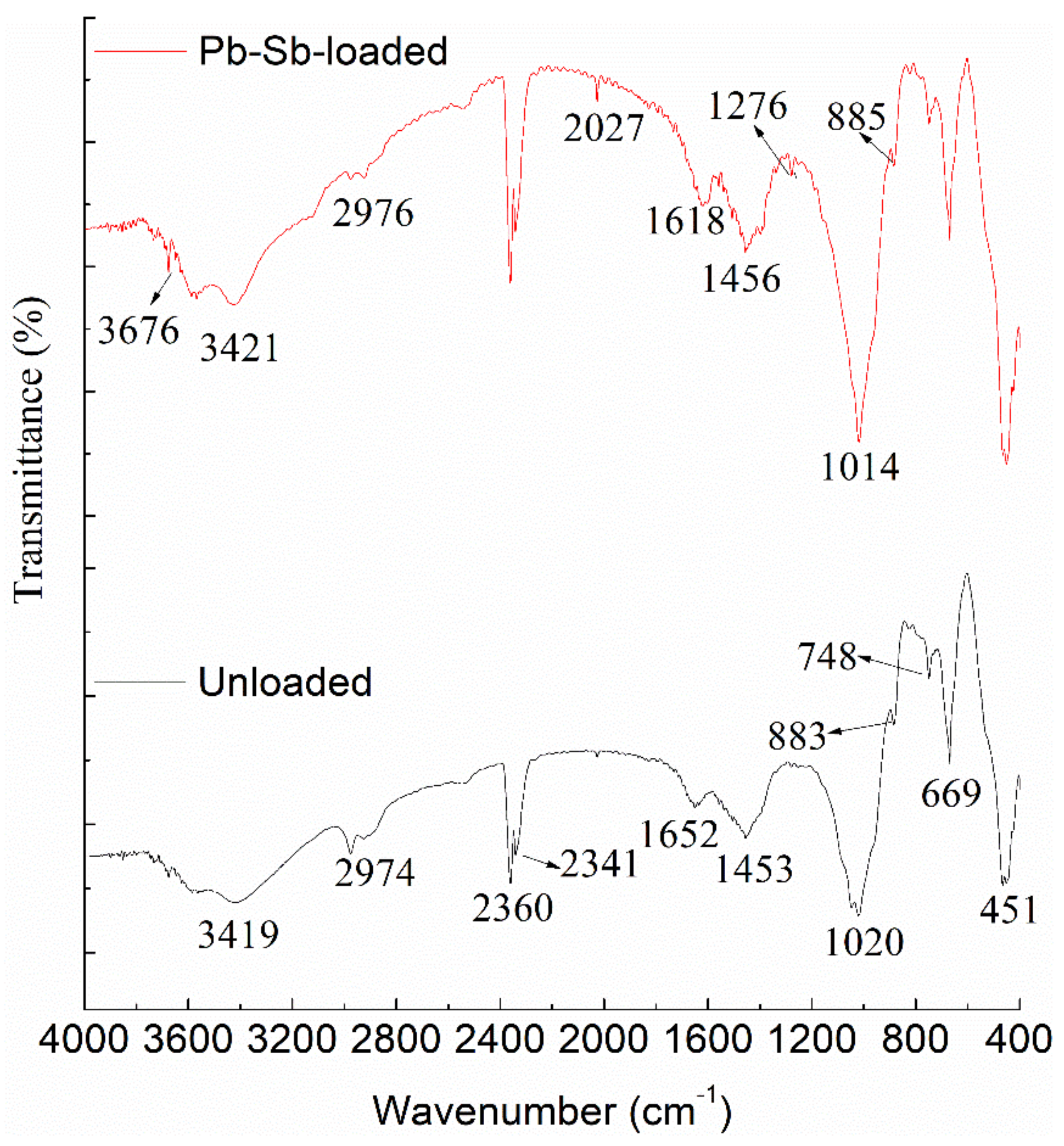
| Wavelength (cm−1) | Unloaded Cell (cm−1) | Pb-Sb-Loaded Cell (cm−1) | Assignment |
|---|---|---|---|
| 3750–3200 | 3419 | 3676, 3421 | bonded -OH or -NH groups |
| 3000–2850 | 2974 | 2976 | -CH stretching vibration. |
| 1680–1550 | 1652 | 1618 | C=O stretching in carboxyl or amide I and amide II groups |
| 1550–1375 | 1453 | 1456 | N-H bending, -CH2 scissoring or -CH3 asymmetrical bending vibration and O-H deformation |
| 1375–1300 | - | 1276 | C-O stretching of -COOH |
| 1200–1000 | 1020 | 1014 | C-O stretching of alcohols and carboxylic acids |
| 1000–800 | 883 | 885 | S=O stretching |
| Isotherm Model | Parameter | Pb(II) | Sb(III) |
|---|---|---|---|
| Langmuir | qmax (mg/g) | 17.43 | 2.32 |
| KL (L/mg) | 0.19 | 0.010 | |
| R2 | 0.98 | 0.20 | |
| Freundlich | kF (L/g)1/n | 1.62 | 19.23 |
| n | 1.68 | 0.78 | |
| R2 | 0.89 | 0.91 | |
| Temkin | a | 1.42 | 0.070 |
| b | 293.76 | 522.47 | |
| R2 | 0.87 | 0.45 | |
| D-R | qmax (mg/g) | 0.099 | 0.046 |
| β | 1.39 | 6.43 | |
| R2 | 0.97 | 0.64 |
| T (°C) | qe, exp | Pseudo First Order Equation | Pseudo Second Order Equation | |||||
|---|---|---|---|---|---|---|---|---|
| (mg/g) | k1 (min−1) | qe (mg/g) | R2 | k2 (g/mg min) | qe (mg/g) | R2 | ||
| Pb(II) | 15 | 3.489 | 0.012 | 0.210 | 0.439 | 0.405 | 3.474 | 0.999 |
| 25 | 3.577 | 0.020 | 0.311 | 0.842 | 0.251 | 3.589 | 0.999 | |
| 35 | 3.737 | 0.030 | 0.695 | 0.957 | 0.124 | 3.772 | 0.999 | |
| 45 | 3.791 | 0.040 | 0.879 | 0.991 | 0.150 | 3.795 | 0.974 | |
| Sb(III) | 15 | 0.322 | 0.0073 | 0.199 | 0.728 | 0.345 | 0.322 | 0.974 |
| 25 | 0.248 | 0.0047 | 0.118 | 0.251 | 2.027 | 0.251 | 0.967 | |
| 35 | 0.328 | −0.0004 | 0.062 | −0.123 | −1.947 | 0.313 | 0.979 | |
| 45 | 0.292 | 0.0014 | 0.127 | −0.089 | −3.340 | 0.281 | 0.976 | |
| Parameter | ∆G (KJ/mol) | ∆H (KJ/mol) | ∆S KJ/(mol·K) | |||
|---|---|---|---|---|---|---|
| Temperature (°C) | Pb | Sb | Pb | Sb | Pb | Sb |
| 15 | −16.13 | −4.23 | 18.10 | 22.37 | 91.85 | 64.56 |
| 25 | −17.09 | −4.43 | ||||
| 35 | −18.58 | −5.38 | ||||
| 45 | −19.59 | −5.63 | ||||
| Pb(II) | Sb(III) | ||||||
|---|---|---|---|---|---|---|---|
| Biosorbent | qm (mg/g) | Biosorption Mechanism | Reference | Biosorbent | qm (mg/g) | Biosorption Mechanism | Reference |
| Bacillus subtilis | 17.34 | L | this study | Bacillus subtilis | 2.32 | F | this study |
| Bacillus cereus | 23.3 | L | [38] | Brown Algae | 5.5 | L | [7] |
| Bacillus pumilus | 29.6 | L | [38] | Cyanobacterium Synechocystis sp. | 4.68 | L | [67] |
| Bacillus cereus | 72.0 | R-P | [68] | Microcystis | 4.88 | L | [69] |
| Bacillus sp. (ATS-1) | 92.3 | L | [70] | ||||
| Bacillus cereus | 36.7 | L | [71] | ||||
| Bacillus subtilis | 57.0 | Not mention | [32] | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, Y.; Li, X.; Liu, D.; Xu, C.; Ai, Y.; Sun, X.; Zhang, M.; Gao, Y.; Zhang, Y.; Yang, T.; et al. A Novel Pb-Resistant Bacillus subtilis Bacterium Isolate for Co-Biosorption of Hazardous Sb(III) and Pb(II): Thermodynamics and Application Strategy. Int. J. Environ. Res. Public Health 2018, 15, 702. https://doi.org/10.3390/ijerph15040702
Cai Y, Li X, Liu D, Xu C, Ai Y, Sun X, Zhang M, Gao Y, Zhang Y, Yang T, et al. A Novel Pb-Resistant Bacillus subtilis Bacterium Isolate for Co-Biosorption of Hazardous Sb(III) and Pb(II): Thermodynamics and Application Strategy. International Journal of Environmental Research and Public Health. 2018; 15(4):702. https://doi.org/10.3390/ijerph15040702
Chicago/Turabian StyleCai, Yue, Xiaoping Li, Dongying Liu, Changlin Xu, Yuwei Ai, Xuemeng Sun, Meng Zhang, Yu Gao, Yuchao Zhang, Tao Yang, and et al. 2018. "A Novel Pb-Resistant Bacillus subtilis Bacterium Isolate for Co-Biosorption of Hazardous Sb(III) and Pb(II): Thermodynamics and Application Strategy" International Journal of Environmental Research and Public Health 15, no. 4: 702. https://doi.org/10.3390/ijerph15040702
APA StyleCai, Y., Li, X., Liu, D., Xu, C., Ai, Y., Sun, X., Zhang, M., Gao, Y., Zhang, Y., Yang, T., Wang, J., Wang, L., Li, X., & Yu, H. (2018). A Novel Pb-Resistant Bacillus subtilis Bacterium Isolate for Co-Biosorption of Hazardous Sb(III) and Pb(II): Thermodynamics and Application Strategy. International Journal of Environmental Research and Public Health, 15(4), 702. https://doi.org/10.3390/ijerph15040702







