The Performance of a Self-Flocculating Microalga Chlorococcum sp. GD in Wastewater with Different Ammonia Concentrations
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgal Strain
2.2. Synthetic Wastewater
2.3. Experimental Set-Up
2.4. Analytical Methods
2.4.1. Flocculating Ability Test
2.4.2. Extraction and Analysis of Extracellular Polymeric Substances (EPS)
2.4.3. Determination of Microalgal Growth
2.4.4. Determination of Water Quality
2.4.5. Determination of Microalgal Lipid
2.4.6. Statistical Analysis
3. Results
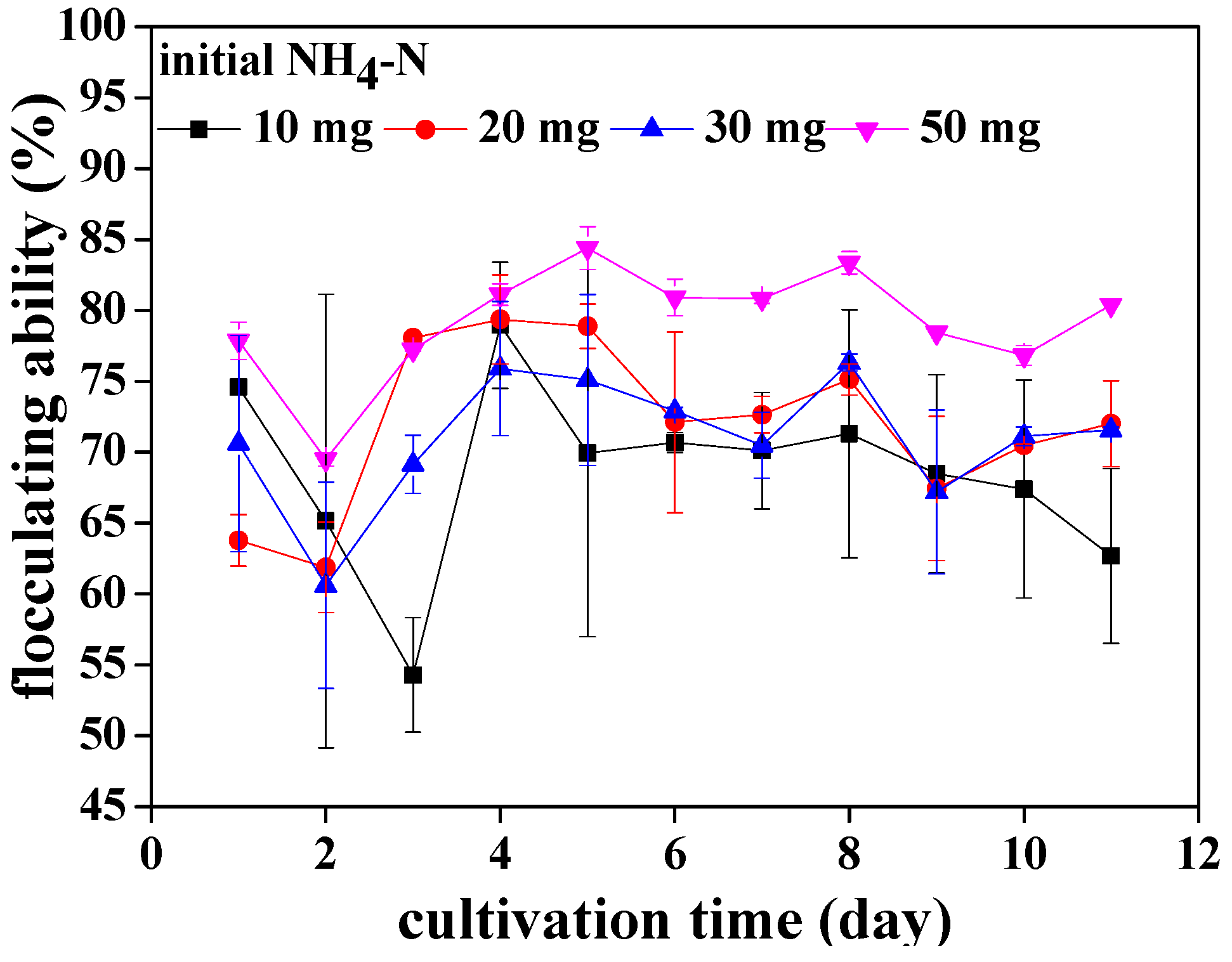
3.1. The Flocculating Property of Chlorococcum sp. GD in Wastewater with Different Ammonia Nitrogen Concentrations
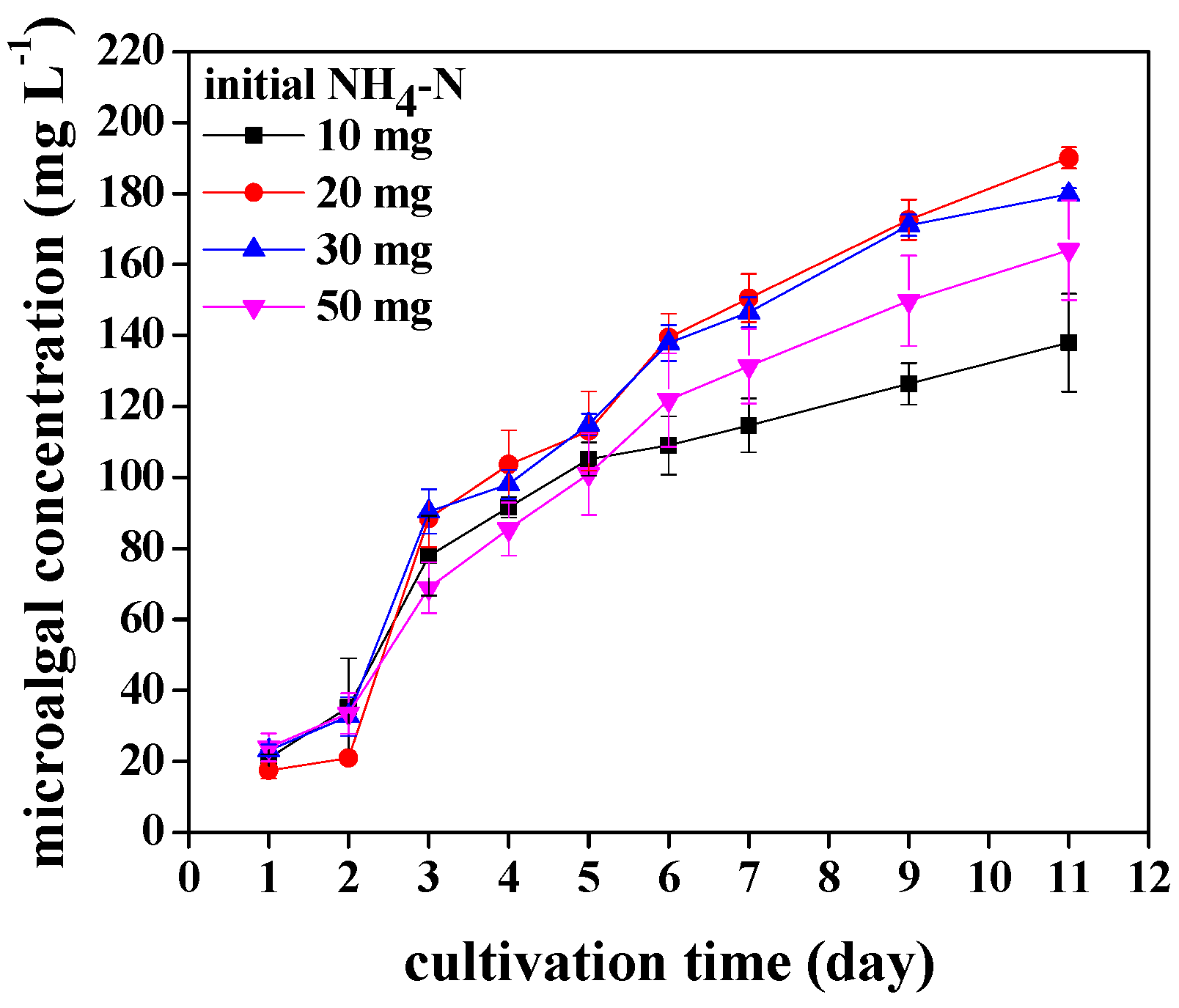
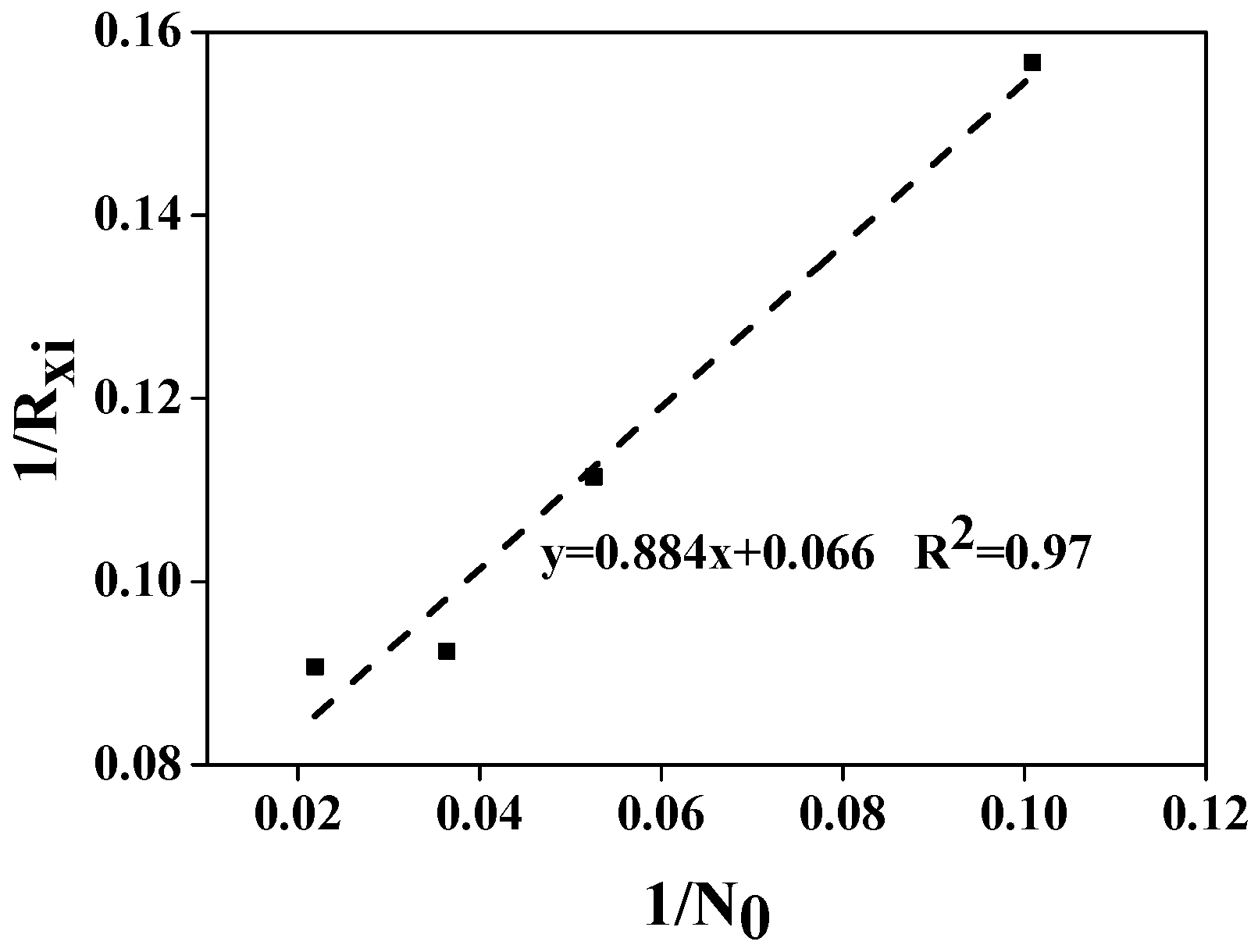
3.2. The Growth and Biomass Production of Chlorococcum sp. GD in Wastewater with Different Ammonia Nitrogen Concentrations
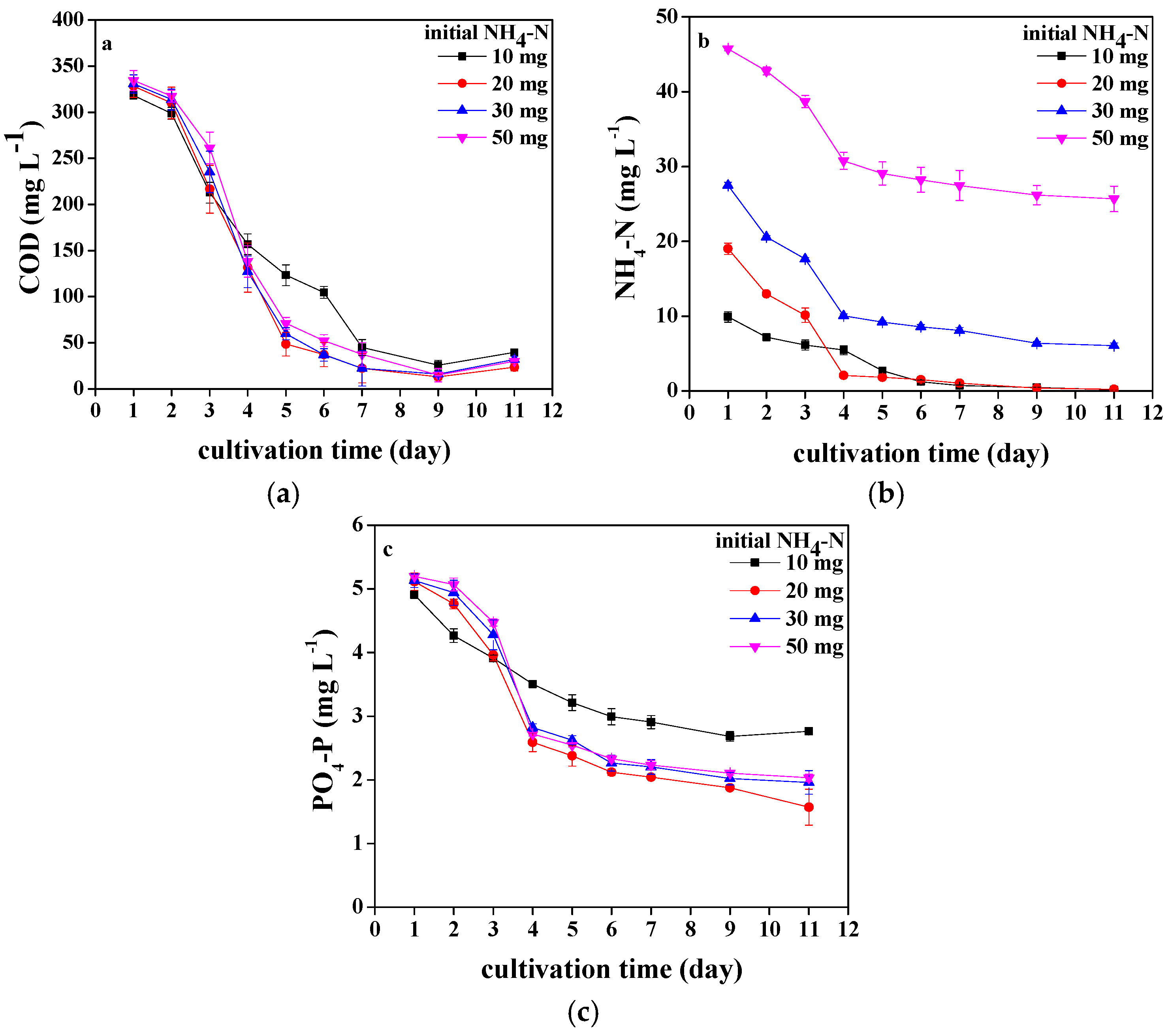
3.3. Pollutants Removal of Chlorococcum sp. GD in Wastewater with Different Ammonia Nitrogen Concentrations
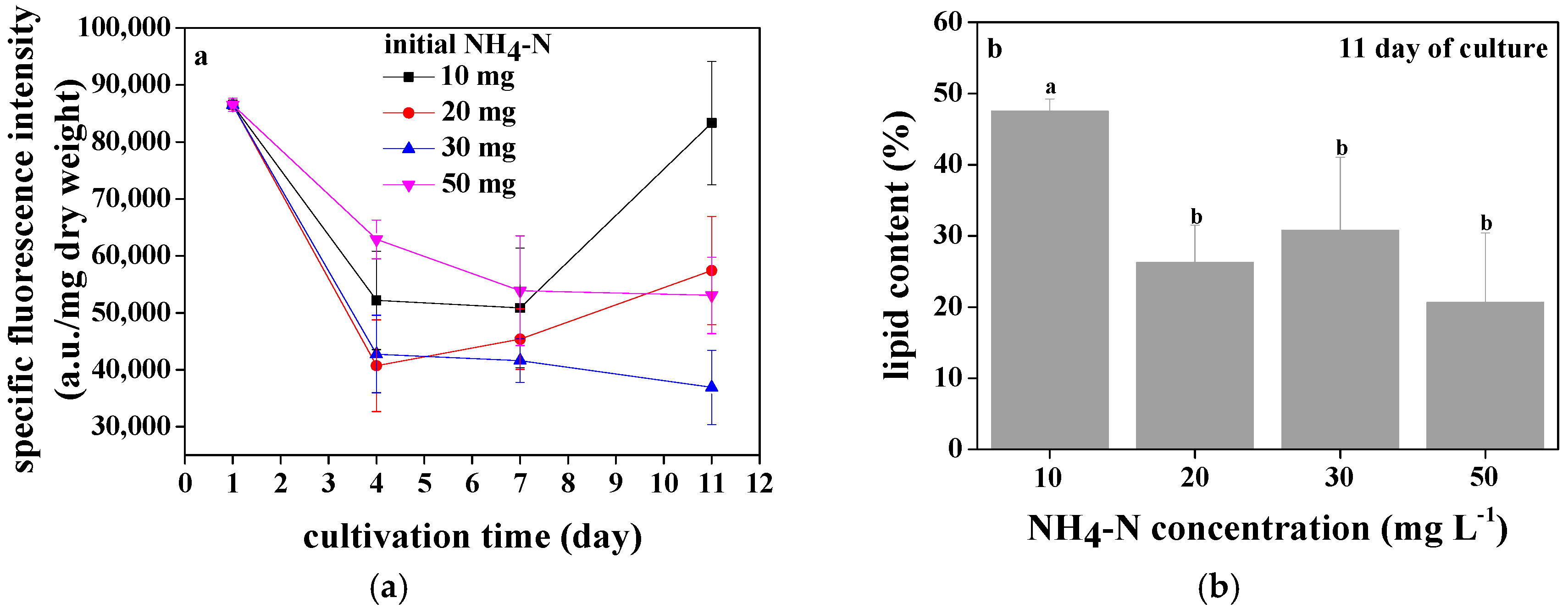
3.4. Lipid Accumulation of Chlorococcum sp. GD in Wastewater with Different Ammonia Nitrogen Concentrations
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, Y.; Zhou, W.; Hu, B.; Min, M.; Chen, P.; Ruan, R.R. Integration of algae cultivation as biodiesel production feedstock with municipal wastewater treatment: Strains screening and significance evaluation of environmental factors. Bioresour. Technol. 2011, 102, 10861–10867. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, Z.; Shu, Q.; Takala, J.; Hiltunen, E.; Feng, P.; Yuan, Z. Nutrient removal and biodiesel production by integration of freshwater algae cultivation with piggery wastewater treatment. Water Res. 2013, 47, 4294–4302. [Google Scholar] [CrossRef] [PubMed]
- Olguín, E.J. Dual purpose microalgae-bacteria-based systems that treat wastewater and produce biodiesel and chemical products within a Biorefinery. Biotechnol. Adv. 2012, 30, 1031–1046. [Google Scholar] [CrossRef] [PubMed]
- Milledge, J.J.; Heaven, S. A review of the harvesting of micro-algae for biofuel production. Rev. Environ. Sci. Biotechnol. 2013, 12, 165–178. [Google Scholar] [CrossRef]
- Bosma, R.; van Spronsen, W.A.; Tramper, J.; Wijffels, R.H. Ultrasound, a new separation technique to harvest microalgae. J. Appl. Phycol. 2003, 15, 143–153. [Google Scholar] [CrossRef]
- Xu, Y.; Purton, S.; Baganz, F. Chitosan flocculation to aid the harvesting of the microalga Chlorella sorokiniana. Bioresour. Technol. 2013, 129, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Zhao, X.Q.; Guo, S.L.; Alam, M.A.; Bai, F.W. Bioflocculant production from Solibacillus silvestris W01 and its application in cost-effective harvest of marine microalga Nannochloropsis oceanica by flocculation. Bioresour. Technol. 2013, 135, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Alam, M.A.; Zhao, X.Q.; Zhang, X.Y.; Guo, S.L.; Ho, S.H.; Chang, J.S.; Bai, F.W. Current progress and future prospect of microalgal biomass harvest using various flocculation technologies. Bioresour. Technol. 2015, 184, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Salim, S.; Bosma, R.; Vermuë, M.H.; Wijffels, R.H. Harvesting of microalgae by bio-flocculation. J. Appl. Phycol. 2011, 23, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Salim, S.; Vermuë, M.H.; Wijffels, R.H. Ratio between autoflocculating and target microalgae affects the energy-efficient harvesting by bio-flocculation. Bioresour. Technol. 2012, 118, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.L.; Zhao, X.Q.; Wan, C.; Huang, Z.Y.; Yang, Y.L.; Alam, M.A.; Ho, S.H.; Bai, F.W.; Chang, J.S. Characterization of flocculating agent from the self-flocculating microalga Scenedesmus obliquus AS-6-1 for efficient biomass harvest. Bioresour. Technol. 2013, 145, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Salim, S.; Shi, Z.; Vermuë, M.H.; Wijffels, R.H. Effect of growth phase on harvesting characteristics, autoflocculation and lipid content of Ettlia texensis for microalgal biodiesel production. Bioresour. Technol. 2013, 138, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.A.; Wan, C.; Guo, S.L.; Zhao, X.Q.; Huang, Z.Y.; Yang, Y.L.; Chang, J.S.; Bai, F.W. Characterization of the flocculating agent from the spontaneously flocculating microalga Chlorella vulgaris JSC-7. J. Biosci. Bioeng. 2014, 118, 29–33. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhou, L.; Xu, X.; Wang, S.; Wang, C.; Dai, B. Uniform design for optimizing biomass and intracellular polysaccharide production from self-flocculating Scenedesmus sp. BH. Ann. Microbiol. 2014, 64, 1779–1787. [Google Scholar] [CrossRef]
- Salim, S.; Kosterink, N.R.; Wacka, N.T.; Vermuë, M.H.; Wijffels, R.H. Mechanism behind autoflocculation of unicellular green microalgae Ettlia Texensis. J. Biotechnol. 2014, 174, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Zhang, X.Y.; Zhao, X.Q.; Bai, F.W. Harvesting microalgae via flocculation: A review. Chin. J. Biotech. 2015, 31, 161–171. (In Chinese) [Google Scholar]
- González-Fernández, C.; Ballesteros, M. Microalgae autoflocculation: An alternative to high-energy consuming harvesting methods. J. Appl. Phycol. 2013, 25, 991–999. [Google Scholar] [CrossRef]
- Tam, N.F.Y.; Wong, Y.S. Effect of ammonia concentrations on growth of Chlorella vulgaris and nitrogen removal from media. Bioresour. Technol. 1996, 57, 45–50. [Google Scholar] [CrossRef]
- Kim, S.; Lee, Y.; Hwang, S.J. Removal of nitrogen and phosphorus by Chlorella sorokiniana cultured heterotrophically in ammonia and nitrate. Int. Biodeter. Biodegr. 2013, 85, 511–516. [Google Scholar] [CrossRef]
- Guo, S.L. Development of Transgenic Flocculation Scenedesmus obliquus and Characterization of the Flocculation of S. obliquus. Ph.D. Thesis, Dalian University of Technology, Dalian, China, 2013; pp. 91–92. (In Chinese). [Google Scholar]
- Lv, J.P.; Guo, J.Y.; Feng, J.; Liu, Q.; Xie, S.L. A comparative study on flocculating ability and growth potential of two microalgae in simulated secondary effluent. Bioresour. Technol. 2016, 205, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Guo, Y.; Zhang, X.; Wang, G.; Lv, J.; Liu, Q.; Xie, S. Identification and characterization of a symbiotic alga from soil bryophyte for lipid profiles. Biol. Open 2016, 5, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Kapdan, I.K. Batch kinetics of nitrogen and phosphorus removal from synthetic wastewater by algae. Ecol. Eng. 2006, 28, 64–70. [Google Scholar] [CrossRef]
- Yang, S.F.; Li, X.Y. Influences of extracellular polymeric substances (EPS) on the characteristics of activated sludge under non-steady-state conditions. Process. Biochem. 2009, 44, 91–96. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for quantification of microgram quantities of protein utilizing principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Gaudy, A.F. Colorimetric determination of protein and carbohydrate. Ind. Water Wastes 1962, 7, 17–22. [Google Scholar]
- APHA-AWWA-WEF. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Park, S.Y.; Li, Y. Nutrient recovery from wastewater streams by microalgae: Status and prospects. Renew. Sustain. Energy Rev. 2013, 19, 360–369. [Google Scholar] [CrossRef]
- Durmaz, B.; Sanin, F.D. Effect of carbon to nitrogen ratio on the composition of microbial extracellular polymers in activated sludge. Water Sci. Technol. 2001, 44, 221–229. [Google Scholar] [PubMed]
- Díaz-Santos, E.; Vila, M.; de la Vega, M.; León, R.; Vigara, J. Study of bioflocculation induced by Saccharomyces bayanus var. uvarum and flocculating protein factors in microalgae. Algal Res. 2015, 8, 23–29. [Google Scholar]
- Díaz-Santos, E.; Vila, M.; Vigara, J.; León, R. A new approach to express transgenes in microalgae and its use to increase the flocculation ability of Chlamydomonas reinhardtii. J. Appl. Phycol. 2016, 28, 1611–1621. [Google Scholar] [CrossRef]
- He, P.J.; Mao, B.; Shen, C.M.; Shao, L.M.; Lee, D.J.; Chang, J.S. Cultivation of Chlorella vulgaris on wastewater containing high levels of ammonia for biodiesel production. Bioresour. Technol. 2013, 129, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Marin, A.; Mendoza-Espinosa, L.G.; Stephenson, T. Growth and nutrient removal in free and immobilized green algae in batch and semi-continuous cultures treating real wastewater. Bioresour. Technol. 2010, 101, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Mennerich, A.; Urban, B. Coupled nutrient removal and biomass production with mixed algal culture: Impact of biotic and abiotic factors. Bioresour. Technol. 2012, 118, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Deng, Z.; Hu, Z.; Wang, Z.; Fan, L. Characterization of Chlorococcum pamirum as a potential biodiesel feedstock. Bioresour. Technol. 2014, 162, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Vyverman, W. Differences in nutrient uptake capacity of the benthic filamentous algae Cladophora sp., Klebsormidium sp. and Pseudanabaena sp. under varying N/P conditions. Bioresour. Technol. 2015, 179, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef] [PubMed]
- Jebali, A.; Acién, F.G.; Gómez, C.; Fernández-Sevilla, J.M.; Mhiri, N.; Karray, F.; Dhouib, F.; Molina-Grima, E.; Sayadi, S. Selection of native Tunisian microalgae for simultaneous wastewater treatment and biofuel production. Bioresour. Technol. 2015, 198, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.M.; Chen, T.Y.; Lin, T.H.; Kao, C.Y.; Lai, J.T.; Chang, J.S.; Lin, C.S. Cultivation of Chlorella sp. GD using piggery wastewater for biomass and lipid production. Bioresour. Technol. 2015, 194, 326–333. [Google Scholar] [CrossRef] [PubMed]

| Ammonia Concentration (mg·L−1) | N/P Ratio | Specific Growth Rate (μ, d−1) | Mean Biomass Productivity (mg DW·L−1·d−1) | Double Time (d−1) |
|---|---|---|---|---|
| 10 | 2 | 0.19 ± 0.02 a | 10.64 ± 1.35 a | 2.37 ± 0.08 a |
| 20 | 4 | 0.24 ± 0.01 b | 15.70 ± 0.48 b | 2.12 ± 0.06 b |
| 30 | 6 | 0.21 ± 0.01 a | 14.26 ± 0.06 b,c | 2.27 ± 0.03 a,b |
| 50 | 10 | 0.19 ± 0.03 a | 12.74 ± 1.61 c | 2.34 ± 0.1 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, J.; Wang, X.; Liu, W.; Feng, J.; Liu, Q.; Nan, F.; Jiao, X.; Xie, S. The Performance of a Self-Flocculating Microalga Chlorococcum sp. GD in Wastewater with Different Ammonia Concentrations. Int. J. Environ. Res. Public Health 2018, 15, 434. https://doi.org/10.3390/ijerph15030434
Lv J, Wang X, Liu W, Feng J, Liu Q, Nan F, Jiao X, Xie S. The Performance of a Self-Flocculating Microalga Chlorococcum sp. GD in Wastewater with Different Ammonia Concentrations. International Journal of Environmental Research and Public Health. 2018; 15(3):434. https://doi.org/10.3390/ijerph15030434
Chicago/Turabian StyleLv, Junping, Xuechun Wang, Wei Liu, Jia Feng, Qi Liu, Fangru Nan, Xiaoyan Jiao, and Shulian Xie. 2018. "The Performance of a Self-Flocculating Microalga Chlorococcum sp. GD in Wastewater with Different Ammonia Concentrations" International Journal of Environmental Research and Public Health 15, no. 3: 434. https://doi.org/10.3390/ijerph15030434
APA StyleLv, J., Wang, X., Liu, W., Feng, J., Liu, Q., Nan, F., Jiao, X., & Xie, S. (2018). The Performance of a Self-Flocculating Microalga Chlorococcum sp. GD in Wastewater with Different Ammonia Concentrations. International Journal of Environmental Research and Public Health, 15(3), 434. https://doi.org/10.3390/ijerph15030434





