Tobacco Harm Reduction with Vaporised Nicotine (THRiVe): The Study Protocol of an Uncontrolled Feasibility Study of Novel Nicotine Replacement Products among People Living with HIV Who Smoke
Abstract
:1. Introduction
2. Materials and Methods
2.1. Trial Aim and Objectives
- Measuring quit attempts.
- Measuring cigarettes smoked per day.
- Evaluating short-term (7-day) abstinence from cigarette smoking (at 4, 8, 12 and 24 weeks post treatment initiation).
- Evaluating medium-term abstinence from cigarette smoking (continuous, abstinence from tobacco smoking for at least 8 weeks), measured at 12 and 24 weeks post treatment initiation.
- Obtaining feedback on the intervention (including, printed information, investigational devices and medicine).
- Obtaining data to accurately describe the sample (e.g., demographic features, history of smoking and quitting), knowledge of health impacts, their level of quitting motivation, quit self-efficacy and level of nicotine dependence.
- Monitoring adverse events and contribute to the literature on the safety of vaping as a method to quit smoking.
2.2. Design
2.3. Research Ethics
2.4. Sample
2.4.1. Participants
2.4.2. Eligibility Criteria
- Inclusion criteria: diagnosis of HIV; aged 18 years, or over; smoke ≥5 cigarettes per day at the time of enrolment into the trial; have been smoking for at least 12 months; have capacity to consent and able to understand study instructions and procedures (e.g., sufficient English language ability); willing to attempt to quit tobacco smoking after study enrolment.
- Exclusion criteria: participating in a smoking-cessation program; pregnant (measured by self-report, with confirmation by self-administered pregnancy test where there is doubt) or planning to become pregnant during trial participation period; breast-feeding (measured by self-report) or planning to be during trial participation period; experienced chest pain, or another cardiovascular event or procedure (e.g., heart attack, stroke, insertion of stent, bypass surgery) in the last month; being treated with oxygen therapy.
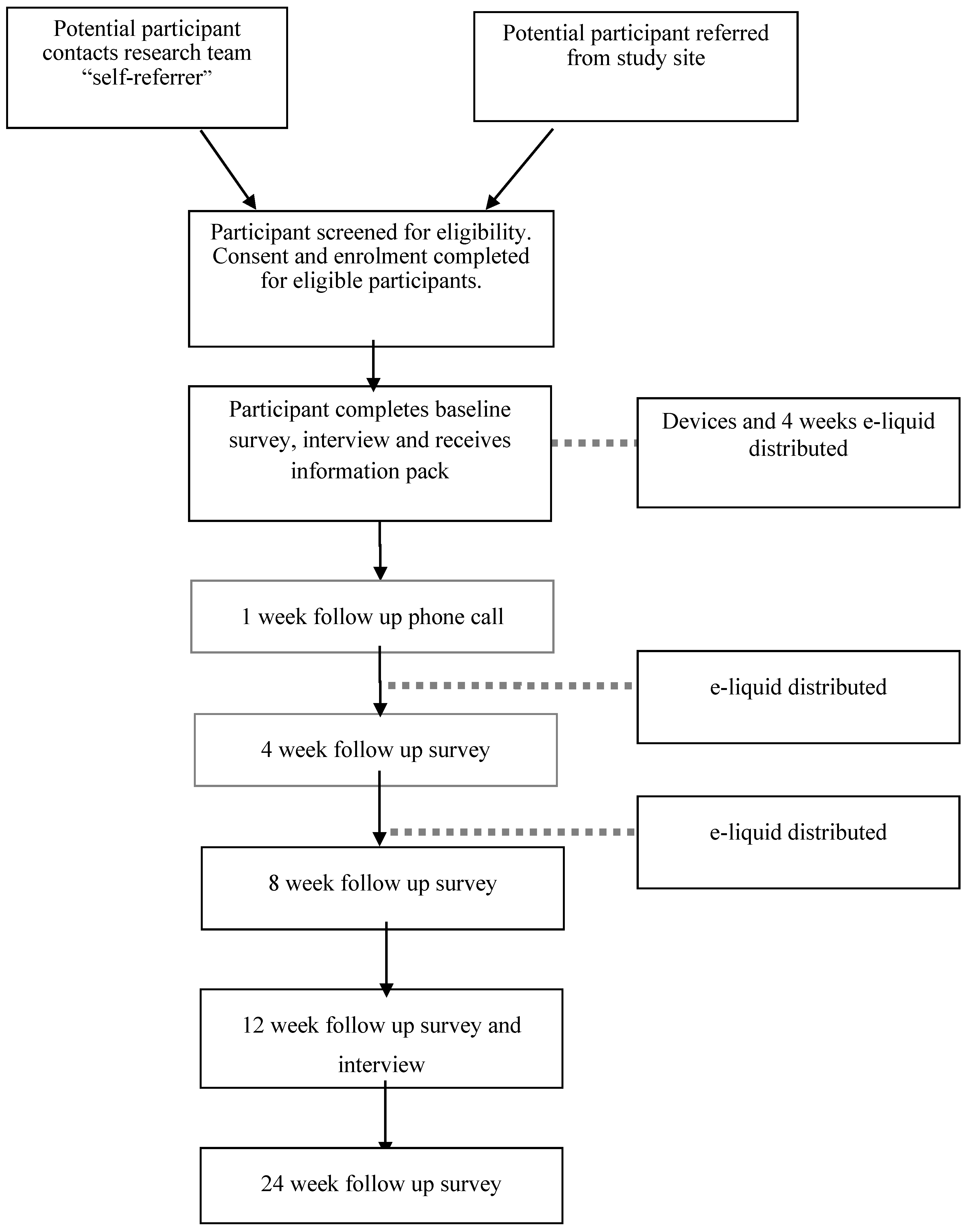
2.4.3. Recruitment
2.5. Intervention Description
- Printed information
- Information and instruction booklet. The booklet outlines instructions on how to use, store and handle the investigational products, information of vaping instead of quitting and information and links on quitting smoking resources supports (such as Quitline) and tips from vapers.
- A “Positively Quitting” booklet. A booklet developed by Queensland Positive People to help PLHIV to quit smoking containing information such as the benefits of quitting and suggestions for managing cigarette cravings.
- Copies of device user manuals.
- A study wallet card confirming participation in the trial.
- Investigational devices
- One Innokin Endura T18® vaporiser kit
- One Innokin Endura T22® vaporiser kit
- Four spare coils
- One wall charger
- Investigational medicine
- Ten 10 mL bottles of Nicophar® 12 mg nicotine e-liquid
2.6. Data Collection Points and Outcome Measures
2.6.1. Primary Outcome Measures
- Familiarity and experience with VNPs
- Attitudes toward VNPs
- Acceptability of trial products and associated reasons (e.g., impact on cravings, assisting with quitting, ease of use)
- Comparison of perceptions and use of trial products with traditional smoking cessation pharmacotherapy (e.g., varenicline, bupropion and NRT).
- Level of interest in short and long-term use of VNPs
- Nature of product use
- Quantity of e-liquid and coils used
- Locations, when used and how often, reasons for use, ease of use
- Barriers and facilitators to VNP use, such as adequacy for reducing withdrawal
2.6.2. Secondary Outcome Measures
- Number of quit attempts
- Tobacco cigarettes smoked per day (CPD)
- Abstinence measures:
- Short-term abstinence or 7 days point prevalence (defined as having not smoked any tobacco in the previous 7 days at assessment) at weeks 4, 8, 12 and 24.
- Medium-term abstinence from tobacco smoking measured by continuous abstinence from tobacco smoking for at least 8 weeks post quit date, with no more than five (tobacco) cigarettes from the start of the abstinence period (measured at weeks 12 and 24).
- Descriptive measures of the sample:
- Demographics
- History of tobacco smoking and quitting
- Knowledge of health effects of smoking and nicotine
- Motivation to quit
- Quitting self-efficacy
- Fagerstrom Test for Nicotine Dependence (FTND) [35]
- A measure of behavioural dependence, using the Glover-Nilsson Smoking Behavioural Questionnaire (GN-SBQ) [36]
- Measures of adverse events.
2.6.3. Participant Reimbursement
- Baseline survey and interview: $50
- Week 4 survey: $20
- Week 8 survey: $20
- Week 12 (end of treatment) survey and interview: $50
- Week 24 (final follow-up) survey: $20
2.7. Safety and Adverse Events
2.8. Participant Attrition
2.9. Data Management and Analysis
3. Discussion
3.1. Strengths/Usefulness of Findings
3.2. Innovation/Novelty and Strengths
3.3. Limitations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nakagawa, F.; May, M.; Phillips, A. Life expectancy living with HIV: Recent estimates and future implications. Curr. Opin. Infect. Dis. 2013, 26, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Petoumenos, K.; Law, M.G. Smoking, alcohol and illicit drug use effects on survival in HIV-positive persons. Curr. Opin. HIV AIDS 2016, 11, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Hunt, P.W. HIV and inflammation: Mechanisms and consequences. Curr. HIV/AIDS Rep. 2012, 9, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Shirley, D.K.; Kaner, R.J.; Glesby, M.J. Effects of smoking on non-AIDS-related morbidity in HIV-infected patients. Clin. Infect. Dis. 2013, 57, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Fuster, M.; Estrada, V.; Fernandez-Pinilla, M.C.; Fuentes-Ferrer, M.E.; Tellez, M.J.; Vergas, J.; Serrano-Villar, S.; Fernandez-Cruz, A. Smoking cessation in HIV patients: Rate of success and associated factors. HIV Med. 2009, 10, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Mdodo, R.; Frazier, E.L.; Dube, S.R.; Mattson, C.L.; Sutton, M.Y.; Brooks, J.T.; Skarbinski, J. Cigarette smoking prevalence among adults with HIV compared with the general adult population in the United States: Cross-sectional surveys. Ann. Intern. Med. 2015, 162, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Petoumenos, K.; Worm, S.; Reiss, P.; de Wit, S.; d’Arminio Monforte, A.; Sabin, C.; Friis-Møller, N.; Weber, R.; Mercie, P.; Pradier, C.; et al. Rates of cardiovascular disease following smoking cessation in patients with HIV infection: Results from the D:A:D study. HIV Med. 2011, 12, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Helleberg, M.; Afzal, S.; Kronborg, G.; Larsen, C.S.; Pedersen, G.; Pedersen, C.; Gerstoft, J.; Nordestgaard, B.G.; Obel, N. Mortality attributable to smoking among HIV-1-infected individuals: A nationwide, population-based cohort study. Clin. Infect. Dis. 2013, 56, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Helleberg, M.; May, M.T.; Ingle, S.M.; Dabis, F.; Reiss, P.; Fätkenheuer, G.; Costagliola, D.; d’Arminio, A.; Cavassini, M.; Smith, C.; et al. Smoking and life expectancy among HIV-infected individuals on antiretroviral therapy in Europe and North America. AIDS 2015, 29, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Humfleet, G.L.; Delucchi, K.; Kelley, K.; Hall, S.M.; Dilley, J.; Harrison, G. Characteristics of HIV-positive cigarette smokers: A sample of smokers facing multiple challenges. AIDS Educ. Prev. 2009, 21, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Vidrine, D.J. Cigarette smoking and HIV/AIDs: Health implications, smoker characteristics and cessation strategies. AIDS Educ. Prev. 2009, 21, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Mamary, E.M.; Bahrs, D.; Martinez, S. Cigarette smoking and the desire to quit among individuals living with HIV. AIDS Patient Care STDS 2002, 16, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Tesoriero, J.M.; Gieryic, S.M.; Carrascal, A.; Lavigne, H.E. Smoking among HIV positive New Yorkers: Prevalence, frequency, and opportunities for cessation. AIDS Behav. 2010, 14, 824–835. [Google Scholar] [CrossRef] [PubMed]
- Shuter, J.; Bernstein, S.L.; Moadel, A.B. Cigarette smoking behaviors and beliefs in persons living with HIV/AIDS. Am. J. Health Behav. 2012, 36, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Burkhalter, J.E.; Springer, C.M.; Chhabra, R.; Ostroff, J.S.; Rapkin, B.D. Tobacco use and readiness to quit smoking in low-income HIV-infected persons. Nicotine Tob. Res. 2005, 7, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Pool, E.R.M.; Dogar, O.; Lindsay, R.P.; Weatherburn, P.; Siddiqi, K. Interventions for tobacco use cessation in people living with HIV and AIDS. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Tobacco: Harm-Reduction Approaches to Smoking; National Institute for Health and Care Excellence (NICE): London, UK, 2013. [Google Scholar]
- McNeill, A.; Munafo, M.R. Reducing harm from tobacco use. J. Psychopharmacol. 2013, 27, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Wodak, A.; McLeod, L. The role of harm reduction in controlling HIV among injecting drug users. AIDS 2008, 22, 81–92. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). A New Health Sector Agenda for HIV/AIDS Global Health Sector Strategy on HIV/AIDS, 2011–2015; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Ball, A.L. HIV, injecting drug use and harm reduction: A public health response. Addiction 2007, 102, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Tobacco Advisory Group of the Royal College of Physicians. Harm Reduction in Nicotine Addiction: Helping People Who Can’t Quit; RCP: London, UK, 2007. [Google Scholar]
- McRobbie, H.; Bullen, C.; Hartmann-Boyce, J.; Hajek, P. Electronic cigarettes for smoking cessation and reduction. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef]
- Royal College of Physicians. Nicotine without Smoke: Tobacco Harm Reduction; RCP: London, UK, 2016. [Google Scholar]
- McNeill, A.; Brose, L.S.; Calder, R.; Hitchman, S.C.; Hajek, P.; McRobbie, H. E-Cigarettes: An Evidence Update a Report Commissioned by Public Health England; Public Health England: London, UK, 2015. [Google Scholar]
- Shiffman, S.; Hughes, J.R.; Pillitteri, J.L.; Burton, S.L. Persistent use of nicotine replacement therapy: An analysis of actual purchase patterns in a population based sample. Tob. Control 2003, 12, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, L.; Turner, J.; Hasna, S.; Soar, K. The electronic-cigarette: Effects on desire to smoke, withdrawal symptoms and cognition. Addict. Behav. 2012, 37, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Wagener, T.L.; Floyd, E.L.; Stepanov, I.; Driskill, L.M.; Frank, S.G.; Meier, E.; Leavens, E.L.; Tackett, A.P.; Molina, N.; Queimado, L. Have combustible cigarettes met their match? The nicotine delivery profiles and harmful constituent exposures of second-generation and third-generation electronic cigarette users. Tob. Control 2017. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Hann, N.; Wilson, A.; Mnatzaganian, G.; Worrall-Carter, L. E-cigarettes and smoking cessation: Evidence from a systematic review and meta-analysis. PLoS ONE 2015, 10, e0122544. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; West, R.; Beard, E.; Michie, S.; Shahab, L.; McNeill, A. Prevalence and characteristics of e-cigarette users in Great Britain: Findings from a general population survey of smokers. Addict. Behav. 2014, 39, 1120–1125. [Google Scholar] [CrossRef] [PubMed]
- West, R.; Brown, J.; Beard, E. Trends in Electronic Cigarette Use in England. Available online: www.smokinginengland.info/ (accessed on 18 October 2014).
- Creswell, J.W. A Concise Introduction to Mixed Methods Research; SAGE Publications: Thousand Oaks, CA, USA, 2015. [Google Scholar]
- Dawkins, L.; Turner, J.; Roberts, A.; Soar, K. ‘Vaping’ profiles and preferences: An online survey of electronic cigarette users. Addiction 2013, 108, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, C.; Gartner, C. Electronic cigarettes. What should you tell your patients? Med. Today 2015, 16, 26–32. [Google Scholar]
- Heatherton, T.F.; Kozlowski, L.T.; Frecker, R.C.; Fagerstrom, K.O. The fagerstrom test for nicotine dependence. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Glover, E.D.; Nilsson, F.; Westin, Å.; Glover, P.N.; Laflin, M.T.; Persson, B. Developmental history of the Glover-Nilsson Smoking Behavioral Questionnaire. Am. J. Health Behav. 2005, 29, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Fagerstrom, K.O.; Tejding, R.; Westin, A.; Lunell, E. Aiding reduction of smoking with nicotine replacement medications: Hope for the recalcitrant smoker? Tob. Control 1997, 6, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Bolliger, C.T.; Zellweger, J.; Danielsson, T.; van Biljon, X.; Robidou, A.; Westin, Å.; Perruchoud, A.; Säwe, U. Smoking reduction with oral nicotine inhalers: Double blind, randomised clinical trial of efficacy and safety. BMJ 2000, 321, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. Ann. Intern. Med. 2010, 152, 726–732. [Google Scholar] [CrossRef] [PubMed]
- IBM Corp. IBM SPSS Statistics for Windows, Version 23.0; IBM Corp.: Armonk, NY, USA, 2015. [Google Scholar]
- Corbin, J.M.; Strauss, A.L. Basics of Qualitative Research: Techniques and Procedures for Developing Grounded Theory; Sage Publications: Thousand Oaks, CA, USA, 2008. [Google Scholar]
- Lloyd-Richardson, E.E.; Stanton, C.A.; Papandonatos, G.D.; Shadel, W.G.; Stein, M.; Tashima, K.; Flanigan, T.; Morrow, K.; Neighbors, C.; Niaura, R. Motivation and patch treatment for HIV + smokers: A randomized controlled trial. Addiction 2009, 104, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Peikes, D.; Geonnotti, K.; Wang, W. Using Pragmatic Clinical Trials to Test the Effectiveness of Patient-Centered Medical Home Models in Real-World Settings; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2013. [Google Scholar]
- Zhu, S.; Sun, J.Y.; Bonnevie, E.; Cummins, S.; Gamst, A.; Yin, L.; Lee, M. Four hundred and sixty brands of e-cigarettes and counting: Implications for product regulation. Tob. Control 2014, 23, iii3–iii9. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Romagna, G.; Tsiapras, D.; Kyrzopoulos, S.; Spyrou, A.; Voudris, V. Impact of flavour variability on electronic cigarette use experience: An internet survey. Int. J. Environ. Res. Public Health 2013, 10, 7272–7282. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.G.; Flanigan, S.S.; LeBlanc, M.; Vallarino, J.; MacNaughton, P.; Stewart, J.H.; Christiani, D.C. Flavoring chemicals in e-cigarettes: Diacetyl, 2,3-pentanedione, and acetoin in a sample of 51 products, including fruit, candy-, and cocktail-flavored e-cigarettes. Environ. Health Perspect. 2016, 124, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Tierney, P.A.; Karpinski, C.D.; Brown, J.E.; Luo, W.; Pankow, J.F. Flavour chemicals in electronic cigarette fluids. Tob. Control 2016, 25, 10–15. [Google Scholar] [CrossRef] [PubMed]
| Contact | Baseline | |||||
|---|---|---|---|---|---|---|
| Week (month) | 0 | 1 (1) | 4 (1) | 8 (2) | 12 (3) | 24 (6) |
| Treatment Period (12 weeks) | X | X | X | X | ||
| Device dispensation | X | |||||
| e-liquid dispensation | X | X | X | |||
| 1 week check in | X | |||||
| Measures: Primary Objectives | ||||||
| Familiarity with vaporized nicotine products (VNPs)—survey | X | |||||
| Attitudes and use of VNPs—survey | X | X | X | X | X | |
| Attitudes towards smoking, quitting and VNPs—interview | X | X | ||||
| Measures: Secondary Objectives | ||||||
| Cigarettes per day (CPD) | X | X | X | X | X | |
| Quit attempts | X | X | X | X | X | |
| Abstinence measures | X | X | X | X | ||
| Demographics | X | |||||
| Smoking and quitting | X | |||||
| Knowledge of harms | X | X | ||||
| Quitting self-efficacy | X | X | ||||
| Fagerstrom Test for Nicotine Dependence (FTND) | X | X | ||||
| Glover Nilsson Smoking Behavioural Questionnaire (GNSBQ) | X | X | ||||
| Adverse events | X | X | X | X | X | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bell, S.; Dean, J.; Gilks, C.; Boyd, M.A.; Fitzgerald, L.; Mutch, A.; Baker, P.; Neilsen, G.; Gartner, C.E. Tobacco Harm Reduction with Vaporised Nicotine (THRiVe): The Study Protocol of an Uncontrolled Feasibility Study of Novel Nicotine Replacement Products among People Living with HIV Who Smoke. Int. J. Environ. Res. Public Health 2017, 14, 799. https://doi.org/10.3390/ijerph14070799
Bell S, Dean J, Gilks C, Boyd MA, Fitzgerald L, Mutch A, Baker P, Neilsen G, Gartner CE. Tobacco Harm Reduction with Vaporised Nicotine (THRiVe): The Study Protocol of an Uncontrolled Feasibility Study of Novel Nicotine Replacement Products among People Living with HIV Who Smoke. International Journal of Environmental Research and Public Health. 2017; 14(7):799. https://doi.org/10.3390/ijerph14070799
Chicago/Turabian StyleBell, Stephanie, Judith Dean, Charles Gilks, Mark A. Boyd, Lisa Fitzgerald, Allyson Mutch, Peter Baker, Graham Neilsen, and Coral E. Gartner. 2017. "Tobacco Harm Reduction with Vaporised Nicotine (THRiVe): The Study Protocol of an Uncontrolled Feasibility Study of Novel Nicotine Replacement Products among People Living with HIV Who Smoke" International Journal of Environmental Research and Public Health 14, no. 7: 799. https://doi.org/10.3390/ijerph14070799
APA StyleBell, S., Dean, J., Gilks, C., Boyd, M. A., Fitzgerald, L., Mutch, A., Baker, P., Neilsen, G., & Gartner, C. E. (2017). Tobacco Harm Reduction with Vaporised Nicotine (THRiVe): The Study Protocol of an Uncontrolled Feasibility Study of Novel Nicotine Replacement Products among People Living with HIV Who Smoke. International Journal of Environmental Research and Public Health, 14(7), 799. https://doi.org/10.3390/ijerph14070799






