1. Introduction
Diarrheal illness was the underlying cause of roughly 1.5 million deaths globally in 2012 [
1]. In low and middle income nations, inadequate access to safe water, poor hygiene, and unimproved sanitation conditions (also known as the WASH paradigm) are responsible for 502,000 deaths per year: 58% of all diarrheal disease-related deaths in these countries [
1]. Much of this burden is the result of diarrheal illness, as poor WASH conditions contribute to the spread of gastrointestinal pathogens via mainly the fecal-oral route [
2]. Concerning microbial water quality, the largest contributor to illness is fecal contamination of drinking water and its sources (thereby adding enteric pathogens). Breaking this transmission cycle requires improvements in hygiene (such as handwashing behaviors), sanitation facilities, and/or improved drinking water sources and treatment.
Fortunately, high-quality WASH interventions have proven to be effective in combatting diarrheal illness. A recent meta-analysis found that hygiene-based interventions, such as hygiene education campaigns and handwashing promotion, may reduce the risk of diarrheal illness by 45%, with a pooled estimated relative risk of 0.55 (95% CI 0.40–0.75) [
3]. However, this protective effect may be reduced in settings with limited access to water [
4]. Further, household, point-of-use water treatment (boiling, chlorination, etc.) in rural areas may reduce diarrheal illness by 39% [
3]. Improved drinking water supplies (such as public taps, boreholes, protected springs, or water piped directly to homes) may help reduce the transmission of enteric pathogens through drinking water treatment or source water protection, but randomized trials studying the effectiveness of water supply interventions are limited [
3,
4]. Unfortunately, of the 663 million people still lacking access to an improved source of drinking water, nearly half live in sub-Saharan Africa [
5].
Over the duration of the Millennium Development Goals (MDGs) (1990–2015), access to improved water sources in rural Cameroon improved from just 34% coverage to 53% [
6]. Access to improved or shared sanitation sources increased by just 3% from 1990 to 2015 in urban Cameroon (from 82% to 85%) and did not improve at all in rural areas of the country, remaining at an estimated 27% throughout the MDG timeframe [
6]. One improvement made in rural areas is the decrease in open defecation from 17% to 12% over the 25-year period [
6]. The combination of low access to safe water and poor sanitation conditions is made evident by Cameroon’s recent history of cholera outbreaks and the burden of disease attributed to diarrheal illness in the country, especially in the Far North region [
7,
8].
Despite Cameroon’s greatest cholera outbreaks often occurring in the Far North region [
7], the majority of water quality studies conducted in Cameroon have taken place outside of this region, near Cameroon’s two largest cities, Yaoundé and Douala [
9,
10,
11,
12,
13,
14]. This disproportionate impact of cholera on the Sahel region is not specific to Cameroon, as Chad’s Guera region reported 1754 cases of cholera during the outbreak in 2011 [
15]. Closer to the Far North, Djaouda et al., measured high and consistent levels of fecal contamination in wells used as a source for drinking water in the North region of Cameroon [
16,
17]. Working in the Logone Valley, at the border of Chad and Cameroon, Sorlini et al., studied drinking water sources in the Far North and noted that current World Health Organization (WHO)/United Nations International Children’s Emergency Fund (UNICEF) Joint Monitoring Program for Water Supply and Sanitation (JMP) definitions of “improved sources” may not actually be protective of human health [
18,
19]. Recently, Healy Profitós et al. investigated water quality in Maroua, the capital city of the Far North, offering insight on water quality degradation across informal distribution systems and health outcomes in an urban setting [
20], as well as a recent microbial source tracking and pathogen detection investigation [
21]. A significant gap remains in the literature of studies simultaneously studying WASH conditions and health outcomes in rural Far North, Cameroon, particularly with respect to nomadic populations.
The current study seeks to first survey the status and relationship between factors of WASH and diarrheal illness among agro-pastoralists living in established villages and transhumant pastoralists living in nomadic camps in rural Far North, Cameroon. The results of this study not only serve as a baseline understanding of these issues in the rural Far North, but also provide insight into the differences between these two rural study populations and those living in the urban center of Maroua. The outcomes of this study can provide a better understanding for future public health initiatives directed at improving WASH conditions in Far North, Cameroon, such as increased access to improved drinking water sources and water handling practices for transhumant pastoralists.
2. Materials and Methods
2.1. Study Site
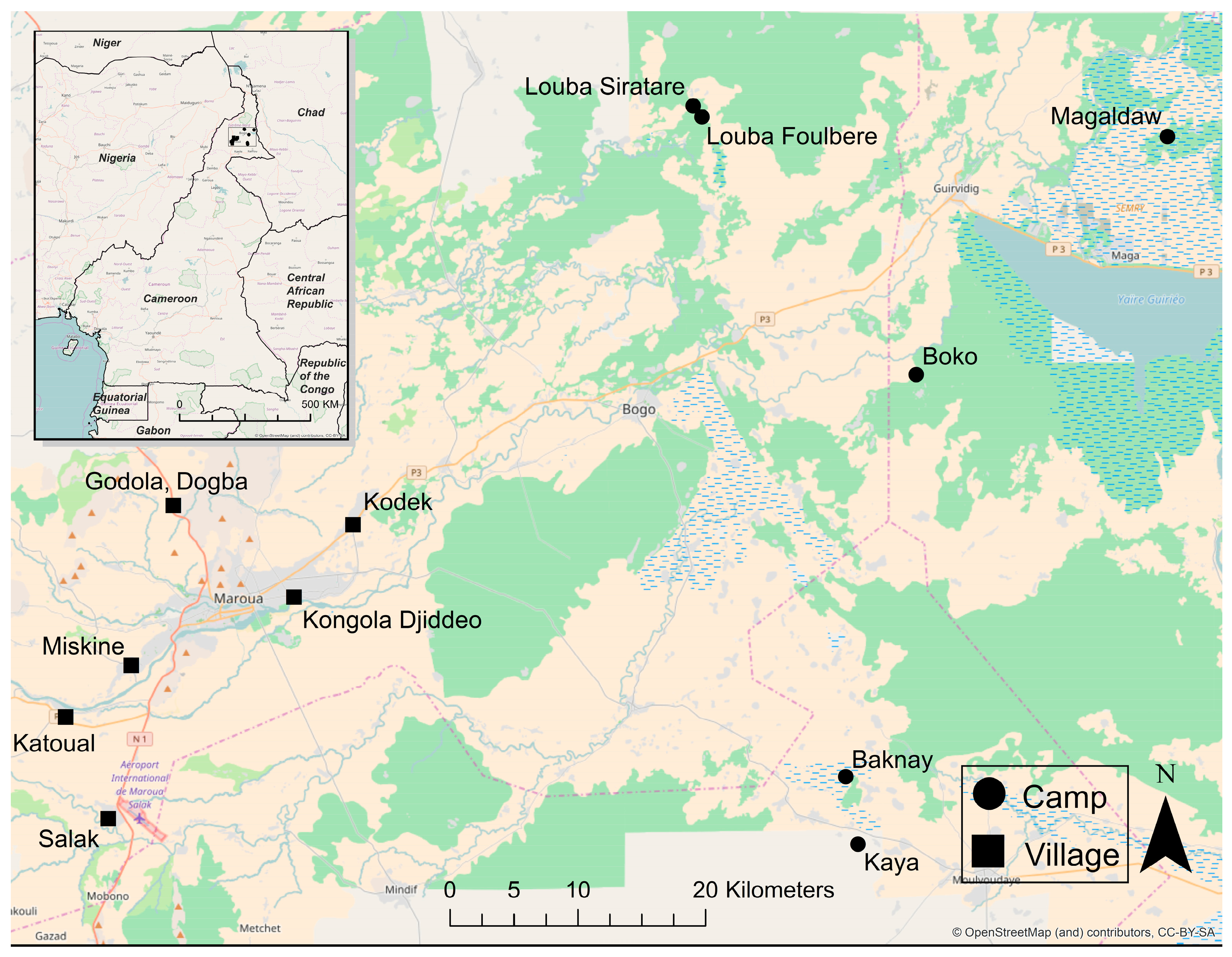
The study was conducted in the Far North of Cameroon, where six transhumant pastoral camps (Boko, Baknay, Kaya, Magaldaw, Louba Foulbere, Louba Siratare) and six permanent agro-pastoralist villages (Meskine, Salak, Katoual, Godala, Kodek, Kongola) are within a triangle formed by the cities of Maroua, Yagoua, and Kaele (
Figure 1). The sites were arbitrarily selected. The climate is Soudan Sahelian type and the vegetation is characterized by savanna shrubs and graminacea. A climate gradient is observed in the region between the dryer areas in the North and those in which a more humid Sudanese climate prevails towards the south. Soils are predominantly made of vertisols, alluvial and hydromophic soils in the flood plain, whereas washed tropical soils, ferriginous soils, fersialitic and lithosoils are prevalent in the dry part. Most water used for drinking and domestic activities comes from wells or community boreholes and is mainly stored in canaries (wide-mouth clay jars) or jerry cans [
20]. Water sampling and survey implementation were conducted between June and July 2014, during the end of the rainy season. The areas are flat with a dry plain to the west and a floodplain to the east.
Two agricultural systems are practiced among the study sites: agro-pastoralism and transhumant pastoralism. Residents of established villages practice agro-pastoralism, relying primarily upon crop farming, including cotton, millet, peanuts, and vegetables. In this area, it is also common for families to have at least one cow, though livestock production is not a primary source of income. In nomadic camps, individuals practice transhumant pastoralism, where livestock rearing is the only source of income and herders travel with their cattle to allow for open grazing. Generally, these pastoral communities are seasonal migrants, traveling from the Kaele area in the south in the rainy season to the Logone Floodplain in the north of the region during the dry season as the flood water recedes.
2.2. Water Sampling
Samples of household water and the reported drinking water source (when available; N = 70) were collected from survey participants into sterile, 800 mL Whirl-Pak bags (Nasco, Fort Atkinson, WI, USA). Among villages, 15 samples were collected from drinking water sources and 29 samples were collected from homes, while 6 water samples were collected from nomadic camp drinking water sources and 20 were collected from camp homes (
Figure 1). Household water—used for drinking, cooking, and washing—was collected directly from the household’s storage container(s). In the case of a central household water storage vessel (such as a canari), the samples were collected using the same method that the residents use to serve water, such as collecting water from the canari using a shared cup or ladle. Source water was collected per the study participants’ main water source, which ranged from surface water in many camps to drilled boreholes in villages. After collection, between 40 and 500 mL of water sampled was concentrated via membrane filtration onto 0.45 µm Millipore membranes (IsoporeTM Membrane Filters, Millipore, Tullagreen, Cork, Ireland) using a manual syringe. The range in the volume of water concentrated was due to membranes clogging before 500 mL could be passed through for highly turbid samples. The total amount filtered was recorded. After concentration, sample storage and transport were conducted as reported by Healy Profitós et al. [
20].
2.3. Molecular Analysis
The filtered membranes were kept frozen and then transported on ice to the lab at The Ohio State University (Columbus, OH, USA). To begin DNA extraction, each membrane was transferred into a sterile 2 mL microcentrifuge tube containing 1.4 mL of ASL buffer (provided in the QIAmp DNA Stool Mini Kit; QIAGEN, Valencia, CA, USA), as well as sterile glass beads 0.1 mm and 0.5 mm in diameter. Bead-beating was then performed for 3 min at 2100 oscillations per minute in a Mini-Beadbeater-96 (Biospec Products, Bartlesville, OK, USA). After bead-beating, the supernatant was transferred to sterile 2 mL microcentrifuge tubes and DNA was extracted using a QIAamp DNA Stool Mini Kit, following the manufacturer’s instructions (QIAGEN, Valencia, CA, USA).
The presence of quantitative PCR (qPCR) inhibitors in water samples was investigated using Sketa22 qPCR assay [
22]. None of the extracted DNA samples demonstrated the presence of PCR inhibitors. In order to investigate sources of water contamination, microbial source tracking was conducted by targeting two human-specific genetic markers and one specific to ruminant fecal contamination. Human markers included: HF183, a 16S rRNA gene of human-specific
Bacteroides spp. [
23,
24] and
gyrB, a target gene of gyrase subunit B from human-specific
Bacteroides fragilis [
25,
26,
27]. Rum2Bac, a
Baceroidales 16S rRNA gene, was chosen for its ruminant gut microbiome specificity [
28].
Quantitative PCR was also used to investigate the presence and concentrations of pathogens both in household water and drinking water source. Virulence factor genes for Shiga toxin types 1 and 2 (
stx1 and
stx2, respectively), from Shiga toxin-producing
Escherichia coli (STEC) were tested [
29]. A second group of pathogens also associated with fecal contamination of water,
Salmonella spp., were investigated using a 16S rRNA gene target [
30]. Lastly, as a preliminary investigation of antimicrobial resistance gene presence, resistance to tetracycline was tested by targeting
tetQ [
31]. Resistance to tetracycline was chosen among the wide variety of antibiotics based on informal observation of local practices during sampling in our previous study of water quality in the region [
20]. All qPCR analyses were conducted using a Bio-Rad CFX96 Touch thermocycler (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
2.4. Health Survey
For each settlement selected (with the exception of a single camp, Lauba Siratare), in-home health surveys were administered, as conducted by Healy Profitós et al. [
20]. Necessary research permissions were obtained through The Ohio State University’s Institutional Review Board (IRB 2010B0004) and Dr. Arabi Mouhaman’s status as faculty at the University of Maroua (Law No. 005 of 16 April 2001; Decree No. 93/035 of 19 January 1993 on the special status of the higher education personnel). Twenty-five village household surveys (including 159 individuals) and 14 camp household surveys (including 86 individuals) were collected during June and July of 2014. Topics covered in the survey included demographics, household-level hygiene behaviors (e.g., handwashing practices), access to improved sanitation, water source and accessibility, and recent health status, including diarrheal incidence. Household water source, water storage vessel type, hygiene behavior, and access to sanitation facilities were surveyed at the household level, but responses were assigned to individual respondents, based on head of household response. This allowed for comparison to national and global rates by accounting for the percentage of the study population, rather than percentage of households. Consent and privacy considerations were handled as previously reported [
20].
2.5. Statistical Analysis
Exploratory data analysis and statistical analyses were conducted using RStudio Version 0.98.1103 (RStudio, Boston, MA, USA; R version 3.2.4, R Core Team, Vienna, Austria); tables and descriptive pie charts were generated in Excel 2016 (Microsoft, Redmond, WA, USA). The R package ggplot2 was used to create bar plots [
32]. Before analysis, gene copy results from qPCR were log-transformed to allow for a more normal distribution of gene copy concentrations and values detected-but-not-quantifiable (DNQ) were set to one-half the limit of quantification [
21]. Further, three of the samples needed to be eliminated from the dataset due to uncertainties in recorded filtered volumes or status as Home or Source water samples, resulting in 67 water samples with qPCR results available for analysis. A Shapiro-Wilk test determined the molecular data to be non-normal, with the exception of
tetQ. Because of the non-normal nature of the molecular data, comparisons between gene copy numbers among sample types were conducted using the Wilcoxon Rank Sum test. Chi-square tests, risk ratios, Fisher’s exact test, and the Cochran-Mantel-Haenszel adjusted odds ratios were conducted by comparing survey results among different settlement types.
4. Discussion
4.1. Rural WASH Conditions
The results of the drinking water source highlight the importance of studying the status of local WASH conditions. Stark differences were seen regarding reliance on surface water between villages and nomadic camps. Comparing the WHO/UNICEF Joint Monitoring Program’s [
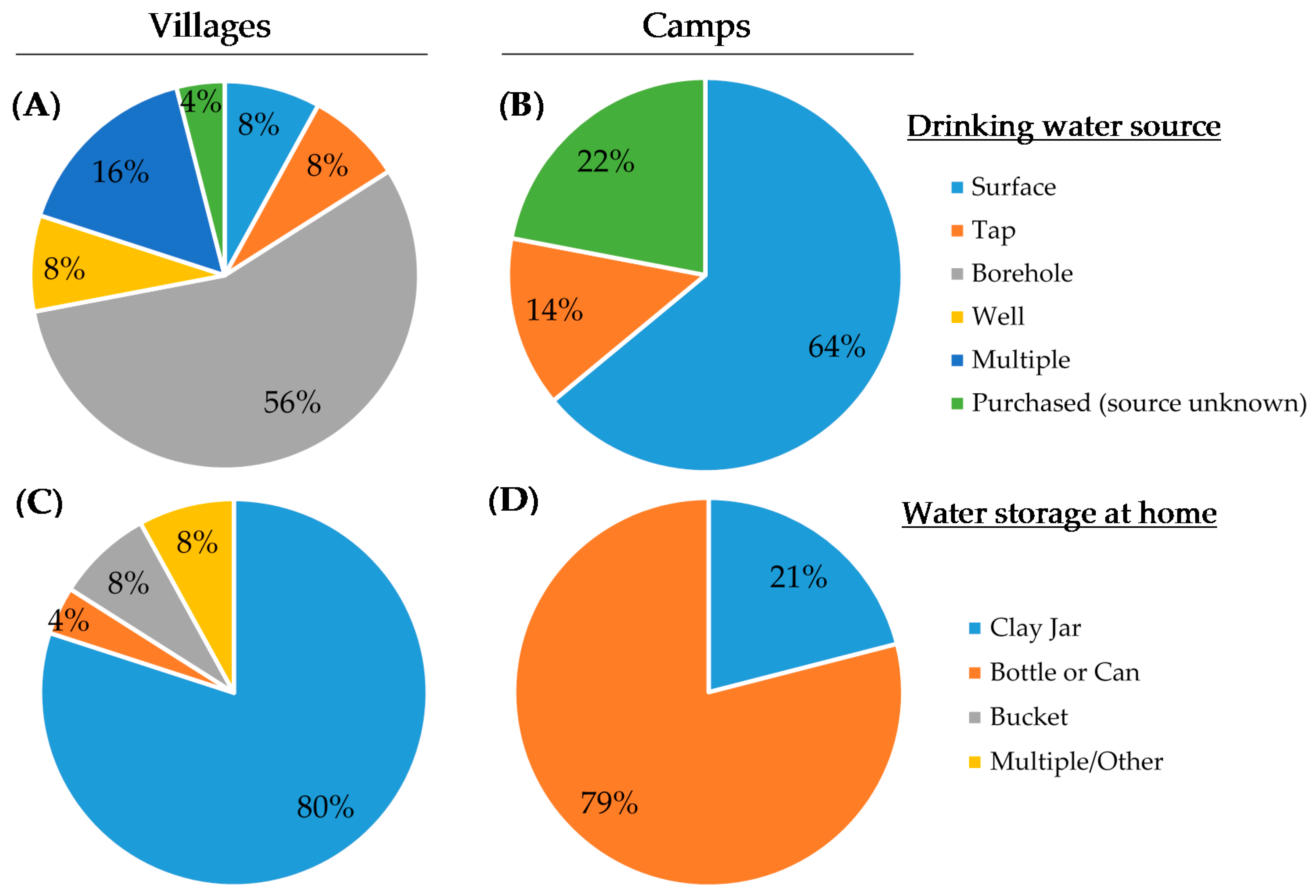
6] estimate of 16% of rural Cameroonians relying on surface water to 64% (camps) and 8% (villages) from this study, one can see the importance of understanding the resources of local communities. Being nomadic, transhumant pastoralists must rely on more easily accessible water sources for themselves and their cattle, which is likely the reason for such a high reliance on surface waters. Water storage trends also reflect this difference in lifestyle, as the jerry cans and bottles used by homes in nomadic camps would be much more portable than the clay jars used by most village homes.
Regarding sanitation and hygiene, general trends suggest worse conditions in camps than in villages. A relatively high open defecation rate in camps (19%) suggests that a difference in access to proper sanitation may exist. The open defecation rate of 4% in villages is much lower than the WHO/UNICEF Joint Monitoring Program’s estimate of 12% of the rural Cameroon population [
33]. Though the differences were not statistically significant, it appears from our study that reported handwashing is more common in villages than camps, especially handwashing with soap (
Table 2).
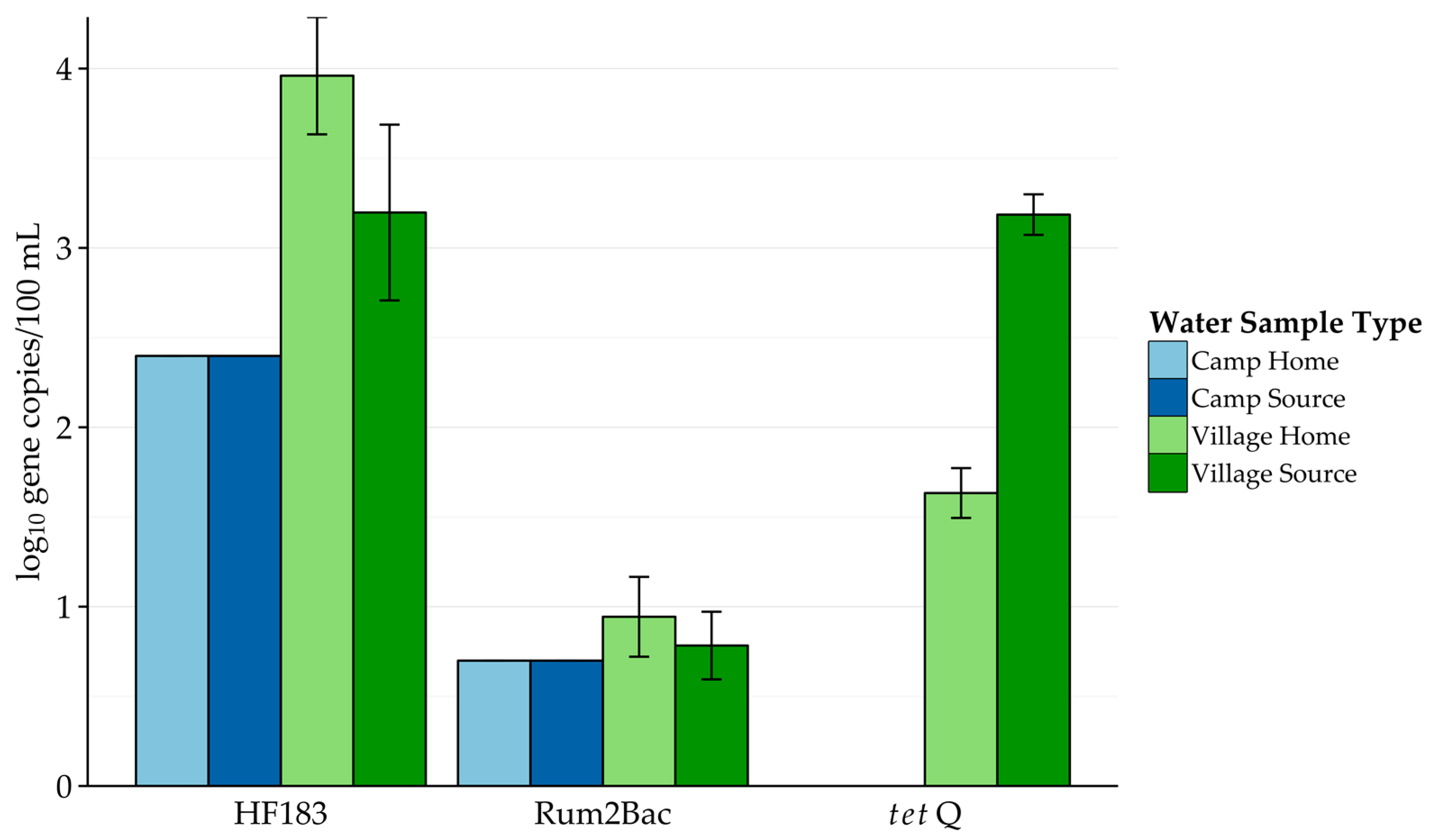
A few major trends can be seen in the molecular analysis of the region’s water quality. First, fecal contamination of water sources by ruminants was very persistent, with detection rates ranging from 65% to 100%. Interestingly, despite higher open defecation rates, human fecal contamination levels appeared to be only slightly higher in camps than in village source water, and were lower among household water samples. The presence of tetQ in villages alone may suggest differences in antibiotic use (for humans and/or animals) in the two settings.
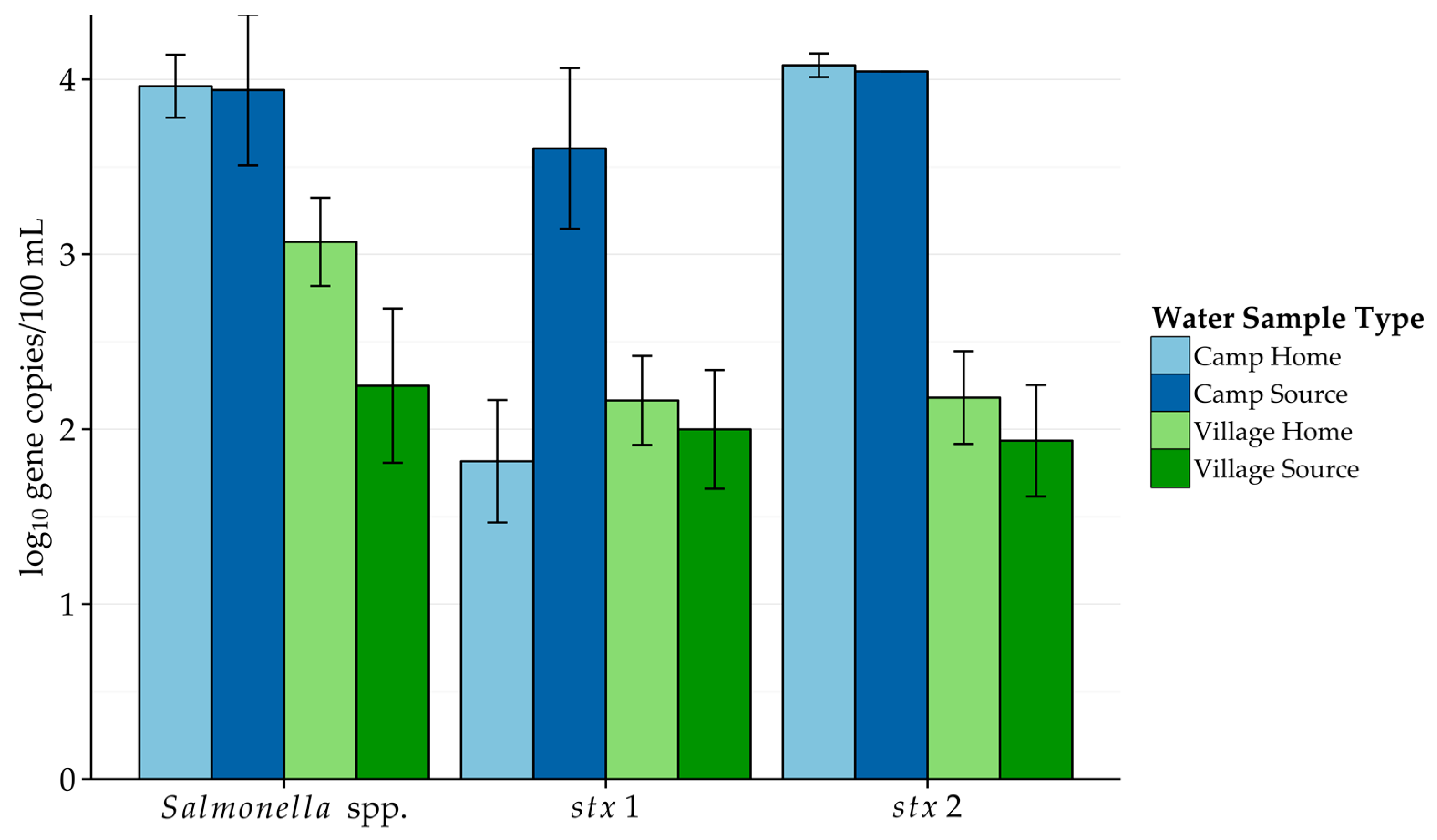
STEC are common zoonotic pathogens associated with fecal contamination from animals (especially cattle) and humans; Shiga toxins are capable of causing gastrointestinal illnesses ranging from diarrhea to the potentially fatal hemolytic uremic syndrome in humans [
34]. Accordingly, Shiga-toxin producing genes
stx1 and
stx2 were studied to assess the potential presence of Shiga-toxin producers.
Salmonella were also investigated due to the importance of Typhoid fever in the region and the recent identification of this genus from a wide variety of water sources in the Logone Valley, which includes Far North, Cameroon [
19]. Thus,
Salmonella spp. was tested as a screening tool for potential presence of
Salmonella-related pathogens. No clear pattern was seen in pathogen detection rates when comparing villages to camps. However, when considering the contamination level among those samples that were positive, camp water sources had higher
stx2 and
Salmonella spp. gene copy numbers. These results may be taken together to say that while source and household water in camps was not more frequently contaminated by
Salmonella spp. and Shiga toxin-producing
E. coli, these water samples had higher contamination levels of these pathogens than similar village water samples.
Considering the impact of WASH characteristics on gastrointestinal illness, one would expect the impact of higher contamination levels in water consumed by individuals in nomadic camps to only be worsened by the lower reported handwashing rates. This effect was not shown at the population level, as diarrheal rates were not significantly different between populations in camps and villages. However, a significant difference in diarrheal incidence was seen among children under 5 years of age when comparing camps to villages (31.3%, 0%;
p-value < 0.05). This is an important finding when considering the impact that diarrheal illness has on the global under 5 mortality rate. Children’s immune systems have not yet adapted to the pathogens encountered in day-to-day life. Because of this, this difference in diarrheal rates may act as an indicator of differences in WASH conditions, whereas the stronger immune systems of adults who have adapted to their individual settings may dilute this effect. Oloruntoba, et al. found improper water handling and insufficient caretaker handwashing to be significant contributors to diarrheal risk among children under five in Ibadan, Nigeria. These may present important intervention points in the reduction of the under-5 diarrheal rate in rural Far North, Cameroon, but additional studies on handwashing behaviors are needed [
35]. Additionally, we measured higher gene concentrations for all three pathogen markers studied in camps as opposed to villages (though not statistically significant), which may also contribute to this difference.
Worth noting, other factors may be driving this difference in diarrheal rates, such as access to medical care to prevent reoccurring illnesses and differences in direct transmission in different households. Additionally, children in nomadic villages are likely in closer proximity to cattle and, therefore, they might have more chances to get exposed to cattle waste when they play outside. This direct contact with fecal pathogens may also contribute to differences in the under-5 diarrheal rates observed. Though not statistically significant, village residents appeared to visit hospitals more frequently, with 14% of respondents having visited a hospital within the past 30 days, compared to just 6% of camp residents. Further, a significant difference was not found between the proportions of respondents having been under the care of a physician between camps (7%) and villages (10%).
4.2. Comparison with Urban Water Quality in Far North, Cameroon
This study serves as a follow-up to previous work conducted by Healy Profitós et al. [
20,
21] who investigated water quality along informal water distribution systems in urban Maroua, Cameroon. Across both rural camps and rural villages, evidence of human fecal contamination suggests rural areas are subject to greater human impacts on microbial water quality, when compared to the urban area of Maroua. While only 7% of urban water samples collected showed HF183 detection [
20], the same marker was detected in 20%–33% of rural samples in this study (
Table 2). Comparing the detection rates of
tetQ, rural villages had the highest
tetQ detection rate among all sample types (47%), urban source water samples were second highest (28%), and similar rates were found among urban homes (13%) and rural village homes (14%). Note that no tetracycline resistance genes were found in the rural camp water samples.
Concerning non-bloody diarrheal rates, the rate among children was lower in rural villages than in Maroua among children under five years old (0% and 23.7%, respectively) [
20]. However, rural camps reported the highest diarrheal rates in this age group, at 31.3%. Diarrheal rates were higher for children 5 years and older in Maroua, as compared to rural camps and villages (11.9%, 0%, and 1.7%, respectively) [
20]. Among adults, non-bloody diarrheal rates were similar across all study areas.
4.3. Study Limitations and Future Work
Study limitations primarily pertain to the scale of this study. Surveys and water samples were collected as a small portion of a larger study in Cameroon, so resources were limited. This factor surfaced regarding the surveys, as respondent numbers were not equal between camps and villages, and in the collection of water samples, where small sample numbers likely contributed to insignificant findings, though trends were observed. Questionnaire response rates were strong, but item non-response on the surveys may present a bias that we were unable to account for. Further, behavior and health outcome measurements in this study relied on self-reported health history and hygiene behavior survey questions. These self-reported data are subject to both recall bias and potential misreporting, where negative perceptions of illness or improper hygiene behaviors may influence survey participants’ responses.
Another limitation of this study pertains to the number of microbial source tracking markers and antibiotic resistance genes investigated. To sufficiently investigate the sources of fecal contamination, further analysis of human- and ruminant-specific markers would be helpful, as geographic difference in gut microbiomes may exist. Thus, an investigation of the best host-specific fecal markers should be conducted, but this analysis was beyond the scope of our study. As mentioned, tetracycline resistance genes were investigated as a snapshot of antibiotic resistance, but tetracycline is not the only antibiotic used in this region for humans or animals. A known limitation of using molecular techniques to study pathogens is that these techniques test for the presence of genes rather than viable organisms; thus, the presence/levels of detected target pathogens should not be directly interpreted as a known risk for pathogen infection.
It is important to note that the villages under study, while rural, are near the provincial capital of Maroua. Given this, our study may underestimate the need for improved WASH conditions in rural villages of Cameroon, as areas just outside of cities are likely to have more access to healthcare services and WASH essentials, such as chlorine and soap.