One-Year Results of a Synthetic Intervention Model for the Primary Prevention of T2D among Elderly Individuals with Prediabetes in Rural China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Research Design
2.2. Sample Size
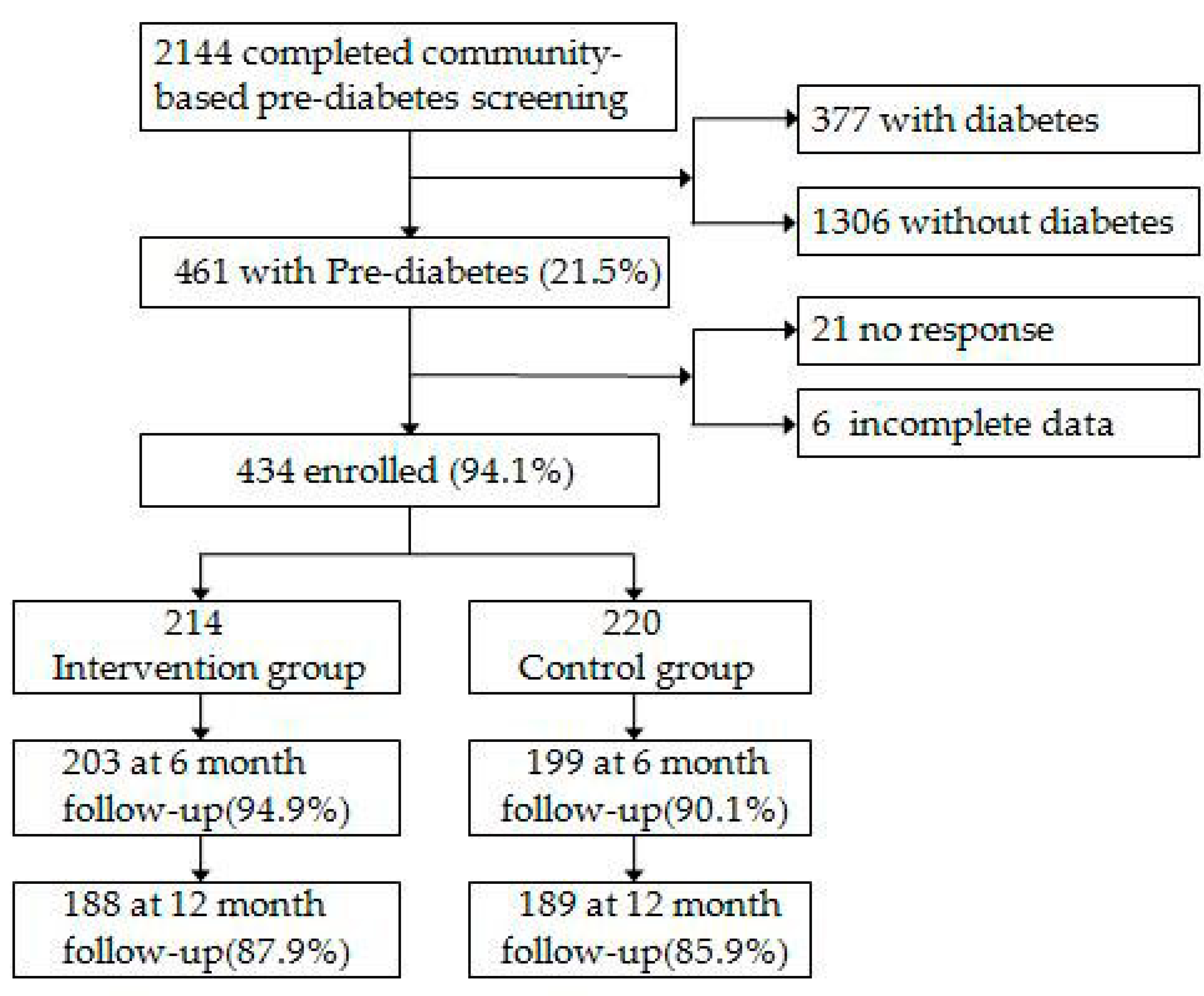
2.3. Population and Procedures
2.4. Baseline Measures
2.5. Primary Outcome Measures
2.6. Intervention
- (1)
- Standard health advice (control group): Subjects were advised to stop smoking, limit the amount of liquor consumed, eat less animal fat and avoid a sedentary lifestyle. These objectives were repeatedly explained every six months by the IST.
- (2)
- Synthetic intervention (intervention group): The synthetic intervention model was formulated through consultations with several experts (12 experts in total) and focus group discussions and included lifestyle education, lifestyle intervention, training for the Self-Monitoring of Blood Glucose (SMBG) and setting up a Help Each Other Group (HEOG) (Table 1).
2.7. Motivation and Adherence
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Participation and Adherence
3.3. Incidence of Diabetes
3.4. Outcome at the 1-Year Follow-Up
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- De Vegt, F.; Dekker, J.M.; Jager, A.; Hienkens, E.; Kostense, P.J.; Stehouwer, C.D.; Nijpels, G.; Bouter, L.M.; Heine, R.J. Relation of impaired fasting and postload glucose with incident type 2 diabetes in a Dutch population: The Hoorn Study. JAMA 2001, 285, 2109–2113. [Google Scholar] [CrossRef] [PubMed]
- Perreault, L.; Pan, Q.; Mather, K.J.; Watson, K.E.; Hamman, R.F.; Kahn, S.E. Effect of regression from prediabetes to normal glucose regulation on long-term reduction in diabetes risk: Results from the Diabetes Prevention Program Outcomes Study. Lancet 2012, 379, 2243–2251. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Luan, J.; Hennings, S.; Wareham, N.J. Incidence of Type 2 diabetes in England and its association with baseline impaired fasting glucose: The Ely study 1990–2000. Diabet. Med. 2007, 24, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M.; Davidson, M.B.; DeFronzo, R.A.; Heine, R.J.; Henry, R.R.; Pratley, R.; Zinman, B. Impaired fasting glucose and impaired glucose tolerance: Implications for care. Diabetes Care 2007, 30, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Gillies, C.L.; Abrams, K.R.; Lambert, P.C.; Cooper, N.J.; Sutton, A.J.; Hsu, R.T.; Khunti, K. Pharmacological and lifestyle interventions to prevent or delay type 2 diabetes in people with impaired glucose tolerance: Systematic review and meta-analysis. BMJ 2007, 334, 299. [Google Scholar] [CrossRef] [PubMed]
- DREAM Trial Investigators; Gerstein, H.C.; Yusuf, S.; Bosch, J.; Pogue, J.; Sheridan, P.; Dinccag, N.; Hanefeld, M.; Hoogwerf, B.; Laakso, M.; et al. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: A randomised controlled trial. Lancet 2006, 368, 1096–1105. [Google Scholar] [PubMed]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [PubMed]
- Tuomilehto, J.; Lindstrom, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, K.F.; Lindgarde, F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise: The 6-year Malmo feasibility study. Diabetologia 1991, 34, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Snehalatha, C.; Mary, S.; Mukesh, B.; Bhaskar, A.D.; Vijay, V. The Indian diabetes prevention programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1). Diabetologia 2006, 49, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Hamman, R.F.; Wing, R.R.; Edelstein, S.L.; Lachin, J.M.; Bray, G.A.; Delahanty, L.; Hoskin, M.; Kriska, A.M.; Mayer-Davis, E.J.; Pi-Sunyer, X.; et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006, 29, 2102–2107. [Google Scholar] [CrossRef] [PubMed]
- Penn, L.; White, M.; Lindstrom, J.; den Boer, A.T.; Blaak, E.; Eriksson, J.G.; Feskens, E.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.M.; Walker, M.; et al. Importance of weight loss maintenance and risk prediction in the prevention of type 2 diabetes analysisof European diabetes prevention study RCT. PLoS ONE 2013, 8, e57143. [Google Scholar] [CrossRef] [PubMed]
- Satterfield, D.W.; Volansky, M.; Caspersen, C.J.; Engelgau, M.M.; Bowman, B.A.; Gregg, E.W.; Geiss, L.S.; Hosey, G.M.; May, J.; Vinicor, F. Community-based lifestyle interventions to prevent type 2 diabetes. Diabetes Care 2003, 26, 2643–2652. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Lovejoy, J.C.; Smith, S.R.; DeLany, J.P.; Lefevre, M.; Hwang, D.; Ryan, D.H.; York, D.A. The influence of different fats and fatty acids on obesity, insulin resistance and inflammation. J. Nutr. 2002, 132, 2488–2491. [Google Scholar] [PubMed]
- Yang, W.; Lu, J.; Weng, J.; Jia, W.; Ji, L.; Xiao, J.; Shan, Z.; Liu, J.; Tian, H.; Ji, Q.; et al. Prevalence of diabetes among men and women in China. N. Engl. J. Med. 2010, 362, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Economic costs of diabetes in the U.S.in 2007. Diabetes Care 2008, 31, 596–615. [Google Scholar]
- Li, G.; Zhang, P.; Wang, J.; Gregg, E.W.; Yang, W.; Gong, Q.; Li, H.; Jiang, Y.; An, Y.; Shuai, Y.; et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: A 20-year follow-up study. Lancet 2008, 371, 1783–1789. [Google Scholar] [CrossRef]
- Pan, X.R.; Li, G.W.; Hu, Y.H.; Wang, J.X.; Yang, W.Y.; An, Z.X.; Hu, Z.X.; Lin, J.; Xiao, J.Z.; Cao, H.B.; et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucosetolerance: The Da Qing IGT and Diabetes Study. Diabetes Care 1997, 20, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Department of Noncommunicable Disease Surveillance. Definition, diagnosis and classification of diabetes mellitus and its complications: Report of a WHO consultation. Part 1: Diagnosis and classification of diabetes mellitus. Diabet. Med. 1998, 15, 539–553. [Google Scholar]
- Qin, L.L.; Xu, H.L. A cross-sectional study of the effect of health literacy on diabetes prevention and control among elderly individuals with prediabetes in rural China. BMJ Open 2016, 6, e11077. [Google Scholar] [CrossRef] [PubMed]
- The Chinese Medical Association. Chinese type 2 diabetes prevention guide. Chin. J. Diabetes Mellit. 2014, 6, 447–498. (In Chinese) [Google Scholar]
- Kosaka, K.; Noda, M.; Kuzuya, T. Prevention of type 2 diabetes by lifestyle intervention: A Japanese trial in IGT males. Diabetes Res. Clin. Pract. 2004, 67, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Gregg, E.W.; Chen, H.; Wagenknecht, L.E.; Clark, J.M.; Delahanty, L.M.; Bantle, J.; Pownall, H.J.; Johnson, K.C.; Safford, M.M.; Kitabchi, A.E.; et al. Association of an intensive lifestyle intervention with remission of type 2 diabetes. JAMA 2012, 308, 2489–2496. [Google Scholar] [CrossRef] [PubMed]
- Diabetes Prevention Program Research Group; Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Christophi, C.A.; Hoffman, H.J.; Brenneman, A.T.; Brown-Friday, J.O.; Goldberg, R.; et al. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet 2009, 374, 1677–1686. [Google Scholar] [PubMed]
- Tabák, A.G.; Herder, C.; Rathmann, W.; Brunner, E.J.; Kivimäki, M. Prediabetes: A high-risk state for diabetes development. Lancet 2012, 379, 2279–2290. [Google Scholar] [CrossRef]
- Ligthart, S.; van Herpt, T.T.; Leening, M.J.; Kavousi, M.; Hofman, A.; Stricker, B.H.; van Hoek, M.; Sijbrands, E.J.; Franco, O.H.; Dehghan, A. Lifetime risk of developing impaired glucose metabolism and eventual progression from prediabetes to type 2 diabetes: A prospective cohort study. Lancet Diabetes Endocrinol. 2016, 4, 44–51. [Google Scholar] [CrossRef]
- Martin, S.; Schneider, B.; Heinemann, L.; Lodwig, V.; Kurth, H.J.; Kolb, H.; Scherbaum, W.A. Self-monitoring of blood glucose in type 2 diabetes and long-term outcome: An epidemiological cohort study. Diabetologia 2006, 49, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Murata, G.H.; Shah, J.H.; Hoffman, R.M.; Wendel, C.S.; Adam, K.D.; Solvas, P.A.; Bokhari, S.U.; Duckworth, W.C. Intensified blood glucose monitoring improves glycemic control in stable, insulin-treated veterans with type 2 diabetes: The Diabetes Outcomes in Veterans Study (DOVES). Diabetes Care 2003, 26, 1759–1763. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Su, Q.; Feng, B.; Shan, Z.; Hu, R.; Xing, X.; Xue, Y. Glycemic control and self-monitoring of blood glucose in Chinese patients with type 2 diabetes on insulin: Baseline results from the COMPASS study. Diabetes Res. Clin. Pract. 2016, 112, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.M.; Yang, E.; Ramanathan, D.; Benjamins, M.R. Community-based diabetes community health worker intervention in an underserved Chicago population. J. Community Health 2016, 41, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Pearce, K.A.; Jarrett, T.D.; Scutchfield, F.D.; Talbert, J.C.; Bolt, W.D.; Barron, M.A.; Houlihan, J.M.; Dignan, M.B. Research partnerships with healthcare providers in rural community health centers: Needs and challenges in diabetes research. Public Health Front. 2015, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.; Kent, J.; Lyons, C. Building partnerships for healthy environments: Research, leadership and education. Health Promot. J. Aust. 2014, 25, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Dunkley, A.J.; Bodicoat, D.H.; Greaves, C.J.; Russell, C.; Yates, T.; Davies, M.J.; Khunti, K. Diabetes prevention in the real world: Effectiveness of pragmatic lifestyle interventions for the prevention of type 2 diabetes and of the impact of adherence to guideline recommendations: A systematic review and meta-analysis. Diabetes Care 2014, 37, 922–933. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, R.T.; Finch, E.A.; Brizendine, E.; Zhou, H.; Marrero, D.G. Translating the diabetes prevention program into the community: The DEPLOY pilot study. Am. J. Prev. Med. 2008, 35, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Katula, J.A.; Vitolins, M.Z.; Rosenberger, E.L.; Blackwell, C.S.; Morgan, T.M.; Lawlor, M.S.; Goff, D.C.J. One-Year Results of a community-based translation of the Diabetes Prevention Program: Healthy-Living Partnerships to Prevent Diabetes (HELP PD) Project. Diabetes Care 2011, 34, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Hall, J.; Byles, J.; Shi, Z. Dietary pattern is associated with obesity in older people in China: Data from China Health and Nutrition Survey (CHNS). Nutrients 2015, 7, 8170–8188. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Buys, N.J.; Hills, A.P. Dietary pattern and its association with the prevalence of obesity, hypertension and other cardiovascular risk factors among Chinese older adults. Int. J. Environ. Res. Public Health 2014, 11, 3956–3971. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.K.; Kriska, A.M.; Venditti, E.M.; Miller, R.G.; Brooks, M.M.; Burke, L.E.; Siminerio, L.M.; Solano, F.X.; Orchard, T.J. Translating the diabetes prevention program: A comprehensive model for prevention training and program delivery. Am. J. Prev. Med. 2009, 37, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Whittemore, R.; Melkus, G.; Wagner, J.; Dziura, J.; Northrup, V.; Grey, M. Translating the diabetes prevention program to primary care: A pilot study. Nurs. Res. 2009, 58, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Wolf, A.M.; Conaway, M.R.; Crowther, J.Q.; Hazen, K.Y.; Nadler, J.L.; Oneida, B.; Bovbjerg, V.E. Translating lifestyle intervention to practice in obese patients with type 2 diabetes: Improving Control with Activity and Nutrition (ICAN) study. Diabetes Care 2004, 27, 1570–1576. [Google Scholar] [CrossRef] [PubMed]
- Fianu, A.; Bourse, L.; Naty, N.; Le Moullec, N.; Lepage, B.; Lang, T.; Favier, F. Long-Term Effectiveness of a Lifestyle Intervention for the Primary Prevention of Type 2 Diabetes in a Low Socio-Economic Community—An Intervention Follow-Up Study on Reunion Island. PLoS ONE 2016, 11, e146095. [Google Scholar] [CrossRef] [PubMed]
- Collins, L.M.; Schafer, J.L.; Kam, C.M. A comparison of inclusive and restrictive strategies in modern missing data procedures. Psychol. Methods 2001, 6, 330–351. [Google Scholar] [CrossRef] [PubMed]
- Gerstein, H.C.; Santaguida, P.; Raina, P.; Morrison, K.M.; Balion, C.; Hunt, D.; Yazdi, H.; Booker, L. Annual incidence and relative risk of diabetes in people with various categories of dysglycemia: A systematic overview and meta-analysis of prospective studies. Diabetes Res. Clin. Pract. 2007, 78, 305–312. [Google Scholar] [CrossRef] [PubMed]
| Time Interval | Intervention Components | Intervention Content |
|---|---|---|
| 3 months | Lifestyle education | 1. Giving out diabetes knowledge manual and leaflets |
| 2. Education curriculum | ||
| 3 months | Lifestyle intervention | 1. Giving instructions about diet and physical activity |
| 2. lifestyle counsel sessions | ||
| 3 months | SMBG | 1. Training method of fasting glucose measurement |
| 2. Reporting SMBG times | ||
| At first intervention | HEOG | 1. Setting HEOG |
| 2. Choose HEOG responsible persons |
| Variables | Intervention Group | Control Group | Value x2 or t | p Value |
|---|---|---|---|---|
| n | 214 | 220 | ||
| Age (years) | 69.2 ± 6.8 | 69.5 ± 6.3 | 0.38 | 0.67 |
| Sex | ||||
| Male | 93 (43.5) | 87 (39.5) | 0.68 | 0.41 |
| Female | 121 (56.5) | 133 (60.5) | ||
| Marriage status | ||||
| Married | 146 (68.2) | 166 (75.5) | 2.81 | 0.09 |
| Non-married | 68 (31.8) | 54 (24.5) | ||
| Education | ||||
| Less than 1 year | 35 (16.4) | 46 (20.9) | 2.47 | 0.29 |
| 1–6 years | 134 (62.6) | 138 (62.7) | ||
| 6 years and above | 45 (21.0) | 36 (16.4) | ||
| History of hyperglycemia * | ||||
| Yes | 11 (5.1) | 17 (7.7) | 1.20 | 0.27 |
| No | 203 (94.9) | 203 (92.3) | ||
| Family history of diabetes | ||||
| Yes | 14 (6.5) | 22 (10.0) | 1.71 | 0.19 |
| No | 200 (93.5) | 198 (90.0) | ||
| Weight (kg) | 59.5 ± 8.8 | 59.7 ± 10.6 | 0.63 | 0.53 |
| BMI (kg/m2) | 23.5 ± 3.2 | 23.9 ± 3.7 | 1.73 | 0.09 |
| Waist circumference (cm) | 84.2 ± 9.8 | 85.6 ± 8.8 | 1.73 | 0.09 |
| Plasma glucose (mg/dL) | ||||
| Fasting | 111.0 ± 10.9 | 109.8 ± 8.8 | 1.38 | 0.17 |
| 2-h glucose | 153.9 ± 18.9 | 156.5 ± 20.4 | 1.41 | 0.16 |
| Serum lipid (mg/dL) | ||||
| Total cholesterol | 99.8 ± 12.8 | 99.0 ± 12.4 | 0.65 | 0.51 |
| HDL-C | 32.3 ± 9.4 | 31.7 ± 7.9 | 0.80 | 0.43 |
| Triglycerides | 36.8 ± 20.3 | 34.8 ± 21.1 | 1.02 | 0.31 |
| HbA1c (mg %) | 5.7 ± 0.9 | 5.8 ±1 .1 | 0.95 | 0.34 |
| Physical activity (MET/week) | 1484.0 ± 959.4 | 1382.8 ± 925.4 | 1.12 | 0.26 |
| DBI-07 score | ||||
| ≤14 | 46 (21.5) | 64 (29.1) | 3.31 | 0.07 |
| ≥15 | 168 (78.5) | 156 (70.9) | ||
| Total kcal/day | 1763.6 ± 235.6 | 1784.2 ± 213.2 | 0.95 | 0.34 |
| Variable | Follow Up 6 Months Intervention Control | Follow Up 12 Months Intervention Control | ||
|---|---|---|---|---|
| Lifestyle Education | ||||
| Attended an Education Curriculum | 162 (79.9) | - | 161 (85.7) | - |
| Lifestyle Intervention | ||||
| Decreased Consumption of Fat | 93 (45.8) | 49 (24.6) † | 156 (83.0) | 56 (29.6) † |
| Increased Consumption of Vegetables | 78 (38.4) | 30 (15.1) † | 98 (52.1) | 32 (16.9) † |
| Increased Consumption of Fruit | 30 (14.8) | 25 (12.6) | 50 (26.6) | 43 (22.8) |
| Decreased Consumption of Alcohol | 43 (21.2) | 40 (20.1) | 55 (29.3) | 44 (23.3) |
| Decreased Consumption of Sugar | 168 (82.8) | 110 (55.3) † | 142 (75.5) | 105 (55.6) † |
| Decreased Calorie Intake | 86 (42.4) | 47 (23.6) † | 75 (39.9) | 28 (14.8) † |
| Increased Walking Time | 76 (37.4) | 40 (20.1) † | 98 (52.1) | 42 (22.2) † |
| Decreased Sitting Time | 49 (24.1) | 33 (16.6) | 57 (30.3) | 35 (18.5) † |
| SMBG | 147 (72.6) | - | 107 (56.7) | - |
| Variable | Intervention Group | Control Group | p-Value |
|---|---|---|---|
| At 6 months | |||
| n | 203 | 199 | |
| Incidence of diabetes †, % (95% CI) | 1.5 (0.0, 3.4) | 2.5 (0.5, 5.0) | 0.452 |
| Adjusted HR †† (95% CI) | 0.6 (0.1, 2.6) | 1.0 | 0.509 |
| At 12 months | |||
| n | 188 | 189 | |
| Incidence of diabetes,% (95% CI) | 4.2 (1.5, 6.7) | 19.7 (13.8, 25.1) | 0.041 |
| Adjusted HR †† (95% CI) | 0.2 (0.1, 0.5) | 1.0 | <0.001 |
| Outcome | Intervention Group | Control Group | p-Value |
|---|---|---|---|
| Change in Weight | |||
| In kilograms | −3.2 (−4.2, −2.3) | 1.7 (0.2, 3.1) | <0.001 |
| Percent change | −4.5 (−6.2, −2.5) | 4.6 (2.0, 7.2) | <0.001 |
| Change in BMI (kg/m2) | −3.2 (−3.8, −2.9) | 0.8 (0.2, 1.4) | <0.001 |
| Change in Waist circumference | |||
| In centimeter | −2.4 (−3.0, −1.9) | 1.0 (0.4, 1.5) | <0.001 |
| Percent change | −2.9 (−3.5, −2.3) | 1.3 (0.6, 1.9) | <0.001 |
| Change in glucose (mg/dL) | |||
| Fasting | −3.9 (−6.2, −1.8) | 2.2 (0.6, 3.8) | <0.001 |
| 2-h glucose | −8.7 (−12.8, −4.7) | −5.4 (−9.2, −1.2) | 0.246 |
| Change in serum lipid (mg/dL) | |||
| Total cholesterol | −9.1 (−11.7, −6.1) | −4.6 (−7.3, −2.1) | 0.014 |
| HDL-C | 3.4 (1.6, 5.3) | 2.4 (1.0, 4.0) | 0.415 |
| Triglycerides | −4.2 (−7.9, −0.5) | −0.7 (−4.6, 3.2) | 0.199 |
| Change in HbA1c (mg %) | −1.0 (−1.2, −0.8) | 0.1 (−0.1, 0.4) | 0.002 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Z.; Qin, L.; Xu, H. One-Year Results of a Synthetic Intervention Model for the Primary Prevention of T2D among Elderly Individuals with Prediabetes in Rural China. Int. J. Environ. Res. Public Health 2017, 14, 417. https://doi.org/10.3390/ijerph14040417
Hu Z, Qin L, Xu H. One-Year Results of a Synthetic Intervention Model for the Primary Prevention of T2D among Elderly Individuals with Prediabetes in Rural China. International Journal of Environmental Research and Public Health. 2017; 14(4):417. https://doi.org/10.3390/ijerph14040417
Chicago/Turabian StyleHu, Zhao, Lulu Qin, and Huilan Xu. 2017. "One-Year Results of a Synthetic Intervention Model for the Primary Prevention of T2D among Elderly Individuals with Prediabetes in Rural China" International Journal of Environmental Research and Public Health 14, no. 4: 417. https://doi.org/10.3390/ijerph14040417
APA StyleHu, Z., Qin, L., & Xu, H. (2017). One-Year Results of a Synthetic Intervention Model for the Primary Prevention of T2D among Elderly Individuals with Prediabetes in Rural China. International Journal of Environmental Research and Public Health, 14(4), 417. https://doi.org/10.3390/ijerph14040417





