Trait Rumination Predicts Elevated Evening Cortisol in Sexual and Gender Minority Young Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Measures
2.3.1. Salivary Cortisol
2.3.2. Trait Rumination
2.3.3. Depressed Mood
2.3.4. Daily Negative Affect
2.3.5. Additional Descriptive Variables and Covariates
2.4. Statistical Analyses
3. Results
3.1. Descriptive Results
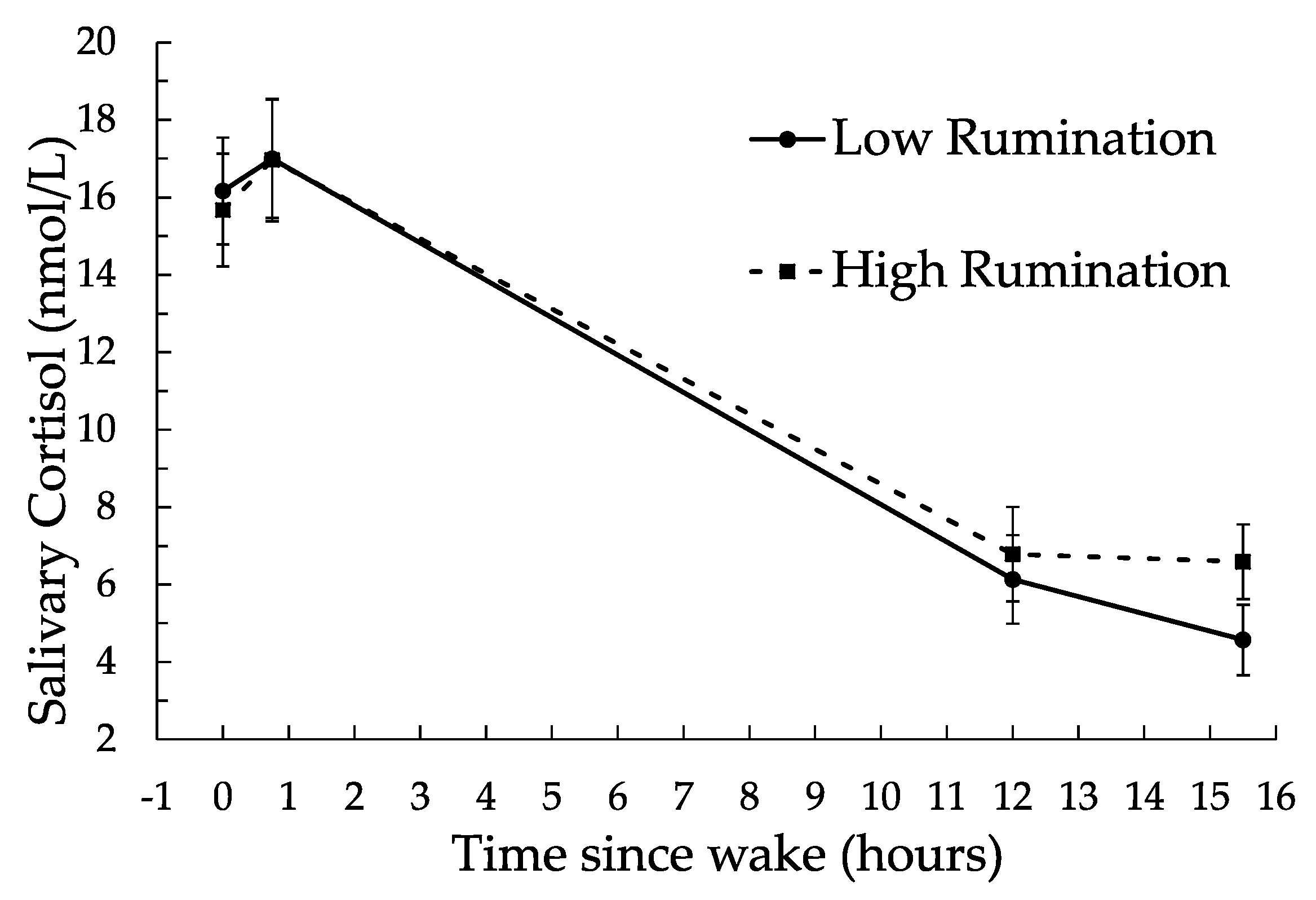
3.2. Rumination and Cortisol Results
3.3. Depressed Mood and Daily Negative Affect
3.4. Additional Exploratory Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Allen, A.P.; Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Biological and psychological markers of stress in humans: Focus on the Trier Social Stress Test. Neurosci. Biobehav. Rev. 2014, 38, 94–124. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Seeman, T. Protective and damaging effects of mediators of stress: Elaborating and testing the concepts of allostasis and allostatic load. Ann. N. Y. Acad. Sci. 1999, 896, 30–47. [Google Scholar] [CrossRef] [PubMed]
- Charmandari, E.; Tsigos, C.; Chrousos, G. Endocrinology of the stress response. Annu. Rev. Physiol. 2005, 67, 259–284. [Google Scholar] [CrossRef] [PubMed]
- Ehlert, U.; Gaab, J.; Heinrichs, M. Psychoneuroendocrinological contributions to the etiology of depression, posttraumatic stress disorder, and stress-related bodily disorders: The role of the hypothalamus-pituitary-adrenal axis. Biol. Psychol. 2001, 57, 141–152. [Google Scholar] [CrossRef]
- Juster, R.-P.; Marin, M.-F.; Sindi, S.; Nair, N.P.V.; Ng, Y.K.; Pruessner, J.C.; Lupien, S.J. Allostatic load associations to acute, 3-year and 6-year prospective depressive symptoms in healthy older adults. Physiol. Behav. 2011, 104, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Mattei, J.; Demissie, S.; Falcon, L.M.; Ordovas, J.M.; Tucker, K. Allostatic load is associated with chronic conditions in the Boston Puerto Rican Health Study. Soc. Sci. Med. 2010, 70, 1988–1996. [Google Scholar] [CrossRef] [PubMed]
- Weitzman, E.D.; Fukushima, D.; Nogeire, C.; Roffwarg, H.; Gallagher, T.F.; Hellman, L. Twenty-four hour pattern of the episodic secretion of cortisol in normal subjects. J. Clin. Endocrinol. Metab. 1971, 33, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Shipley, M.; Stafford, M.; Kivimaki, M. Association of diurnal patterns in salivary cortisol with all-cause and cardiovascular mortality: Findings from the Whitehall II Study. J. Clin. Endocrinol. Metab. 2011, 96, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- Sephton, S.E.; Sapolsky, R.M.; Kraemer, H.C.; Spiegel, D. Diurnal cortisol rhythm as a predictor of breast cancer survival. J. Natl. Cancer Inst. 2000, 92, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Sephton, S.E.; Lush, E.; Dedert, E.A.; Floyd, A.R.; Rebholz, W.N.; Dhabhar, F.S.; Spiegel, D.; Salmon, P. Diurnal cortisol rhythm as a predictor of lung cancer survival. Brain Behav. Immun. 2013, 30, S163–S170. [Google Scholar] [CrossRef] [PubMed]
- Hackett, R.A.; Kivimäki, M.; Kumari, M.; Steptoe, A. Diurnal cortisol patterns, future diabetes, and impaired glucose metabolism in the Whitehall II Cohort Study. J. Clin. Endocrinol. Metab. 2016, 101, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Segerstrom, S.C.; Stanton, A.L.; Alden, L.E.; Shortridge, B.E. A multidimensional structure for repetitive thought: What’s on your mind, and how, and how much? J. Personal. Soc. Psychol. 2003, 85, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.M.; Alloy, L.B. A roadmap to rumination: A review of the definition, assessment, and conceptualization of this multifaceted construct. Clin. Psychol. Rev. 2009, 29, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Brosschot, J.F.; Gerin, W.; Thayer, J.F. The perseverative cognition hypothesis: A review of worry, prolonged stress-related physiological activation, and health. J. Psychosom. Res. 2006, 60, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Quas, J.A.; Yim, I.S. Salivary cortisol responses to a psychosocial laboratory stressor and later verbal recall of the stressor: The role of trait and state rumination. Stress 2010, 13, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Just, N.; Alloy, L.B. The response styles theory of depression: Tests and an extension of the theory. J. Abnorm. Psychol. 1997, 106, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Dickerson, S.S.; Lam, S. Eliciting and maintaining ruminative thought: The role of social-evaluative threat. Emotion 2012, 12, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Nolen-Hoeksema, S.; Morrow, J.; Fredrickson, B.L. Response styles and the duration of episodes of depressed mood. J. Abnorm. Psychol. 1993, 102, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Dickerson, S.S. Assessing the relationship between rumination and cortisol: A review. J. Psychosom. Res. 2012, 73, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Dickerson, S.S. Extending the recovery window: Effects of trait rumination on subsequent evening cortisol following a laboratory performance stressor. Psychoneuroendocrinology 2015, 58, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Rydstedt, L.W.; Cropley, M.; Devereux, J.J.; Michalianou, G. The effects of gender, long-term need for recovery and trait inhibition-rumination on morning and evening saliva cortisol secretion. Anxiety Stress Coping 2009, 22, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Cropley, M.; Rydstedt, L.W.; Devereux, J.J.; Middleton, B. The relationship between work-related rumination and evening and morning salivary cortisol secretion. Stress Health 2015, 31, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Kuehner, C.; Holzhauer, S.; Huffziger, S. Decreased cortisol response to awakening is associated with cognitive vulnerability to depression in a nonclinical sample of young adults. Psychoneuroendocrinology 2007, 32, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Hilt, L.M.; Sladek, M.R.; Doane, L.D.; Stroud, C.B. Daily and trait rumination: Diurnal cortisol patterns in adolescent girls. Cogn. Emot. 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Huffziger, S.; Ebner-Priemer, U.; Zamoscik, V.; Reinhard, I.; Kirsch, P.; Kuehner, C. Effects of mood and rumination on cortisol levels in daily life: An ambulatory assessment study in remitted depressed patients and healthy controls. Psychoneuroendocrinology 2013, 38, 2258–2267. [Google Scholar] [CrossRef] [PubMed]
- Meyer, I.H. Prejudice, social stress, and mental health in lesbian, gay, and bisexual populations: Conceptual issues and research evidence. Psychol. Bull. 2003, 129, 674–697. [Google Scholar] [CrossRef] [PubMed]
- Pascoe, E.A.; Smart Richman, L. Perceived discrimination and health: A meta-analytic review. Psychol. Bull. 2009, 135, 531–554. [Google Scholar] [CrossRef] [PubMed]
- Parra, L.A.; Benibgui, M.; Helm, J.L.; Hastings, P.D. Minority stress predicts depression in lesbian, gay, and bisexual emerging adults via elevated diurnal cortisol. Emerg. Adulthood 2016, 4, 365–372. [Google Scholar] [CrossRef]
- Juster, R.-P.; Smith, N.G.; Ouellet, É.; Sindi, S.; Lupien, S.J. Sexual orientation and disclosure in relation to psychiatric symptoms, diurnal cortisol, and allostatic load. Psychosom. Med. 2013, 75, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Floyd, F.J.; Bakeman, R. Coming-out across the life course: Implications of age and historical context. Arch. Sex. Behav. 2006, 35, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, S.S.; Kemeny, M.E. Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychol. Bull. 2004, 130, 355–391. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, S.S.; Mycek, P.J.; Zaldivar, F. Negative social evaluation, but not mere social presence, elicits cortisol responses to a laboratory stressor task. Health Psychol. 2008, 27, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Cheung, M.S.-P.; Gilbert, P.; Irons, C. An exploration of shame, social rank and rumination in relation to depression. Personal. Individ. Differ. 2004, 36, 1143–1153. [Google Scholar] [CrossRef]
- Joireman, J. Empathy and the self-absorption paradox II: Self-rumination and self-reflection as mediators between shame, guilt, and empathy. Self Identity 2004, 3, 225–238. [Google Scholar] [CrossRef]
- Hatzenbuehler, M.L. How does sexual minority stigma “get under the skin”? A psychological mediation framework. Psychol. Bull. 2009, 135, 707–730. [Google Scholar] [CrossRef] [PubMed]
- Swank, E.; Frost, D.M.; Fahs, B. Rural location and exposure to minority stress among sexual minorities in the United States. Psychol. Sex. 2012, 3, 226–243. [Google Scholar] [CrossRef]
- Browne, K. Snowball sampling: Using social networks to research non-heterosexual women. Int. J. Soc. Res. Methodol. 2005, 8, 47–60. [Google Scholar] [CrossRef]
- Sadler, G.R.; Lee, H.-C.; Lim, R.S.-H.; Fullerton, J. Recruitment of hard-to-reach population subgroups via adaptations of the snowball sampling strategy. Nurs. Health Sci. 2010, 12, 369–374. [Google Scholar] [CrossRef] [PubMed]
- MacArthur Research Network on Socioeconomic Status and Health. Salivary Cortisol Measurement. Available online: http://www.macses.ucsf.edu/research/allostatic/salivarycort.php (accessed on 28 September 2017).
- Garde, A.H.; Hansen, Å.M. Long-term stability of salivary cortisol. Scand. J. Clin. Lab. Investig. 2005, 65, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Clements, A.D.; Parker, C.R. The relationship between salivary cortisol concentrations in frozen versus mailed samples. Psychoneuroendocrinology 1998, 23, 613–616. [Google Scholar] [CrossRef]
- Roger, D.; Najarian, B. The construction and validation of a new scale for measuring emotion control. Personal. Individ. Differ. 1989, 10, 845–853. [Google Scholar] [CrossRef]
- Kohout, F.J.; Berkman, L.F.; Evans, D.A.; Cornoni-Huntley, J. Two shorter forms of the CES-D depression symptoms index. J. Aging Health 1993, 5, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef] [PubMed]
- Fries, E.; Dettenborn, L.; Kirschbaum, C. The cortisol awakening response (CAR): Facts and future directions. Int. J. Psychophysiol. 2009, 72, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Fekedulegn, D.B.; Andrew, M.E.; Burchfiel, C.M.; Violanti, J.M.; Hartley, T.A.; Charles, L.E.; Miller, D.B. Area under the curve and other summary indicators of repeated waking cortisol measurements. Psychosom. Med. 2007, 69, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.; Plessow, F.; Rauh, M.; Gröschl, M.; Kirschbaum, C. Comparison of salivary cortisol as measured by different immunoassays and tandem mass spectrometry. Psychoneuroendocrinology 2013, 38, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, C.; Thayer, J.F.; Verkuil, B.; Lonigro, A.; Medea, B.; Couyoumdjian, A.; Brosschot, J.F. Physiological concomitants of perseverative cognition: A systematic review and meta-analysis. Psychol. Bull. 2016, 142, 231–259. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Dickerson, S.S.; Zaldivar, F.P. Rumination and cortisol responses to laboratory stressors. Psychosom. Med. 2008, 70, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Rusting, C.L.; Larsen, R.J. Diurnal patterns of unpleasant mood: Associations with neuroticism, depression, and anxiety. J. Personal. 1998, 66, 85–103. [Google Scholar] [CrossRef]
- Moberly, N.J.; Watkins, E.R. Ruminative self-focus, negative life events, and negative affect. Behav. Res. Ther. 2008, 46, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Andresen, E.M.; Malmgren, J.A.; Carter, W.B.; Patrick, D.L. Screening for depression in well older adults: Evaluation of a short form of the CES-D (Center for Epidemiologic Studies Depression Scale). Am. J. Prev. Med. 1994, 10, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Gerin, W.; Davidson, K.W.; Christenfeld, N.J.S.; Goyal, T.; Schwartz, J.E. The role of angry rumination and distraction in blood pressure recovery from emotional arousal. Psychosom. Med. 2006, 68, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Woody, A.; Figueroa, W.S.; Benencia, F.; Zoccola, P.M. Trait reflection predicts interleukin-6 response to a social-evaluative stressor. Brain Behav. Immun. 2016, 52, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Zoccola, P.M.; Figueroa, W.S.; Rabideau, E.M.; Woody, A.; Benencia, F. Differential effects of poststressor rumination and distraction on cortisol and C-reactive protein. Health Psychol. 2014, 33, 1606–1609. [Google Scholar] [CrossRef] [PubMed]
- Querstret, D.; Cropley, M. Assessing treatments used to reduce rumination and/or worry: A systematic review. Clin. Psychol. Rev. 2013, 33, 996–1009. [Google Scholar] [CrossRef] [PubMed]
- DuBois, L.Z.; Powers, S.; Everett, B.G.; Juster, R.-P. Stigma and diurnal cortisol among transitioning transgender men. Psychoneuroendocrinology 2017, 82, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Huebner, D.M.; Davis, M.C. Gay and bisexual men who disclose their sexual orientations in the workplace have higher workday levels of salivary cortisol and negative affect. Ann. Behav. Med. 2005, 30, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Stawski, R.S.; Cichy, K.E.; Piazza, J.R.; Almeida, D.M. Associations among daily stressors and salivary cortisol: Findings from the National Study of Daily Experiences. Psychoneuroendocrinology 2013. [Google Scholar] [CrossRef] [PubMed]
- Cochran, S.D.; Mays, V.M. Physical health complaints among lesbians, gay men, and bisexual and homosexually experienced heterosexual individuals: Results from the California Quality of Life Survey. Am. J. Public Health 2007, 97, 2048–2055. [Google Scholar] [CrossRef] [PubMed]
- Przedworski, J.M.; McAlpine, D.D.; Karaca-Mandic, P.; VanKim, N.A. Health and health risks among sexual minority women: An examination of 3 subgroups. Am. J. Public Health 2014, 104, 1045–1047. [Google Scholar] [CrossRef] [PubMed]
- Farmer, G.W.; Jabson, J.M.; Bucholz, K.K.; Bowen, D.J. A population-based study of cardiovascular disease risk in sexual-minority women. Am. J. Public Health 2013, 103, 1845–1850. [Google Scholar] [CrossRef] [PubMed]
- Conron, K.J.; Mimiaga, M.J.; Landers, S.J. A population-based study of sexual orientation identity and gender differences in adult health. Am. J. Public Health 2010, 100, 1953–1960. [Google Scholar] [CrossRef] [PubMed]
- Segerstrom, S.C.; Boggero, I.A.; Smith, G.T.; Sephton, S.E. Variability and reliability of diurnal cortisol in younger and older adults: Implications for design decisions. Psychoneuroendocrinology 2014, 49, 299–309. [Google Scholar] [CrossRef] [PubMed]
| Variables of Interest | Waking | 45 min Post-Wake | 12 h Post-Wake | Before Bed | AUCg | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Estimate | SE | Estimate | SE | Estimate | SE | Estimate | SE | Estimate | SE | |
| Intercept | 2.52 ** | 0.06 | 2.67 ** | 0.06 | 1.30 ** | 0.08 | 1.00 ** | 0.09 | 4.99 ** | 0.06 |
| Rumination | 0.00 | 0.02 | 0.00 | 0.02 | 0.03 | 0.02 | 0.07 * | 0.02 | 0.01 | 0.02 |
| Depressed mood | 0.01 | 0.01 | −0.01 | 0.01 | 0.00 | 0.01 | 0.03 † | 0.02 | −0.01 | 0.01 |
| Negative affect | 0.06 | 0.07 | 0.03 | 0.07 | 0.27 ** | 0.10 | 0.16 | 0.10 | 0.12 | 0.07 |
| Wake time | 0.01 | 0.02 | −0.07 ** | 0.02 | −0.07 ** | 0.02 | 0.01 | 0.03 | −0.10 ** | 0.02 |
| Day of the week | −0.14 | 0.08 | −0.25 ** | 0.08 | −0.18 † | 0.10 | 0.12 | 0.11 | −0.21 | 0.07 |
| Sex | 0.12 | 0.13 | 0.13 | 0.12 | −0.02 | 0.16 | −0.17 | 0.20 | 0.07 | 0.13 |
| Bedtime | 0.01 | 0.02 | −0.00 | 0.02 | 0.00 | 0.02 | 0.03 | 0.03 | 0.01 | 0.03 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zoccola, P.M.; Manigault, A.W.; Figueroa, W.S.; Hollenbeck, C.; Mendlein, A.; Woody, A.; Hamilton, K.; Scanlin, M.; Johnson, R.C. Trait Rumination Predicts Elevated Evening Cortisol in Sexual and Gender Minority Young Adults. Int. J. Environ. Res. Public Health 2017, 14, 1365. https://doi.org/10.3390/ijerph14111365
Zoccola PM, Manigault AW, Figueroa WS, Hollenbeck C, Mendlein A, Woody A, Hamilton K, Scanlin M, Johnson RC. Trait Rumination Predicts Elevated Evening Cortisol in Sexual and Gender Minority Young Adults. International Journal of Environmental Research and Public Health. 2017; 14(11):1365. https://doi.org/10.3390/ijerph14111365
Chicago/Turabian StyleZoccola, Peggy M., Andrew W. Manigault, Wilson S. Figueroa, Cari Hollenbeck, Anna Mendlein, Alex Woody, Katrina Hamilton, Matt Scanlin, and Ryan C. Johnson. 2017. "Trait Rumination Predicts Elevated Evening Cortisol in Sexual and Gender Minority Young Adults" International Journal of Environmental Research and Public Health 14, no. 11: 1365. https://doi.org/10.3390/ijerph14111365
APA StyleZoccola, P. M., Manigault, A. W., Figueroa, W. S., Hollenbeck, C., Mendlein, A., Woody, A., Hamilton, K., Scanlin, M., & Johnson, R. C. (2017). Trait Rumination Predicts Elevated Evening Cortisol in Sexual and Gender Minority Young Adults. International Journal of Environmental Research and Public Health, 14(11), 1365. https://doi.org/10.3390/ijerph14111365





