Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Graphene Oxide (GO) Sheets
2.2. Preparation of Silver Nanoparticles Anchored Graphene Oxide (GO-Ag)
2.3. Characterization of GO and GO-Ag
2.4. Cell Culture
2.5. Viruses and Virus Titration
2.6. Virus Inhibition Assay
2.7. Determination of Cytotoxicity of GO or GO-Ag
3. Results
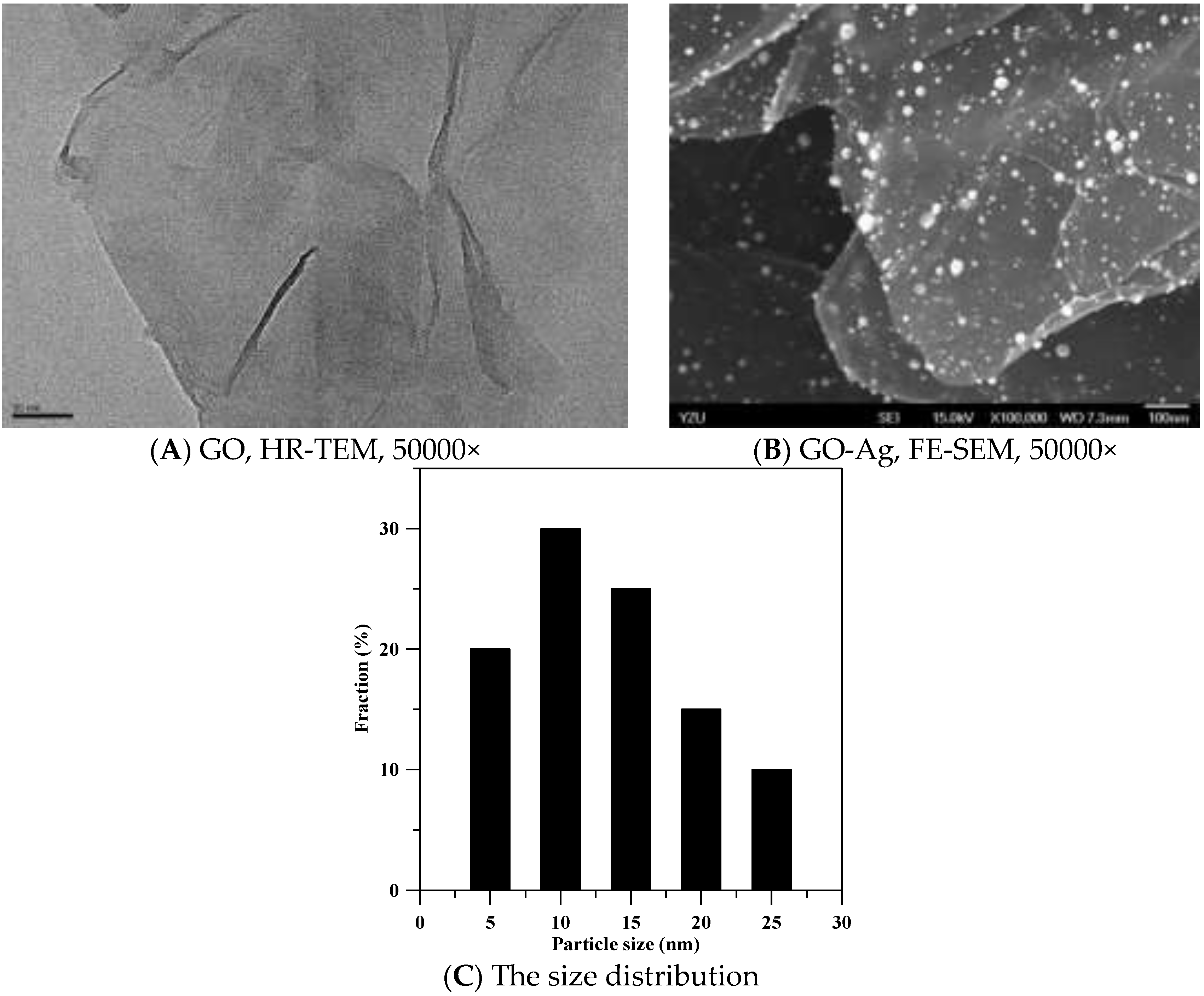
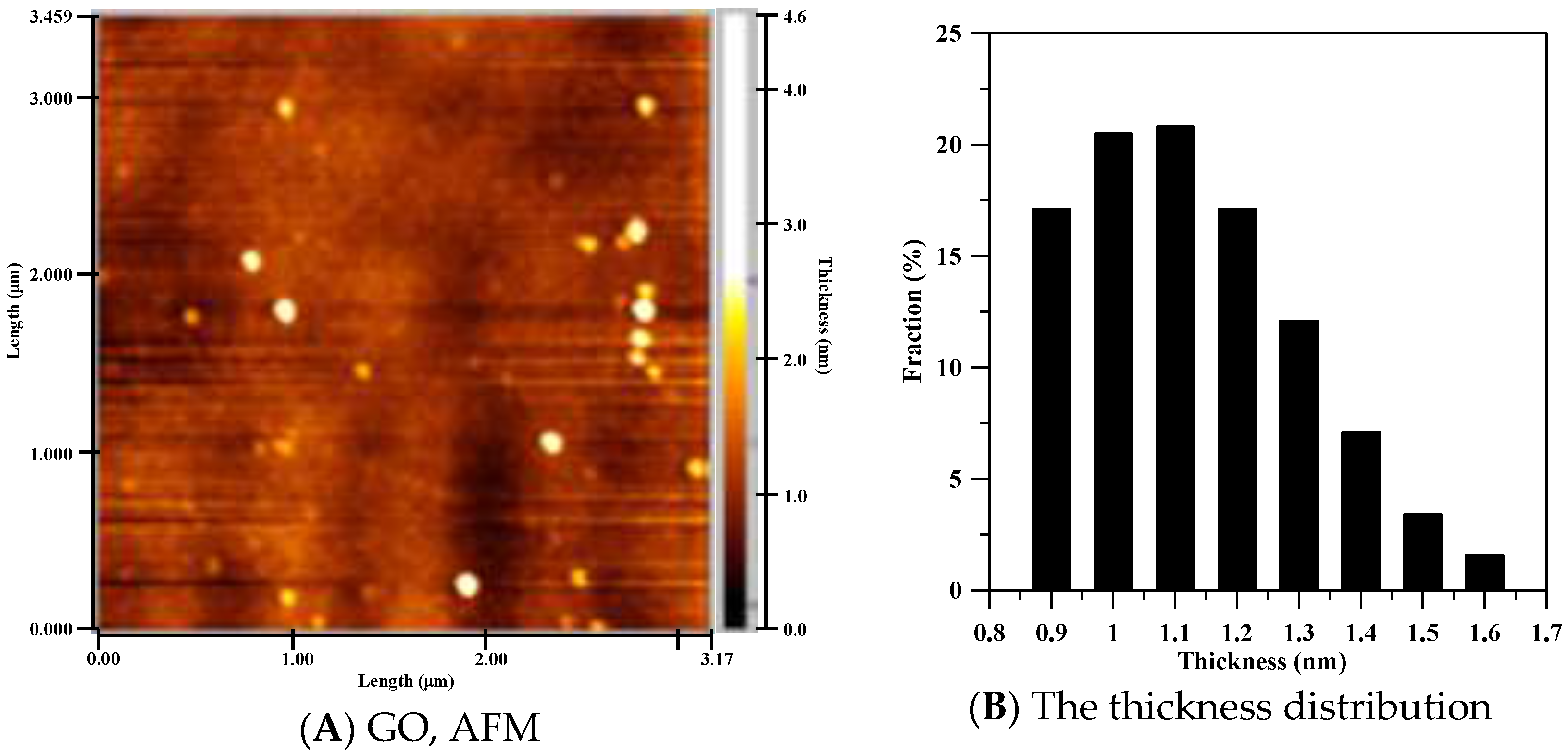
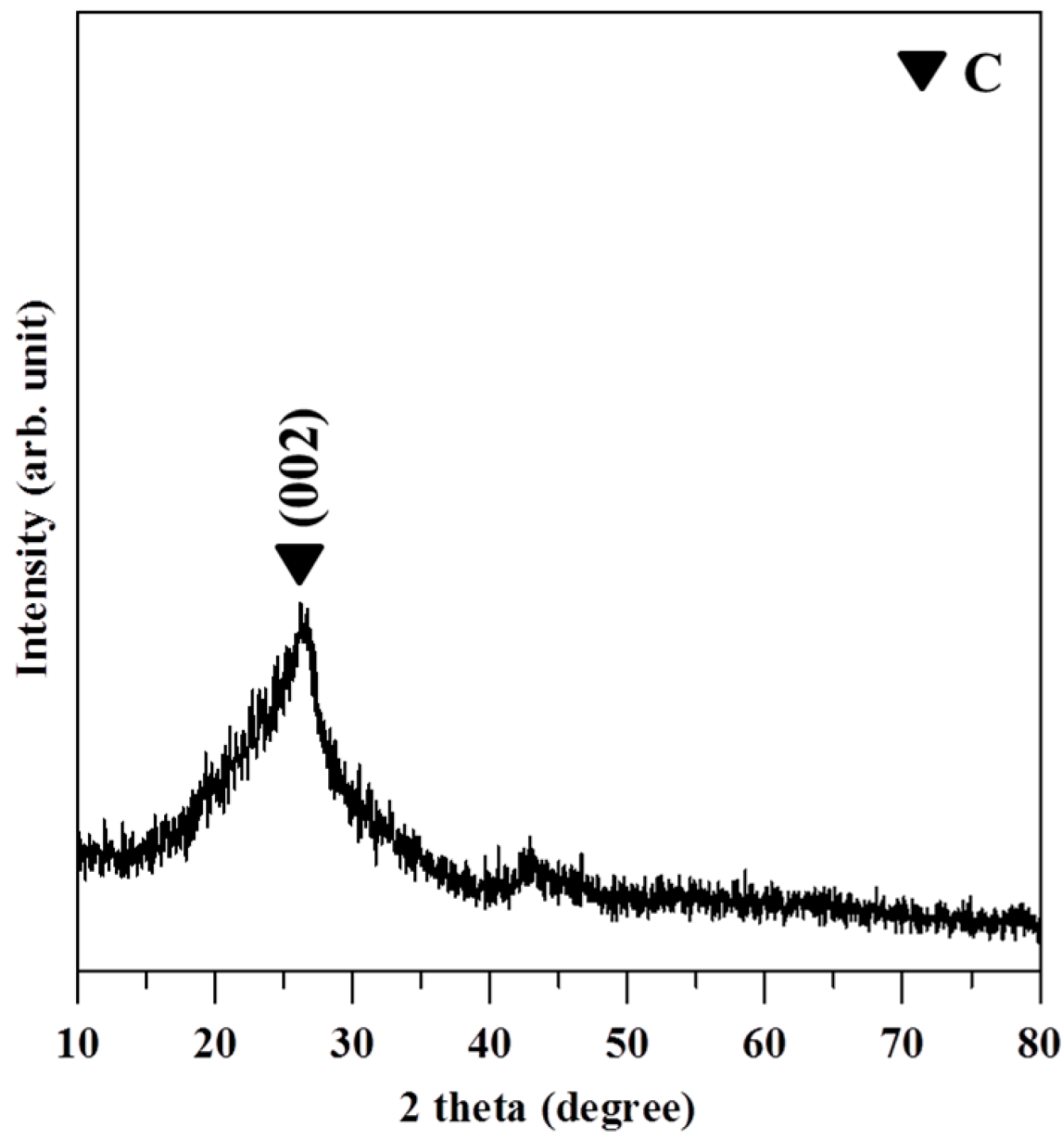
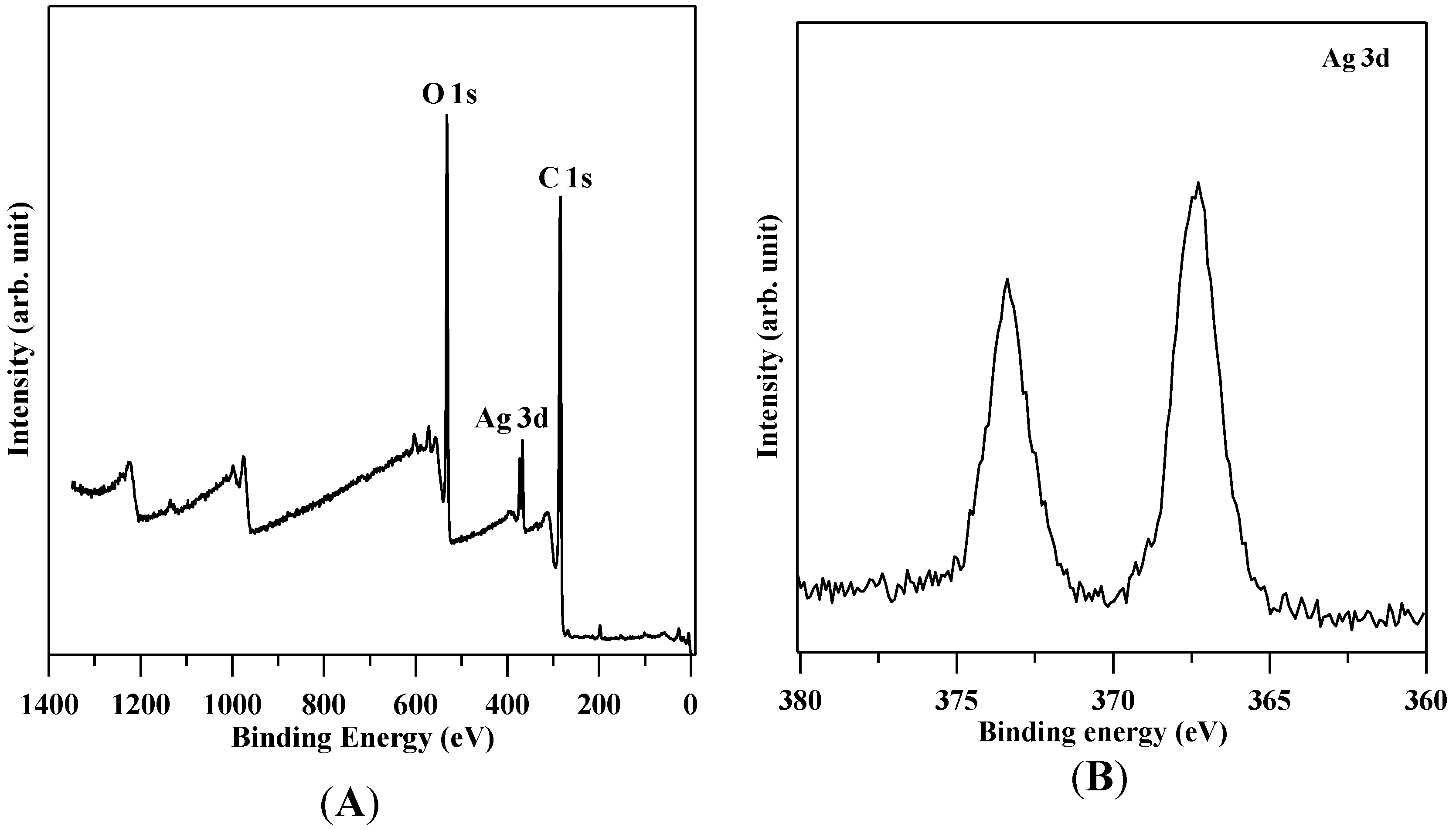
3.1. Characterization of GO and GO-Ag
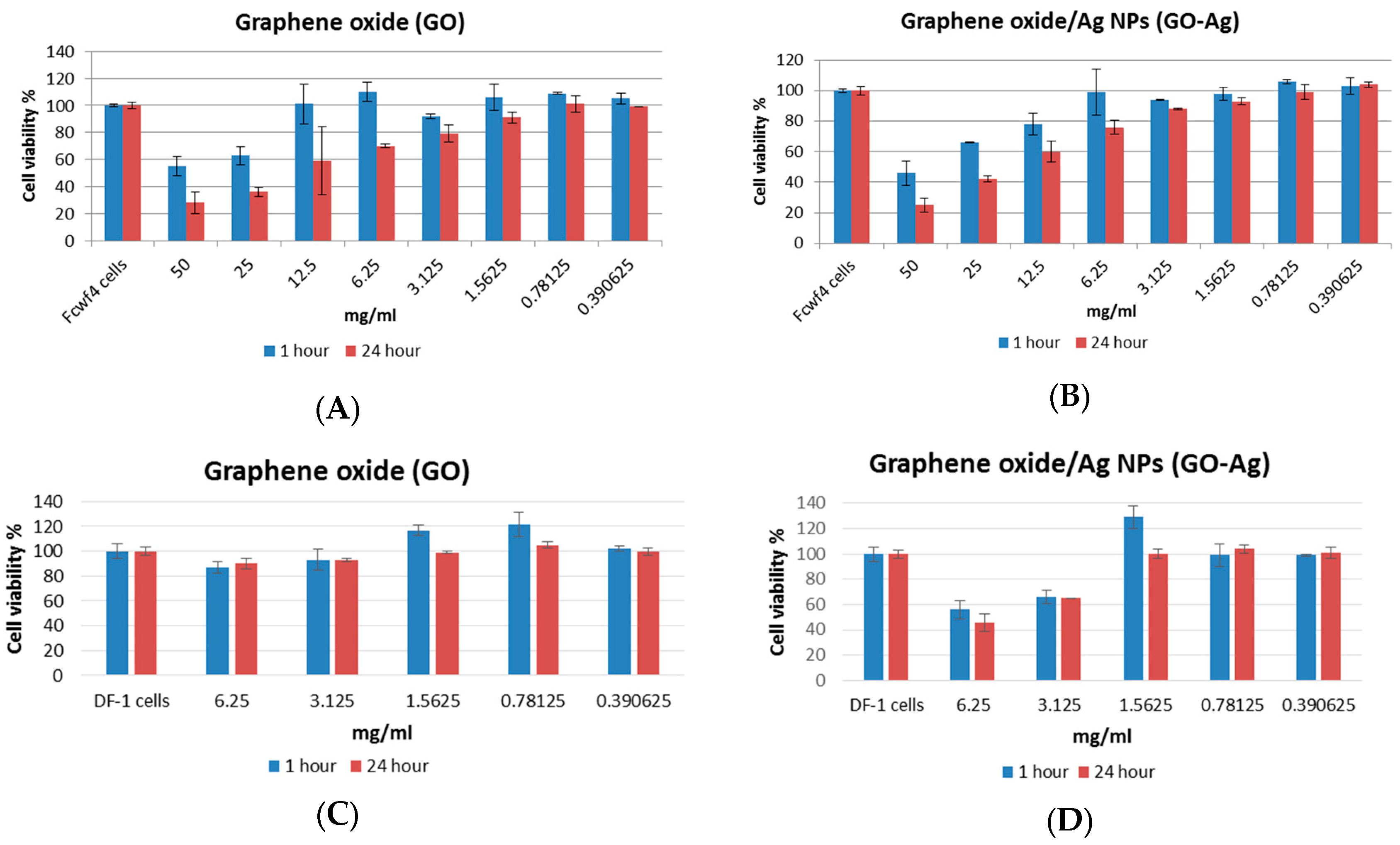
3.2. Cytotoxicity of GO and GO-Ag
3.3. Cytopathic Effect (CPE) of the Viruses
3.4. Virus Inhibition of GO and GO-Ag
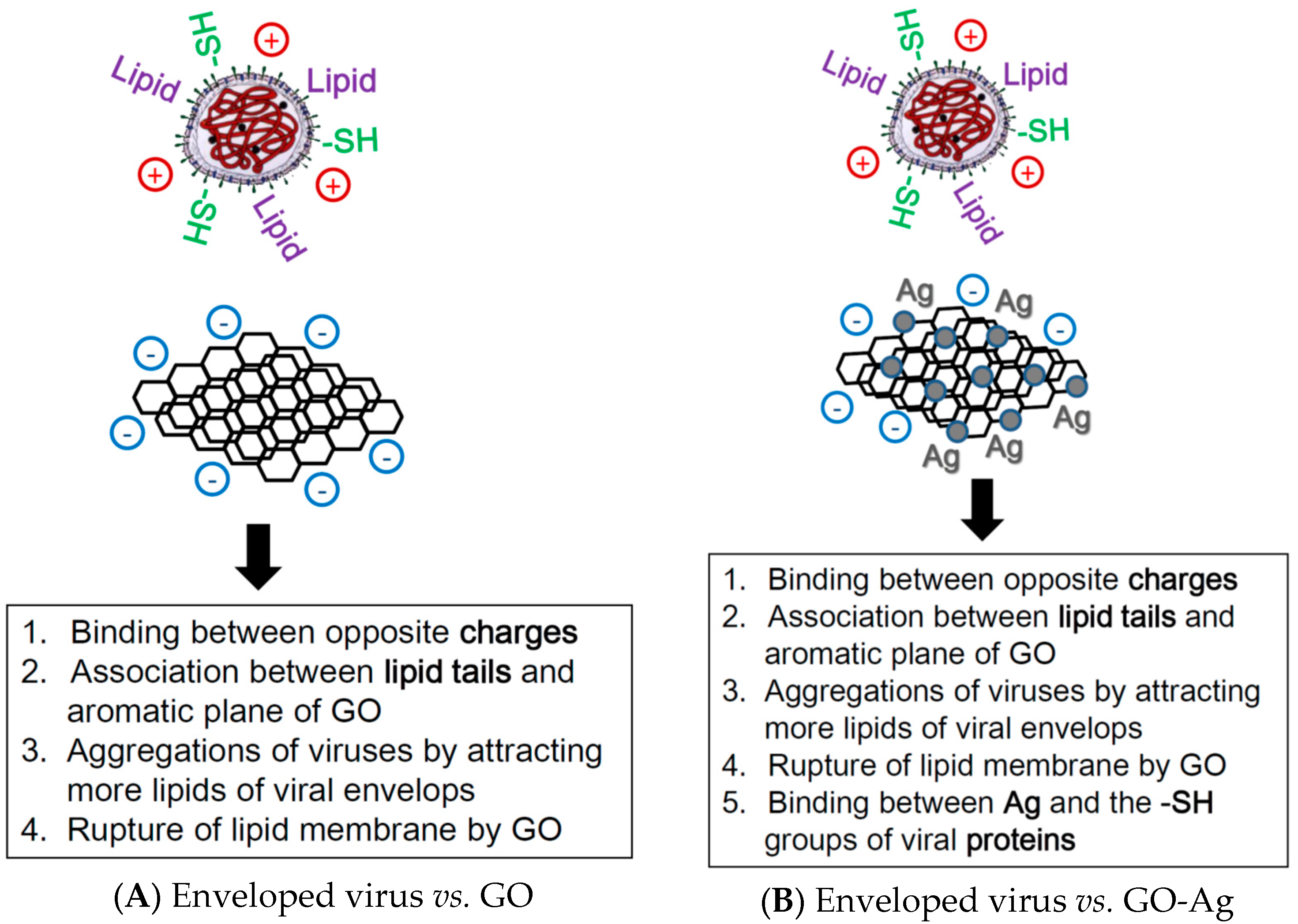
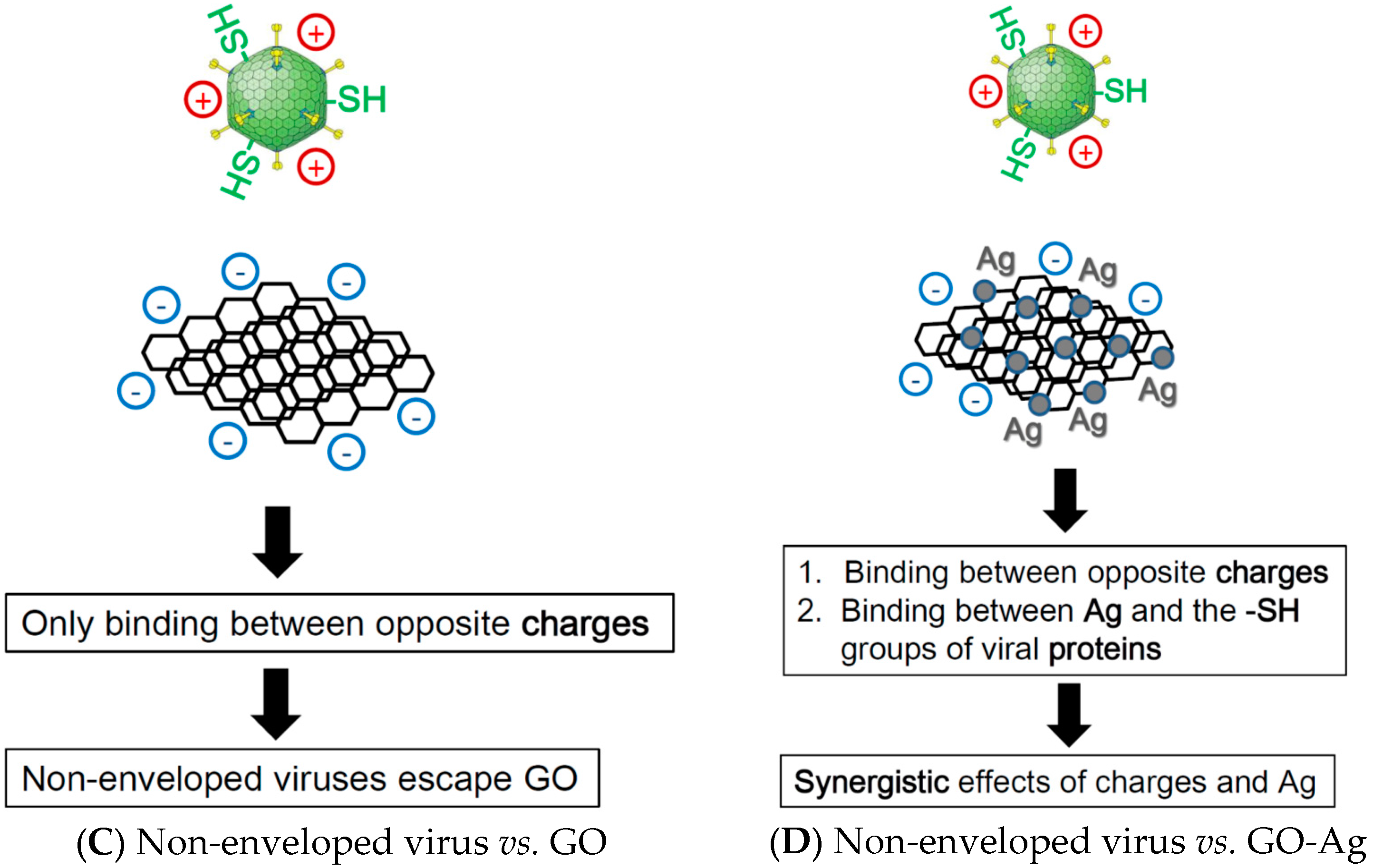
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Albrecht, M.A.; Evan, C.W.; Raston, C.L. Green chemistry and the health implications of nanoparticles. Green Chem. 2006, 8, 417–432. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.H.; Park, S.J.; Lee, H.J. Antimicrobial effects of silver nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Gong, P.; Li, H.; He, X.; Wang, K.; Hu, J.; Tan, W. Preparation and antibacterial activity of Fe3O4@Ag nanoparticles. Nanotechnology 2007, 18, 604–611. [Google Scholar] [CrossRef]
- Elechiguerra, J.L.; Burt, J.L.; Morones, J.R.; Camacho-Bragado, A.; Gao, X.; Lara, H.H.; Yacaman, M.J. Interaction of silver nanoparticles with HIV-1. J. Nanobiotechnol. 2005, 3, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Ayala-Nunez, N.; Ixtepan-Turrent, L.; Rodriguez-Padilla, C. Mode of antiviral action of silver nanoparticles against HIV-1. J. Nanobiotechnol. 2010, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Ixtepan-Turrent, L.; Garza-Trevino, E.N.; Rodriguez-Padilla, C. PVP-coated silver nanoparticles block the transmission of cell-free and cell-associated HIV-1 in human cervical culture. J. Nanobiotechnol. 2010, 8, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Sun, R.W.; Chen, R.; Hui, C.K.; Ho, C.M.; Luk, J.M.; Lau, G.K.; Che, C.M. Silver nanoparticles inhibit hepatitis B virus replication. Antivir. Ther. 2008, 13, 253–262. [Google Scholar] [PubMed]
- Baram-Pinto, D.; Shukla, S.; Perkas, N.; Gedanken, A.; Sarid, R. Inhibition of herpes simplex virus type 1 infection by silver nanoparticles capped with mercaptoethane sulfonate. Bioconj. Chem. 2009, 20, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Singh, A.K.; Vig, K.; Pillai, S.R.; Singh, S.R. Silver nanoparticles inhibit replication of respiratory syncytial virus. J. Biomed. Nanotechnol. 2008, 4, 149–158. [Google Scholar]
- Rogers, J.V.; Parkinson, C.V.; Choi, Y.W.; Speshock, J.L.; Hussain, S.M. A preliminary assessment of silver nanoparticle inhibition of monkeypox virus plaque formation. Nanoscale Res. Lett. 2008, 3, 129–133. [Google Scholar] [CrossRef]
- Speshock, J.L.; Murdock, R.C.; Braydich-Stolle, L.K.; Schrand, A.M.; Hussain, S.M. Interaction of silver nanoparticles with Tacaribe virus. J. Nanobiotechnol. 2010, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Mehrbod, P.; Motamed, N.; Tabatabaian, M.; Soleimani, E.R.; Amini, E.; Shahidi, M.; Kheiri, M.T. In vitro antiviral effect of “Nanosilver” on influenza virus. DARU 2009, 17, 88–93. [Google Scholar]
- Mori, Y.; Ono, T.; Miyahira, Y.; Nguyen, V.Q.; Matsui, T.; Ishihara, M. Antiviral activity of silver nanoparticles/chitosan composites against H1N1 influenza A virus. Nanoscale Res. Lett. 2013, 8, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Xiang, D.; Chen, Q.; Pang, L.; Zheng, C.-L. Inhibitory effects of silver nanoparticles on H1N1 influenza A virus in vitro. J. Virol. Methods 2011, 178, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.N.R.; Sood, A.K.; Voggu, R.; Subrahmanyam, K.S. Some novel attributes of graphene. J. Phys. Chem. Lett. 2010, 1, 572–580. [Google Scholar] [CrossRef]
- Faria, A.F.; Martinez, D.S.T.; Meira, S.M.M.; Moraes, A.C.M.; Brandelli, A.; Filho, A.G.S.; Alves, O.L. Anti-adhesion and antibacterial activity of silver nanoparticles supported on graphene oxide sheets. Colloid Surf. B Biointerfaces 2014, 113, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Han, J.W.; Dayem, A.A.; Eppakayala, V.; Kim, J.-H. Oxidative stress-mediated antibacterial activity of graphene oxide and reduced graphene oxide in Pseudomonas aeruginosa. Int. J. Nanomed. 2012, 7, 5901–5914. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hu, M.; Zeng, T.H.; Wu, R.; Jiang, R.; Wei, J.; Wang, L.; Kong, J.; Chen, Y. Lateral dimension-dependent antibacterial activity of graphene oxide sheets. Langmuir 2012, 28, 12364–12372. [Google Scholar] [CrossRef] [PubMed]
- Sametband, M.; Kalt, I.; Gedanken, A.; Sarid, R. Herpes simplex virus type-1 attachment inhibition by functionalized graphene oxide. ACS Appl. Mater. Interfaces 2014, 6, 1228–1235. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Shao, K.; Li, Z.; Guo, N.; Zuo, Y.; Li, Q.; Lu, Z.; Chen, L.; He, Q.; Han, H. Antiviral activity of graphene oxide: How sharp edged structure and charge matter. ACS Appl. Mater. Interfaces 2015, 7, 21571–21579. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.C.; Black, J.W.; Boyle, J.F.; Evermann, J.F.; McKeirnan, A.J.; Ott, R.L. Pathogenic differences between various feline coronavirus isolates. Adv. Exp. Med. Biol. 1984, 173, 365–380. [Google Scholar] [PubMed]
- Rekha, K.; Sivasubramanian, C.; Chung, I.-M.; Thiruvengadam, M. Growth and replication of infectious bursal disease virus in the DF-1 cell line and chicken embryo fibroblasts. Biomed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Tang, J.; Chen, Q.; Xu, L.; Zhang, S.; Feng, L.; Cheng, L.; Xu, H.; Liu, Z.; Peng, R. Graphene oxide-silver nanocomposites as a highly effective antibacterial agent with species-specific mechanisms. ACS Appl. Mater. Interfaces 2013, 5, 3867–3874. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Leung, P.; Yao, L.; Song, Q.W.; Newton, E. Antimicrobial effect of surgical masks coated with nanoparticles. J. Hosp. Infect. 2006, 62, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Frost, R.; Jonsson, G.E.; Chakarov, D.; Svedhem, S.; Kasemo, B. Graphene oxide and lipid membranes: Interactions and nanocomposite structures. Nano Lett. 2012, 12, 3356–3362. [Google Scholar] [CrossRef] [PubMed]
- Rui, L.; Liu, J.; Li, J.; Weng, Y.; Dou, Y.; Yuan, B.; Yang, K.; Ma, Y. Reduced graphene oxide directed self-assembly of phospholipid monolayers in liquid and gel phases. Biochim. Biophys. Acta 2015, 1848, 1203–1211. [Google Scholar] [CrossRef] [PubMed]


| mg/mL * | TCID50/mL | Inhibition % | ||
|---|---|---|---|---|
| Exp 1 | FCoV | 106 | ||
| FCoV + GO | 100 | 105 | 17 | |
| 10 | 4.7 × 105 | 5.5 | ||
| 1 | 3.7 × 105 | 7.2 | ||
| FCoV + GO-Ag | 100 | 105 | 17 | |
| 10 | 1.6 × 105 | 13 | ||
| 1 | 3.2 × 105 | 8.4 | ||
| Exp 2 | FCoV | 4.7 × 104 | ||
| FCoV + GO | 0.1 | 8.1 × 103 | 16.3 | |
| FCoV + GO-Ag | 0.1 | 3.7 × 103 | 24.8 | |
| Exp 3 | IBDV | 9 × 105 | ||
| IBDV + GO | 1 | 9.5 × 105 | −0.4 | |
| IBDV + GO-Ag | 1 | 4 × 104 | 22.7 |
| Concentration (mg/mL) | |||||
|---|---|---|---|---|---|
| 1 | 0.5 | 0.25 | 0.125 | 0.0625 | |
| IBDV: 9 × 103 TCID50/mL | |||||
| IBDV + GO | 100% | 100% | 100% | 100% | N.A. |
| IBDV + GO-Ag | 75% | 87.5% | 100% | 100% | N.A. |
| IBDV: 9 × 102 TCID50/mL | |||||
| IBDV + GO | 100% | 100% | 100% | 100% | 100% |
| IBDV + GO-Ag | 0% | 0% | 0% | 0% | 37.5% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-N.; Hsueh, Y.-H.; Hsieh, C.-T.; Tzou, D.-Y.; Chang, P.-L. Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses. Int. J. Environ. Res. Public Health 2016, 13, 430. https://doi.org/10.3390/ijerph13040430
Chen Y-N, Hsueh Y-H, Hsieh C-T, Tzou D-Y, Chang P-L. Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses. International Journal of Environmental Research and Public Health. 2016; 13(4):430. https://doi.org/10.3390/ijerph13040430
Chicago/Turabian StyleChen, Yi-Ning, Yi-Huang Hsueh, Chien-Te Hsieh, Dong-Ying Tzou, and Pai-Ling Chang. 2016. "Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses" International Journal of Environmental Research and Public Health 13, no. 4: 430. https://doi.org/10.3390/ijerph13040430
APA StyleChen, Y.-N., Hsueh, Y.-H., Hsieh, C.-T., Tzou, D.-Y., & Chang, P.-L. (2016). Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses. International Journal of Environmental Research and Public Health, 13(4), 430. https://doi.org/10.3390/ijerph13040430







