Characterization and Feasibility Assessment of Recycled Paper Mill Sludges for Land Application in Relation to the Environment
Abstract
:1. Introduction
2. Experimental Section
2.1. Recycled Paper Mill Sludge Sampling
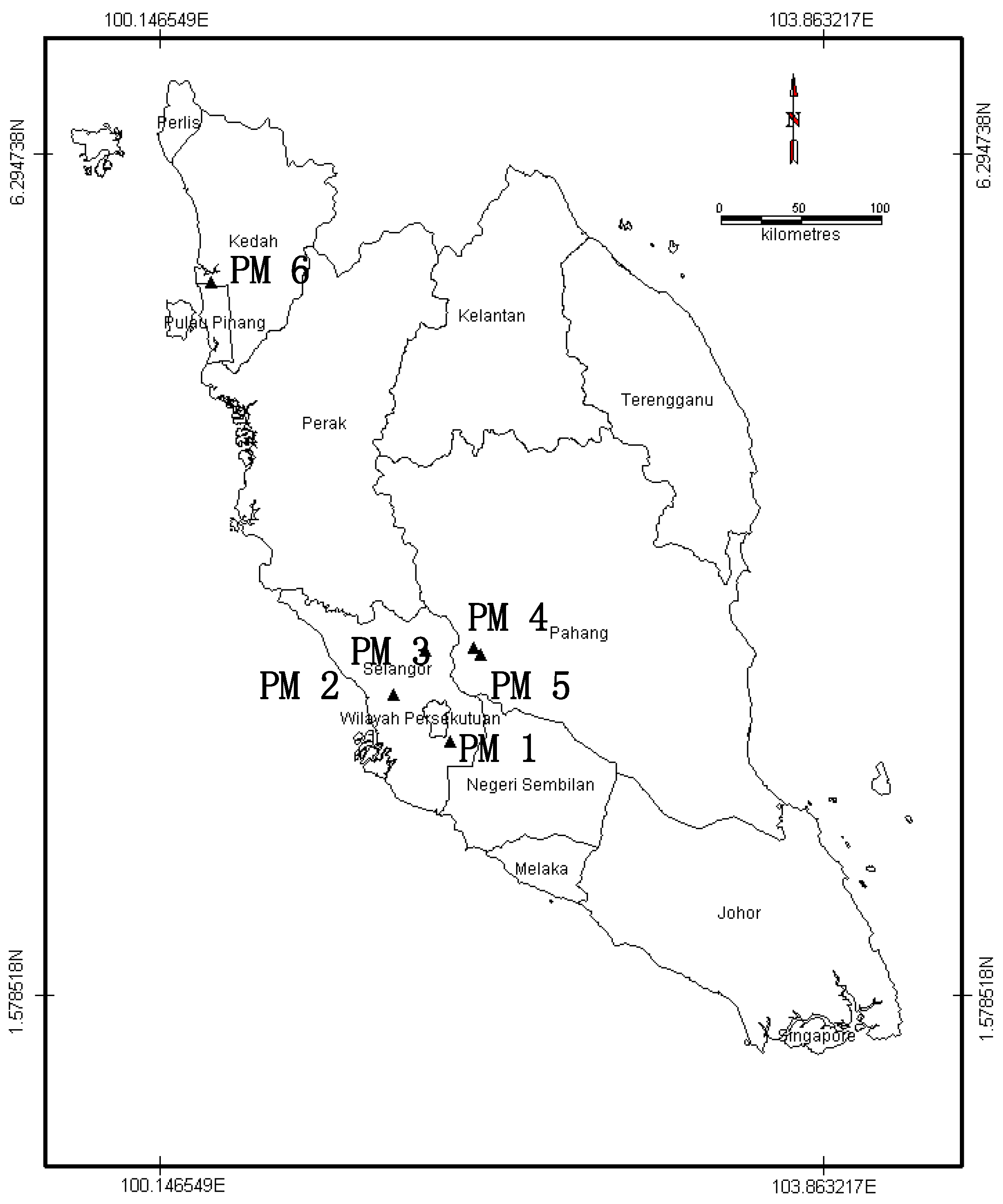
2.2. Physical and Chemical Characteristics of Paper Mill Sludges
2.3. Determination of Total Heavy Metals in Recycled Paper Mill Sludge
2.4. Analytical Procedure for Polycyclic Aromatic Hydrocarbons
2.5. Analytical Procedure for Dioxin/Furan
3. Results and Discussion
3.1. Physico-Chemical Characteristics of Paper Mill Sludges

| No. | Parameters | * PM 1 | PM 2 | PM 3 | PM 4 | PM 5 | PM 6 | Min. | Max. | Mean |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Moisture (%) | 69.32 | 78.32 | 59.08 | 68.68 | 69.34 | 45.78 | 45.78 | 78.32 | 65.08 |
| 2. | pH | 6.45 | 7.84 | 7.32 | 7.54 | 6.33 | 7.08 | 6.33 | 7.84 | 7.09 |
| 3. | EC, mS/cm | 1.12 | 3.08 | 0.51 | 0.61 | 1.97 | 0.56 | 0.51 | 3.08 | 1.30 |
| 4. | Nitrogen, % | 1.29 | 4.05 | 1.51 | 0.31 | 1.32 | 0.49 | 0.31 | 4.05 | 1.49 |
| 5. | Carbon, % | 24.39 | 33.67 | 18.92 | 23.40 | 31.36 | 21.92 | 18.92 | 33.67 | 25.61 |
| 6. | C/N ratio | 18.91 | 8.31 | 12.52 | 75.48 | 23.75 | 44.73 | 8.31 | 75.48 | 30.61 |
| 7. | Organic matter, % | 41.95 | 57.91 | 32.54 | 40.24 | 53.93 | 37.70 | 32.54 | 57.91 | 44.04 |
| 8. | CEC, cmol(+) kg−1 | 25.00 | 28.07 | 4.39 | 3.33 | 18.66 | 7.11 | 3.33 | 28.07 | 14.43 |
| 9. | Phosphorous, % | 0.07 | 0.78 | 0.02 | 0.12 | 0.09 | 0.02 | 0.02 | 0.78 | 0.18 |
| 10. | Potassium, % | 0.13 | 0.42 | 0.02 | 0.05 | 0.07 | 0.06 | 0.02 | 0.42 | 0.12 |
| 11. | Calcium, % | 0.54 | 0.53 | 0.54 | 1.28 | 0.74 | 0.36 | 0.36 | 1.28 | 0.66 |
| 12. | Magnesium, % | 0.73 | 0.45 | 0.42 | 0.41 | 1.06 | 0.57 | 0.41 | 1.06 | 0.61 |
| 13. | Sodium, % | 0.69 | 0.78 | 0.58 | 0.64 | 0.45 | 0.88 | 0.64 | 0.88 | 0.67 |
| 14. | Aluminum, % | 1.65 | 2.76 | 1.09 | 1.39 | 1.73 | 1.45 | 1.09 | 2.76 | 1.68 |
| No. | Parameters | * PM 1 | PM 2 | PM 3 | PM 4 | PM 5 | PM 6 | Min. | Max. | Mean | Zarcinas, (2004) ** | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| # Class 1 | # Class 2 | # Class 3 | ||||||||||||
| 1. | Cadmium, mg·kg−1 | 2.01 | 4.09 | 2.07 | 2.47 | 1.3 | 1.38 | 1.3 | 4.09 | 2.34 | 3 | 20 | 20 | 0.3 |
| 2. | Chromium, mg·kg−1 | 18.92 | 37.01 | 7.44 | 17.36 | 26.56 | 12.92 | 7.44 | 37.01 | 20.58 | 100 | 1060 | 1060 | 60 |
| 3. | Copper, mg·kg−1 | 199 | 102 | 119 | 102 | 156 | 83 | 83 | 199 | 130.38 | 100 | 757 | 757 | 50 |
| 4. | Manganese, mg·kg−1 | 241 | 329 | 85 | 343 | 102 | 103 | 85 | 329 | 203.88 | - | - | - | - |
| 5. | Lead, mg·kg−1 | 81 | 328 | 43 | 61 | 73 | 55 | 43 | 328 | 126.5 | 150 | 500 | 500 | 65 |
| 6. | Zinc, mg·kg−1 | 358 | 287 | 257 | 277 | 365 | 351 | 257 | 365 | 314.63 | 500 | 1850 | 1850 | 95 |
| 7. | Nickel, mg·kg−1 | 41.6 | 29.11 | 13.20 | 10.14 | 15.96 | 10.78 | 10.14 | 4.16 | 21.56 | 62 | 180 | 180 | 45 |
| 8. | Iron, % | 0.41 | 0.38 | 0.19 | 0.42 | 0.37 | 0.37 | 0.19 | 0.42 | 0.34 | - | - | - | - |
3.2. Heavy Metals Concentrations of Recycled Paper Mill Sludges
3.3. Organic Contaminants of Polycyclic Aromatic Hydrocarbons
3.4. Dioxins and Furans
| No. | * PAHs | # PM 1 | PM 2 | PM 3 | PM 4 | PM 5 | PM 6 | Class 1 | Class 2 | Class 3 |
|---|---|---|---|---|---|---|---|---|---|---|
| (µg·g−1) | ||||||||||
| 1. | Phenanthrene | 113.76 | 39.10 | 532.72 | n.d | 77.33 | n.d | 0.1 | 50 | 50 |
| 2. | Anthracene | 91.09 | 61.92 | 703.13 | n.d | 159.24 | n.d | 0.1 | 10 | 10 |
| 3. | 3-Methylphenanthrene | 158.79 | 18.55 | 265.65 | 256.47 | 244.21 | 211.84 | 0.1 | 10 | 10 |
| 4. | 2-Methylphenanthrene | 152.61 | 16.24 | 279.95 | 351.67 | 301.30 | 80.21 | 0.1 | 10 | 10 |
| 5. | 2-Methylanthracene | 26.80 | n.d | 53.40 | n.d | 59.55 | n.d | 0.1 | 10 | 10 |
| 6. | 9-Methylphenanthrene | 15.43 | n.d | 251.34 | 192.97 | 243.00 | n.d | 0.1 | 10 | 10 |
| 7. | 1-Methylphenanthrene | 101.79 | n.d | 193.56 | 137.95 | 202.51 | n.d | 0.1 | 10 | 10 |
| 8. | Fluoranthene | 168.95 | 13.98 | 86.54 | 4.32 | 84.33 | 40.25 | 0.1 | 10 | 10 |
| 9. | Pyrene | 278.38 | 14.68 | 165.03 | 23.33 | 146.77 | 58.27 | 0.1 | 100 | 100 |
| 10. | 1-Methylpyrene | 478.92 | 37.00 | 337.59 | 58.13 | 273.87 | 360.86 | 0.1 | 10 | 10 |
| 11. | Chrysene | 56.79 | n.d | 23.22 | 7.99 | 19.86 | 18.42 | 0.1 | 10 | 10 |
| 12. | Benzo(a)antharene | 138.98 | 16.57 | 38.32 | 11.65 | 46.79 | 46.87 | 0.1 | 10 | 10 |
| 13. | Benzo(k)fluoranthene | 0.79 | n.d | 0.68 | 0.87 | 1.41 | 0.88 | 0.1 | 10 | 10 |
| 14. | Benzo(e)acephenanthrylene | 1.05 | n.d | 0.64 | 0.67 | 1.63 | 1.01 | 0.1 | 10 | 10 |
| 15. | Benzo(e)pyrene | 0.79 | n.d | 0.99 | 1.51 | 1.06 | 1.30 | 0.1 | 10 | 10 |
| 16. | Benzo(a)pyrene | 2.62 | n.d | 0.95 | 0.68 | 2.62 | 3.15 | 0.1 | 10 | 10 |
| 17. | Dibenzo(a,h)antharacene | 5.92 | n.d | 19.71 | 2.98 | 23.60 | 23.27 | 0.1 | 10 | 10 |
| Total PAHs | 1793.45 | 218.04 | 3646.67 | 1051.20 | 1889.10 | 846.33 | ||||
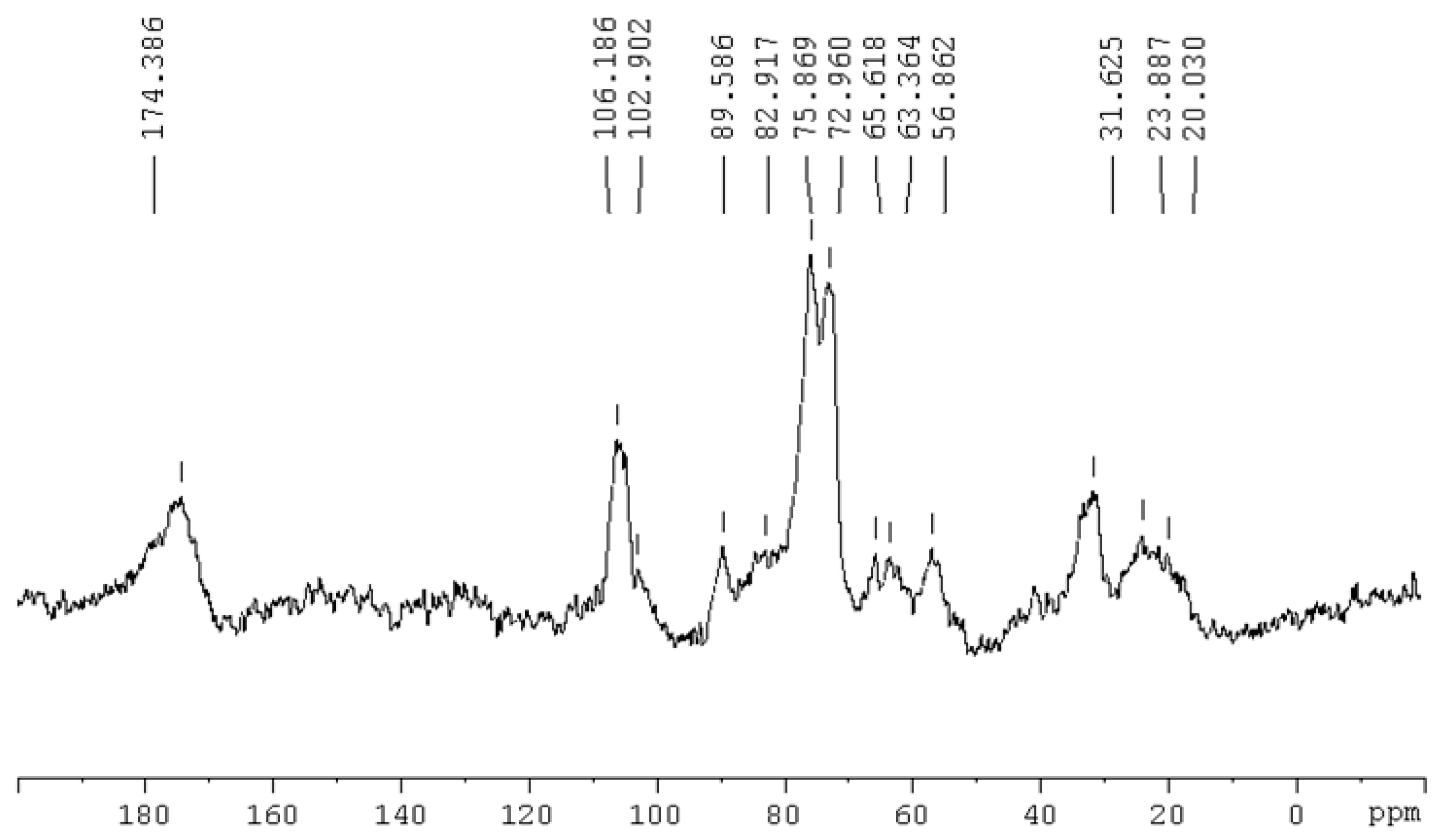
3.5. 13C-NMR Spectra of Paper Mill Sludge
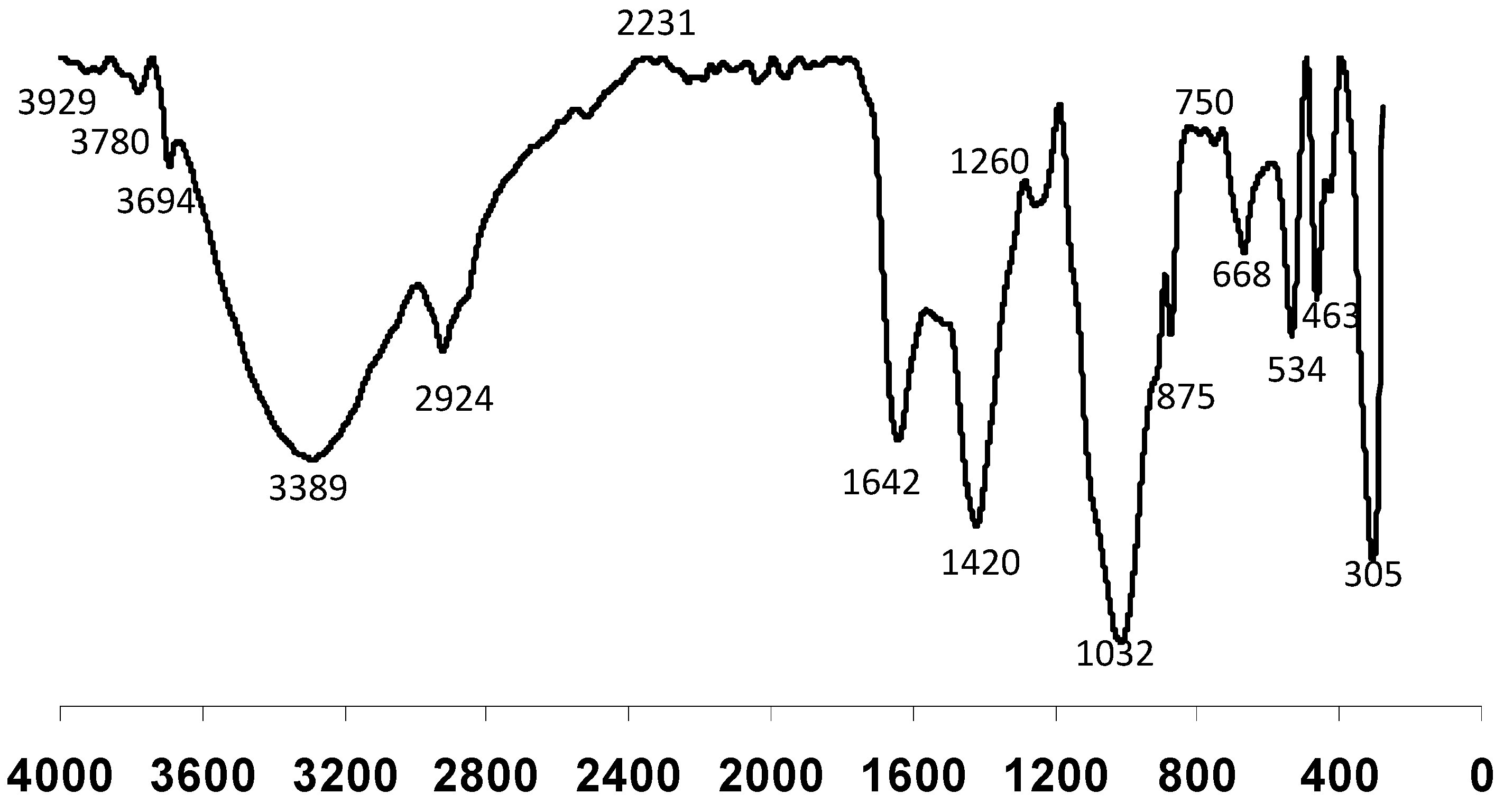
3.6. FTIR Spectra of Paper Mill Sludge
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Suriyanarayanan, S.; Mailappa, A.S.; Jayakumar, D.; Nanthakumar, K.; Karthikeyan, K.; Balasubramanian, S. Studies on the Characterization and Possibilities of Reutilization of Solid Wastes from a Waste Paper Based Paper Industry. Glob. J. Environ. Res. 2010, 4, 18–22. [Google Scholar]
- Xu, G.; Yang, X.; Spinosa, L. Development of sludge-based adsorbents: Preparation, characterization, utilization and its feasibility assessment. J. Environ. Manag. 2015, 151, 221–232. [Google Scholar]
- Nasir, M.H.; Kamil, M.Y.; Azmin, S.N.; Rakmi, A.R. Issues and problems of solid waste management. In National Review on Environmental Quality Management in Malaysia; Towards the Next Two Decades; Institute for Environment and Development, Universiti Kebangsaan Malaysia: Bangi, Malaysia, 1998; pp. 179–225. [Google Scholar]
- Hojamberdiev, M.; Kameshima, Y.; Nakajima, A.; Okada, K.; Kadirova, Z. Preparation and sorption properties of materials from paper sludge. J. Hazard. Mater. 2008, 151, 710–719. [Google Scholar]
- Feldkirchner, D.C.; Wang, C.; Gower, S.T.; Kruger, E.L.; Ferris, J. Effects of nutrient and paper mill biosolids amendments on the growth and nutrient status of hardwood forests. For. Ecol. Manag. 2003, 177, 95–116. [Google Scholar]
- Abdul Nasir, A.Z. Institutionalising Solid Waste Management in Malaysia; Department of National Solid Waste Management, Ministry of Housing and Local Government Malaysia: Kuala Lumpur, Malaysia, 2007.
- Wan Rasidah, K.; Mahmudin, S.; Wan Asma, I.; Rosazlin, A. Properties of sludge from paper mill processing recycle paper. In Proceedings of the 7th Pacific Rim Bio-Based Composites Symposium, Nanjing, China, 31 October–2 November; 2004; pp. 53–58. [Google Scholar]
- Merry, R.H.; Spouncer, L.S. The measurement of carbon in soils using a microprocessor-controlled resistance furnace. Comm. Soil Sci. Plant Anal. 1988, 19, 707–720. [Google Scholar]
- Bremner, J.M.; Mulvaney, C.S. Nitrogen-total. In Methods of Soil Analysis (Part 2). Chemical and Microbiological Properties-Agronomy, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Monograph No. 9; American Society of Agronomy: Madison, WI, USA, 1982; pp. 595–622. [Google Scholar]
- Thomas, G.W. Exchangeable cations. In Methods of Soil Analysis (Part 2). Chemical and Microbiological Properties-Agronomy, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Monograph No. 9; American Society of Agronomy: Madison, WI, USA, 1982; pp. 159–165. [Google Scholar]
- Zarcinas, B.A.; Cartwright, B.; Spouncer, L.R. Nitric acid digestion and multi-element analysis of plant material by inductively coupled plasma spectrometry. Comm. Soil Sci. Plant Anal. 1987, 18, 131–146. [Google Scholar] [CrossRef]
- Esakku, S.; Selvam, A.; Kurian, J.; Palanivelu, K. Assessment of heavy metal species in decomposed municipal solid waste. Chem. Spec. Bioavailab. 2005, 17, 95–102. [Google Scholar] [CrossRef]
- Zakaria, M.P.; Takada, H.; Tsutsumi, S.; Ohno, K.; Yamada, J.; Kouno, E.; Kumata, H. Distribution of Polycyclic Aromatic Hydrocarbons (PAHs) in rivers and estuaries in Malaysia: A Widespread Input of Petrogenic PAHs. Environ. Sci. Technol. 2002, 36, 1907–1918. [Google Scholar] [CrossRef] [PubMed]
- Barkey, A.V. Composition and uses of compos. In Agricultural Uses of By-Products and Wastes; ACS Symposium Series 668; Rechcig, J.E., MacKinnon, H.C., Eds.; ACS: Washington, DC, USA, 1997; pp. 141–162. [Google Scholar]
- Field, G.; Carroll, B.; Fasth, W. Beneficial use of primary sludges from a recycle/tmp newsprint mill—A case study. In Proceedings of TAPPI Environmental Confenrence; TAPPI Press: Atlanta, GA, USA, 1996; pp. 539–544. [Google Scholar]
- Feagley, S.E.; Valdez, M.S.; Hudnall, W.H. Bleached primary papermill sludge effect on bermudagrass grown on a mine soil. Soil Sci. 1994a, 157, 389–397. [Google Scholar] [CrossRef]
- Campbell, A.G.; Zhang, X.; Tripepi, R.R. Composting and evaluating a pulp and paper sludge for use as a soil amendment/mulch. Comp. Sci. Utilization. 1995, 3, 84–95. [Google Scholar] [CrossRef]
- Cavaleri, M.A.; Gilmore, D.W.; Mozaffari, M.; Rosen, C.J.; Halbach, T.R. Hybrid poplar and forest soil response to municipal and industrial by-products: A greenhouse study. J. Environ. Qual. 2004, 33, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Fierro, A.; Angers, A.A.; Beauchamp, C.J. Restoration of ecosystem function in an abandoned sand pit: Plant and soil response to paper de-inking sludge. J. Appl. Ecol. 1999, 36, 244–253. [Google Scholar] [CrossRef]
- Bellamy, K.L.; Chong, C.; Cline, R.A. Paper sludge utilization in agriculture and container nursery culture. J. Environ. Qual. 1995, 24, 1074–1082. [Google Scholar] [CrossRef]
- Camberato, J.J.; Vance, E.D.; Someshwar, A.V. Composition and land application of paper manufacturing residuals. In Agricultural Uses of By-Products and Wastes; Rechcig, J.E., MacKinnon, H.C., Eds.; ACS: Washington, DC, USA, 1997. [Google Scholar]
- Mezenner, N.Y.; Bensmaili, A. Kinetics and thermodynamic study of phosphate adsorption on iron hydroxide-eggshell waste. Chem. Eng. J. 2009, 147, 87–96. [Google Scholar] [CrossRef]
- NCASI (National Council of the Paper Industry for Air and Stream Improvement). The Land Application and Related Utilization of Pulp and Papermill Sludges; Technical Bulletin. 439; NCASI: New York, NY, USA, 1984. [Google Scholar]
- Feagley, S.E.; Valdez, M.S.; Hudnall, W.H. Papermill sludge, phosphorus, potassium, and lime effect on clover grown on a mine soil. J. Environ. Qual. 1994b, 23, 759–765. [Google Scholar] [CrossRef]
- Organix Waste Solutions Inc. Final Report—Pulp and Paper Mill Residual Chemistry and Options for Regulations; Prepared for the British Columbia Pulp and Paper Association; Organix Waste Solutions Inc.: Vancouver, BC, Canada, 2000; p. 20. [Google Scholar]
- Zarcinas, B.A.; Che Fauziah, I.; McLaughlin, M.J.; Cozens, G. Heavy metals in soils and crop in Southeast Asia. Environ. Geochem. Health. 2004, 26, 343–357. [Google Scholar] [CrossRef] [PubMed]
- Pichtel, J.R.; Dick, W.A.; Sutton, P. Comparison of amendments and management practices for long-term reclamation of abandoned mine lands. J. Environ. Qual. 1994, 23, 766–772. [Google Scholar] [CrossRef]
- Howe, J.; Wagner, M. The effect of papermill wastewater and organic amendments on sodium accumulation by potted cottonwoods. Environ. Poll. 1996, 92, 113–118. [Google Scholar] [CrossRef]
- Shepard, R.K. Sludge and ash. In Maine Agricultural and Forest Experiment Station Miscellaneous Report 397; University of Maine: Orono, ME, USA, 1996. [Google Scholar]
- Thacker, W.E. Silvicultural land application of wastewater and sludge from the pulp and paper industry. In The forest Alternative for Treatment and Utilization of Municipal and Industrial Wastes; Cole, D.W., Henry, C.L., Nutter, W.L., Eds.; University of Washington Press: Seattle, WA, USA, 1986; pp. 41–54. [Google Scholar]
- Beauchamp, C.J.; Charest, M.H.; Gosselin, A. Examination of environmental quality of raw and composting de-inking paper sludge. Chemosphere 2002, 46, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Murray, H.H. Clay. In Pigments for Paper; Hagemeyer, R.W., Ed.; TAPPI Press: Atlanta, GA, USA, 1984; pp. 95–141. [Google Scholar]
- Kabata-Pendias, A.; Pendias, H. Trace Elements in Soils and Plants, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Watson, M.E.; Hoitink, H.A.J. Utilizing papermill sudge: Longterm effects in stripmine reclamation. BioCycle. 1985, volume, 52–53. [Google Scholar]
- Cabral, F.; Vasconcelos, E. Agricultural use of combined primary/secondary pulpmill sludge. Agrochimica. 1993, 37, 409–417. [Google Scholar]
- Li, D.; Wu, Y.; Feng, L.; Zhang, L. Surface properties of SAC and its adsorption mechanisms for phenol and nitrobenzene. Bioresour. Technol. 2012, 113, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zhang, W.; Yang, Y.; Huang, X.; Wang, S.; Qiu, R. Relative distribution of Pb2þ sorption mechanisms by sludge-derived biochar. Water Res. 2012, 46, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Webber, M.D.; Goodin, J.D. Studies on the fate of organic contaminants in sludge treated soils. In Effects of Organic Contaminants in Sewage Sludge on Soil Fertility, Plants and Animals; Hall, J.E., Sauerbeck, D.R., L’Hermite, P., Eds.; Commission of the European Communities: Luxembourg, Luxembourg, 1992; pp. 54–69. [Google Scholar]
- Maine Department of Environmental Protection. Chapter 567. In Rules for Land Application of Sludge and Residuals; Maine Department of Environmental Protection: Augusta, ME, USA, 1986. [Google Scholar]
- Diamantis, V.; Erguder, T.H.; Aivasidis, A.; Verstraete, W.; Voudrias, E. Wastewater disposal to landfill-sites: A synergistic solution for centralized management of olive mill wastewater and enhanced production of landfill gas. J. Environ. Manag. 2013, 128, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.J.; Line, M.A. Organic composition of a pulp and paper mill sludge determined by FTIR, 13C CP MAS NMR ad chemical extraction techniques. J. Agri. Food Chem. 1997, 45, 2354–2358. [Google Scholar] [CrossRef]
- Sierra, M.M.D.; Giovanela, M.; Parlanti, E.; Esteves, V.I.; Duarte, A.C.; Fransozo, A.; Soriano-Sierra, E.J. Structural description of humic substances from subtropical coastal environments using elemental analysis, FT-IR and 13C-Solid state NMR data. J. Coastal Res. 2005, 42, 370–382. [Google Scholar]
- Schmidth, M.W.I.; Knicker, H.; Hatcher, P.G.; Kogel Knabner, I. Improvement of 13C and 15N NMR spectra of bulk soil, particle size fraction and organic material by treatment with hydrofluoric acid (10%). Eur. J. Soil Sci. 1997, 48, 319–328. [Google Scholar] [CrossRef]
- Amir, S.; Hafidi, M.; Merlina, G.; Hamdi, H.; Revel, J.C. Elemental analysis, FTIR and 13C-NMR of humic acids from sewage sludge composting. Agronomic. 2004, 24, 13–18. [Google Scholar] [CrossRef]
- Giovanella, M.; Parlanti, E.; Soriano-sierra, J.; Soldi, M.S.; Sierra, M.D. Elemental compositions, FT-IR spectra thermal behavior of sedimentary fulvic and humic acids from aquatic and terrestrial environments. Geochem. J. 2004, 38, 255–264. [Google Scholar] [CrossRef]
- Matias, M.C.; Orden, M.U.; Sanchez, S.G.; Urreaga, J.M. Comparative spectroscopic study of the modification of cellulose materials with different coupling agents. J. Appl. Pol. Sci. 2000, 75, 256–266. [Google Scholar] [CrossRef]
- Reig, F.B.; Adelantado, J.V.G.; Moreno, M.C.M.M. FTIR quantitative analysis of calcium carbonate (calcite) and silica (quartz) mixtures using the constant ratio method. Application to geological samples. Talanta. 2002, 58, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Muruganantham, S.; Anbalagan, G.; Ramamurthy, N. FT-IR and SEM-EDS comparative analysis of medicinal plants, Eclipta Alba Hassk and Eclipta Prostrata Linn. Romanian J. Biophys. 2009, 19, 285–294. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah, R.; Ishak, C.F.; Kadir, W.R.; Bakar, R.A. Characterization and Feasibility Assessment of Recycled Paper Mill Sludges for Land Application in Relation to the Environment. Int. J. Environ. Res. Public Health 2015, 12, 9314-9329. https://doi.org/10.3390/ijerph120809314
Abdullah R, Ishak CF, Kadir WR, Bakar RA. Characterization and Feasibility Assessment of Recycled Paper Mill Sludges for Land Application in Relation to the Environment. International Journal of Environmental Research and Public Health. 2015; 12(8):9314-9329. https://doi.org/10.3390/ijerph120809314
Chicago/Turabian StyleAbdullah, Rosazlin, Che Fauziah Ishak, Wan Rasidah Kadir, and Rosenani Abu Bakar. 2015. "Characterization and Feasibility Assessment of Recycled Paper Mill Sludges for Land Application in Relation to the Environment" International Journal of Environmental Research and Public Health 12, no. 8: 9314-9329. https://doi.org/10.3390/ijerph120809314
APA StyleAbdullah, R., Ishak, C. F., Kadir, W. R., & Bakar, R. A. (2015). Characterization and Feasibility Assessment of Recycled Paper Mill Sludges for Land Application in Relation to the Environment. International Journal of Environmental Research and Public Health, 12(8), 9314-9329. https://doi.org/10.3390/ijerph120809314





