Cr(VI) Sorption/Desorption on Pine Sawdust and Oak Wood Ash
Abstract
:1. Introduction
2. Material and Methods
2.1. Characterization of pine sawdust and oak wood ash
| Parameter | Oak Ash | Pine Sawdust |
|---|---|---|
| pHwater | 11.19 | 4.91 |
| pHKCl | 10.57 | 4.56 |
| C (%) | 6.69 | 46.13 |
| N (%) | 0.10 | 0.03 |
| POlsen (mg·kg−1) | 958.9 | 11.47 |
| CaT (mg·kg−1) | 81,031 | 8087 |
| MgT (mg·kg−1) | 24,505 | 164.4 |
| NaT (mg·kg−1) | 8095 | 98.35 |
| KT (mg·kg−1) | 70,661 | 540.6 |
| AsT (mg·kg−1) | 4.00 | 0.39 |
| CdT (mg·kg−1) | 0.18 | 0.39 |
| CrT (mg·kg−1) | 66.67 | 5.19 |
| CuT (mg·kg−1) | 590.5 | 14.87 |
| NiT (mg·kg−1) | 51.51 | 0.00 |
| ZnT (mg·kg−1) | 728.9 | 50.82 |
| AlT (mg·kg−1) | 20.218 | 260.6 |
| FeT (mg·kg−1) | 41.425 | 234.2 |
| MnT (mg·kg−1) | 6778 | 97.18 |
| Alox (mg·kg−1) | 8722 | 122.5 |
| Feox (mg·kg−1) | 5239 | 15.62 |
| Cae (cmol·kg−1) | 9.81 | 5.39 |
| Mge (cmol·kg−1) | 8.49 | 1.37 |
| Nae (cmol·kg−1) | 20.53 | 0.66 |
| Ke (cmol·kg−1) | 152.44 | 1.55 |
| Ale (cmol·kg−1) | 0.00 | 0.05 |
2.2. Chromium Sorption and Desorption
3. Results and Discussion
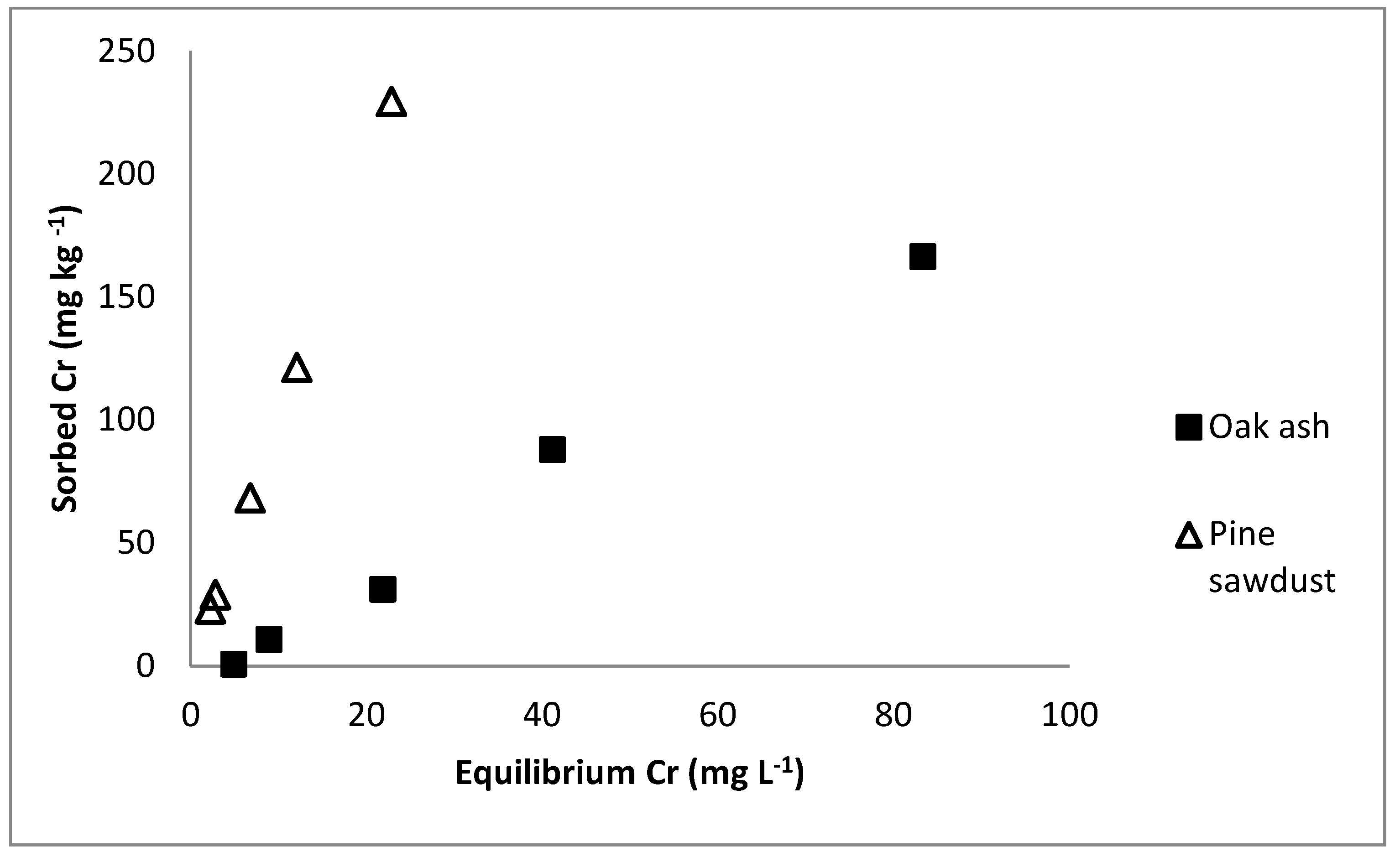
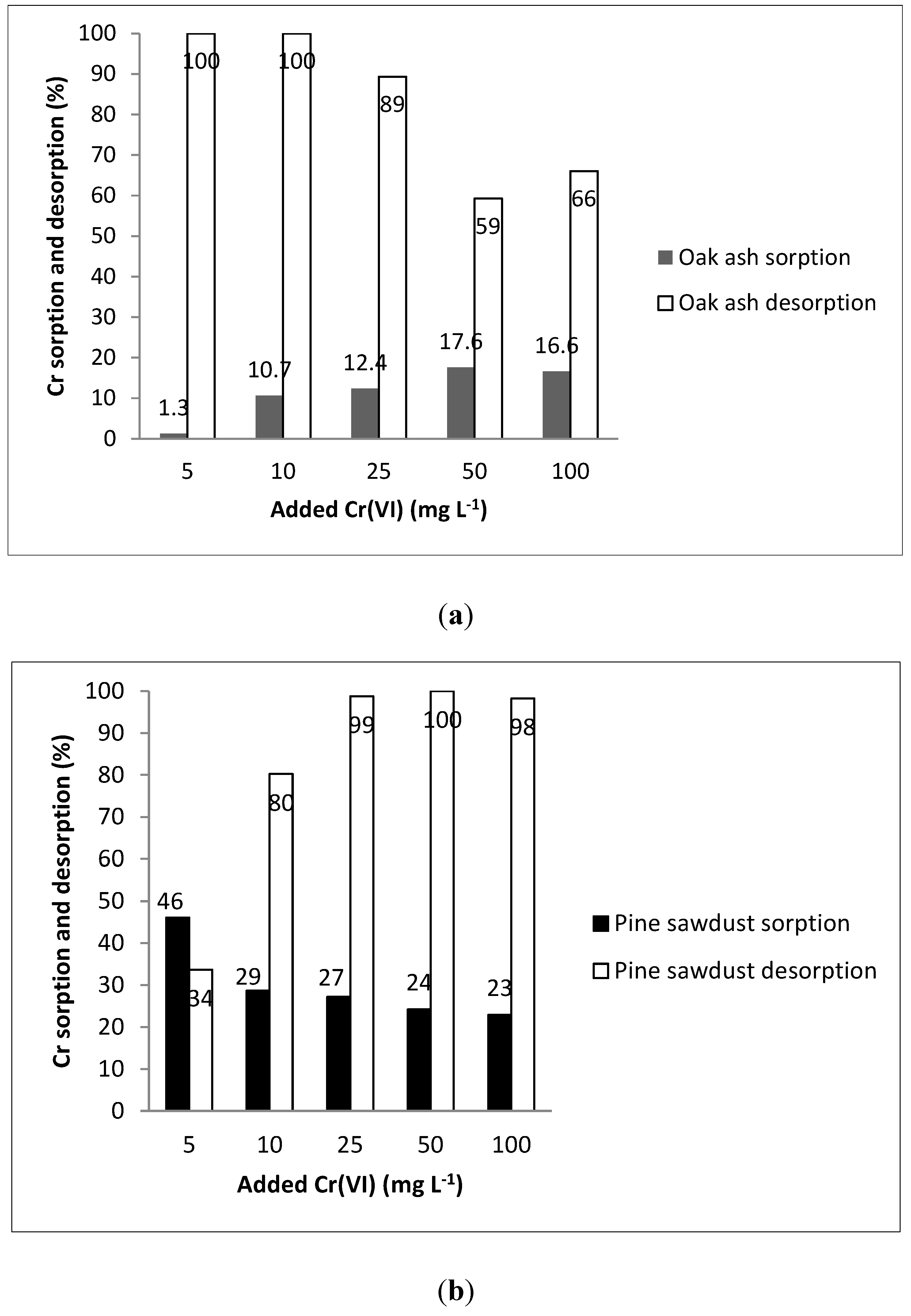
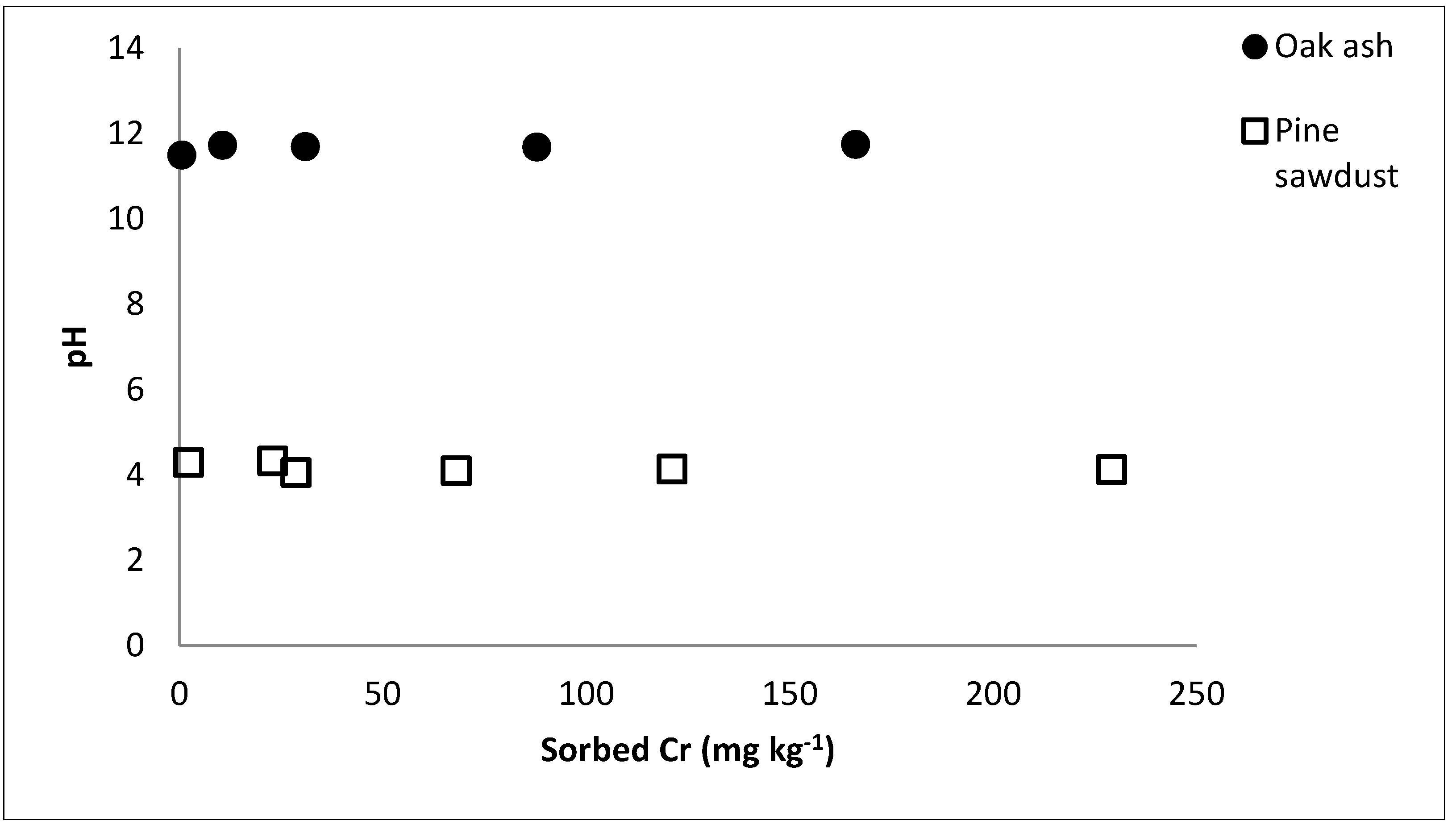
| Sorbent | Freundlich | Lineal | |||||
|---|---|---|---|---|---|---|---|
| KF (Ln g−1·mg(1−n)) | n | R2 | a (mg·kg−1) | Ka (L·kg−1) | R2 | ||
| Oak ash | 1.920 | 1.117 | 0.990 | −9.313 | 2.140 | 0.993 | |
| Pine sawdust | 3.141 | 0.842 | 0.997 | 10.148 | 2.873 | 0.996 | |
| Adsorbents | KF | n | Reference |
|---|---|---|---|
| Pine biomass | 38.38 | 2.86 | Ucun et al. [25] |
| Rhizopus nigrificans | 12.06 | 0.31 | Sudha-Bai and Abraham [40] |
| Rhizopus arrhizus | 10.99 | 5.55 | Prakasham et al. [41] |
| Anaerobic activated sludge | 3.62 | 0.79 | Aksu and Akpinar [42] |
| Synechocystis sp. | 1.54 | 0.71 | Cetinkaya-Donmez et al. [43] |
| Scenedesmus obliquus | 0.68 | 0.70 | Cetinkaya-Donmez et al. [43] |
| Chlorella vulgaris | 0.48 | 0.79 | Cetinkaya-Donmez et al. [43] |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Selvi, K.; Pattabhi, S.; Kadirvelu, K. Removal of Cr(VI) from aqueous solution by adsorption onto activated carbon. Bioresour. Technol. 2001, 80, 87–89. [Google Scholar] [CrossRef]
- Choppala, G.; Bolan, N.; Lamb, D.; Kunhikrishnan, A. Comparative sorption and mobility of Cr(III) and Cr(VI) species in a range of soils: Implications to bioavailability. Water Air Soil. Pollut. 2013, 224, 1699–1711. [Google Scholar] [CrossRef]
- Bielicka, A.; Bojanowska, I.; Wiśniewski, A. Two faces of chromium—Pollutant and bioelement. Pol. J. Environ. Stud. 2005, 14, 5–10. [Google Scholar]
- Fernández-Calviño, D.; Garrido-Rodríguez, B.; Cutillas-Barreiro, L.; Araújo-Nespereira, P.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Influence of mussel shell on As and Cr competitive and non-competitive sorption—Desorption kinetics in a mine soil: Stirred flow chamber experiments. Geoderma 2014, 232–234, 300–308. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Pendias, H. Trace Elements in Soils and Plants, 3rd ed.; CRC Press LLC: Boca Raton, FL, USA, 2001. [Google Scholar]
- Panda, S.K.; Choudhury, S. Chromium stress in plants. Braz. J. Plant. Physiol. 2005, 17, 95–102. [Google Scholar] [CrossRef]
- Fernández-Pazos, M.T.; Garrido-Rodriguez, B.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A.; Álvarez, E. Cr(VI) adsorption and desorption on soils and biosorbents. Water Air Soil Pollut. 2013, 224. [Google Scholar] [CrossRef]
- WHO. Chromium in drinking-water. In Background Document for Preparation of WHO Guidelines for Drinking-Water Quality; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Gode, F.; Pehlivan, E. Removal of Cr(VI) from aqueous solution by two Lewatit-anion exchange resins. J. Hazard. Mater. 2005, 119, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.W.; Ku, Y.; Bhattacharyya, D. Evaluation of recent treatment techniques for removal of heavy metals from industrial wastewater. AICHE Symp. Ser. 1985, 812, 165–203. [Google Scholar]
- Demiral, H.; Demiral, I.; Tumsek, F.; Karabacakoglu, B. Adsorption of chromium(VI) from aqueous solution by activated carbon derived from olive bagasse and applicability of different adsorption models. Chem. Eng. J. 2008, 144, 188–196. [Google Scholar] [CrossRef]
- Cimino, G.; Passerini, A.; Toscano, G. Removal of toxic cations and Cr(VI) from aqueous solution by halzelnut shell. Water Res. 2000, 34, 2955–2962. [Google Scholar] [CrossRef]
- Faghihian, H.; Bowman, R.S. Adsorption of Chromate by clinoptilolite exchanged with various metal cations. Water Res. 2005, 39, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Rastogi, A.; Nayak, A. Adsorption studies on the removal of hexavalent chromium from aqueous solution using a low cost fertilizer industry waste material. J. Colloid. Interface Sci. 2010, 34, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Ali, I.; Saleh, T.A.; Siddiqui, M.N.; Agarwal, S. Chromium removal from water by activated carbon developed from waste rubber tires. Environ. Sci. Pollut. Res. 2013, 20, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Khezami, L.; Capart, R. Removal of chromium(VI) from aqueous solution by activated carbons: Kinetic and equilibrium studies. J. Hazard. Mater. 2005, 123, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Mor, S.; Ravindra, K.; Bishnoi, N.R. Adsorption of chromium from aqueous solution by activated Alumina and activated Charcoal. Bioresour. Technol. 2007, 98, 954–957. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Marín, A.B.; Meseguer-Zapata, V.; Ortuño, J.F.; Aguilar, M.; Sáez, J.; Lloréns, M. Removal of cadmium from aqueous solutions by adsorption onto orange waste. J. Hazard. Mater. 2007, 139, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Dakiky, M.; Khamis, M.; Manassra, A.; Mer’eb, M. Selective adsorption of chromium(VI) in industrial wastewater using low-cost abundantly available adsorbents. Adv. Environ. Res. 2002, 6, 533–540. [Google Scholar] [CrossRef]
- Park, D.; Lim, S.R.; Yun, Y.S.; Park, J.M. Reliable evidences that the removal mechanism of hexavalent chromium by natural biomaterials is adsorption-coupled reduction. Chemosphere. 2007, 70, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Miretzkya, P.; Fernandez-Cirelli, A. Cr(VI) and Cr(III) removal from aqueous solution by raw and modified lignocellulosic materials, a review. J. Hazard. Mater. 2010, 180, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Seco-Reigosa, N.; Bermúdez-Couso, A.; Garrido-Rodríguez, B.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. As(V) retention on soils and forest by-products and other waste materials. Environ. Sci. Pollut. Res. 2013, 20, 6574–6583. [Google Scholar] [CrossRef] [PubMed]
- Arnesen, A.K.M.; Krogstad, T. Sorption and desorption of fluoride in soil polluted from the aluminium smelter at Ardal in western Norway. Water Air Soil. Pollut. 1998, 103, 357–373. [Google Scholar] [CrossRef]
- Ucun, H.; Bayhan, Y.K.; Kaya, Y.; Cakici, A.; Algur, O.F. Biosorption of chromium (VI) form aqueous solution by cone biomass of Pinus sylvestris. Bioresour. Technol. 2002, 85, 155–158. [Google Scholar] [CrossRef]
- Vinodhini, V.; Nilanjana, D. Biowaste materials as sorbents to remove chromium (VI) from aqueous environment a comparative study. J. Agric. Biol. Sci. 2009, 4, 19–23. [Google Scholar]
- Giles, C.H.; MacEwan, T.H.; Nakhwa, S.N.; Smith, D. A system of classification of solution adsorption isotherms and its use in diagnosis of adsorption mechanisms and in measurement of specific surface area of solids. J. Chem. Soc. 1960, 111, 3973–3993. [Google Scholar] [CrossRef]
- Marin, J.; Ayele, J. Removal of some heavy metal cations from aqueous solutions by spruce sawdust. I. Study of the binding mechanism through batch experiments. Environ. Technol. 2002, 23, 1157–1171. [Google Scholar] [CrossRef] [PubMed]
- Marin, J.; Ayele, J. Removal of some heavy metal cations from aqueous solutions by spruce sawdust. II. Adsorptiondesorption through column experiments. Environ. Technol. 2003, 24, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Kalmykova, Y.; Strömwall, A.M.; Steenari, B.M. Alternative materials for adsorption of heavy metals and petroleum hydrocarbons from contaminated leachates. Environ. Technol. 2008, 29, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, T.A.; Chan, G.Y.S.; Lo, W.; Babel, S. Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals. Sci. Total Environ. 2006, 366, 409–426. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.A.; Jain, N.; Joshi, H.C.; Prasad, S. Agricultural and agro-processing wastes as low cost adsorbents for metal removal from wastewater. A review. J. Sci. Ind. Res. 2008, 67, 647–658. [Google Scholar]
- Dong, D.; Zhao, X.; Hua, X.; Liu, J.; Gao, M. Investigation of the potential mobility of Pb Cd and Cr(VI) from moderately contaminated farmland soil to groundwater in northeast China. J. Hazard. Mater. 2009, 162, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Boddu, V.M.; Abburi, K.; Talbott, J.L.; Smith, E.D. Removal of hexavalent chromium from wastewater using a new composite chitosan biosorbent. Environ. Sci. Technol. 2003, 37, 4449–4456. [Google Scholar] [CrossRef] [PubMed]
- Schmuhl, R.; Krieg, H.M.; Keizer, K. Adsorption of Cu (II) and Cr(VI) ions by chitosan kinetics and equilibrium studies. Water SA 2001, 27, 1–8. [Google Scholar] [CrossRef]
- Wang, X.S.; Li, Z.Z.; Tao, S.R. Removal of chromium (VI) from aqueous solution using walnut hull. J. Environ. Manag. 2009, 90, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Griffin, R.; Au, A.K.; Frost, R. Effect of pH on adsorption of chromium from landfill—Leachate by clay minerals. J. Environ. Sci. Health. 1977, 12, 431–449. [Google Scholar] [CrossRef]
- Bower, C.A.; Hatcher, J.T. Adsorption of fluoride by soils and minerals. Soil. Sci. 1967, 103, 151–154. [Google Scholar] [CrossRef]
- Gago, C.; Romar, A.; Fernández-Marcos, M.L.; Álvarez, E. Fluorine sorption by soils developed from various parent materials in Galicia (NW Spain). J. Colloid. Interface Sci. 2012, 374, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Sudha-Bai, R.; Abraham, T.E. Biosorption of Cr(VI) from aqueous solution by Rhizopus. nigrificans. Bioresour. Technol. 2001, 79, 73–81. [Google Scholar]
- Prakasham, R.S.; Merrie, J.S.; Sheela, R.; Saswathi, N.; Ramakrisha, S.V. Biosorption of chromium(VI) by free and immobilized Rhizopus arrhizus. Environ. Pollut. 1999, 104, 421–427. [Google Scholar] [CrossRef]
- Aksu, Z.; Akpinar, D. Competitive biosorption of phenol and chromium (VI) from binary mixtures onto dried anaerobic activated sludge. Biochem. Eng. J. 2001, 7, 183–193. [Google Scholar] [CrossRef]
- Cetinkaya-Donmez, G.; Aksu, Z.; Ozturk, A.; Kutsal, T. A comparative study on heavy metal biosorption characteristics of some algae. Process. Biochem. 1999, 34, 885–892. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Cutillas-Barreiro, L.; Nóvoa-Muñoz, J.; Arias-Estévez, M. Cr(VI) Sorption/Desorption on Pine Sawdust and Oak Wood Ash. Int. J. Environ. Res. Public Health 2015, 12, 8849-8860. https://doi.org/10.3390/ijerph120808849
Núñez-Delgado A, Fernández-Sanjurjo MJ, Álvarez-Rodríguez E, Cutillas-Barreiro L, Nóvoa-Muñoz J, Arias-Estévez M. Cr(VI) Sorption/Desorption on Pine Sawdust and Oak Wood Ash. International Journal of Environmental Research and Public Health. 2015; 12(8):8849-8860. https://doi.org/10.3390/ijerph120808849
Chicago/Turabian StyleNúñez-Delgado, Avelino, María José Fernández-Sanjurjo, Esperanza Álvarez-Rodríguez, Laura Cutillas-Barreiro, JuanCarlos Nóvoa-Muñoz, and Manuel Arias-Estévez. 2015. "Cr(VI) Sorption/Desorption on Pine Sawdust and Oak Wood Ash" International Journal of Environmental Research and Public Health 12, no. 8: 8849-8860. https://doi.org/10.3390/ijerph120808849
APA StyleNúñez-Delgado, A., Fernández-Sanjurjo, M. J., Álvarez-Rodríguez, E., Cutillas-Barreiro, L., Nóvoa-Muñoz, J., & Arias-Estévez, M. (2015). Cr(VI) Sorption/Desorption on Pine Sawdust and Oak Wood Ash. International Journal of Environmental Research and Public Health, 12(8), 8849-8860. https://doi.org/10.3390/ijerph120808849










