Prototype Early Warning Systems for Vector-Borne Diseases in Europe
Abstract
:1. Introduction
2. The European Environment and Epidemiology (E3) Network
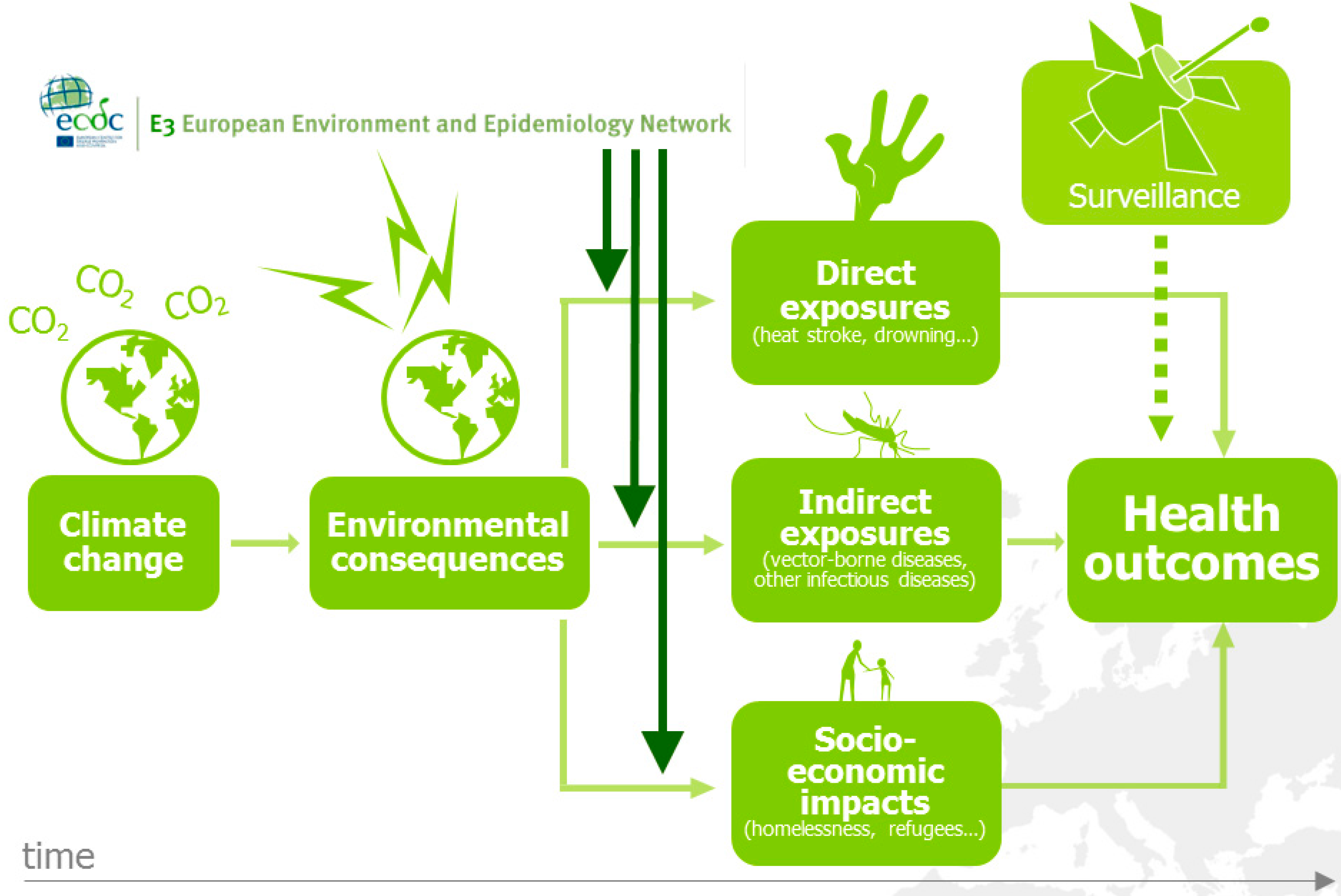
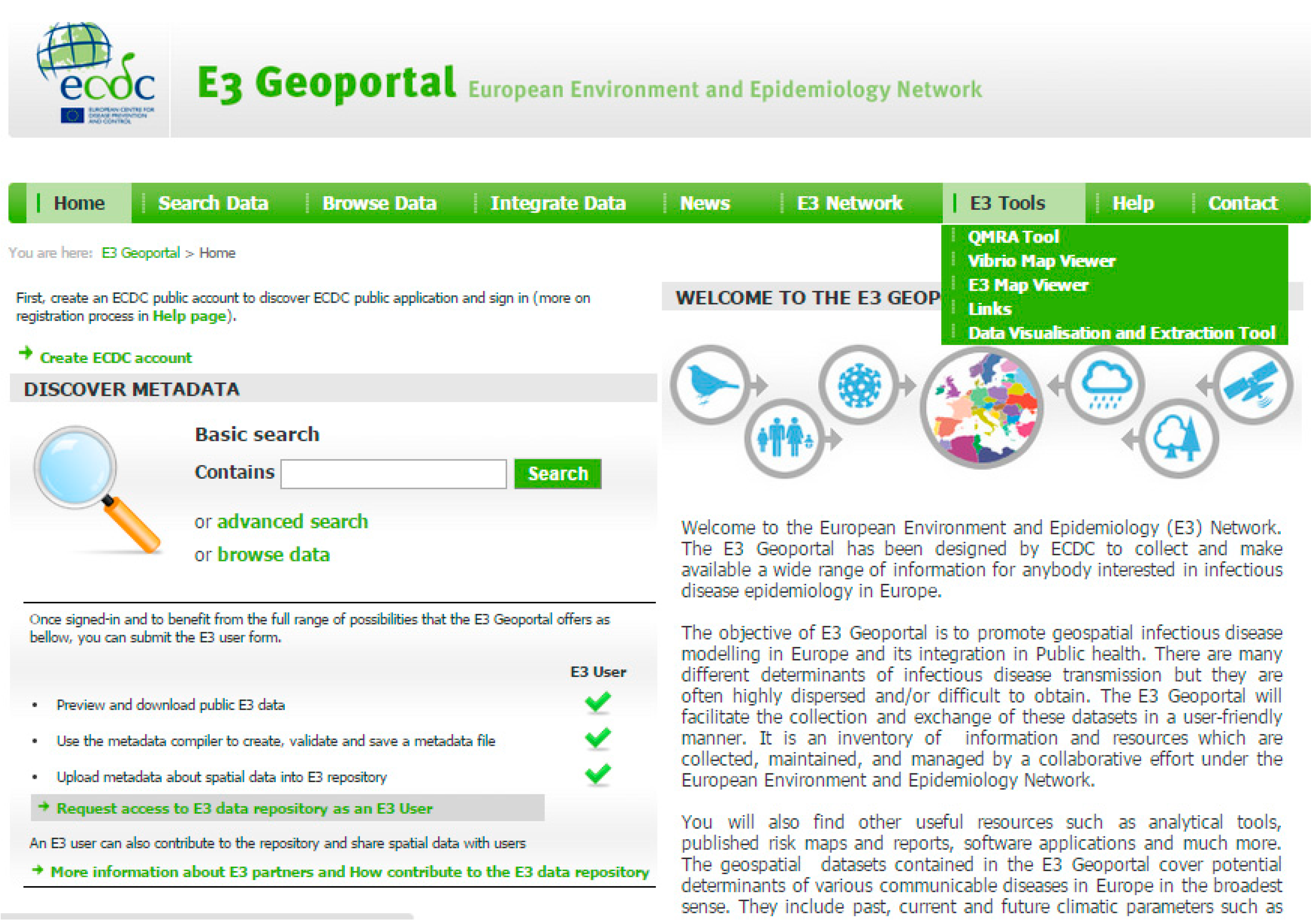
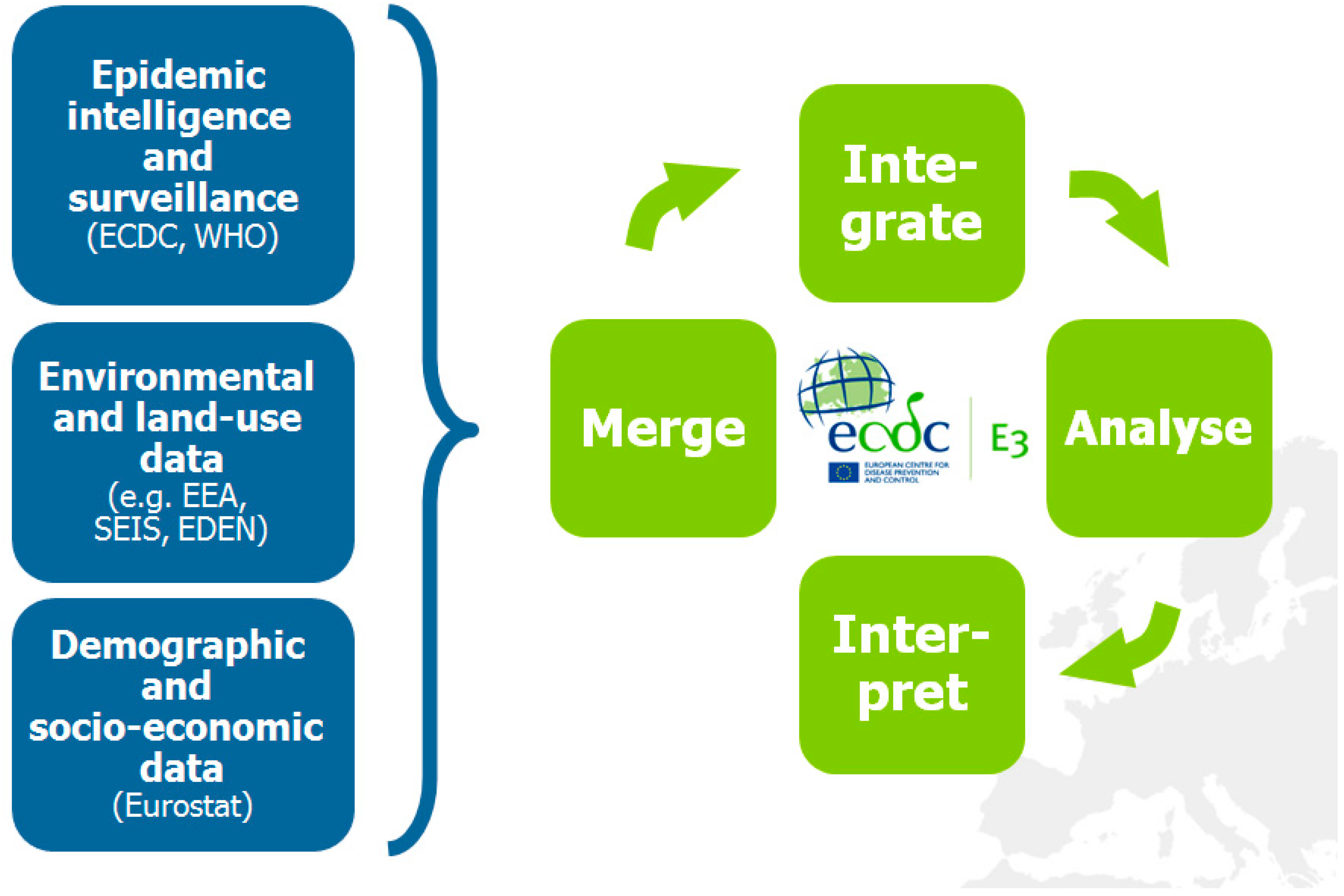
3. Intercepting Vector-borne Disease Emergence and Spread: Case Studies
3.1. Environmental Suitability of Malaria Transmission in Greece
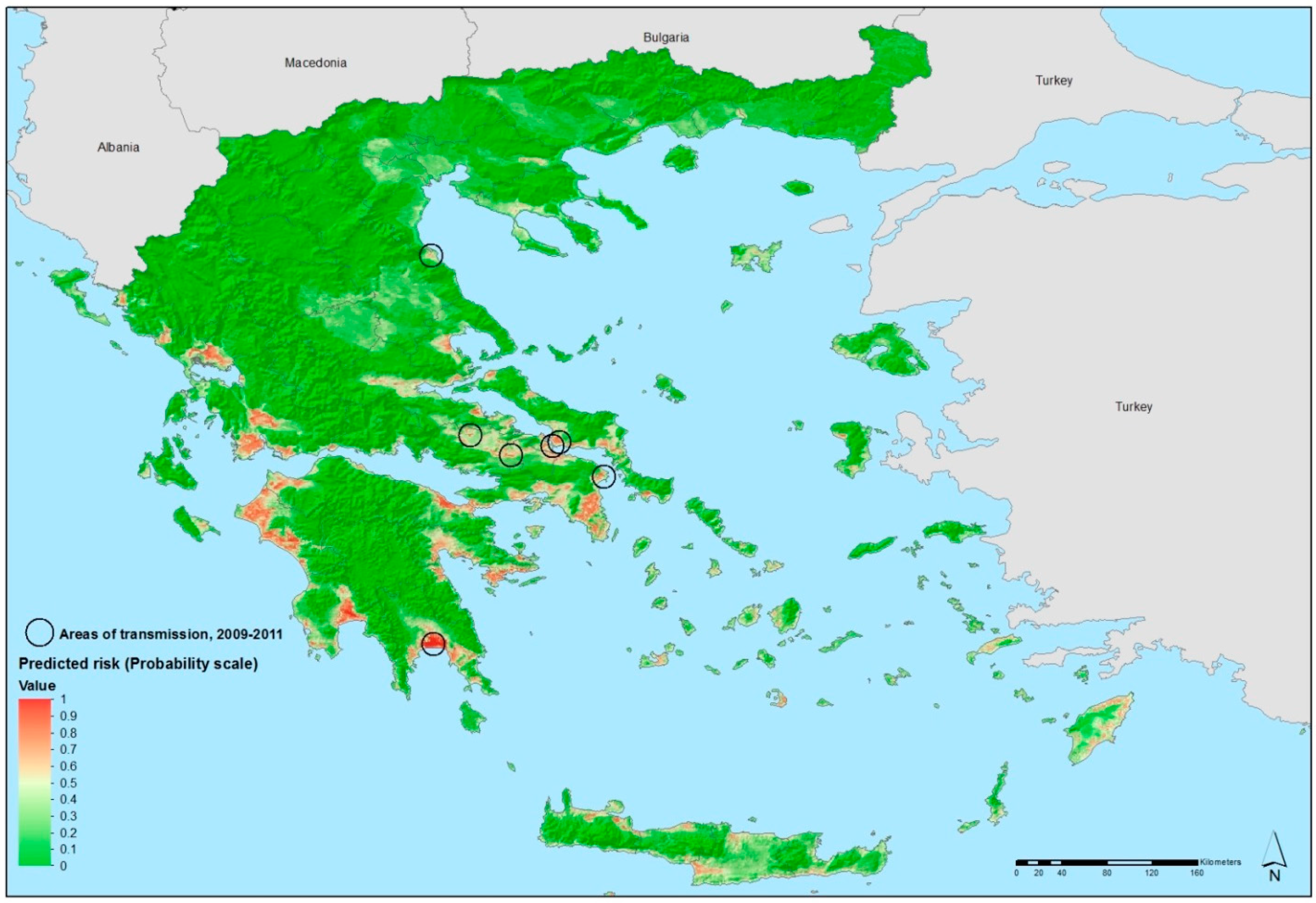
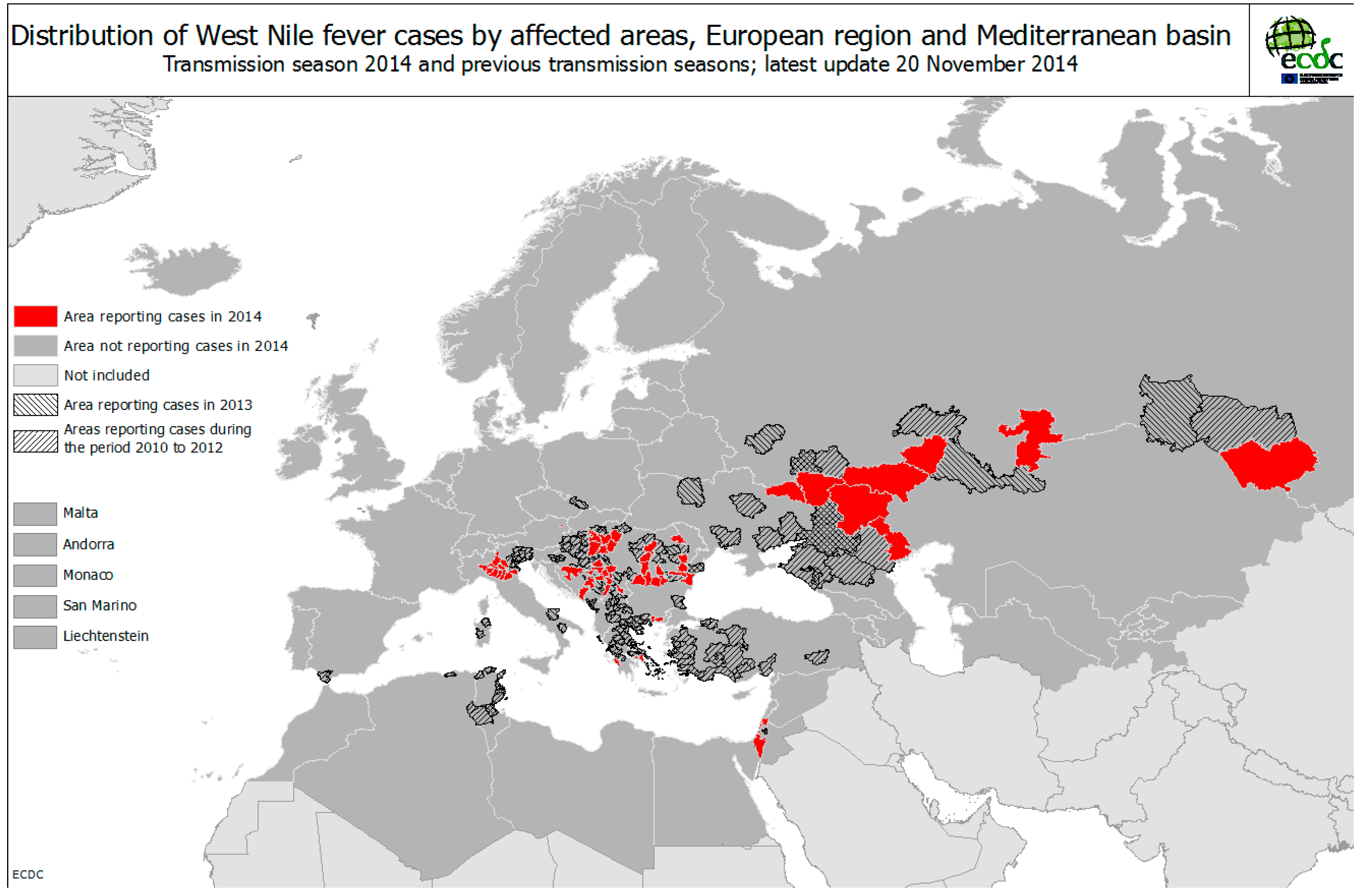
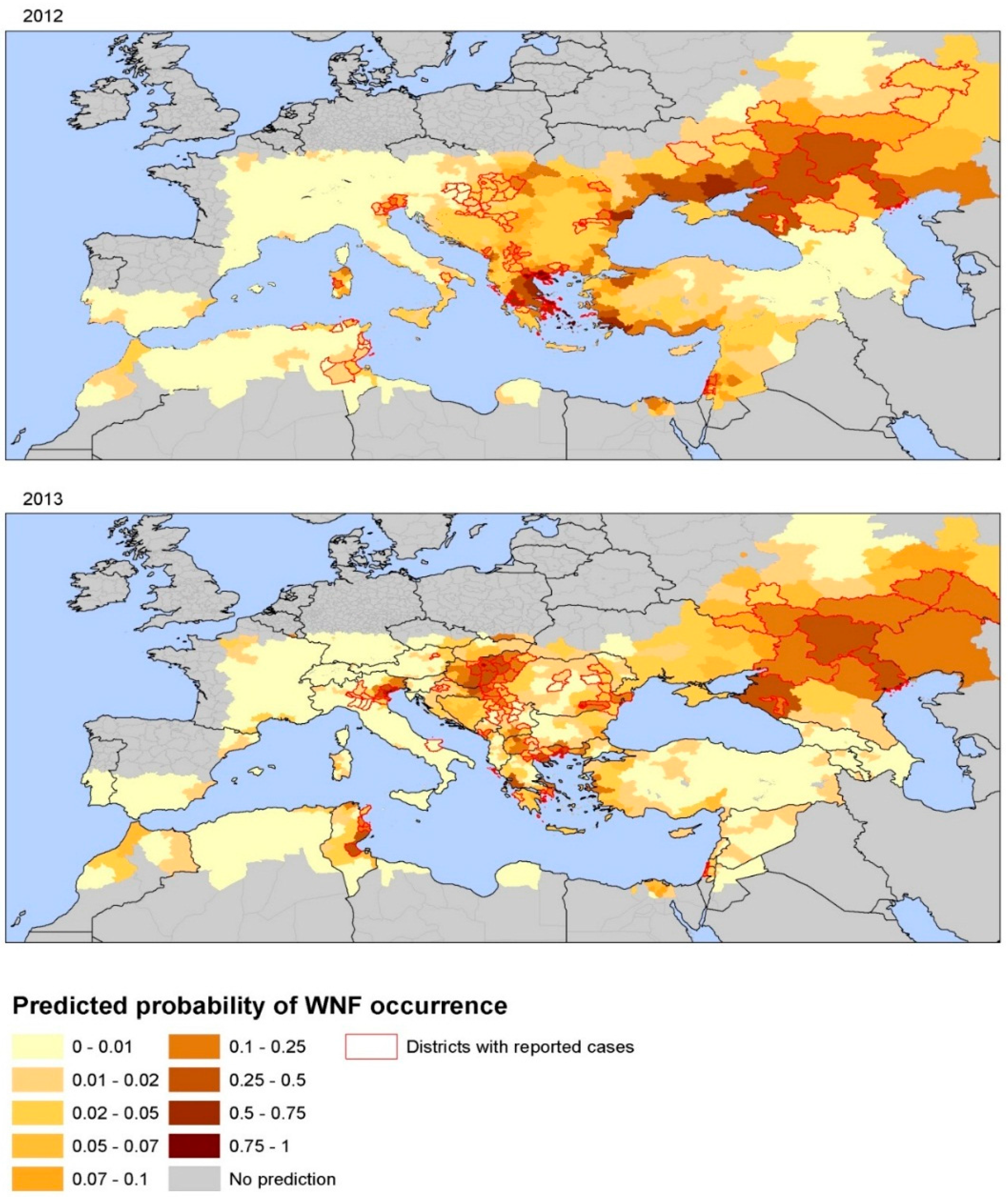
3.2. Environmental Determinants of West Nile Virus Transmission
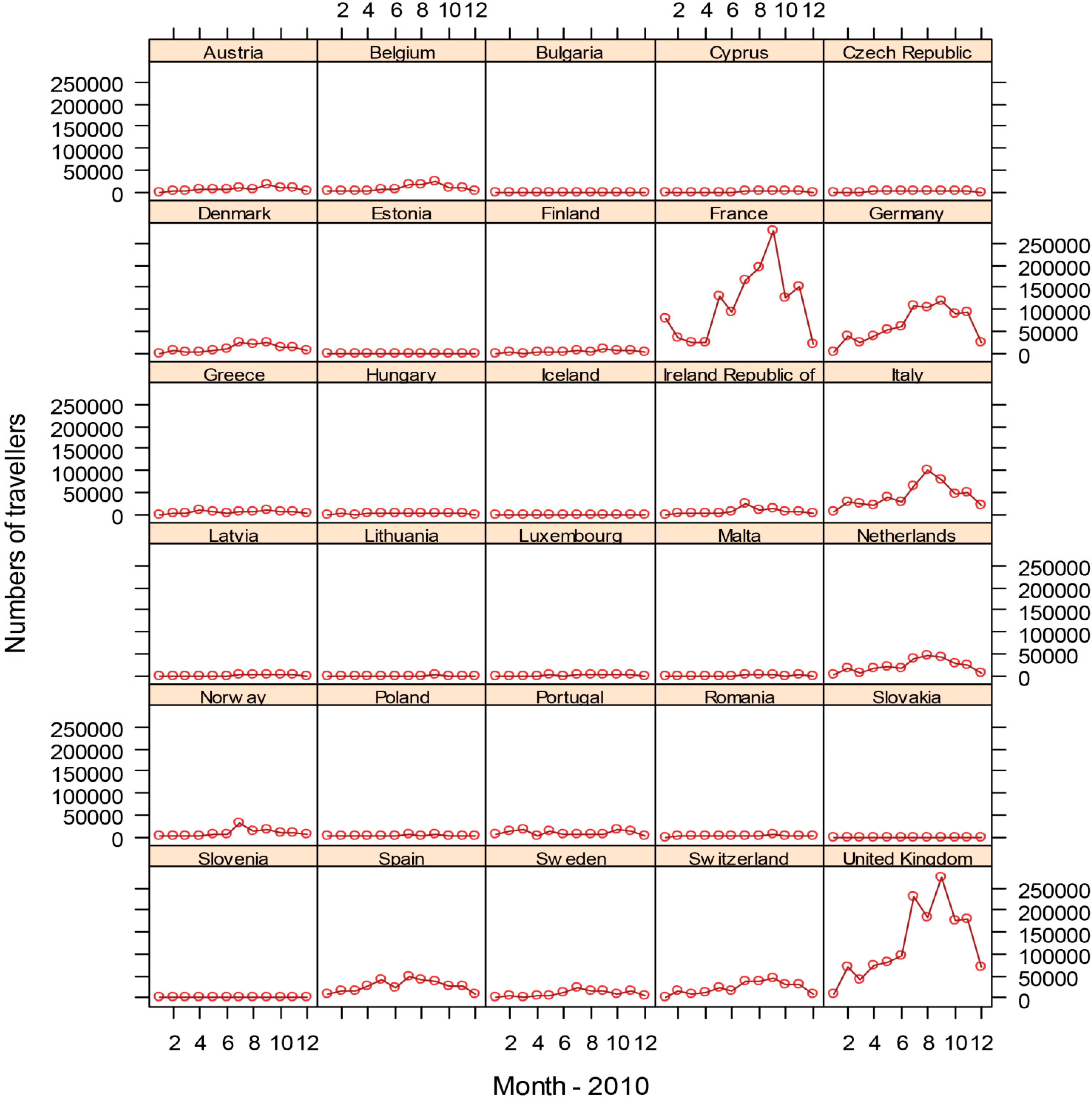
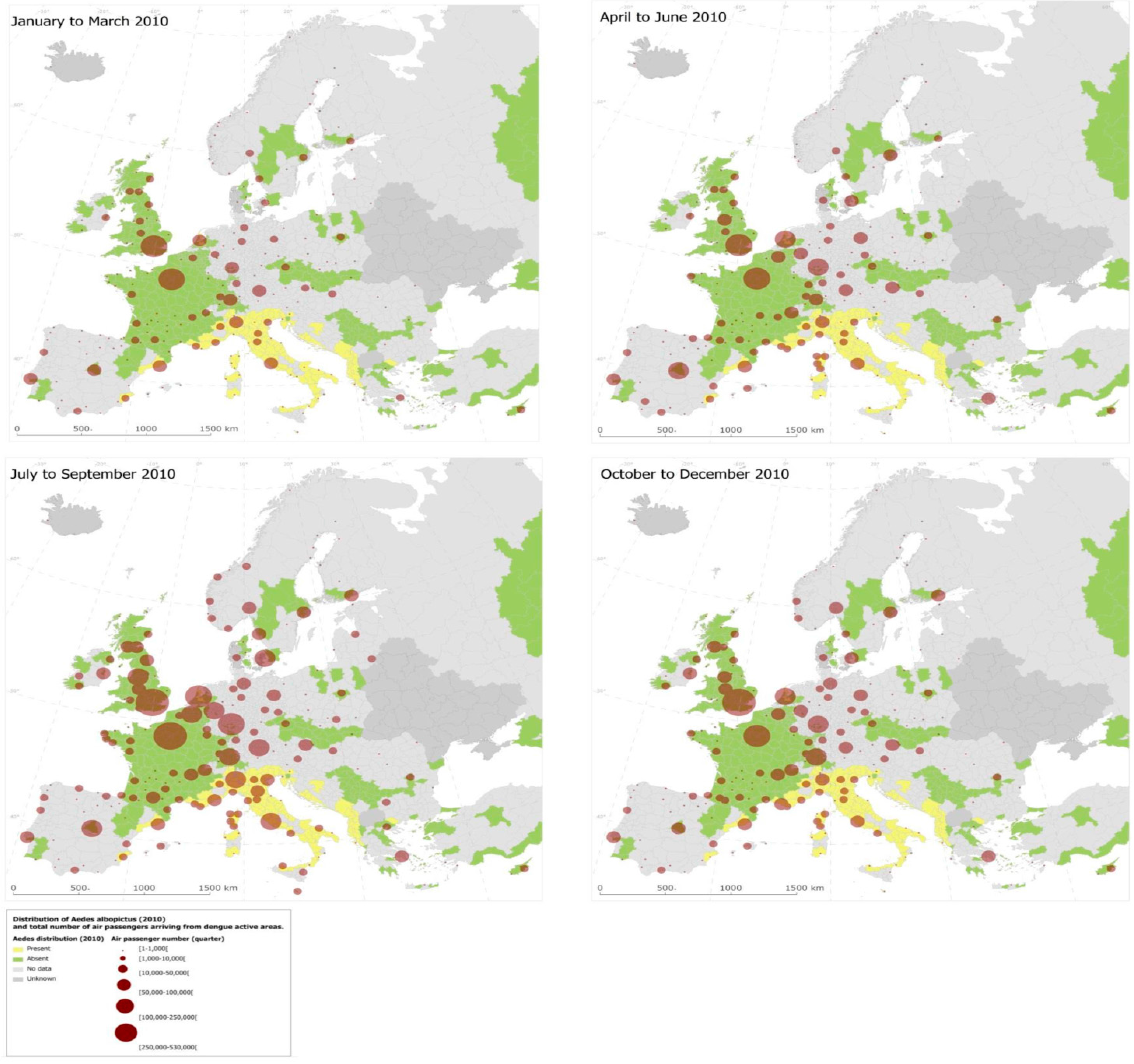
3.3. Dengue Dispersal through Air Traffic
4. Conclusions
Acknowledgements
Conflicts of Interest
References
- Habegger, B. Strategic foresight in public policy: Reviewing the experiences of the UK, Singapore, and the Netherlands. Futures 2010, 42, 49–58. [Google Scholar] [CrossRef]
- Suk, J.E.; Semenza, J.C. Future infectious disease threats to Europe. Amer. J. Public Health. 2011, 101, 2068–2079. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Sudre, B.; Miniota, J.; Rossi, M.; Hu, W.; Kossowsky, D.; Suk, J.E.; van Bortel, W.; Khan, K. International dispersal of dengue through air travel: Importation risk for Europe. PLoS Neglect. Trop. Dis. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.M.; Tjaden, N.B.; van den Bos, S.; Beierkuhnlein, C. Implementing cargo movement into climate based risk assessment of vector-borne diseases. Int. J. Environ. Res. Public Health 2014, 11, 3360–3374. [Google Scholar] [CrossRef] [PubMed]
- Randolph, S.E.; Rogers, D.J. The arrival, establishment and spread of exotic diseases: Patterns and predictions. Nature Rev. Microbiol. 2010, 8, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Karesh, W.B.; Dobson, A.; Lloyd-Smith, J.O.; Lubroth, J.; Dixon, M.A.; Bennett, M.; Aldrich, S.; Harrington, T.; Formenty, P.; Loh, E.H.; et al. Ecology of zoonoses: Natural and unnatural histories. Lancet 2012, 380, 1936–1945. [Google Scholar] [CrossRef] [PubMed]
- Patz, J.A.; Daszak, P.; Tabor, G.M.; Aguirre, A.A.; Pearl, M.; Epstein, J.; Wolfe, N.D.; Kilpatrick, A.M.; Foufopoulos, J.; Molyneux, D.; et al. Unhealthy landscapes: Policy recommendations on land use change and infectious disease emergence. Environ. Health Perspect. 2004, 112, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature. 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.J. Globalization, climate change, and human health. N. Engl. J. Med. 2013, 368, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, E.; Andersson, Y.; Suk, J.E.; Sudre, B.; Semenza, J.C. Public health. Monitoring EU emerging infectious disease risk due to climate change. Science 2012, 336, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Confalonieri, U.; Menne, B.; Akhtar, R.; Ebi, K.L.; Hauengue, M.; Kovats, R.S.; Revich, B.; Woodward, A. Human health. In Climate Change 2007: Impacts, Adaptation and Vulnerability Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Parry, M.L., Canziani, O.F., Palutik, J.P., van der Linden, P.J., Hanson, C.E., Eds.; Cambridge University Press: Cambridge, MA, USA, 2007. [Google Scholar]
- Altizer, S.; Ostfeld, R.S.; Johnson, P.T.J.; Kutz, S.; Harvell, C.D. Climate change and infectious diseases: From evidence to a predictive framework. Science 2013, 341, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Menne, B. Climate change and infectious diseases in Europe. Lancet Infect. Dis. 2009, 9, 365–375. [Google Scholar] [CrossRef]
- Semenza, J.C.; Suk, J.E.; Estevez, V.; Ebi, K.L.; Lindgren, E. Mapping climate change vulnerabilities to infectious diseases in Europe. Environ. Health Perspect. 2012, 20, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Giesecke, J. Intervening to reduce inequalities in infections in Europe. Amer. J. Public Health 2008, 98, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Suhrcke, M.; Stuckler, D.; Suk, J.E.; Desai, M.; Senek, M.; McKee, M.; Tsolova, S.; Basu, S.; Abubakar, I.; et al. The impact of economic crises on communicable disease transmission and control: A systematic review of the evidence. PloS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Stefanoff, P.; Rosinska, M.; Samuels, S.; White, D.J.; Morse, D.L.; Randolph, S.E. A national case-control study identifies human socio-economic status and activities as risk factors for tick-borne encephalitis in Poland. PloS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Reintjes, R.; Dedushaj, I.; Gjini, A.; Jorgensen, T.R.; Cotter, B.; Lieftucht, A.; D’Ancona, F.; Dennis, D.T.; Kosoy, M.A.; Mulliqi-Osmani, G.; Grunow, R.; Kalaveshi, A.; Gashi, L.; Humolli, I. Tularemia outbreak investigation in Kosovo: Case control and environmental studies. Emerg. Infect. Dis. 2002, 8, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Reisen, W.K.; Takahashi, R.M.; Carroll, B.D.; Quiring, R. Delinquent mortgages, neglected swimming pools, and West Nile virus, California. Emerg. Infect. Dis. 2008, 14, 1747–1749. [Google Scholar] [CrossRef] [PubMed]
- Semenza, J.C.; Domanović, D. Blood supply under threat. Nature Climate Change 2013, 3, 432–435. [Google Scholar] [CrossRef]
- Semenza, J.C.; Sudre, B.; Oni, T.; Suk, J.E.; Giesecke, J. Linking environmental drivers to infectious diseases: The European environment and epidemiology network. PLoS Neglect. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Lourenco, J.; Recker, M. The 2012 Madeira dengue outbreak: Epidemiological determinants and future epidemic potential. PLoS Neglect. Trop. Dis. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Paz, S.; Semenza, J.C. Environmental drivers of West Nile fever epidemiology in Europe and Western Asia—A review. Int. J. Environ. Res. Public Health 2013, 10, 3543–3562. [Google Scholar] [CrossRef] [PubMed]
- Sudre, B.; Rossi, M.; van Bortel, W.; Danis, K.; Baka, A.; Vakalis, N.; Semenza, J.C. Mapping environmental suitability for malaria transmission, Greece. Emerg. Infect. Dis. 2013, 19, 784–786. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.J. Impediments to comprehensive research on climate change and health. Int. J. Environ. Res. Public Health 2013, 10, 6096–6105. [Google Scholar] [CrossRef] [PubMed]
- Butler, C.D.; Harley, D. Primary, secondary and tertiary effects of eco-climatic change: The medical response. Postgraduate Med. J. 2010, 86, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M. How to make predictions about future infectious disease risks. Phil. Trans. Roy. Soc. London Ser. B Biol. Sci. 2011, 366, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Directive I. Available online: http://inspire.ec.europa.eu/ (accessed on 20 May 2015).
- ECDC. E3 Geoportal 2014. Available online: http://e3devint.ecdcnet.europa.eu/SitePages/Vibrio%20Risk%20Map.aspx (accessed on 20 May 2015).
- Sabatinelli, G.; Ejov, M.; Joergensen, P. Malaria in the WHO European Region (1971–1999). Eur. Surveill. 2001, 6, 61–65. [Google Scholar]
- Danis, K.; Baka, A.; Lenglet, A.; van Bortel, W.; Terzaki, I.; Tseroni, M.; Detsis, M.; Papanikolaou, E.; Balaska, A.; Gewehr, S.; et al. Autochthonous Plasmodium vivax malaria in Greece, 2011. Euro Surveill. 2011, 16. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19993 (accessed on 26 May 2015). [Google Scholar]
- Vakali, A.; Patsoula, E.; Spanakos, G.; Danis, K.; Vassalou, E.; Tegos, N.; Economopoulou, A.; Baka, A.; Pavli, A.; Koutis, C.; et al. Malaria in Greece, 1975 to 2010. Eur. Surveill. 2012, 17. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20322 (accessed on 26 May 2015). [Google Scholar]
- Kampen, H.; Maltezos, E.; Pagonaki, M.; Hunfeld, K.P.; Maier, W.A.; Seitz, H.M. Individual cases of autochthonous malaria in Evros Province, northern Greece: Serological aspects. Parasit. Res. 2002, 88, 261–266. [Google Scholar] [CrossRef]
- Kampen, H.; Proft, J.; Etti, S.; Maltezos, E.; Pagonaki, M.; Maier, W.A.; Seitz, H.M. Individual cases of autochthonous malaria in Evros Province, northern Greece: Entomological aspects. Parasit. Res. 2003, 89, 252–258. [Google Scholar] [CrossRef]
- Scharlemann, J.P.; Benz, D.; Hay, S.I.; Purse, B.V.; Tatem, A.J.; Wint, G.R.; Rogers, D.J. Global data for ecology and epidemiology: A novel algorithm for temporal fourier processing MODIS data. PLoS ONE 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Global 30 Arc-Second Elevation Dataset (GTOPO30). Available online: http://eros.usgs.gov/ (accessed on 20 May 2015).
- European Environment Agency. Corine Land Cover 2000 Seamless Vector Data—Version 15 (08/2011). 2011. Available online: http://www.eea.europa.eu/data-and-maps/data/corine-land-cover-2000-clc2000-seamless-vector-database-3 (accessed on 20 May 2015).
- European Environment Agency. Raster Data on Population Density Using Corine Land Cover 2000 Inventory. 2009. Available online: http://www.eea.europa.eu/data-and-maps/data/population-density-disaggregated-with-corine-land-cover-2000–2 (accessed on 20 May 2015).
- David Morley, D.R.; Luigi, S. Tala Research Group. Available online: http://www.tala.ox.ac.uk/ (accessed on 28 May 2015).
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Effects of temperature on the transmission of West Nile virus by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2006, 43, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Paz, S.; Albersheim, I. Influence of warming tendency on Culex pipiens population abundance and on the probability of West Nile fever outbreaks (Israeli Case Study: 2001–2005). EcoHealth 2008, 5, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Savage, H.M.; Ceianu, C.; Nicolescu, G.; Karabatsos, N.; Lanciotti, R.; Vladimirescu, A.; Laiv, L.; Ungureanu, A.; Romanca, C.; Tsai, T.F. Entomologic and avian investigations of an epidemic of West Nile fever in Romania in 1996, with serologic and molecular characterization of a virus isolate from mosquitoes. Amer. J. Trop. Med. Hyg. 1999, 61, 600–611. [Google Scholar]
- Paz, S.; Malkinson, D.; Green, M.S.; Tsioni, G.; Papa, A.; Danis, K.; Sirbu, A.; Ceianu, C.; Katalin, K.; Ferenczi, E.; et al. Permissive summer temperatures of the 2010 European West Nile fever upsurge. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Epstein, P.R. West Nile virus and the climate. J. Urban Health Bull. New York Acad. Med. 2001, 78, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Pecoraro, H.L.; Day, H.L.; Reineke, R.; Stevens, N.; Withey, J.C.; Marzluff, J.M.; Meschke, J.S. Climatic and landscape correlates for potential West Nile virus mosquito vectors in the Seattle region. J. Vector Ecol. J. Soc.Vec. Ecol. 2007, 32, 22–28. [Google Scholar] [CrossRef]
- Soverow, J.E.; Wellenius, G.A.; Fisman, D.N.; Mittleman, M.A. Infectious disease in a warming world: how weather influenced West Nile virus in the United States (2001–2005). Environ. Health Perspect. 2009, 117, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Ozdenerol, E.; Taff, G.N.; Akkus, C. Exploring the spatio-temporal dynamics of reservoir hosts, vectors, and human hosts of West Nile virus: A review of the recent literature. Int. J. Environ. Res. Public Health 2013, 10, 5399–5432. [Google Scholar] [CrossRef] [PubMed]
- ECDC. West Nile Fever Maps 2015. Available online: http://ecdc.europa.eu/en/healthtopics/west_nile_fever/West-Nile-fever-maps/pages/index.aspx (accessed on 1 June 2015).
- Tran, A.; Sudre, B.; Paz, S.; Rossi, M.; Desbrosse, A.; Chevalier, V.; Semenza, J.C. Environmental predictors of West Nile fever risk in Europe. Int. J. Health Geogr. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Xu, H. Modification of normalised difference water index (NDWI) to enhance open water features in remotely sensed imagery. Int. J. Remote Sens. 2006, 27, 3025–3033. [Google Scholar] [CrossRef]
- Marcantonio, M.; Rizzoli, A.; Metz, M.; Rosa, R.; Marini, G.; Chadwick, E.; Neteler, M. Identifying the environmental conditions favouring West Nile Virus outbreaks in Europe. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Kwan, J.L.; Park, B.K.; Carpenter, T.E.; Ngo, V.; Civen, R.; Reisen, W.K. Comparison of enzootic risk measures for predicting West Nile disease, Los Angeles, California, USA, 2004–2010. Emerg. Infect. Dis. 2012, 8, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Semenza , J.C.; Zeller, H. Integrated surveillance for prevention and control of emerging vector-borne diseases in Europe. Eur. Surveill. 2014, 19. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20757 (accessed on 26 May 2015).
- Simmons, C.P.; Farrar, J.J.; Nguyen, V.V.; Wills, B. Dengue. N. Engl. J. Med. 2012, 366, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Capeding, M.; Tran, N.; Hadinegoro, S.; Ismail, H.; Chotpitayasunondh, T.; Chua, M.; Luong, C.; Rusmi, L.K.; Wirawan, D.; Nallusamy, R.; et al. Clinical effi cacy and safety of a novel tetravalent dengue vaccine in healthy children in Asia: A phase 3, randomised, observer-masked, placebo-controlled trial. Lancet 2014, 384, 1358–1365. [Google Scholar] [CrossRef]
- Gubler, D.J. The global emergence/resurgence of arboviral diseases as public health problems. Arch. Med. Res. 2002, 33, 330–342. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Gubler, D.J.; Petersen, L.R. Emerging flaviviruses: The spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nature Med. 2004, 10, S98–S109. [Google Scholar] [CrossRef] [PubMed]
- WHO. Dengue 2014. Available online: http://www.who.int/denguecontrol/en/ (accessed on 26 May 2015).
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, L.; Scott, T.W.; Gubler, D.J. Consequences of the expanding global distribution of Aedes albopictus for dengue virus transmission. PLoS Neglect. Trop. Dis. 2010, 4. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control (ECDC). Dengue Outbreak in Madeira, Portugal 2012. Available online: http://www.ecdc.europa.eu/en/press/news/Lists/News/ECDC_DispForm.aspx?List=32e43ee8%2De230%2D4424%2Da783%2D85742124029a&ID=866&RootFolder=%2Fen%2Fpress%2Fnews%2FLists%2FNews (accessed on 26 May 2015).
- Giatropoulos, A.; Emmanouel, N.; Koliopoulos, G.; Michaelakis, A. A study on distribution and seasonal abundance of Aedes albopictus (Diptera: Culicidae) population in Athens, Greece. J. Med. Entomol. 2012, 49, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Tran, A.; L’Ambert, G.; Lacour, G.; Benoit, R.; Demarchi, M.; Cros, M.; Cailly, P.; Aubry-Kientz, M.; Balenghien, T.; Ezanno, P. A rainfall- and temperature-driven abundance model for Aedes albopictus populations. Int. J. Environ. Res. Public Health 2013, 10, 1698–1719. [Google Scholar] [CrossRef] [PubMed]
- Zitko, T.; Merdic, E. Seasonal and spatial oviposition activity of Aedes albopictus (Diptera: Culicidae) in Adriatic Croatia. J. Med. Entomol. 2014, 51, 760–768. [Google Scholar] [CrossRef]
- Gardner, L.M.; Fajardo, D.; Waller, S.T.; Wang, O.; Sarkar, S. A predictive spatial model to quantify the risk of air-travel-associated dengue importation into the United States and Europe. J. Trop. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, D.W.; Green, S.; Kalayanarooj, S.; Innis, B.L.; Nimmannitya, S.; Suntayakorn, S.; Endy, T.P.; Raengsakulrach, B.; Rothman, A.L.; Ennis, F.A.; et al. Dengue viremia titer, antibody response pattern, and virus serotype correlate with disease severity. J. Infect. Dis. 2000, 181, 2–9. [Google Scholar] [CrossRef] [PubMed]
- La Ruche, G.; Souares, Y.; Armengaud, A.; Peloux-Petiot, F.; Delaunay, P.; Despres, P.; Lenglet, A.; Jourdain, F.; Leparc-Goffart, I.; Charlet, F.; et al. First two autochthonous dengue virus infections in metropolitan France, September 2010. Eur. Surveill. 2010, 15. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19676 (accessed on 26 May 2015). [Google Scholar]
- Gjenero-Margan, I.; Aleraj, B.; Krajcar, D.; Lesnikar, V.; Klobucar, A.; Pem-Novosel, I.; Kurecic-Filipovic, S.; Komparak, S.; Martic, R.; Duricic, S.; et al. Autochthonous dengue fever in Croatia, August–September 2010. Eur. Surveill. 2011, 16. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19805 (accessed on 26 May 2015). [Google Scholar]
- Hufnagel, L.; Brockmann, D.; Geisel, T. Forecast and control of epidemics in a globalized world. Proc. Nat. Acad. Sci. USA 2004, 101, 15124–15129. [Google Scholar] [CrossRef] [PubMed]
- Suk, J.E.; Semenza, J.C. From global to local: Vector-borne disease in an interconnected world. Eur. J. Public Health 2014, 24, 531–532. [Google Scholar] [CrossRef] [PubMed]
- Nichols, G.L.; Andersson, Y.; Lindgren, E.; Devaux, I.; Semenza, J.C. European monitoring systems and data for assessing environmental and climate impacts on human infectious diseases. Int. J. Environ. Res. Public Health 2014, 11, 3894–3936. [Google Scholar] [CrossRef] [PubMed]
- Han, B.A.; Schmidt, J.P.; Bowden, S.E.; Drake, J.M. Rodent reservoirs of future zoonotic diseases. Proc. Nat. Acad. Sci. USA 2015. [Google Scholar] [CrossRef]
- Communities Cote. Human, Animal and Plant Health Impacts of Climate Change Brussels. Available online: http://ec.europa.eu/health/ph_threats/climate/docs/com_2009–147_en.pdf (accessed on 26 May 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Semenza, J.C. Prototype Early Warning Systems for Vector-Borne Diseases in Europe. Int. J. Environ. Res. Public Health 2015, 12, 6333-6351. https://doi.org/10.3390/ijerph120606333
Semenza JC. Prototype Early Warning Systems for Vector-Borne Diseases in Europe. International Journal of Environmental Research and Public Health. 2015; 12(6):6333-6351. https://doi.org/10.3390/ijerph120606333
Chicago/Turabian StyleSemenza, Jan C. 2015. "Prototype Early Warning Systems for Vector-Borne Diseases in Europe" International Journal of Environmental Research and Public Health 12, no. 6: 6333-6351. https://doi.org/10.3390/ijerph120606333
APA StyleSemenza, J. C. (2015). Prototype Early Warning Systems for Vector-Borne Diseases in Europe. International Journal of Environmental Research and Public Health, 12(6), 6333-6351. https://doi.org/10.3390/ijerph120606333




