The Impact of Different Proportions of a Treated Effluent on the Biotransformation of Selected Micro-Contaminants in River Water Microcosms
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
| Name (Application) | Structure | Molecular Weight | Log KOW | pKa |
|---|---|---|---|---|
| Caffeine (stimulant) | 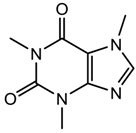 | 194.19 | −0.628 ± 0.753 | 0.52 ± 0.70 a |
| Carbamazepine (anticonvulsant) | 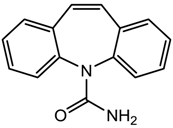 | 236.27 | 1.895 ± 0.597 | −0.49 ± 0.20 a 13.94 ± 0.20 b |
| Metoprolol (antihypertensive) | 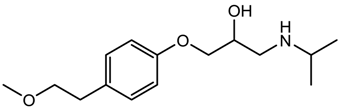 | 267.36 | 1.632 ± 0.263 | 9.43 ± 0.10 a 13.89 ± 0.20 b |
| Paracetamol (analgesic) | 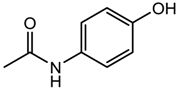 | 151.16 | 0.475 ± 0.210 | 1.72 ± 0.50 a 9.86 ± 0.13 b |
| Valsartan (antihypertensive) | 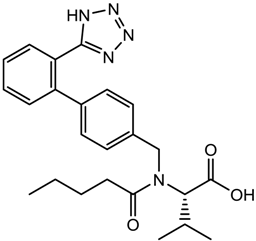 | 435.52 | 4.022 ± 0.606 | 0.60 ± 0.10 a 3.56 ± 0.10 b |
2.2. Sampling and Pretreatment of Matrix Components
2.3. Preparation and Sampling of Batch Experiments
2.3.1. Main Experiment
2.3.2. Additional Experiment
2.4. Analysis
| Compound | Type | Quantifier | Cap U [V] a | CE [V] b | Qualifier | Cap U [V] a | CE [V] b | IS c |
|---|---|---|---|---|---|---|---|---|
| Atenolol | PC | 267 > 145 | 55 | −20.0 | 267 > 190 | 55 | −11.0 | 1 |
| Atenolol acid | Atenolol/Metoprolol TP | 268 > 191 | 60 | −12.0 | 268 > 145 | 60 | −17.5 | 1 |
| Metoprolol | PC | 268 > 116 | 55 | −11.0 | 268 > 191 | 55 | −10.0 | 2 |
| 1-Methylxanthine | Caffeine TP | 165 > 108 | −55 | 19.0 | 165 > 80 | −55 | 25.0 | 3 |
| 3-Methylxanthine | Caffeine TP | 165 > 122 | −55 | 19.0 | 165 > 150 | −55 | 18.0 | 3 |
| 7-Methylxanthine | Caffeine TP | 167 > 124 | 55 | −13.0 | 167 > 150 | 55 | −12.0 | 3 |
| Caffeine | PC | 195 > 138 | 55 | −9.5 | 195 > 110 | 55 | −9.0 | 4 |
| Paraxanthine | Caffeine TP | 181 > 124 | 60 | −8.0 | 181 > 96 | 60 | −10.5 | 5 |
| Theobromine | Caffeine TP | 181 > 138 | 55 | −9.5 | 181 > 110 | 55 | −13.0 | 3 |
| Theophylline | Caffeine TP | 181 > 124 | 60 | −8.0 | 181 > 96 | 60 | −10.5 | 5 |
| Paracetamol | PC | 152 > 110 | 40 | −11.0 | 152 > 93 | 40 | −18.5 | 6 |
| Carbamazepine | PC | 237 > 194 | 45 | −11.0 | 237 > 179 | 45 | −27.0 | 7 |
| Irbesartan | PC | 429 > 207 | 50 | −22.5 | 429 > 195 | 50 | −21.5 | 8 |
| Losartan | PC | 423 > 207 | 40 | −21.0 | 423 > 180 | 40 | −36.0 | 9 |
| Valsartan | PC | 434 > 179 | −60 | 23.0 | 434 > 350 | −60 | 19.0 | 10 |
| Valsartan acid | Sartan TP | 265 > 165 | −40 | 17.0 | 265 > 193 | −40 | 13.5 | 10 |
| No. c | Internal standards | |||||||
| 1 | Atenolol-D7 | 274 > 145 | 55 | −17.5 | ||||
| 2 | Metoprolol-D7 | 275 > 123 | 55 | −11.5 | ||||
| 3 | Theobromine-D6 | 187 > 144 | 55 | −13.5 | ||||
| 4 | Caffeine-D9 | 204 > 144 | 60 | −8.5 | ||||
| 5 | Paraxanthine-D6 | 187 > 127 | 60 | −9.0 | ||||
| 6 | Paracetamol-D4 | 156 > 114 | 40 | −11.0 | ||||
| 7 | Carbamazepine-D10 | 247 > 204 | 45 | −13.0 | ||||
| 8 | Irbesartan-D7 | 436 > 207 | 50 | −22.5 | ||||
| 9 | Losartan-D4 | 427 > 211 | 40 | −21.0 | ||||
| 10 | Valsartan-D9 | 443 > 179 | −60 | 22.5 |
2.5. Background Concentrations of Relevant Parameters (Main Experiment)
3. Results and Discussion
3.1. Background Concentrations
3.2. pH and O2 Measurement in the Microcosms
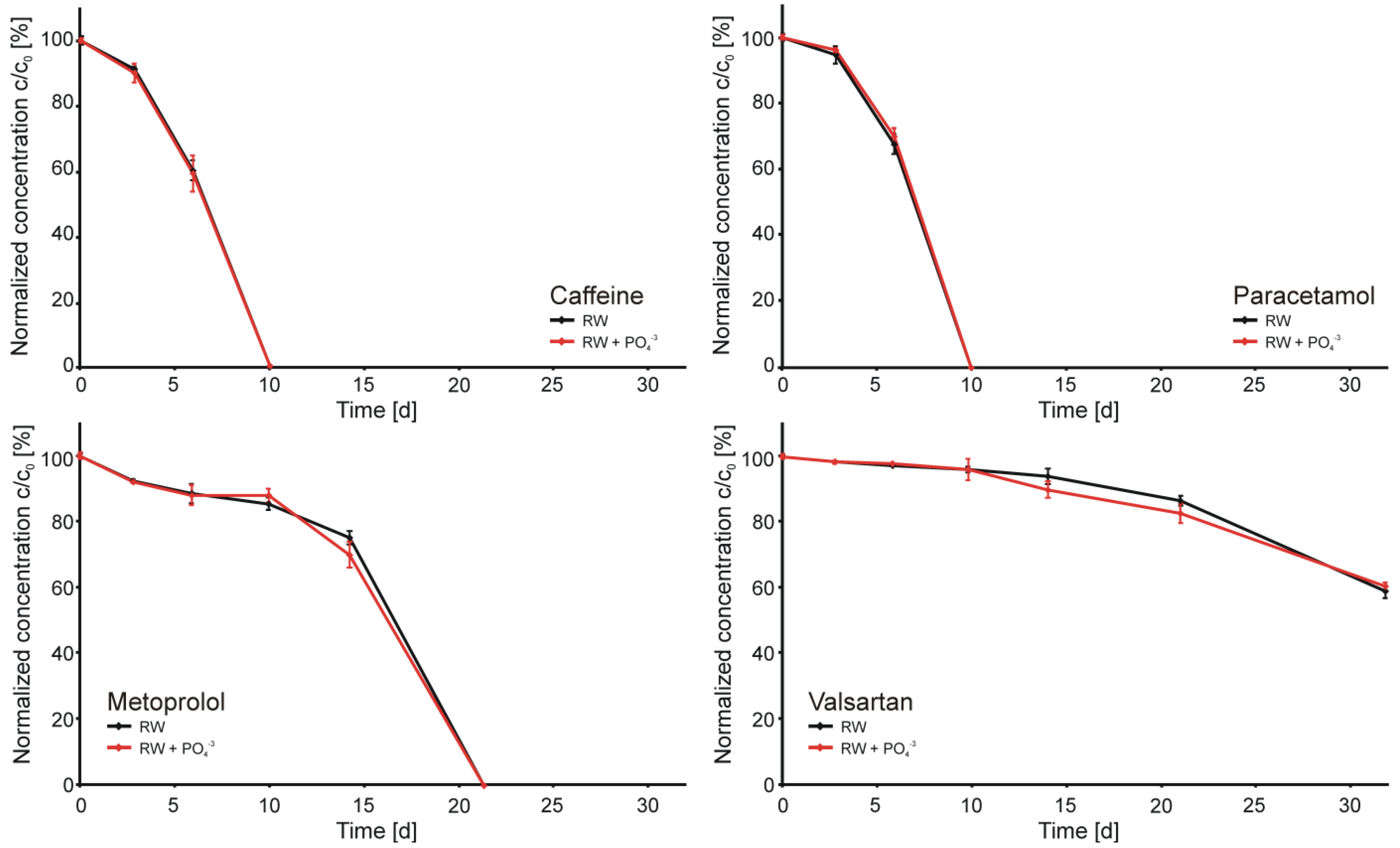
3.3. Abiotic Microcosms
3.4. Biotic Microcosms
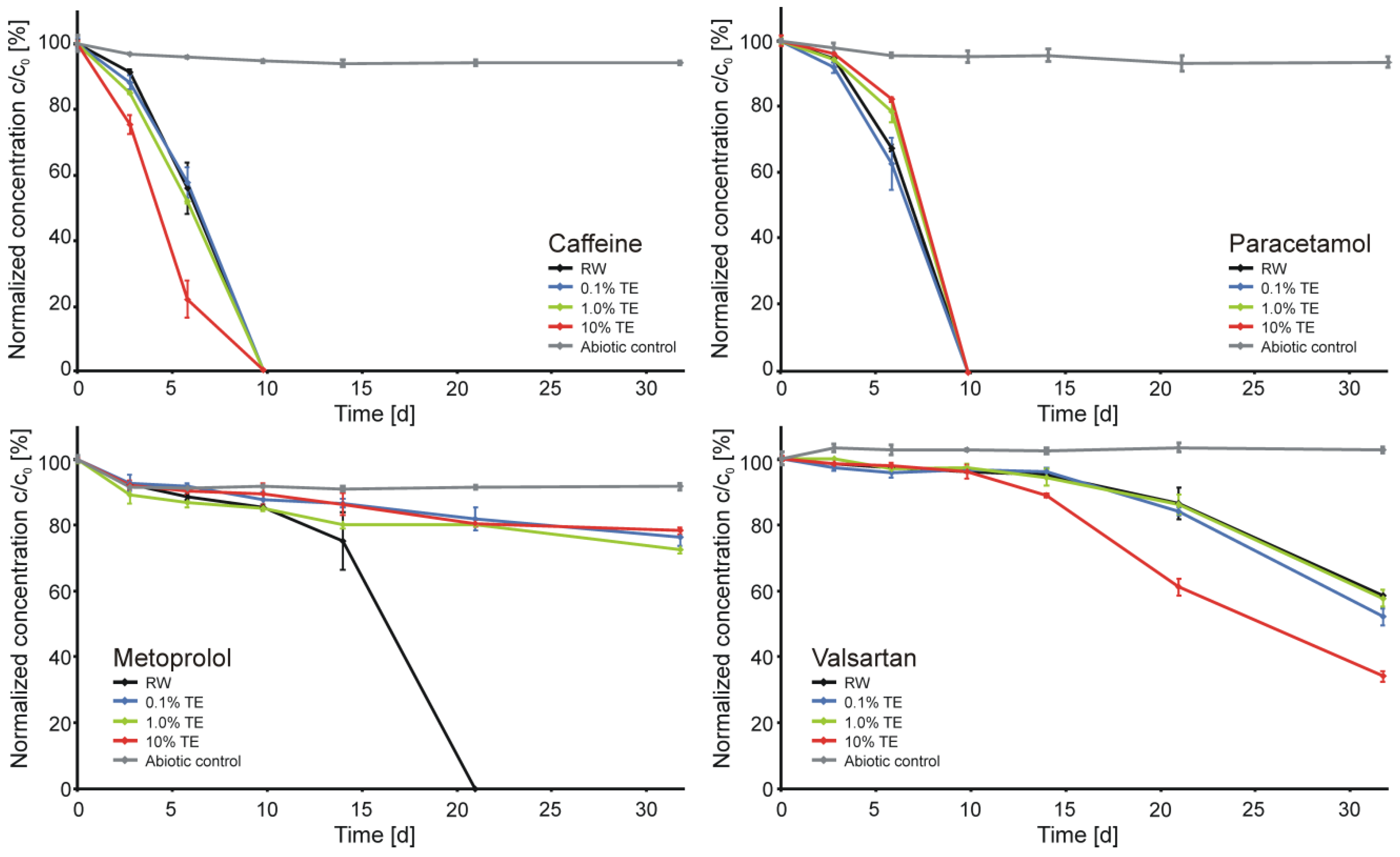
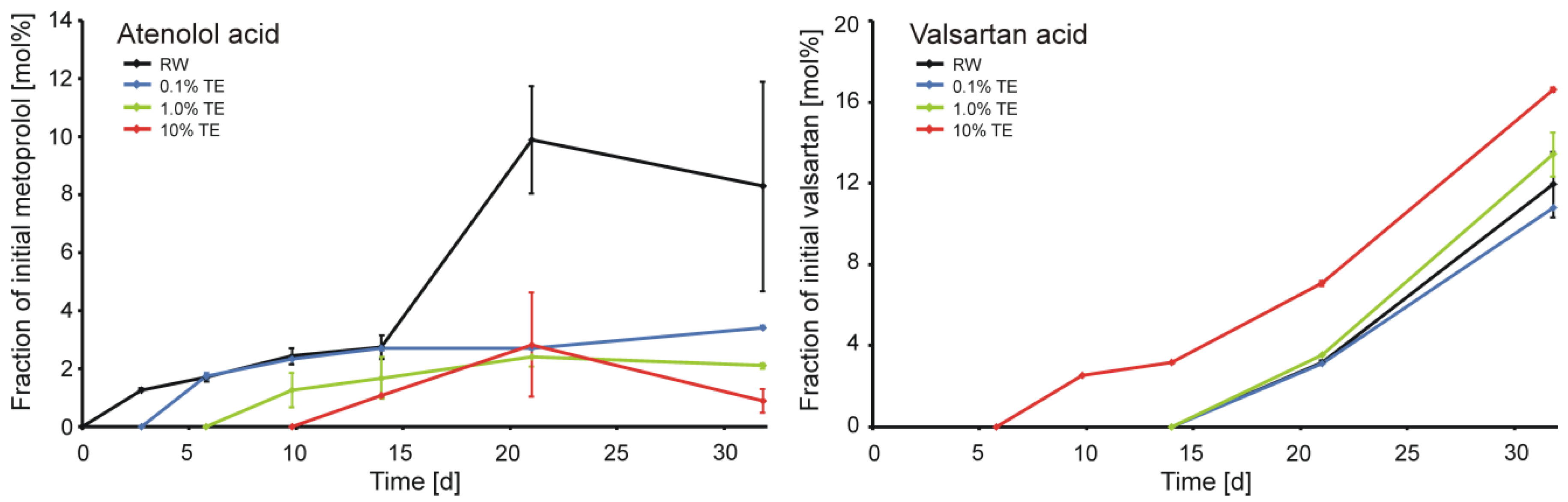
3.5. Environmental Relevance of the Study
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Loos, R.; Carvalho, R.; António, D.C.; Comero, S.; Locoro, G.; Tavazzi, S.; Paracchini, B.; Ghiani, M.; Lettieri, T.; Blaha, L.; et al. EU-wide monitoring survey on emerging polar organic contaminants in wastewater treatment plant effluents. Water Res. 2013, 47, 6475–6487. [Google Scholar]
- Buerge, I.J.; Poiger, T.; Müller, M.D.; Buser, H.-R. Combined sewer overflows to surface waters detected by the anthropogenic marker caffeine. Environ. Sci. Technol. 2006, 40, 4096–4102. [Google Scholar]
- Hughes, S.R.; Kay, P.; Brown, L.E. Global synthesis and critical evaluation of pharmaceutical data sets collected from river systems. Environ. Sci. Technol. 2013, 47, 661–677. [Google Scholar]
- Buser, H.-R.; Poiger, T.; Müller, M.D. Occurrence and fate of the pharmaceutical drug diclofenac in surface waters: Rapid photodegradation in a lake. Environ. Sci. Technol. 1998, 32, 3449–3456. [Google Scholar]
- Daneshvar, A.; Svanfelt, J.; Kronberg, L.; Prévost, M.; Weyhenmeyer, G.A. Seasonal variations in the occurrence and fate of basic and neutral pharmaceuticals in a Swedish river-lake system. Chemosphere 2010, 80, 301–309. [Google Scholar]
- Kunkel, U.; Radke, M. Fate of pharmaceuticals in rivers: Deriving a benchmark dataset at favorable attenuation conditions. Water Res. 2012, 46, 5551–5565. [Google Scholar]
- Radke, M.; Ulrich, H.; Wurm, C.; Kunkel, U. Dynamics and attenuation of pharmaceuticals along a river stretch. Environ. Sci. Technol. 2010, 44, 2968–2974. [Google Scholar]
- Alexander, M. Environmental and microbiological problems arising from recalcitrant molecules. Microbial Ecol. 1975, 2, 17–27. [Google Scholar]
- Hoppe-Jones, C.; Dickenson, E.R.V.; Drewes, J.E. The role of microbial adaptation and biodegradable dissolved organic carbon on the attenuation of trace organic chemicals during groundwater recharge. Sci. Total Environ. 2012, 437, 137–144. [Google Scholar]
- Grenni, P.; Patrolecco, L.; Ademollo, N.; Tolomei, A.; Caracciolo, A.B. Degradation of gemfibrozil and naproxen in a river water ecosystem. Microchem. J. 2013, 107, 158–164. [Google Scholar]
- Hillebrand, O.; Musallam, S.; Scherer, L.; Nödler, K.; Licha, T. The challenge of sample-stabilisation in the era of multi-residue analytical methods: A practical guideline for the stabilisation of 46 organic micropollutants in aqueous samples. Sci. Total Environ. 2013, 454–455, 289–298. [Google Scholar]
- Gawlik, B.M.; Loos, R.; Bidoglio, G.; Fauler, G.; Guo, X.; Lankmayr, E.; Linsinger, T. Testing sample stability in short-term isochronous stability studies for EU-wide monitoring surveys of polar organic contaminants in water. TrAC Trends Anal. Chem. 2012, 36, 36–46. [Google Scholar]
- Dalton, H.; Stirling, D.I.; Quayle, J.R. Co-metabolism. Philos. Trans. R. Soc. B 1982, 297, 481–496. [Google Scholar]
- Quintana, J.S.; Weiss, S.; Reemtsma, T. Pathways and metabolites of microbial degradation of selected acidic pharmaceutical and their occurrence in municipal wastewater treated by a membrane bioreactor. Water Res. 2005, 39, 2654–2664. [Google Scholar]
- Clara, M.; Strenn, B.; Kreuzinger, N. Carbamazepine as a possible anthropogenic marker in the aquatic environment: Investigations on the behavior of carbamazepine in wastewater treatment and during ground water infiltration. Water Res. 2004, 38, 947–954. [Google Scholar]
- Margot, J.; Kienle, C.; Magnet, A.; Weil, M.; Rossi, L.; de Alencastro, L.F.; Abegglen, C.; Thonney, D.; Chèvre, N.; Schärer, M.; et al. Treatment of micropollutants in municipal wastewater: Ozone or powdered activated carbon? Sci. Total Environ. 2013, 461–462, 480–498. [Google Scholar]
- Onesios, K.M.; Yu, J.T.; Bouwer, E.J. Biodegradation and removal of pharmaceuticals and personal care products in treatment systems: A review. Biodegradation 2009, 20, 441–466. [Google Scholar]
- Oosterhuis, M.; Sacher, F.; ter Laak, T.L. Prediction of concentration levels of metformin and other high consumption pharmaceuticals in wastewater and regional surface water based on sales data. Sci. Total Environ. 2013, 442, 380–388. [Google Scholar]
- Nödler, K.; Hillebrand, O.; Idzik, K.; Strathmann, M.; Schiperski, F.; Zierlewagen, J.; Licha, T. Occurrence and fate of the angiotensin II receptor antagonist transformation product valsartan acid in the water cycle—A comparative study with selected β-blockers and the persistent anthropogenic wastewater indicators carbamazepine and acesulfame. Water Res. 2013, 47, 6650–6659. [Google Scholar]
- Hebig, K.; Nödler, K.; Licha, T.; Scheytt, T. Impact of materials used in lab and field experiments on the recovery of organic micropollutants. Sci. Total Environ. 2014, 473–474, 125–131. [Google Scholar]
- Nödler, K.; Licha, T.; Fischer, S.; Wagner, B.; Sauter, M. A case study on the correlation of micro-contaminants and potassium in the Leine River (Germany). Appl. Geochem. 2011, 26, 2172–2180. [Google Scholar]
- Bester, K. Quantification with HPLC-MS/MS for environmental issues: Quality assurance and quality assessment. Anal. Bioanal. Chem. 2008, 391, 15–20. [Google Scholar]
- Barbieri, M.; Licha, T.; Nödler, K.; Carrera, J.; Ayora, C.; Sanchez-Vila, X. Fate of β-blockers in aquifer material under nitrate reducing conditions: Batch experiments. Chemosphere 2012, 89, 1272–1277. [Google Scholar]
- Kern, S.; Baumgartner, R.; Helbling, D.E.; Hollender, J.; Singer, H.; Loos, M.J.; Schwarzenbach, R.P.; Fenner, K. A tiered procedure for assessing the formation of biotransformation products of pharmaceuticals and biocides during activated sludge treatment. J. Environ. Monitor. 2010, 12, 2100–2111. [Google Scholar]
- Nödler, K.; Licha, T.; Bester, K.; Sauter, M. Development of a multi-residue analytical method, based on liquid chromatography-tandem mass spectrometry, for the simultaneous determination of 46 micro-contaminants in aqueous samples. J. Chromatogr. 2010, 1217, 6511–6521. [Google Scholar]
- Buerge, I.J.; Buser, H.-R.; Kahle, M.; Müller, M.D.; Poiger, T. Ubiquitous occurrence of the artificial sweetener acesulfame in the aquatic environment: An ideal chemical marker of domestic wastewater in groundwater. Environ. Sci. Technol. 2009, 43, 4381–4385. [Google Scholar]
- ISO 6878:2004. In Water quality—Determination of Phosphorus—Ammonium Molybdate Spectrometric Method; ISO: Geneva, Switzerland, 2004.
- ISO 11348–1:2007. In Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (Luminescent Bacteria Test)—Part 1: Method Using Freshly Prepared Bacteria; ISO: Geneva, Switzerland, 2007.
- Lange, F.T.; Scheurer, M.; Brauch, H.-J. Artificial sweeteners—A recently recognized class of emerging environmental contaminants: A review. Anal. Bioanal. Chem. 2012, 403, 2503–2518. [Google Scholar]
- Schaffer, M.; Licha, T. A guideline for the identification of environmentally relevant, ionizable organic molecule species. Chemosphere 2014, 103, 12–25. [Google Scholar]
- Schaffer, M.; Börnick, H.; Nödler, K.; Licha, T.; Worch, E. Role of cation exchange processes on the sorption influenced transport of cationic β-blockers in aquifer sediments. Water Res. 2012, 46, 5472–5482. [Google Scholar]
- Hillebrand, O.; Nödler, K.; Licha, T.; Sauter, M.; Geyer, T. Identification of the attenuation potential of a Karst aquifer by an artificial dualtracer experiment with caffeine. Water Res. 2012, 46, 5381–5388. [Google Scholar]
- Granger, D.A.; Butler, B.J.; Barker, J.F. A case of phosphorus limiting monoaromatic hydrocarbon biodegradation in groundwater. Biorememdiat. J. 1999, 3, 213–221. [Google Scholar]
- Madigan, M.T.; Martinko, J.M.; Parker, J. Wastewater treatment, water purification, and waterborne microbial diseases. In Brock—Biology of Microorganisms, 10th ed.; Pearson Education, Inc.: Upper Saddle River, NJ, USA, 2003; pp. 934–347. [Google Scholar]
- Szczepanowski, R.; Linke, B.; Krahn, I.; Gartemann, K.-H.; Gützkow, T.; Eichler, W.; Pühler, A.; Schlüter, A. Detection of 140 clinically relevant antibiotic-resistance genes in the plasmid metagenome of wastewater treatment plant bacteria showing reduced susceptibility to selected antibiotics. Microbiology 2009, 155, 2306–2319. [Google Scholar]
- Barker, D.J.; Stuckey, D.C. A review of soluble microbial products (SMP) in wastewater treatment systems. Water Res. 1999, 33, 3063–3082. [Google Scholar]
- Burgos, W.D.; Pisutpaisal, N.; Tuntoolavest, M.; Chorover, J.; Unz, R.F. Biodegradation of 1-naphthol in the presence of humic acid. Environ. Eng. Sci. 2000, 17, 343–351. [Google Scholar]
- Loos, R.; Gawlik, B.M.; Locoro, G.; Rimaviciute, E.; Contini, S.; Bidoglio, G. EU-wide survey of polar organic persistent pollutants in European river waters. Environ. Pollut. 2009, 157, 561–568. [Google Scholar]
- Rubirola, A.; Llorca, M.; Rodriguez-Mozaz, S.; Casas, N.; Rodriguez-Roda, I.; Barceló, D.; Buttiglieri, G. Characterization of metoprolol biodegradation and its transformation products generated in activated sludge batch experiments and in full scale WWTPs. Water Res. 2014, 63, 21–32. [Google Scholar]
- Radke, M.; Maier, M.P. Lessons learned from water/sediment-testing of pharmaceuticals. Water Res. 2014, 55, 63–73. [Google Scholar]
Appendix
| Element | TE | RW | Element | TE | RW |
|---|---|---|---|---|---|
| Li7 | 8.09 | 4.70 | Rb85 | 11.20 | 1.04 |
| B11 | 108 | 45.20 | Sr88 | 704.00 | 526.00 |
| Na23 | 89,400 | 19,600 | Cs133 | 0.06 | 0.001 |
| Mg25 | 11,400 | 9,650 | Ba137 | 9.10 | 21.60 |
| Al27 | 35.10 | 5.04 | La139 | 0.003 | 0.006 |
| P31 | 384.00 | 67.10 | Ce140 | 0.005 | 0.007 |
| K39 | 21,100 | 4530 | Pr141 | 0.001 | 0.001 |
| Ca43 | 21,100 | 21,000 | Nd143 | 0.006 | 0.007 |
| Sc45 | 0.31 | 0.27 | Sm147 | <0.001 | 0.001 |
| Ti49 | 2.46 | 1.24 | Eu151 | <0.005 | <0.005 |
| V51 | <20 | <20 | Gd157 | 0.424 | 0.004 |
| Cr53 | <20 | <20 | Tb159 | <0.001 | <0.001 |
| Mn55 | 8.69 | 9.49 | Dy163 | 0.001 | 0.002 |
| Fe57 | 179.00 | 177.00 | Ho165 | <0.001 | <0.001 |
| Co59 | 0.26 | 0.15 | Er166 | 0.001 | 0.001 |
| Ni60 | 2.06 | 1.31 | Tm169 | <0.001 | <0.001 |
| Cu63 | 1.94 | 1.19 | Yb174 | 0.003 | 0.001 |
| Zn66 | 5.56 | 0.82 | Lu175 | <0.001 | <0.001 |
| As75 | <2 | <2 | Hf178 | 0.001 | 0.003 |
| Se77 | <5 | <5 | Ta181 | <0.006 | <0.006 |
| Y89 | 0.01 | 0.01 | W184 | 0.162 | 0.044 |
| Zr90 | 0.05 | 0.19 | Tl205 | 0.001 | 0.001 |
| Nb93 | 0.01 | 0.02 | Pb208 | <2 | <2 |
| Mo98 | 4.47 | 0.39 | Bi209 | <0.002 | <0.002 |
| Cd114 | <0.001 | <0.001 | Th232 | 0.001 | 0.002 |
| Sb121 | 0.37 | 0.12 | U238 | 0.75 | 0.59 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nödler, K.; Tsakiri, M.; Licha, T. The Impact of Different Proportions of a Treated Effluent on the Biotransformation of Selected Micro-Contaminants in River Water Microcosms. Int. J. Environ. Res. Public Health 2014, 11, 10390-10405. https://doi.org/10.3390/ijerph111010390
Nödler K, Tsakiri M, Licha T. The Impact of Different Proportions of a Treated Effluent on the Biotransformation of Selected Micro-Contaminants in River Water Microcosms. International Journal of Environmental Research and Public Health. 2014; 11(10):10390-10405. https://doi.org/10.3390/ijerph111010390
Chicago/Turabian StyleNödler, Karsten, Maria Tsakiri, and Tobias Licha. 2014. "The Impact of Different Proportions of a Treated Effluent on the Biotransformation of Selected Micro-Contaminants in River Water Microcosms" International Journal of Environmental Research and Public Health 11, no. 10: 10390-10405. https://doi.org/10.3390/ijerph111010390
APA StyleNödler, K., Tsakiri, M., & Licha, T. (2014). The Impact of Different Proportions of a Treated Effluent on the Biotransformation of Selected Micro-Contaminants in River Water Microcosms. International Journal of Environmental Research and Public Health, 11(10), 10390-10405. https://doi.org/10.3390/ijerph111010390




