Swinholide J, a Potent Cytotoxin from the Marine Sponge Theonella swinhoei
Abstract
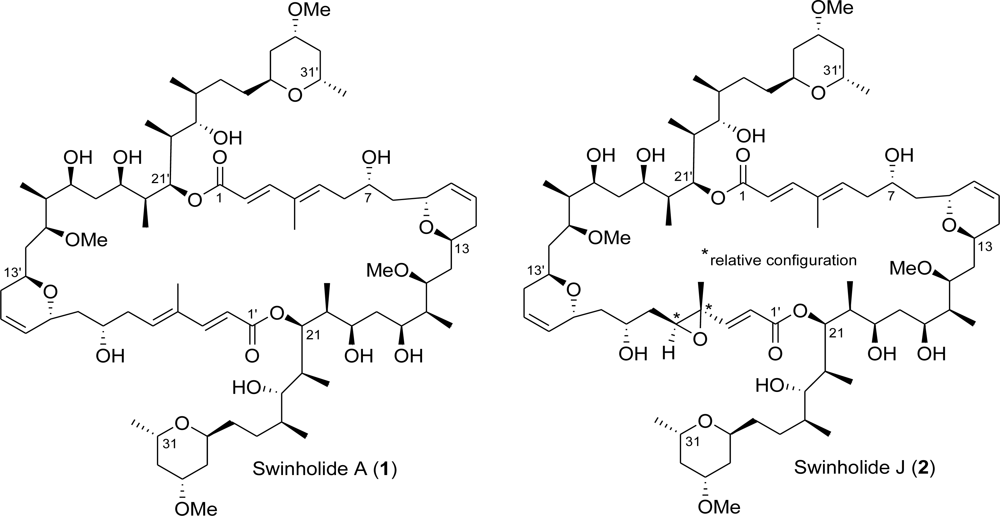
:1. Introduction
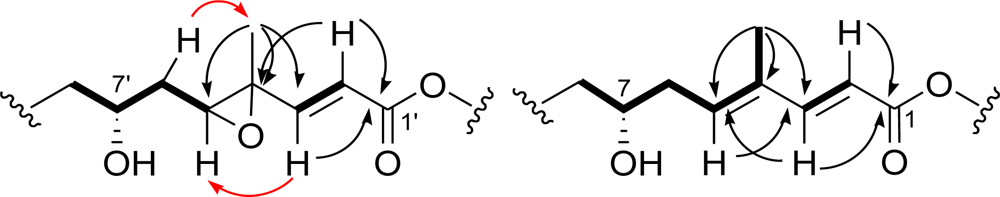
2. Results and Discussion
3. Experimental Section
3.1. General Procedures
3.2. Sponge Material and Separation of Individual Macrolides
3.3. Characteristic Data for Each Compound
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References and Notes
- Carmely, S; Kashman, Y. Structure of swinholide A, a new macrolide from the marine sponge. Theonella swinhoei Tetrahedron Lett 1985, 26, 511–514. [Google Scholar]
- Andrianasolo, EH; Gross, H; Goeger, D; Musafija-Girt, M; McPhail, K; Leal, RM; Mooberry, SL; Gerwick, WH. Isolation of swinholide A and related glycosylated derivatives from two field collections of marine cyanobacteria. Org Lett 2005, 7, 1375–1378. [Google Scholar]
- Kobayashi, M; Tanaka, J; Katori, T; Matsuura, M; Kitagawa, I. Structure of swinholide A, a potent cytotoxic macrolide from the Okinawan marine sponge. Theonella swinhoei Tetrahedron Lett 1989, 30, 2963–2966. [Google Scholar]
- Kobayashi, M; Tanaka, J; Katori, T; Matsuura, M; Yamashita, M; Kitagawa, I. Marine natural products. XXII. The absolute stereostructure of swinholide A, a potent cytotoxic dimeric macrolide from the Okinawan marine sponge. Theonella swinhoei Chem Pharm Bull 1990, 38, 2409–2418. [Google Scholar]
- Kitagawa, I; Kobayashi, M; Katori, T; Yamashita, M; Tanaka, J; Doi, M; Ishida, T. Absolute stereostructure of swinholide A, a potent cytotoxic macrolide from the Okinawan marine sponge. Theonella swinhoei J Am Chem Soc 1990, 112, 3710–3712. [Google Scholar]
- Doi, M; Ishida, T; Kobayashi, M; Kitagawa, I. Molecular conformation of swinholide A, a potent cytotoxic dimeric macrolide from the Okinawan marine sponge Theonella swinhoei: X-ray crystal structure of its diketone derivative. J Org Chem 1991, 56, 3629–3632. [Google Scholar]
- Klenchin, VA; King, R; Tanaka, J; Marriott, G; Rayment, I. Structural basis of swinholide A binding to actin. Chem Biol 2005, 12, 287–291. [Google Scholar]
- Allingham, JS; Zampella, A; D’Auria, MV; Rayment, I. Structures of microfilament destabilizing toxins bound to actin provide insight into toxin design and activity. PNAS 2005, 102, 14527–14532. [Google Scholar]
- Stanley, FM. Insulin-Increased prolactin gene expression requires actin treadmilling: Potential pole for P21 activated kinase. Endocrinology 2007, 148, 5874–5883. [Google Scholar]
- Braet, F; Soon, L; Vekemans, K; Thordarson, P; Spector, I. Actin-binding drugs: An elegant tool to dissect subcellular processes in endothelial and cancer cells. Protein Rev 2008, 8, 37–49. [Google Scholar]
- Sakai, R; Higa, T; Kashman, Y. Misakinolide-A, an antitumor macrolide from the marine sponge Theonella sp. Chem Lett 1986, 9, 1499–1502. [Google Scholar]
- Kato, Y; Fusetani, N; Matsunaga, S; Hashimoto, K; Sakai, R; Higa, T; Kashman, Y. Bioactive marine metabolites. Part XXIII. Antitumor macrodiolides isolated from a marine sponge Theonella sp.: Structure revision of misakinolide A. Tetrahedron Lett 1987, 28, 6225–6228. [Google Scholar]
- Kobayashi, J; Tsukamoto, S; Tanabe, A; Sasaki, T; Ishibashi, M. New congeners of bistheonellides from Okinawan marine sponges of the Genus. Theonella J Chem Soc Perkin Trans 1 1991, 2379–2383. [Google Scholar] [CrossRef]
- Youssef, DTA; Mooberry, LS. Hurghadolide A and swinholide I, potent actin-microfilament disrupters from the red sea sponge. Theonella swinhoei J Nat Prod 2006, 69, 154–157. [Google Scholar]
- Kobayashi, M; Tanaka, J; Katori, T; Kitagawa, I. Marine natural products. XXIII. Three new cytotoxic dimeric macrolides, swinholides B and C and isoswinholide A, congeners of swinholide A, from the Okinawan marine sponge. Theonella swinhoei Chem Pharm Bull 1990, 38, 2960–2966. [Google Scholar]
- Todd, JS; Alvi, KA; Crews, P. The isolation of a monomeric carboxylic acid of swinholide a from the indo-pacific sponge. Theonella swinhoei Tetrahedron Lett 1992, 33, 441–442. [Google Scholar]
- Tsukamoto, S; Ishibashi, M; Sasaki, T; Kobayashi, J. New congeners of swinholides from the Okinawan marine sponge Theonella sp. J Chem Soc Perkin Trans 1 1991, 3185–3188. [Google Scholar] [CrossRef]
- Dumdei, EJ; Blunt, JW; Munro, MHG; Pannell, LK. Isolation of Calyculins, Calyculinamides, and Swinholide H from the New Zealand Deep-Water Marine Sponge Lamellomorpha strongylata. J Org Chem 1997, 62, 2635–2639. [Google Scholar]
- Festa, C; De Marino, S; Sepe, V; Monti, MC; Luciano, P; D’Auria, MV; Debitus, C; Bucci, M; Vellecco, V; Zampella, A. Perthamides C and D, two new potent anti-inflammatory cyclopeptides from a Solomon Lithistid sponge. Theonella swinhoei Tetrahedron 2009, 65, 10424–10429. [Google Scholar]
- Sepe, V; D’Auria, MV; Bifulco, G; Ummarino, R; Zampella, A. Concise synthesis of AHMHA unit in perthamide C. Structural and stereochemical revision of perthamide C. Tetrahedron 2010, 66, 7520–7526. [Google Scholar]
- Festa, C; De Marino, S; D’Auria, MV; Bifulco, G; Renga, B; Fiorucci, S; Petek, S; Zampella, A. Solomonsterols A and B from Theonella swinhoei. The First Example of C-24 and C-23 Sulfated Sterols from a Marine Source Endowed with a PXR Agonistic Activity. J Med Chem 2011, 54, 401–405. [Google Scholar]
- Festa, C; De Marino, S; Sepe, V; D’Auria, MV; Bifulco, G; Debitus, C; Bucci, M; Vellecco, V; Zampella, A. Solomonamides A and B, new anti-inflammatory peptides from Theonella swinhoei. Org Lett 2011, 13, 1532–1535. [Google Scholar]
- De Marino, S; Ummarino, R; D’Auria, MV; Chini, MG; Bifulco, G; Renga, B; D’Amore, C; Fiorucci, S; Debitus, C; Zampella, A. Theonellasterols and conicasterols from Theonella swinhoei. Novel marine natural ligands for human nuclear receptors. J Med Chem 2011, 54, 3065–3075. [Google Scholar]
- De Marino, S; Sepe, V; D’Auria, MV; Bifulco, G; Renga, B; Petek, S; Fiorucci, S; Zampella, A. Towards new ligands of nuclear receptors. Discovery of malaitasterol A, an unique bis-secosterol from marine sponge. Theonella swinhoei Org Biomol Chem 2011, 9, 4856–4862. [Google Scholar]
- The scarcity of isolated material hampered further pharmacological investigation.
- Zampella, A; Sepe, V; Bellotta, F; Luciano, P; D’Auria, MV; Cresteil, T; Debitus, C; Petek, S; Poupat, C; Ahon, A. Homophymines B–E and A1–E1a family of bioactive cyclodepsipeptides from the sponge Homophymia sp. Org Biomol Chem 2009, 7, 4037–4044. [Google Scholar]
| 1 a | 2 a | |||||
|---|---|---|---|---|---|---|
| Position | Type | δH/δH′ (J in Hz) | δC/δC′ | δH/δH′ (J in Hz) | δC/δC′ | HMBC b |
| 1/1′ | C | - | 170.6 | - | 170.2/168.7 | |
| 2/2′ | CH | 5.84 d (15.7) | 115.6 | 5.90 d (15.6)/6.14 d (15.7) | 115.9/122.2 | C1, C4 |
| C1′, C4′ | ||||||
| 3/3′ | CH | 7.43 d (15.7) | 152.3 | 7.46 d (15.6)/6.82 d (15.7) | 152.0/152.4 | C1, C5 |
| C1′ | ||||||
| 4/4′ | C | - | 135.5 | - | 135.5/59.7 | |
| 4/4′-Me | CH3 | 1.77 s | 12.4 | 1.85 s/1.42 s | 12.7/15.5 | C3, C4, C5 |
| C3′, C4′, C5′ | ||||||
| 5/5′ | CH | 6.14 t (7.3) | 140.5 | 6.16 t (7.2)/3.22 dd (4.3, 7.4) | 140.6/64.6 | C3, 4-Me |
| 6/6′ | CH2 | 2.40 t (6.9) | 38.8 | 2.44 m/1.81 m, 1.63 m | 38.7/37.9 | C4, C5, C7, C8 |
| 7/7′ | CH | 4.02 m | 68.1 | 3.99 ovl/4.12 m | 68.2/66.5 | |
| 8/8′ | CH2 | 1.28 m | 41.0 | 1.87 m, 1.37 m/ | 41.8/41.6 | |
| 1.76 m | 1.78 m, 1.37 m | |||||
| 9/9′ | CH | 4.47 br d (10.5) | 70.5 | 4.48 br d (10.2) | 70.4 | |
| 10/10′ | CH | 5.65 dd (1.8, 10.5) | 130.9 | 5.67 br d (10.3) | 130.8 | |
| 11/11′ | CH | 5.81 m | 124.9 | 5.83 m | 124.8 | |
| 12/12′ | CH2 | 1.94 m | 32.2 | 1.96 m | 32.2 | |
| 13/13′ | CH | 3.49 m | 65.3 | 3.54 m | 65.4 | |
| 14/14′ | CH2 | 1.58 m | 37.2 | 1.58 m | 36.9 | |
| 1.77 m | 1.82 m | |||||
| 15/15′ | CH | 3.76 m | 78.2 | 3.76 m/3.83 m | 78.4/77.9 | |
| 15/15′-OMe | CH3 | 3.32 s | 56.7 | 3.34 s/3.35 s | 56.9/57.2 | C15/C15′ |
| 16/16′ | CH | 1.52 m | 43.8 | 1.55 m | 43.3 | |
| 16/16′-Me | CH3 | 0.83 d (6.7) | 8.8 | 0.83 d (7.0)/0.84 d (7.0) | 9.2 | C15, C16, C17 |
| C15′, C16′, C17′ | ||||||
| 17/17′ | CH | 3.61 m | 73.2 | 3.60 m | 73.2 | |
| 18/18′ | CH2 | 1.63 m | 39.0 | 1.61 m | 39.2 | |
| 1.74 m | 1.75 m | |||||
| 19/19′ | CH | 3.97 ovl | 70.1 | 3.97 ovl/3.88 m | 70.0/70.1 | |
| 20/20′ | CH | 1.94 m | 39.4 | 1.94 m | 39.4 | |
| 20/20′-Me | CH3 | 0.91 d (7.0) | 8.9 | 0.90 d (7.1)/0.91 d (7.1) | 9.1 | |
| 21/21′ | CH | 5.46 d (10.5) | 75.6 | 5.45 t (10.3)/5.46 t (10.3) | 75.7/76.3 | |
| 22/22′ | CH | 1.98 m | 37.9 | 1.97 m | 38.0 | |
| 22/22′-Me | CH3 | 0.94 d (6.9) | 9.6 | 0.92 d (7.0)/0.93 d (7.0) | 9.7 | C21, C22, C23 |
| C21′, C22′, C23′ | ||||||
| 23/23′ | CH | 3.11 dd (1.8, 9.5) | 77.2 | 3.10 m | 77.3 | |
| 24/24′ | CH | 1.70 m | 34.4 | 1.70 m | 34.5 | |
| 24/24′-Me | CH3 | 0.98 d (6.7) | 17.7 | 0.97 d (6.7)/0.98 d (6.7) | 17.9 | C23, C24, C25 |
| C23′, C24′, C25′ | ||||||
| 25/25′ | CH2 | 1.24 m | 25.1 | 1.23 m, 1.41 m | 25.1 | |
| 1.42 m | ||||||
| 26/26′ | CH2 | 1.27 m | 29.7 | 1.28 m, 1.94 m | 29.8 | |
| 1.94 m | ||||||
| 27/27′ | CH | 3.99 ovl | 72.7 | 3.98 ovl | 72.8 | |
| 28/28′ | CH2 | 1.52 m | 35.8 | 1.52 m | 35.8 | |
| 1.87 br d (12.8) | 1.87 br d (12.5) | |||||
| 29/29′ | CH | 3.61 m | 74.2 | 3.61 m | 74.3 | C29-OMe |
| C29′-OMe | ||||||
| 29/29′-OMe | CH3 | 3.34 s | 55.3 | 3.34 s | 55.3 | C29 |
| C29′ | ||||||
| 30/30′ | CH2 | 1.09 dd (10.4, 12.6) | 39.7 | 1.09 dd (10.4, 12.6) | 39.7 | |
| 2.01 br d (12.6) | 2.02 br d (12.6) | |||||
| 31/31′ | CH | 3.74 m | 65.7 | 3.74 m | 65.8 | |
| 31/31′-Me | CH3 | 1.19 d (6.2) | 21.8 | 1.19 d (6.2) | 21.8 | |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Marino, S.D.; Festa, C.; D’Auria, M.V.; Cresteil, T.; Debitus, C.; Zampella, A. Swinholide J, a Potent Cytotoxin from the Marine Sponge Theonella swinhoei. Mar. Drugs 2011, 9, 1133-1141. https://doi.org/10.3390/md9061133
Marino SD, Festa C, D’Auria MV, Cresteil T, Debitus C, Zampella A. Swinholide J, a Potent Cytotoxin from the Marine Sponge Theonella swinhoei. Marine Drugs. 2011; 9(6):1133-1141. https://doi.org/10.3390/md9061133
Chicago/Turabian StyleMarino, Simona De, Carmen Festa, Maria Valeria D’Auria, Thierry Cresteil, Cecile Debitus, and Angela Zampella. 2011. "Swinholide J, a Potent Cytotoxin from the Marine Sponge Theonella swinhoei" Marine Drugs 9, no. 6: 1133-1141. https://doi.org/10.3390/md9061133
APA StyleMarino, S. D., Festa, C., D’Auria, M. V., Cresteil, T., Debitus, C., & Zampella, A. (2011). Swinholide J, a Potent Cytotoxin from the Marine Sponge Theonella swinhoei. Marine Drugs, 9(6), 1133-1141. https://doi.org/10.3390/md9061133






