Deoxyuridines from the Marine Sponge Associated Actinomycete Streptomyces microflavus
Abstract
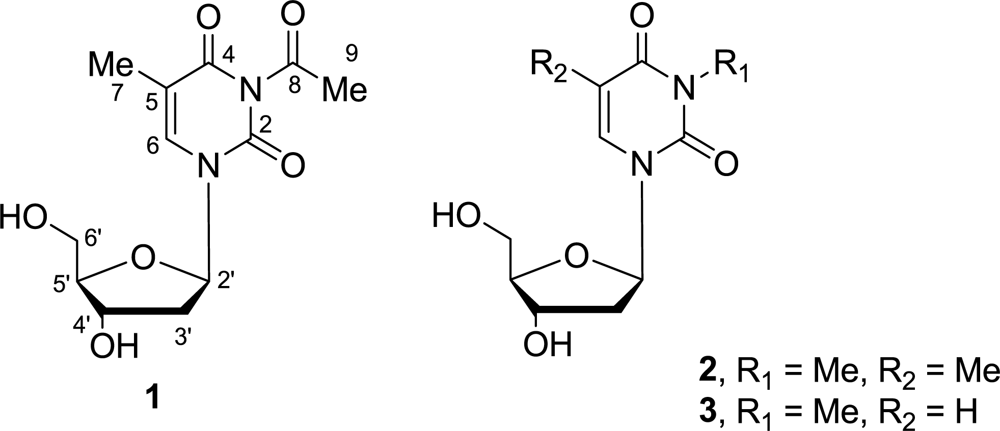
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. General
3.2. Microorganism Material
3.3. Extraction and Isolation
4. Conclusions
Acknowledgments
- Samples Availability: Samples of compounds 1–3 are available from the authors.
References
- Blunt, JW; Copp, BR; Munro, MHG; Northcote, PT; Prinsep, MR. Marine natural products. Nat Prod Rep 2010, 27, 165–237. [Google Scholar]
- Fortman, JL; Sherman, DH. Utilizing the power of microbial genetics to bridge the gap between the promise and the application of marine natural products. Chembiochem 2005, 6, 960–978. [Google Scholar]
- Kennedy, J; Baker, P; Piper, C; Cotter, PD; Walsh, M; Mooij, MJ; Bourke, MB; Rea, MC; O’Connor, PM; Ross, RP; et al. Isolation and analysis of bacteria with antimicrobial activities from the marine sponge Haliclona simulans collected from Irish water. Mar Biotechnol 2009, 11, 384–396. [Google Scholar]
- Li, Z. Advances in marine microbial symbionts in the China Sea and related pharmaceutical metabolites. Mar Drugs 2009, 7, 113–129. [Google Scholar]
- Lin, Z; Antemano, RR; Hughen, RW; Tianero, MDB; Peraud, O; Haygood, MG; Concepcion, GP; Olivera, BM; Light, A; Schmidt, EW. Pulicatins A–E, neuroactive thiazoline metabolites from cone snail-associate bacteria. J Nat Prod 2010, 73, 1922–1926. [Google Scholar]
- Oh, DC; Poulsen, M; Currie, CR; Clardy, J. Sceliphrolactam, a polyene macrocyclic lactam from a wasp-associated Streptomyces sp. Org Lett 2011, 13, 752–755. [Google Scholar]
- Piel, J; Butzke, D; Fusetani, N; Hui, D; Platzer, M; Wen, G; Matsunaga, S. Exploring the chemistry of uncultivated bacterial symbionts: Antitumor polyketides of the Pederin family. J Nat Prod 2005, 68, 472–479. [Google Scholar]
- Izumikawa, M; Khan, ST; Takagi, M; Shin-Ya, K. Sponge-derived Streptomyces producing isoprenoids via the mevalonate pathway. J Nat Prod 2010, 73, 208–212. [Google Scholar]
- Ashworth, DJ; Chang, C. Chemical modification of polynucleotides. Quantitiative studies of polycytidylic acid by nuclear magnetic resonance spectroscopy and secondary-ion mass spectrometry. J Org Chem 1981, 46, 4770–4773. [Google Scholar]
- Chang, C; Gomes, JD; Byrn, SR. Chemical modification of deoxyribonucleic acids: A direct study by carbon-13 nuclear magnetic resonance spectroscopy. J Org Chem 1983, 48, 5151–5160. [Google Scholar]
- Lidgren, G; Bohlin, L. Studies of Swedish marine organisms, part X. biologically active compounds from the marine sponge Geodia Baretti. J Nat Prod 1988, 51, 1277–1280. [Google Scholar]
- Zhang, H; Lee, YK; Zhang, W; Lee, HK. Culturable actinobacteria from the marine sponge Hymeniacidon perleve: Isolation and phylogenetic diversity by 16S rRNA gene-RFLP analysis. Antonie van Leeuwenhoek 2006, 90, 159–169. [Google Scholar]
- Xin, Y; Wu, P; Deng, M; Zhang, W. Phylogenetic diversity of the culturable rare actinomycetes in marine sponge Hymeniacidon perlevis by improved isolation media. Acta Microbiol Sin 2009, 49, 859–866. [Google Scholar]
- Cao, VT; Phan, VK; Bui, VH. Actinomycin X2, an antibiotic component from the antibiotic biosynthesis fermentation by. Streptomyces microflavus Tap Chi Hoa Hoc 2010, 48, 469–474. [Google Scholar]
- Uyeda, M; Yokomizo, K; Miyamoto, Y; Habib, E. Fattiviracin A1, a novel antiherpetic agent produced by Streptomyces microflavus strain No. 2445. I. Taxonomy, fermentation, isolation, physicochemical properties and structure elucidation. J Antibiot 1998, 51, 823–828. [Google Scholar]
- Yokomizo, K; Miyamoto, Y; Nagao, K; Kumagae, E; Suzuki, K; Harada, S; Uyeda, M. Fattiviracin A1, a novel antiviral agent produced by Streptomyces microflavus strain No. 2445. II. Biological properties. J Antibiot 1998, 51, 1035–1039. [Google Scholar]
- Habib, E; Yokomizo, K; Murata, K; Uyeda, M. Structures of fattiviracin family, antiviral antibiotics. J Antibiot 2000, 53, 1420–1423. [Google Scholar]
- el-Naggar, MY; el-Kersh, MA; el-Sharaky, AS. Correlation of actinomycin X2 to the lipid profile in static and shaken cultures of Streptomyces nasri strain YG62. Microbios 1999, 100, 117–127. [Google Scholar]
- Srivastav, NC; Shakya, N; Mak, M; Agrawal, B; Tyrrell, DL; Kumar, R. Antiviral activity of various 1-(2′-deoxy-β-d-lyxofuranosyl), 1-(2′-fluoro-β-d-xylofuranosyl), 1-(3′-fluoro-β-d-arabinofuranosyl), and 2′-fluoro-2′,3′-didehydro-2′,3′-dideoxyribose pyrimidine nucleoside analogues against duct hepatitis B virus (DHBV) and human hepatitis B virus (HBV) replication. J Med Chem 2010, 53, 7156–7166. [Google Scholar]
- Chacko, AM; Qu, W; Kung, HF. Synthesis and in vitro evaluation of 5-[18F]fluoroalkyl pyrimidine nucleosides for molecular imaging of herpes simplex virus type 1 thymidine kinase reporter gene expression. J Med Chem 2008, 51, 5690–5701. [Google Scholar]
- Kumar, R; Rai, D; Sharma, SK; Saffran, HA; Blush, R; Tyrrell, DL. Synthesis and antiviral activity of novel 5-(1-cyanamido-2-haloethyl) and 5-(1-hydeoxy (or methoxy)-2-azidoethyl) analogues of uracil nucleosides. J Med Chem 2001, 44, 3531–3538. [Google Scholar]
- Uyeda, M. Fattiviracins, antiviral antibiotics produced by an actinomycete. Actinomycetologica 2003, 17, 57–66. [Google Scholar]

© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, K.; Li, Q.-L.; Ji, N.-Y.; Liu, B.; Zhang, W.; Cao, X.-P. Deoxyuridines from the Marine Sponge Associated Actinomycete Streptomyces microflavus. Mar. Drugs 2011, 9, 690-695. https://doi.org/10.3390/md9050690
Li K, Li Q-L, Ji N-Y, Liu B, Zhang W, Cao X-P. Deoxyuridines from the Marine Sponge Associated Actinomycete Streptomyces microflavus. Marine Drugs. 2011; 9(5):690-695. https://doi.org/10.3390/md9050690
Chicago/Turabian StyleLi, Ke, Qiao-Lian Li, Nai-Yun Ji, Bo Liu, Wei Zhang, and Xu-Peng Cao. 2011. "Deoxyuridines from the Marine Sponge Associated Actinomycete Streptomyces microflavus" Marine Drugs 9, no. 5: 690-695. https://doi.org/10.3390/md9050690
APA StyleLi, K., Li, Q.-L., Ji, N.-Y., Liu, B., Zhang, W., & Cao, X.-P. (2011). Deoxyuridines from the Marine Sponge Associated Actinomycete Streptomyces microflavus. Marine Drugs, 9(5), 690-695. https://doi.org/10.3390/md9050690




