Antinociceptive and Anti-Inflammatory Activity from Algae of the Genus Caulerpa
Abstract
:1. Introduction
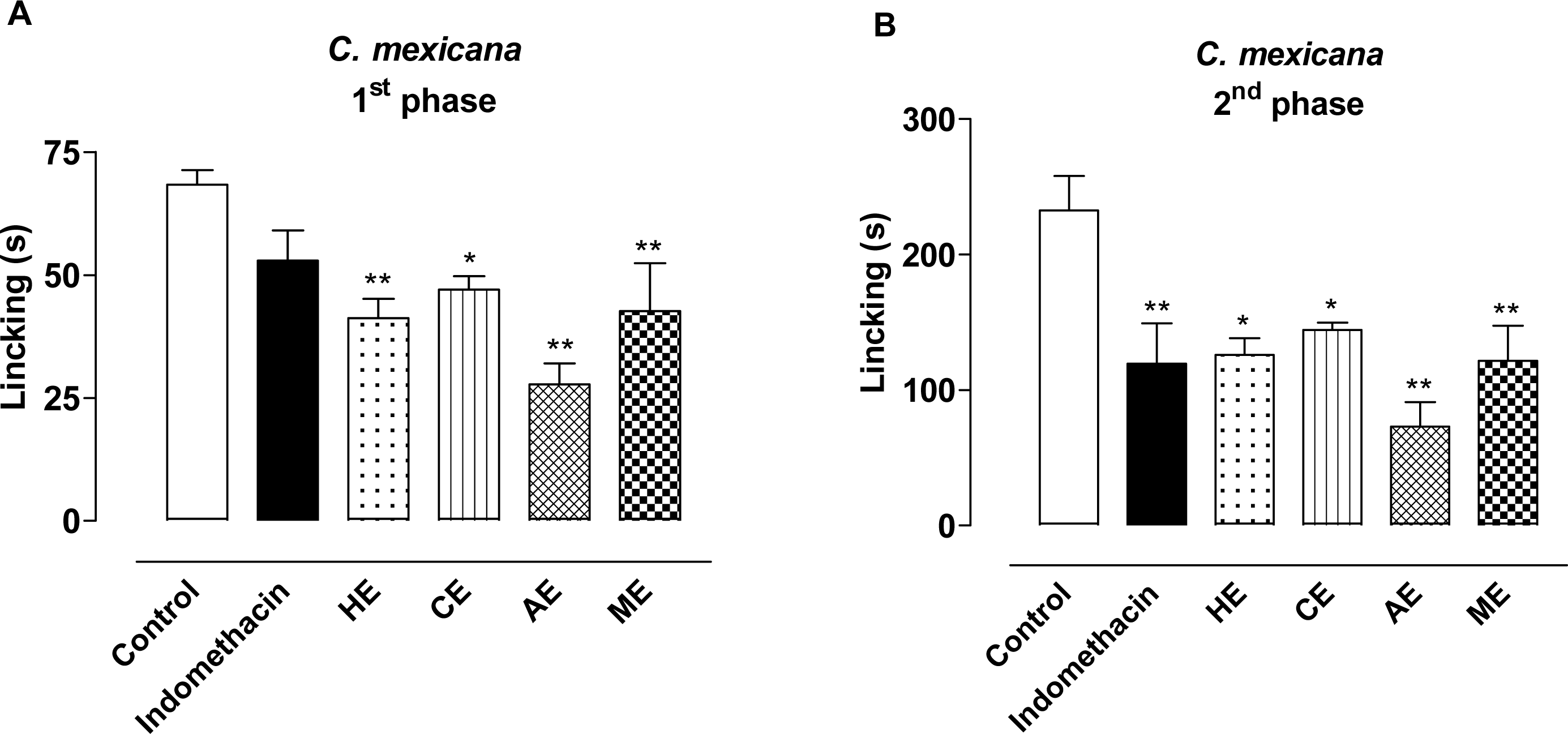
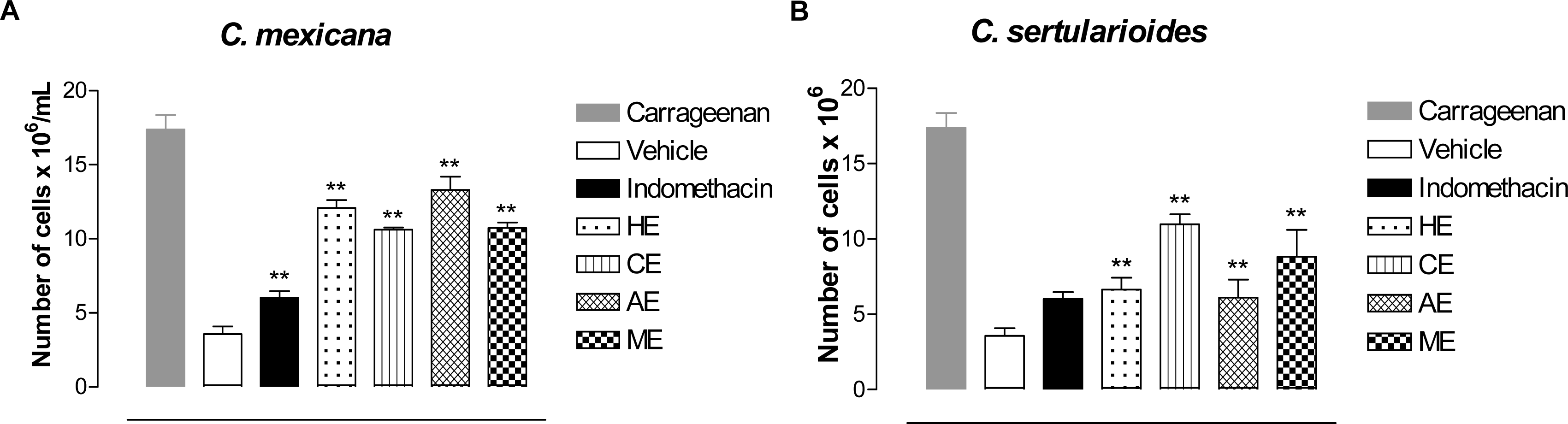
2. Results and Discussion
3. Experimental Section
3.1. Extraction and Isolation
3.2. Biological Activity Tests
3.2.1. Drugs and Reagents
3.2.2. Animals
3.2.3. Acetic Acid-Induced Writhing Test
3.2.4. Hot Plate Test
3.2.5. Formalin-Induced Nociception
3.2.6. Carrageenan-Induced Peritonitis in Mice
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Konig, GM; Wright, AD. Marine natural products research: current directions and future potential. Planta Med 1995, 62, 193–211. [Google Scholar]
- Sousa, FCF; Melo, CTV; Citó, MCO; Félix, FHC; Vasconcelos, SMM; Fonteles, MMF; Barbosa Filho, JM; Viana, GSB. Medicinal plants and their bioactive constituents: A scientific review of bioactivity and potential benefits in the anxiety disorders in animal models. Braz J Pharmacogn 2008, 18, 642–654. [Google Scholar]
- Corrêa, MFP; Melo, GO; Costa, SS. Natural products from plant origin potentially useful in the asthma therapy. Braz J Pharmacogn 2008, 18, 785–797. [Google Scholar]
- Barbosa-Filho, JM; Alencar, AA; Nunes, XP; Tomaz, ACA; Sena-Filho, JG; Athayde-Filho, PF; Silva, MS; Souza, MFV; Cunha, EVL. Sources of alpha-, beta-, gamma-, delta- and epsilon-carotenes: a twentieth century review. Braz J Pharmacogn 2008, 18, 135–154. [Google Scholar]
- Rocha, L; Almeida, JRGS; Macêdo, RO. A review of natural products with antileishmanial activity. Phytomedicine 2005, 12, 514–535. [Google Scholar]
- Silva, JSE; Moura, MD; Oliveira, RAG; Diniz, MFFM; Barbosa-Filho, JM. Natural products inhibitors of ovarian neoplasia. Phytomedicine 2003, 10, 221–232. [Google Scholar]
- Vuorella, P; Leinonenb, M; Saikkuc, P; Tammelaa, P; Rauhad, JP; Wenneberge, T; Vuorella, H. Natural products in the process of finding new drug candidates. Curr Med Chem 2004, 11, 1375–1389. [Google Scholar]
- Agra, MF; Silva, KN; Basílio, IJLD; França, PF; Barbosa-Filho, JM. Survey of medicinal plants used in the region Northeast of Brazil. Braz J Pharmacogn 2008, 18, 472–508. [Google Scholar]
- Almeida, RN; Navarro, DS; Barbosa-Filho, JM. Plants with central analgesic activity. Phytomedicine 2001, 8, 310–322. [Google Scholar]
- Truti, MCT; Ferreira, ICP; Zamuner, MLM; Nakamura, CV; Sarragiotto, MH; Souza, MC. Antiprotozoal and moluscicidal actives of five Brasilians plants. Braz J Med Biol Res 2005, 38, 1873–1878. [Google Scholar]
- Quintans-Júnior, LJ; Almeida, JRGS; Lima, JT; Nunes, XP; Siqueira, JS; Oliveira, LEG; Almeida, RN; Athayde-Filho, PF; Barbosa-Filho, JM. Plants with anticonvulsant properties—a review. Braz J Pharmacogn 2008, 18, 798–819. [Google Scholar]
- Mariath, IR; Falcão, HS; Barbosa-Filho, JM; Sousa, LCF; Tomaz, ACA; Batista, LM; Diniz, MFFM; Athayde-Filho, PF; Tavares, JF; Silva, MS; Cunha, EVL. Plants of the American continent with antimalarial activity. Braz J Pharmacogn 2009, 19, 158–192. [Google Scholar]
- Iannitti, T; Palmieri, B. An update on the therapeutic role of alkylglycerols. Mar Drugs 2010, 8, 2267–2300. [Google Scholar]
- Cabrita, MT; Vale, C; Rauter, AP. Halogenated compounds from marine algae. Mar Drugs 2010, 8, 2301–2317. [Google Scholar]
- Zhang, JL; Xia, WS; Liu, P; Cheng, QY; Tahirou, T; Gu, WX; Li, B. Chitosan modification and pharmaceutical/biomedical applications. Mar Drugs 2010, 8, 1962–1987. [Google Scholar]
- O’Sullivan, L; Murphy, B; McLoughlin, P; Duggan, P; Lawlor, PG; Hughes, H; Gardiner, GE. Prebiotics from marine macroalgae for human and animal health applications. Mar Drugs 2010, 8, 2038–2064. [Google Scholar]
- Aratake, S; Trianto, A; Hanif, N; de Voogd, NJ; Tanaka, J. A new polyunsaturated brominated fatty acid from a Haliclona sponge. Mar Drugs 2009, 7, 523–527. [Google Scholar]
- Lebouvier, N; Jullian, V; Desvignes, I; Maurel, S; Parenty, A; Dorin-Semblat, D; Doerig, C; Sauvain, M; Laurent, D. Antiplasmodial activities of homogentisic acid derivative protein kinase inhibitors isolated from a Vanuatu marine sponge Pseudoceratina sp. Mar Drugs 2009, 7, 640–653. [Google Scholar]
- Truti, MCT; Ferreira, ICP; Zamuner, MLM; Nakamura, CV; Sarragiotto, MH; Souza, MC. Antiprotozoal and moluscicidal actives of five Brasilians plants. Braz J Med Biol Res 2005, 38, 1873–1878. [Google Scholar]
- Medeiros, JD. A biotecnologia e a extinção de espécies, crises da modernidade. Rev Biotecnol Cienc Desenvolv 2003, 30, 109–113. [Google Scholar]
- Arif, JM; Al-Hazzani, AA; Kunhi, M; Al-Khodairy, F. Marine compounds: Anticancer or genotoxic. J Biomed Biotechnol 2004, 2, 93–98. [Google Scholar]
- Viegas, C, Jr; Bolzani, VS; Barreiro, EJ. Os produtos naturais e a química medicinal moderna. Quim Nova 2006, 29, 326–337. [Google Scholar]
- Cen-Pacheco, F; Nordstrom, L; Souto, ML; Martin, MN; Fernandez, JJ; Daranas, AH. Studies on polyethers produced by red algae. Mar Drugs 2010, 8, 1178–1188. [Google Scholar]
- Guven, KC; Percot, A; Sezik, E. Alkaloids in marine algae. Mar Drugs 2010, 8, 269–284. [Google Scholar]
- Stafford, HA. Flavonoid evolution—an enzymatic approach. Plant Physiol 1991, 96, 680–685. [Google Scholar]
- Serrano, J; Puupponen-Pimia, R; Dauer, A; Aura, AM; Saura-Calixto, F. Tannins: Current knowledge of food sources, intake, bioavailability and biological effects. Mol Nutr Food Res 2009, 53, S310–S329. [Google Scholar]
- Narkowicz, CK; Blackman, AJ. Further acetogenins from Tasmanian collections of Caulocystis cephalornithos demonstrating chemical variability. Biochem Syst Ecol 2006, 34, 635–641. [Google Scholar]
- Teixeira, VL; Kelecom, A; Gottlieb, OR. Produtos naturais de algas marinhas. Quim Nova 1991, 14, 83–90. [Google Scholar]
- Pereira, SMB; Oliveira-Carvalho, MF; Angeiras, JAP; Oliveira, NMB; Torres, J; Gestinari, LM; Badeira-Pedrosa, ME; Cocentino, ALM; Santos, MD; Nascimento, PRF; Cavalcanti, DR. Algas bentônicas do Estado de Pernambuco. In Diagnóstico da Biodiversidade de Pernambuco; Tabarelli, M, Silva, JMC, Eds.; Massagana e SECTMA: Recife, Brazil, 2002; pp. 97–124. [Google Scholar]
- Lima-Filho, JVM; Carvalho, AFFU; Freitas, SM; Melo, VMM. Antibacterial activity of extracts of six macroalgae from the Northeastern Brazilian coast. Braz J Microbiol 2002, 33, 311–313. [Google Scholar]
- Li, ZY. Advances in marine microbial symbionts in the China sea and related pharmaceutical metabolites. Mar Drugs 2009, 7, 113–129. [Google Scholar]
- Machado, FLD; Kaiser, CR; Costa, SS; Gestinari, LM; Soares, AR. Braz J Pharmacogn 2010, 20, 441–452.
- Taskin, E; Caki, Z; Ozturk, M; Taskin, E. Assessment of in vitro antitumoral and antimicrobial activities of marine algae harvested from the Eastern Mediterranean sea. Afr J Biotechnol 2010, 9, 4272–4277. [Google Scholar]
- Dias, PF; Siqueira, JM; Vendruscolo, LF; Neiva, TD; Gagliardi, AR; Maraschin, M; Ribeiro-Do-Valle, RM. Antiangiogenic and antitumoral properties of a polysaccharide isolated from the seaweed Sargassum stenophyllum. Cancer Chemother Pharmacol 2005, 56, 436–446. [Google Scholar]
- Matsubara, K; Xue, C; Zhao, X; Mori, M; Sugawara, T; Hirata, T. Effects of middle molecular weight fucoidans on in vitro and ex vivo angiogenesis of endothelial cells. Int J Mol Med 2005, 15, 695–699. [Google Scholar]
- Romanos, MTV; Andrada-Serpa, MJ; Santos, MGM; Ribeiro, ACF; Yoneshiguevalentin, Y; Costa, SS; Wigg, MD. Inhibitory effect of extracts of Brazilian marine algae on human T-cell lymphotropic virus type 1 (HTLV-1) induced syncytium formation in vitro. Cancer Investig 2002, 20, 46–54. [Google Scholar]
- Mayer, AMS; Rodriguez, AD; Berlinck, RGS; Hamann, MT. Marine pharmacology in 2005–6: Marine compounds with anthelmintic, antibacterial, anticoagulant, antifungal, anti-inflammatory, antimalarial, antiprotozoal, antituberculosis, and antiviral activities; affecting the cardiovascular, immune and nervous systems, and other miscellaneous mechanisms of action. Biochim Biophys Acta Gen Subj 2009, 1790, 283–308. [Google Scholar]
- Genovese, G; Tedone, L; Hamann, MT; Morabito, M. The mediterranean red alga Asparagopsis: A source of compounds against. Leishmania Mar Drugs 2009, 7, 361–366. [Google Scholar]
- Rocha, FD; Pereira, RC; Kaplan, MAC; Teixeira, VL. Natural products from marine seaweeds and their antioxidant potential. Braz J Pharmacogn 2007, 17, 631–639. [Google Scholar]
- Souza, ET; Queiroz, AC; Miranda, GEC; Lorenzo, VP; Silva, EF; Freire-Dias, TLM; Cupertino-Silva, YK; Melo, GMA; Chaves, MCO; Barbosa-Filho, JM; Athayde-Filho, PF; Santos, BVO; Alexandre-Moreira, MS. Antinociceptive activities of crude methanolic extract and phases, n-butanolic, chloroformic and ethyl acetate from Caulerpa racemosa (Caulerpaceae). Braz J Pharmacogn 2009, 19, 115–120. [Google Scholar]
- Bars, D; Gozariu, M; Cadden, SW. Animal models of nociception. Pharmacol Rev 2001, 53, 597–652. [Google Scholar]
- Duarte, IDG; Nakamura, M; Ferreira, SH. Participation of the sympathetic system in acetic acid-induced writhing in mice. Braz J Med Biol Res 1988, 21, 341–343. [Google Scholar]
- Collier, HOJ; Dinneen, JC; Johnson, CA; Schneider, C. The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br J Pharmacol Chemother 1968, 32, 295–310. [Google Scholar]
- Ribeiro, RA; Vale, ML; Thomazzi, SM; Paschoalato, AB; Poole, S; Ferreira, SH; Cunha, FQ. Involvement of resident macrophages and mast cells in the writhing nociceptive response induced by zymosan and acetic acid in mice. Eur J Pharmacol 2000, 387, 111–118. [Google Scholar]
- Chu, C; Huang, Y; Chen, YF; Wu, JH; Rahman, K; Zheng, HC; Qin, LP. Anti-nociceptive activity of aqueous fraction from the MeOH extracts of Paederia scandens in mice. J Ethnopharmacol 2008, 118, 177–180. [Google Scholar]
- Dickenson, AH; Sullivan, AF. Peripheral origins and central modulation of subcutaneous formalin-induced activity of rat dorsal horn neurons. Neurosci Lett 1987, 83, 207–211. [Google Scholar]
- Yaksh, TL; Ozaki, G; McCumber, D; Rathbun, M; Svensson, C; Malkmus, S; Yaksh, MC. An automated flinch detecting system for use in the formalin nociceptive bioassay. J Appl Physiol 2001, 90, 2386–2402. [Google Scholar]
- García, MD; Fernandez, MA; Alvarez, A; Saenz, MT. Anti-nociceptive and anti-inflammatory effect of the aqueous extract from leaves of Pimenta racemosa var. ozua (Mirtaceae). J Ethnopharmacol 2004, 91, 69–73. [Google Scholar]
- Zimmermann, M. Ethical guidelines for investigation of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar]
- Koster, R; Anderson, M; De-Beer, EJ. Acetic acid analgesic screen. Fed Proc 1959, 18, 418–420. [Google Scholar]
- Eddy, NB; Leimbach, D. Synthetic analgesics. II. Dithienylbutenyland dithienylbutylamines. J Pharmacol Exp Ther 1953, 107, 385–393. [Google Scholar]
- Hunskaar, S; Hole, K. The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar]
- Tjolsen, A; Hole, K. Animal models of analgesia. In The Pharmacology of Pain; Dickenson, A, Besson, J, Eds.; Springer Verlag: Berlin, Germany, 1997; Volume 130, pp. 1–20. [Google Scholar]
- Ferrándiz, ML; Alcaraz, MJ. Antiinflammatory activity and inhibition of arachidonic acidmetabolism by flavonoids. Inflamm Res 1991, 32, 283–288. [Google Scholar]

| C. mexicana | C. sertularioides | |||
|---|---|---|---|---|
| Number of writhing | ||||
| Treatment | Mean ± S.E.M. a | I (%) b | Mean ± S.E.M. a | I (%) b |
| Vehicle | 35.5 ± 1.6 | – | 34.9 ± 1.9 | – |
| Dypirone | 8.0 ± 2.3 | 86.5 ** | 5.9 ± 1.7 | 83.2 ** |
| HE | 6.0 ± 1.4 | 83.1 ** | 11.7 ± 2.0 | 66.5 ** |
| CE | 8.2 ± 2.3 | 77.0 ** | 11.5 ± 0.9 | 67.0 ** |
| AE | 9.5 ± 1.4 | 73.2 ** | 13.7 ± 2.8 | 60.7 ** |
| ME | 7.7 ± 0.7 | 78.4 ** | 11.5 ± 1.8 | 67.0 ** |
| Post-treatment (min) a | ||||||
|---|---|---|---|---|---|---|
| Animal Group | 0 min | 30 min | 60 min | 90 min | 120 min | 150 min |
| Time latency (s) | ||||||
| Control | 1.4 ± 0.3 | 2.2 ± 0.6 | 1.8 ± 0.2 | 3.2 ± 0.3 | 2.8 ± 0.3 | 2.6 ± 0.5 |
| Morphine | 6.9 ± 0.4 | 5.8 ± 0.3 | 12.8 ± 0.4 ** | 10.3 ± 0.8 ** | 9.7 ± 0.7 ** | 9.7 ± 0.9 ** |
| ME | 3.5 ± 0.5 | 3.0 ± 0.4 | 4.5 ± 0.3 | 5.4 ± 0.6 * | 5.1 ± 0.8 | 5.4 ± 0.4 * |
| AE | 1.7 ± 0.4 | 3.4 ± 0.3 | 3.1 ± 0.5 | 5.3 ± 0.9 ** | 4.1 ± 0.5 | 5.9 ± 1.3 ** |
| CE | 2.7 ± 0.2 | 3.7 ± 0.5 | 4.4 ± 1.0 | 5.7 ± 0.7 * | 3.6 ± 0.6 | 3.9 ± 1.0 |
| HE | 2. 7 ± 0.3 | 2.8 ± 0.6 | 4.6 ± 0.7 | 5.8 ± 0.9 * | 5.9 ± 1.0 * | 5.1 ± 0.8 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brito da Matta, C.B.; De Souza, É.T.; De Queiroz, A.C.; De Lira, D.P.; De Araújo, M.V.; Cavalcante-Silva, L.H.A.; De Miranda, G.E.C.; De Araújo-Júnior, J.X.; Barbosa-Filho, J.M.; De Oliveira Santos, B.V.; et al. Antinociceptive and Anti-Inflammatory Activity from Algae of the Genus Caulerpa. Mar. Drugs 2011, 9, 307-318. https://doi.org/10.3390/md9030307
Brito da Matta CB, De Souza ÉT, De Queiroz AC, De Lira DP, De Araújo MV, Cavalcante-Silva LHA, De Miranda GEC, De Araújo-Júnior JX, Barbosa-Filho JM, De Oliveira Santos BV, et al. Antinociceptive and Anti-Inflammatory Activity from Algae of the Genus Caulerpa. Marine Drugs. 2011; 9(3):307-318. https://doi.org/10.3390/md9030307
Chicago/Turabian StyleBrito da Matta, Carolina Babosa, Éverton Tenório De Souza, Aline Cavalcanti De Queiroz, Daysianne Pereira De Lira, Morgana Vital De Araújo, Luiz Henrique Agra Cavalcante-Silva, George Emmanuel C. De Miranda, João Xavier De Araújo-Júnior, José Maria Barbosa-Filho, Bárbara Viviana De Oliveira Santos, and et al. 2011. "Antinociceptive and Anti-Inflammatory Activity from Algae of the Genus Caulerpa" Marine Drugs 9, no. 3: 307-318. https://doi.org/10.3390/md9030307
APA StyleBrito da Matta, C. B., De Souza, É. T., De Queiroz, A. C., De Lira, D. P., De Araújo, M. V., Cavalcante-Silva, L. H. A., De Miranda, G. E. C., De Araújo-Júnior, J. X., Barbosa-Filho, J. M., De Oliveira Santos, B. V., & Alexandre-Moreira, M. S. (2011). Antinociceptive and Anti-Inflammatory Activity from Algae of the Genus Caulerpa. Marine Drugs, 9(3), 307-318. https://doi.org/10.3390/md9030307




