Effects and Interactions of Medium Components on Laccase from a Marine-Derived Fungus Using Response Surface Methodology
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Microorganism and culture conditions
3.2. Culture medium
3.3. Screening of significant components using Plackett Burman Design
3.4. Effect of carbon and nitrogen interaction using Central Composite Design
3.5. Impact of addition of CuSO4 and Tween 80
3.6. Effect of flask capacity
3.7. Experimental design and statistical analysis
3.8. Responses measured
4. Conclusions
Abbreviations:
| ABTS | [2,2′-azino-bis-(3-ethylbenzothazoline-6-sulfonate)]; |
| CCD | Central Composite Design; |
| PBD | Plackett Burman Design |
Acknowledgments
- Samples Availability: The fungus Cerrena unicolor MTCC 5159 is a patented culture under the Budapest Treaty.
References
- Thurston, CF. The structure and function of fungal laccases. Microbiology 1994, 140, 19–26. [Google Scholar]
- Mayer, AM; Staples, RC. Laccase: new functions for an old enzyme. Phytochemistry 2002, 60, 551–565. [Google Scholar]
- D’Souza-Ticlo, D; Sharma, D; Raghukumar, C. A thermostable metal-tolerant laccase with bioremediation potential from a marine-derived fungus. Mar Biotechnol 2009. [Google Scholar]
- D’Souza, DT; Tiwari, R; Sah, AK; Raghukumar, C. Enhanced production of laccase by a marine fungus during treatment of colored effluents and synthetic dyes. Enzyme Microb Technol 2006, 38, 504–511. [Google Scholar]
- Gao, H; Liu, M; Liu, J; Dai, H; Zhou, X; Liu, X; Zhuo, Y; Zhang, W; Zhang, L. Medium optimization for the production of avermectin B1a by Streptomyces avermitilis 14–12A using response surface methodology. Bioresour Technol 2009, 100, 4012–4016. [Google Scholar]
- Martin, C; Corvini, PF; Vinken, R; Junghanns, C; Krauss, G; Schlosser, D. Quantification of the influence of extracellular laccase and intracellular reactions on the isomer-specific biotransformation of the xenoestrogen technical nonylphenol by the aquatic hyphomycete Clavariopsis aquatica. Appl Environ Microbiol 2009, 75, 4398–4409. [Google Scholar]
- Junghanns, C; Pecyna, MJ; Bohm, D; Jehmlich, N; Martin, C; von Bergen, M; Schauer, F; Hofrichter, M; Schlosser, D. Biochemical and molecular genetic characterisation of a novel laccase produced by the aquatic ascomycete Phoma sp. UHH 5-1-03. Appl Microbiol Biotechnol 2009, 84, 1095–1105. [Google Scholar]
- Pointing, SB; Hyde, KD. Lignocellulose-degrading marine fungi. Biofouling 2000, 15, 221–229. [Google Scholar]
- Li, X; Kondo, R; Sakai, K. Studies on hypersaline-tolerant white-rot fungi. II. Biodegradation of sugarcane bagasse with marine fungus Phlebia sp. MG-60. J Wood Sci 2002, 48, 159–162. [Google Scholar]
- Jensen, PR; Fenical, W. Secondary metabolites from marine fungi. In Fungi in Marine Environments; Hyde, KD, Ed.; Fungal Diversity Press: Hong Kong, China, 2002; pp. 293–315. [Google Scholar]
- Verma, AK; Raghukumar, C; Verma, P; Shouche, YS; Naik, CG. Four marine-derived fungi for bioremediation of raw textile mill effluents. Biodegradation.
- D’Souza-Ticlo, DT. The Lignin-Degrading Enzyme, Laccase from Marine Fungi; Biochemical and Molecular Approaches. . PhD Thesis, Goa University, Dona Paula, Goa, India, 2008. [Google Scholar]
- Stepanova, EV; Pegasova, TV; Gavrilova, VP; Landesman, EO; Koroleva, OV. Extracellular Laccases from Cerrena unicolor 059, Cerrena unicolor 0784, and Pleurotus ostreatus 0432: A Comparative Assay. Appl Biochem Microbiol 2002, 39, 375–381. [Google Scholar]
- Michniewicz, A; Ullrich, R; Ledakowicz, S; Hofrichter, M. The white-rot fungus Cerrena unicolor strain 137 produces two laccase isoforms with different physico-chemical and catalytic properties. Appl Biochem Microbiol 2006, 69, 682–688. [Google Scholar]
- Arora, DS; Gill, PK. Laccase production by some white rot fungi under different nutritional conditions. Bioresour Technol 2000, 73, 283–285. [Google Scholar]
- Vasconcelos, AF; Barbosa, AM; Dekker, RFH; Scarminio, IS; Rezende, MI. Optimization of laccase production by Botryosphaeria sp. in the presence of veratryl alcohol by the response-surface method. Process Biochem 2000, 35, 1131–1138. [Google Scholar]
- Zadrazil, F; Gonser, A; Lang, E. Influence of incubation temperature on the secretion of extracellular ligninolytic enzymes of Pleurotus sp. and Dichomitus squalens into soil. Proceedings of the conference on Enzymes in the environment: Activity, Ecology and Applications, Granada, Spain, July 1999.
- Baldrian, P. Interactions of heavy metals with white-rot fungi. Enzyme Microb Technol 2003, 32, 78–91. [Google Scholar]
- Palmieri, G; Giardina, P; Bianco, C; Fontanella, B; Sannia, G. Copper induction of laccase isoenzymes in the ligninolytic fungus Pleurotus ostreatus. Appl Environ Microbiol 2000, 66, 920–924. [Google Scholar]
- Eggert, C; Temp, U; Karl-Erik; Eriksson, K-EI. The ligninolytic system of the white rot fungus Pycnoporus cinnabarinus: purification and characterization of the laccase. Appl Environ Microbiol 1996, 62, 1151–1158. [Google Scholar]
- Monteiro, M; de Carvalho, M. Pulp bleaching using laccase from Trametes versicolor under high temperature and alkaline conditions. Appl Biochem Biotechnol 1998, 70–72, 983–993. [Google Scholar]
- Bollag, J-M; Leonowicz, A. Comparative studies of extracellular fungal laccases. Appl Environ Microbiol 1984, 48, 849–854. [Google Scholar]
- D’Souza-Ticlo, D; Verma, AK; Mathew, M; Raghukumar, C. Effect of nutrient nitrogen on laccase production, its isozyme pattern and effluent decolorization by a fungus isolated from mangrove wood. Indian J Mar Sci 2006, 35, 364–372. [Google Scholar]
- Keyser, P; Kirk, TK; Zeikus, JG. Ligninolytic enzyme system of Phanerochaete chrysosporium: Synthesized in the absence of lignin in response to nitrogen starvation. J Bacteriol 1978, 135, 790–797. [Google Scholar]
- Hou, H; Zhou, J; Wang, J; Du, C; Yan, B. Enhancement of laccase production by Pleurotus ostreatus and its use for the decolorization of anthraquinone dye. Process Biochem 2004, 39, 1415–1419. [Google Scholar]
- Froehner, ST; Eriksson, K-E. Induction of Neurosporo crassa laccase with protein synthesis inhibitors. J Bacteriol 1974, 120, 450–457. [Google Scholar]
- Lee, I-Y; Jung, K-H; Lee, C-H; Park, Y-H. Enhanced production of laccase in Trametes versicolor by the addition of ethanol. Biotechnol Lett 1999, 21, 965–968. [Google Scholar]
- Sannia, G; Faraco, V; Giardina, P; Palmieri, G. Metal-activated laccase promoters. Proceedings of the 8th International Conference on Biotechnology in the Pulp and Paper Industry, Helsinki, Finland, June 2001.
- Cserhati, T. Alkyl ethoxylated and alkylphenol ethoxylated nonionic surfactants: Interaction with bioactive compounds and biological effects. Environ Health Perspect 1995, 103, 358–364. [Google Scholar]
- Heinzkill, M; Bech, L; Halkier, T; Schneider, P; Anke, T. Characterization of laccase and peroxidase from wood-rotting fungi (family Coprinaceae). Appl Environ Microbiol 1998, 64, 1601–1606. [Google Scholar]
- Xavier, AMRB; Evtuguin, DV; Ferreira, RMP; Amado, FL. Laccase production for lignin oxidative activity. Proceedings of the 8th International Conference on Biotechnology in the Pulp and Paper Industry, Helsinki, Finland, June 2001.
- Bourbonnais, R; Paice, MG; Reid, ID; Lanthier, P; Yaguchi, M. Lignin oxidation by laccase isozymes from Trametes versicolor and role of the mediator 2,2′-azinobis (3-thylbenzthiazoline-6-sulfonate) in kraft lignin depolymerization. Appl Environ Microbiol 1995, 61, 1876–1880. [Google Scholar]
- Butt, TM; Jackson, CW; Magen, N. Fungi as Biocontrol Agents; Progress, Problems and Potential; CABI Publishing: Wallingford, UK, 2001. [Google Scholar]
- Kohlmeyer, J; Kohlmeyer, E. Marine Mycology: The Higher Fungi; Academic Press: New York, NY, USA, 1979. [Google Scholar]
- Tien, M; Kirk, TK. Lignin peroxidase of Phanerochaete chrysosporium. Meth Enzymol 1988, 161, 238–249. [Google Scholar]
- Niku-Paavola, ML; Karhuner, E; Salola, P; Raunio, V. Lignolytic enzymes of the white-rot fungus. Phlebia radiata Biochem J 1988, 254, 877–884. [Google Scholar]
- Bradford, MM. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976, 72, 248–254. [Google Scholar]
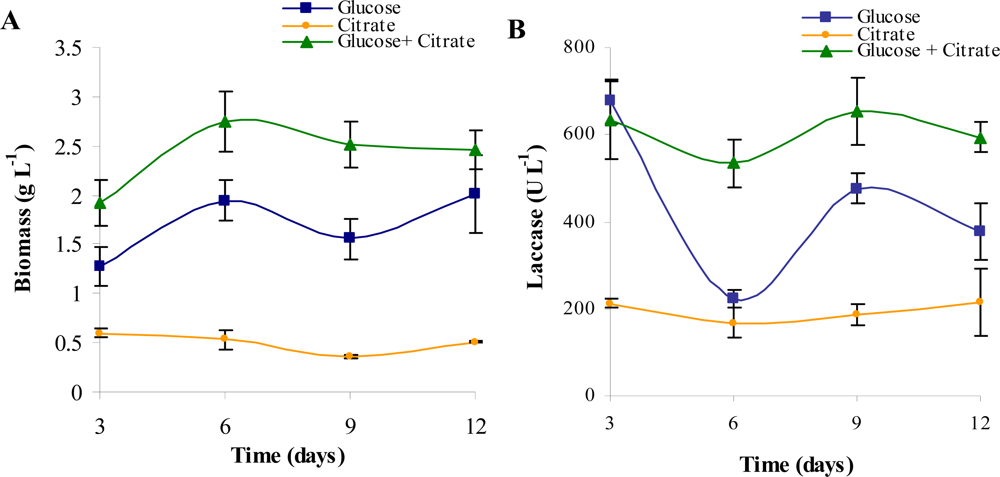

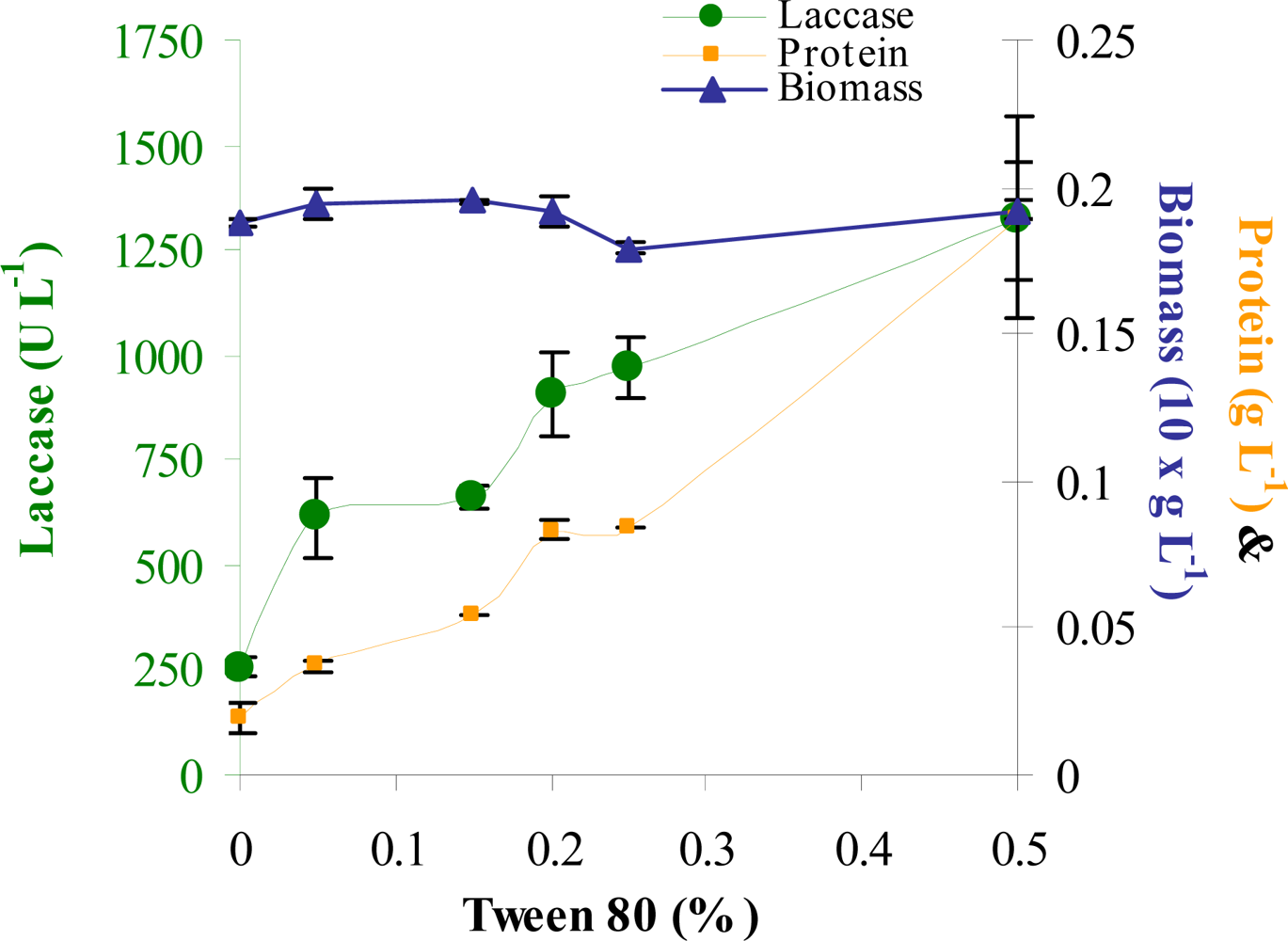
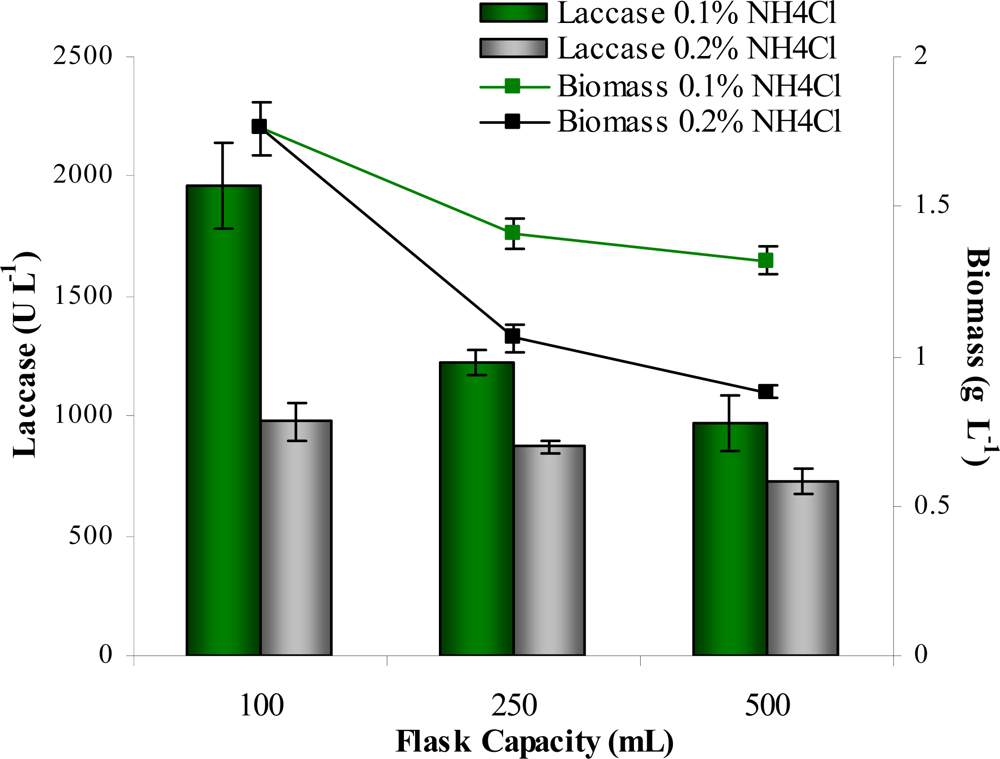
| Trial | Variables | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Coded | Uncoded | |||||||||
| Glucose | NH4Cl | CuSO4 | Tween 80 | NaCl | Glucose (%) | NH4Cl (%) | CuSO4 (mM) | Tween 80 (%) | NaCl (%) | |
| A | − | − | − | − | + | 0.9 | 0.2044 | 0.0375 | 0.05 | 1 |
| B | − | − | + | + | − | 0.9 | 0.2044 | 0.375 | 0.25 | 0.1 |
| C | − | + | − | + | − | 0.9 | 1.022 | 0.0375 | 0.25 | 0.1 |
| D | − | + | + | − | + | 0.9 | 1.022 | 0.375 | 0.05 | 1 |
| E | + | − | − | + | + | 4.5 | 0.2044 | 0.0375 | 0.25 | 1 |
| F | + | − | + | − | − | 4.5 | 0.2044 | 0.375 | 0.05 | 0.1 |
| G | + | + | − | − | − | 4.5 | 1.022 | 0.0375 | 0.05 | 0.1 |
| H | + | + | + | + | + | 4.5 | 1.022 | 0.375 | 0.25 | 1 |
| Set | Biomass (g L−1) | Laccase (U L−1) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Sampling Interval (Days) | ||||||||||
| 3 | 6 | 9 | 12 | 15 | 3 | 6 | 9 | 12 | 15 | ||
| Unspiked | A | 0.96 | 1.21 | 1.30 | 1.08 | 1.70 | 807 | 901 | 888 | 48 | 3 |
| B | 0.48 | 2.59 | 3.37 | 3.36 | 2.44 | 17 | 2926 | 2358 | 1393 | 760 | |
| C | 0.57 | 1.00 | 1.25 | 1.32 | 1.55 | 809 | 1651 | 792 | 3 | 220 | |
| D | 0.38 | 0.51 | 0.62 | 0.34 | 1.15 | 8 | 33 | 908 | 3 | 84 | |
| E | 0.89 | 1.85 | 2.60 | 2.52 | 2.12 | 6 | 1122 | 805 | 1070 | 156 | |
| F | 1.00 | 2.52 | 3.68 | 3.47 | 2.56 | 10 | 1815 | 1744 | 1787 | 662 | |
| G | 1.08 | 0.93 | 1.61 | 1.49 | 1.80 | 802 | 541 | 178 | 178 | 122 | |
| H | 0.30 | 1.17 | 1.79 | 1.75 | 1.72 | 3 | 253 | 825 | 624 | 434 | |
| Spiked | A’ | 0.96 | 1.21 | 0.93 | 1.21 | 1.50 | 807 | 901 | 502 | 254 | 145 |
| B’ | 0.48 | 2.59 | 3.62 | 3.12 | 2.41 | 17 | 2926 | 320 | 74 | 67 | |
| C’ | 0.57 | 1.00 | 1.08 | 1.10 | 1.24 | 809 | 1651 | 288 | 82 | 72 | |
| D’ | 0.38 | 0.51 | 0.11 | 0.36 | 0.76 | 8 | 33 | 301 | 88 | 73 | |
| E’ | 0.89 | 1.85 | 2.44 | 2.26 | 2.09 | 6 | 1122 | 302 | 62 | 65 | |
| F’ | 1.00 | 2.52 | 3.64 | 3.51 | 2.62 | 10 | 1815 | 110 | 91 | 71 | |
| G’ | 1.08 | 0.93 | 1.15 | 1.62 | 1.55 | 802 | 541 | 100 | 102 | 82 | |
| H’ | 0.30 | 1.17 | 1.58 | 1.43 | 1.37 | 3 | 253 | 67 | 54 | 45 | |
| Variables | Sampling Interval (Days) | ||||||||||||||
| 3 | 6 | 9 | 12 | 15 | |||||||||||
| B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | |
| Intercept | 1.250 | 0.016 | 0.0000 | 1.916 | 0.016 | 0.0000 | 2.310 | 0.060 | 0.0000 | 2.107 | 0.092 | 0.0000 | 2.142 | 0.152 | 0.0000 |
| Glucose | 0.061 | 0.003 | 0.0000 | 0.081 | 0.003 | 0.0000 | 0.217 | 0.010 | 0.0000 | 0.217 | 0.016 | 0.0000 | 0.095 | 0.026 | 0.0043 |
| NH4Cl | −0.305 | 0.012 | 0.0000 | −1.393 | 0.012 | 0.0000 | −1.739 | 0.045 | 0.0000 | −1.692 | 0.069 | 0.0000 | −0.794 | 0.113 | 0.0000 |
| CuSO4 | −0.994 | 0.029 | 0.0000 | 1.327 | 0.030 | 0.0000 | 1.999 | 0.108 | 0.0000 | 1.854 | 0.167 | 0.0000 | 0.525 | 0.275 | 0.0849 |
| Tween 80 | −1.469 | 0.048 | 0.0000 | 1.798 | 0.050 | 0.0000 | 2.242 | 0.182 | 0.0000 | 3.213 | 0.282 | 0.0000 | 0.780 | 0.464 | 0.1235 |
| NaCl | −0.172 | 0.011 | 0.0000 | −0.639 | 0.011 | 0.0000 | −1.002 | 0.041 | 0.0000 | −1.096 | 0.063 | 0.0000 | −0.465 | 0.103 | 0.0011 |
| Adjusted r2 = 0.9958 | Adjusted r2 = 0.999 | Adjusted r2 = 0.9952 | Adjusted r2 = 0.9890 | Adjusted r2 = 0.84900 | |||||||||||
| S.E.E.: 0.0193 | S.E.E.: 0.020 | S.E.E.: 0.0729 | S.E.E.: 0.1129 | S.E.E.: 0.18545 | |||||||||||
| Sampling Interval (Days) | |||||||||||||||
| Variables | A spike of 0.375 mM CuSO4 was troduced on day 6. | 9 | 12 | 15 | |||||||||||
| B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | |||||||
| Intercept | 2.126 | 0.092 | 0.0000 | 2.364 | 0.160 | 0.0000 | 2.141 | 0.1604 | 0.0000 | ||||||
| Glucose | 0.213 | 0.016 | 0.0000 | 0.211 | 0.027 | 0.0000 | 0.119 | 0.0272 | 0.0014 | ||||||
| NH4Cl | −2.053 | 0.069 | 0.0000 | −1.710 | 0.120 | 0.0000 | −1.130 | 0.1197 | 0.0000 | ||||||
| CuSO4 | 2.481 | 0.167 | 0.0000 | 1.653 | 0.290 | 0.0002 | 0.578 | 0.2900 | 0.0741 | ||||||
| Tween 80 | 3.605 | 0.282 | 0.0000 | 1.502 | 0.489 | 0.0118 | 0.840 | 0.4894 | 0.1170 | ||||||
| NaCl | −1.231 | 0.063 | 0.0000 | −1.137 | 0.109 | 0.0000 | −0.583 | 0.1088 | 0.0003 | ||||||
| Adjusted r2 = 0.9919 | Adjusted r2 = 0.96483 | Adjusted r2 = 0.90251 | |||||||||||||
| S.E.E.: 0.1129 | S.E.E.: 0.19546 | S.E.E.: 0.19576 | |||||||||||||
| Variables | Sampling Interval (Days) | |||||||||||||||
| 3 | 6 | 9 | 12 | 15 | ||||||||||||
| B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | ||
| Intercept | 952 | 146 | 0.000 | 2375 | 30 | 0.000 | 1471 | 26 | 0.000 | 540 | 86 | 0.000 | 198 | 37 | 0.000 | |
| Glucose | −57 | 25 | 0.044 | −124 | 5 | 0.000 | −97 | 4 | 0.000 | 154 | 15 | 0.000 | 21 | 6 | 0.007 | |
| NH4Cl | 239 | 109 | 0.053 | −1310 | 23 | 0.000 | −946 | 19 | 0.000 | −1067 | 64 | 0.000 | −221 | 28 | 0.000 | |
| CuSO4 | −1768 | 264 | 0.000 | 602 | 55 | 0.000 | 2350 | 46 | 0.000 | 1858 | 155 | 0.000 | 1067 | 67 | 0.000 | |
| Tween 80 | −991 | 446 | 0.051 | 3329 | 92 | 0.000 | 1329 | 78 | 0.000 | 1343 | 262 | 0.000 | 873 | 113 | 0.000 | |
| NaCl | −226 | 99 | 0.046 | −1284 | 20 | 0.000 | −457 | 17 | 0.000 | −449 | 58 | 0.000 | −302 | 25 | 0.000 | |
| Adjusted r2 = 0.7997 | Adjusted r2 = 0.998 | Adjusted r2 = 0.998 | Adjusted r2 = 0.9761 | Adjusted r2 = 0.9725 | ||||||||||||
| S.E.E.: 178.5200 | S.E.E.: 36.884 | S.E.E.: 31.245 | S.E.E.: 104.7401 | S.E.E.: 45.1227 | ||||||||||||
| Variables | Sampling Interval (Days) | |||||||||||||||
| A spike of 0.375 mM CuSO4 was introduced on day 6. | 9 | 12 | 15 | |||||||||||||
| B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | B | Std. Err. of B | p-level | ||||||||
| Intercept | 507 | 9.5 | 0.000 | 228 | 29 | 0.000 | 143 | 10 | 0.000 | |||||||
| Glucose | −58 | 1.6 | 0.000 | −13 | 5 | 0.024 | −7 | 2 | 0.003 | |||||||
| NH4Cl | −146 | 7.1 | 0.000 | −47 | 22 | 0.054 | −23 | 7 | 0.010 | |||||||
| CuSO4 | −292 | 17.1 | 0.000 | −143 | 53 | 0.022 | −80 | 18 | 0.001 | |||||||
| Tween 80 | −45 | 28.9 | 0.151 | −329 | 89 | 0.004 | −153 | 30 | 0.000 | |||||||
| NaCl | 98 | 6.4 | 0.000 | 30 | 20 | 0.155 | 10 | 7 | 0.161 | |||||||
| Adjusted r2 = 0.9956 | Adjusted r2 = 0.66959 | Adjusted r2 = 0.82375 | ||||||||||||||
| S.E.E.: 11.5611 | S.E.E.: 35.46043 | S.E.E.: 11.88795 | ||||||||||||||
| Trial | Variables | Values | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Coded | Uncoded | Experimental | Predicted | Residual | ||||||
| Glucose | NH4Cl | Glucose (%) | NH4Cl (%) | Biomass (g L−1) | Laccase (U L−1) | Biomass (g L−1) | Laccase (U L−1) | Biomass (g L−1) | Laccase (U L−1) | |
| 1 | −1 | −1 | 1.25 | 0.25 | 2.0 | 374 | 2.01 | 271 | −0.01 | 103 |
| 2 | −1 | +1 | 1.25 | 0.75 | 1.56 | 254 | 1.80 | 372 | −0.24 | −118 |
| 3 | +1 | −1 | 3.75 | 0.25 | 2.62 | 917 | 2.38 | 690 | 0.24 | 227 |
| 4 | +1 | +1 | 3.75 | 0.75 | 2.24 | 135 | 1.81 | −2 | 0.43 | 137 |
| 5 | 0 | 0 | 2.5 | 0.5 | 1.93 | 282 | 2.09 | 233 | −0.16 | 49 |
| 6 | − α | 0 | 0.7 | 0.5 | 1.87 | 156 | 1.76 | 249 | 0.11 | −93 |
| 7 | + α | 0 | 4.3 | 0.5 | 2.43 | 153 | 2.03 | 281 | 0.4 | −128 |
| 8 | 0 | − α | 2.5 | 0.14 | 2.26 | 416 | 2.38 | 621 | −0.12 | −205 |
| 9 | 0 | + α | 2.5 | 0.86 | 1.79 | 325 | 1.82 | 196 | −0.03 | 129 |
| 10 | 0 | 0 | 2.5 | 0.5 | 2.0 | 271 | 2.09 | 233 | −0.09 | 38 |
| 11 | 0 | 0 | 2.5 | 0.5 | 1.9 | 286 | 2.09 | 233 | −0.19 | 53 |
| 12 | 0 | 0 | 2.5 | 0.5 | 1.89 | 243 | 2.09 | 233 | −0.2 | 10 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
D’Souza-Ticlo, D.; Garg, S.; Raghukumar, C. Effects and Interactions of Medium Components on Laccase from a Marine-Derived Fungus Using Response Surface Methodology. Mar. Drugs 2009, 7, 672-688. https://doi.org/10.3390/md7040672
D’Souza-Ticlo D, Garg S, Raghukumar C. Effects and Interactions of Medium Components on Laccase from a Marine-Derived Fungus Using Response Surface Methodology. Marine Drugs. 2009; 7(4):672-688. https://doi.org/10.3390/md7040672
Chicago/Turabian StyleD’Souza-Ticlo, Donna, Sandeep Garg, and Chandralata Raghukumar. 2009. "Effects and Interactions of Medium Components on Laccase from a Marine-Derived Fungus Using Response Surface Methodology" Marine Drugs 7, no. 4: 672-688. https://doi.org/10.3390/md7040672
APA StyleD’Souza-Ticlo, D., Garg, S., & Raghukumar, C. (2009). Effects and Interactions of Medium Components on Laccase from a Marine-Derived Fungus Using Response Surface Methodology. Marine Drugs, 7(4), 672-688. https://doi.org/10.3390/md7040672




