Overview of Primary and Secondary Metabolites of Rugulopteryx okamurae Seaweed: Assessing Bioactivity, Scalability, and Molecular Mechanisms
Abstract
1. Introduction
1.1. Biology and Ecology of Rugulopteryx okamurae
1.2. Invasion Dynamics and Environmental Impact
2. Methodology
2.1. Focus Question
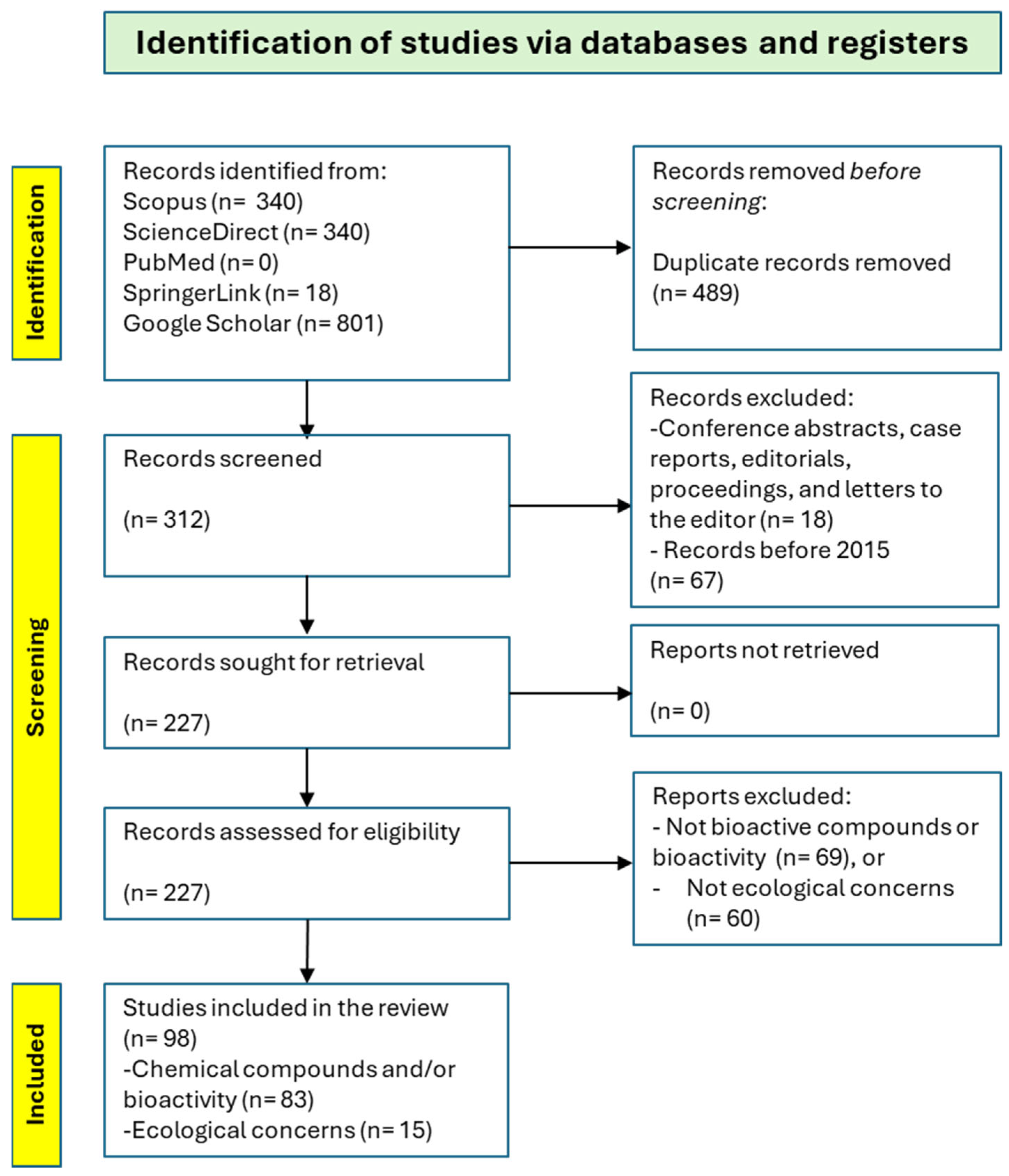
2.2. Search Strategy
2.3. Eligibility Criteria
- Population: Species Rugulopteryx okamurae
- Intervention: Chemical composition and bioactivity
- Comparison: Positive effects vs. negative actions
- Outcome: Rugulopteryx okamurae, bioactive compounds, and bioactivity
2.4. Inclusion Criteria
3. Overview of Primary and Secondary Metabolites
3.1. Polysaccharides
3.1.1. Alginates
3.1.2. Fucoidans
3.1.3. Laminarans
3.1.4. Comparison Among Polysaccharides of Brown Algae
3.1.5. Applications of Polysaccharides Extracted from R. okamurae
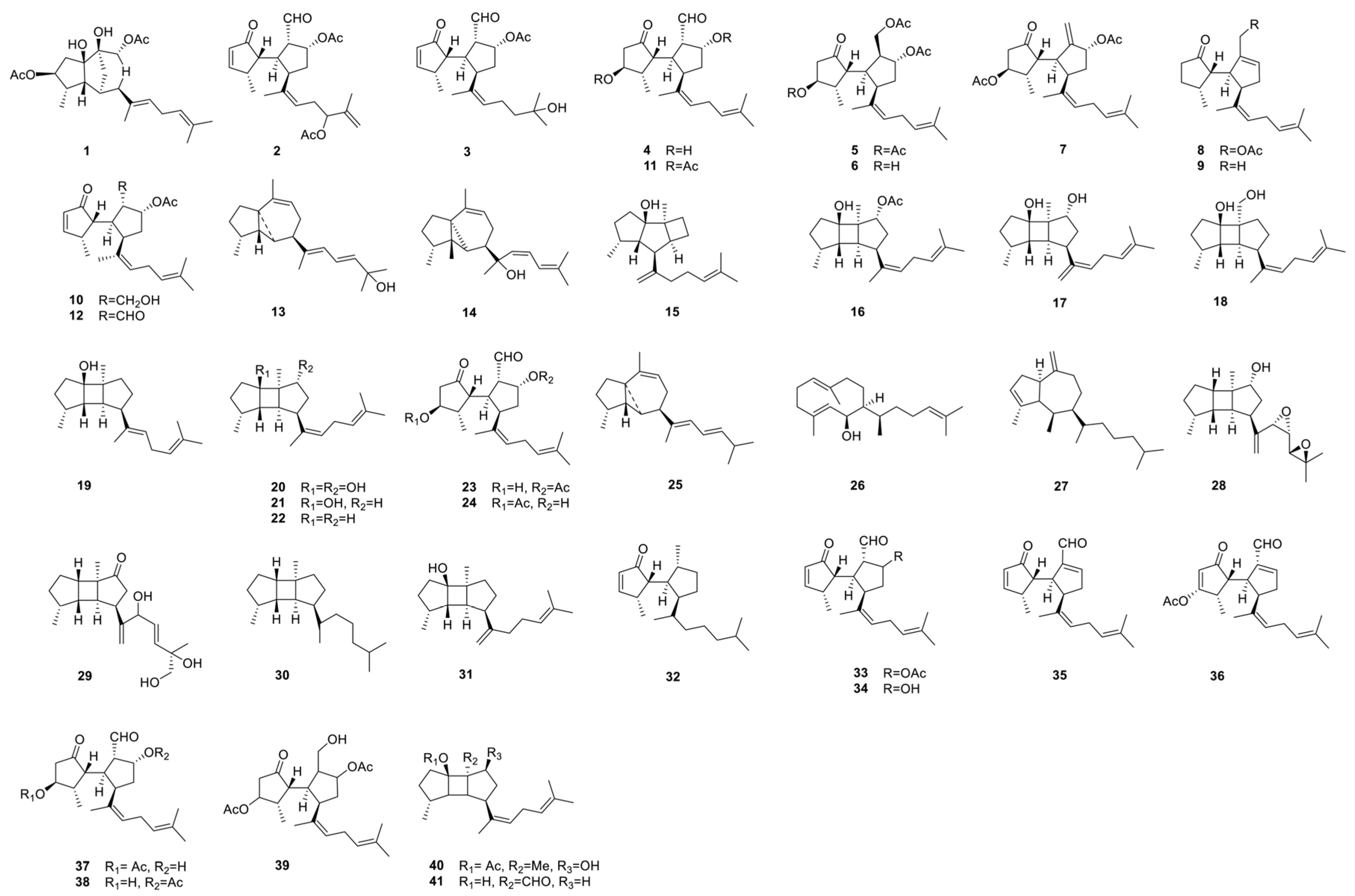
3.2. Terpenoids
3.3. Phenolic Compounds
Phenolic Content
3.4. Lipids and Fatty Acids of R. okamurae
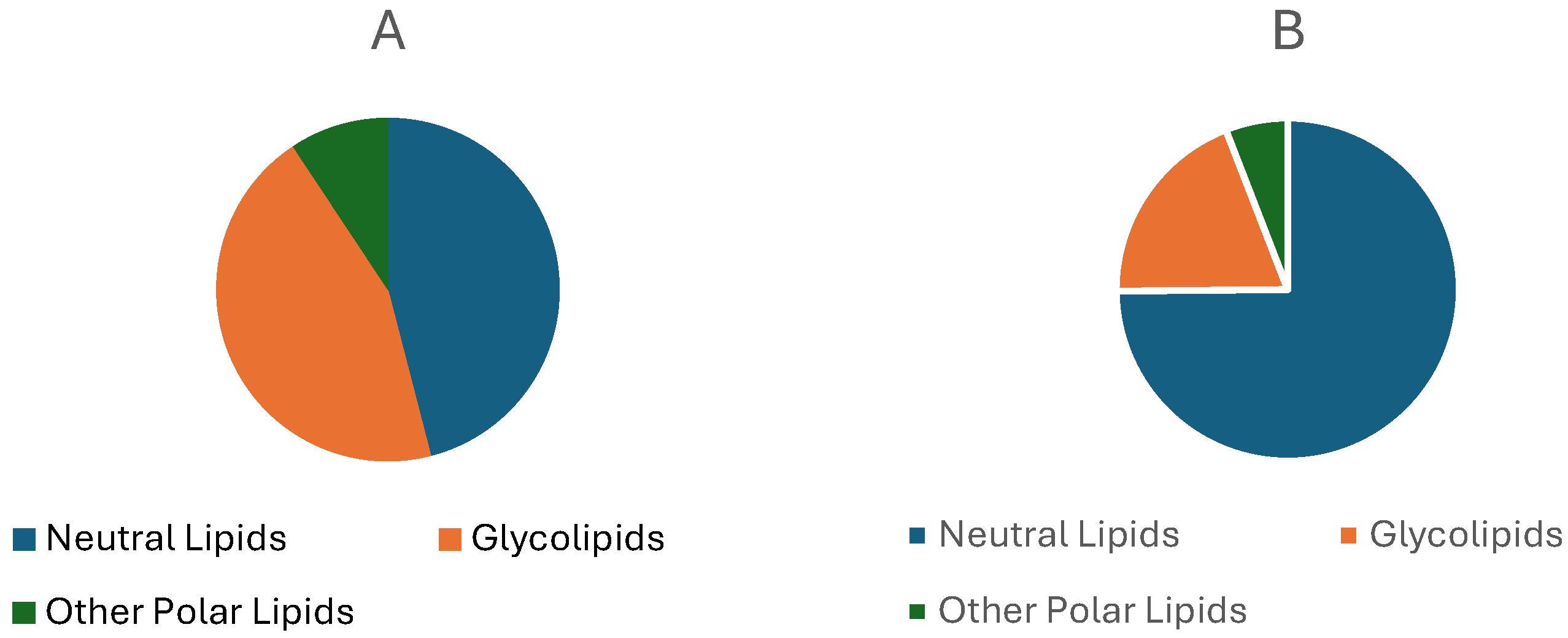
3.4.1. Lipids Content
3.4.2. Fatty Acid Profiles
3.4.3. Lipidic Fraction
3.4.4. Fatty Acid Profiles of R. okamurae as a Chemotaxonomical Tool
3.5. Volatile Compounds
3.6. Peptides
4. Biological Activity of R. okamurae
4.1. Antioxidant Activity
4.2. Anti-Inflammatory Activity
4.3. Antimicrobial Activity
4.4. Cytotoxic and Anticancer Potential
4.5. Anticoagulant and Immunomodulatory Effects
| Property | R. okamurae | Other Marine Polysaccharides (e.g., Fucoidans) | References |
|---|---|---|---|
| Anticoagulant Activity | Not directly studied | Extends plasma coagulation time; antithrombotic | [88,91] |
| Immunomodulatory Activity | High polyphenol content; antioxidant activity | Enhances cytokine production; immune cell activation | [32,89,90,91] |
5. Mechanisms of Action and Bioactivity Correlations
5.1. Structure–Activity Relationships
5.2. Cellular and Molecular Targets
5.3. Synergistic and Additive Effects Among Chemical Compounds
6. Limitations and Outlook of This Review
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chandrasekhar, T.; Riazunnisa, K.; Lakshmi, D.V.; Prasanna, V.A.; Bramhachari, P.V. Exploration of Bioactive Functional Molecules from Marine Algae: Challenges and Applications in Nutraceuticals. In Marine Bioactive Molecules for Biomedical and Pharmacotherapeutic Applications; Springer: Singapore, 2024; pp. 187–196. [Google Scholar] [CrossRef]
- Khora, S.S.; Navya, P. Bioactive polysaccharides from marine macroalgae. In Encyclopedia of Marine Biotechnology; Wiley: Hoboken, NJ, USA, 2020; pp. 121–145. [Google Scholar] [CrossRef]
- Wijesekara, I.; Kim, S.K. Application of Marine Algae Derived Nutraceuticals in the Food Industry. In Marine Algae Extracts: Processes, Products, and Applications; Wiley: Hoboken, NJ, USA, 2015; Volume 2, pp. 627–638. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, J.E.; Kim, K.H.; Kang, N.J. Beneficial effects of marine algae-derived carbohydrates for skin health. Mar. Drugs 2018, 16, 459. [Google Scholar] [CrossRef]
- Rengasamy, K.R.R.; Kulkarni, M.G.; Stirk, W.A.; Van Staden, J. Bioactive metabolites and value-added products from marine macroalgae. In Seafood Processing By-Products: Trends and Applications; Springer: New York, NY, USA, 2013; pp. 423–454. ISBN 978-1-4614-9590-1. [Google Scholar] [CrossRef]
- Cuevas, B.; Arroba, A.I.; de los Reyes, C.; Gómez-Jaramillo, L.; González-Montelongo, M.C.; Zubía, E. Diterpenoids from the brown alga Rugulopteryx okamurae and their anti-inflammatory activity. Mar. Drugs 2021, 19, 677. [Google Scholar] [CrossRef]
- Cuevas, B.; Arroba, A.I.; de los Reyes, C.; Zubía, E. Rugulopteryx-Derived Spatane, Secospatane, Prenylcubebane and Prenylkelsoane Diterpenoids as Inhibitors of Nitric Oxide Production. Mar. Drugs 2023, 21, 252. [Google Scholar] [CrossRef]
- Rincón-Cervera, M.A.; de Burgos-Navarro, I.; Chileh-Chelh, T.; Belarbi, E.H.; Álvarez-Corral, M.; Carmona-Fernández, M.; Ezzaitouni, M.; Guil-Guerrero, J.L. The Agronomic Potential of the Invasive Brown Seaweed Rugulopteryx okamurae: Optimisation of Alginate, Mannitol, and Phlorotannin Extraction. Plants 2024, 13, 3539. [Google Scholar] [CrossRef]
- Borriglione, M.; Ruitton, S.; Boyer, F.; Thibault, D.; Blanfuné, A.; Guillemain, D.; Verlaque, M.; Boudouresque, C.F.; Thibaut, T. Impact of the invasive brown alga Rugulopteryx okamurae on the benthic communities in the Northwestern Mediterranean Sea. Estuar. Coast. Shelf Sci. 2024, 310, 109010. [Google Scholar] [CrossRef]
- Faria, J.; Prestes, A.C.L.; Moreu, I.; Cacabelos, E.; Martins, G.M. Dramatic changes in the structure of shallow-water marine benthic communities following the invasion by Rugulopteryx okamurae (Dictyotales, Ochrophyta) in Azores (NE Atlantic). Mar. Pollut. Bull. 2022, 175, 113358. [Google Scholar] [CrossRef]
- García-Gómez, J.C.; Florido, M.; Olaya-Ponzone, L.; Sempere-Valverde, J.; Megina, C. The Invasive Macroalga Rugulopteryx okamurae: Substrata Plasticity and Spatial Colonization Pressure on Resident Macroalgae. Front. Ecol. Evol. 2021, 9, 631754. [Google Scholar] [CrossRef]
- Mercado, J.M.; Gómez-Jakobsen, F.; Korbee, N.; Aviles, A.; Bonomi-Barufi, J.; Muñoz, M.; Reul, A.; Figueroa, F.L. Analyzing environmental factors that favor the growth of the invasive brown macroalga Rugulopteryx okamurae (Ochrophyta): The probable role of the nutrient excess. Mar. Pollut. Bull. 2022, 174, 113315. [Google Scholar] [CrossRef] [PubMed]
- Román, S.; Vázquez, R. Assessment of the Rugulopteryx okamurae invasion in Northeastern Atlantic and Mediterranean bioregions: Colonisation status, propagation hypotheses and temperature tolerance thresholds. Mar. Environ. Res. 2025, 207, 107093. [Google Scholar] [CrossRef] [PubMed]
- Rueda, J.L.; Mena-Torres, A.; Gallardo-Núñez, M.; González-García, E.; Martín-Arjona, A.; Valenzuela, J.; García-Ruiz, C.; González-Aguilar, M.; Mateo-Ramírez, Á.; García, M. Spatial distribution and potential impact of drifted Thalli of the invasive Alga Rugulopteryx okamurae in Circalittoral and Bathyal habitats of the Northern Strait of Gibraltar and the Alboran Sea. Diversity 2023, 15, 1206. [Google Scholar] [CrossRef]
- Bachot, X.; Riera, R. How the invasive algae Rugulopteryx okamurae affect coastal biodiversity? Insights from coastal fish communities of gran Canaria (NE Atlantic Ocean). J. Sea Res. 2025, 204, 102568. [Google Scholar] [CrossRef]
- Figueroa, F.L.; Vega, J.; Flórez-Fernández, N.; Mazón, J.; Torres, M.D.; Domínguez, H.; Pereira, L. Challenges and opportunities of the exotic invasive macroalga Rugulopteryx okamurae (Phaeophyceae, Heterokontophyta). J. Appl. Phycol. 2025, 37, 579–595. [Google Scholar] [CrossRef]
- Hachero-Cruzado, I.; Castilla-Gavilán, M.; Muñoz-Martinez, M.; Zuasti, E.; Jimenez, T. Assessing grazing of Rugulopteryx okamurae (Ochrophyta) by the sea urchin Paracentrotus lividus (Echinodermata): A prospect for the biocontrol of the alien species? Eur. J. Phycol. 2024, 59, 332–349. [Google Scholar] [CrossRef]
- Nagarajan, D.; Senthilkumar, G.; Chen, C.-W.; Karmegam, N.; Praburaman, L.; Kim, W.; Dong, C.-D. Sustainable bioplastics from seaweed polysaccharides: A comprehensive review. Polym. Adv. Technol. 2024, 35, e6536. [Google Scholar] [CrossRef]
- Marletta, G.; Lombardo, A.; Serio, D. First Record of the Invasive Alien Species Rugulopteryx okamurae (Phaeophyceae, Dictyotales) along the Eastern Coast of Sicily (Italy, Mediterranean Sea): Is It Ready to Expand into the Ionian Sea? Diversity 2024, 16, 424. [Google Scholar] [CrossRef]
- Roca, M.; Dunbar, M.B.; Román, A.; Caballero, I.; Zoffoli, M.L.; Gernez, P.; Navarro, G. Monitoring the marine invasive alien species Rugulopteryx okamurae using unmanned aerial vehicles and satellites. Front. Mar. Sci. 2022, 9, 1004012. [Google Scholar] [CrossRef]
- Mogollón, S.L.; Zilio, M.I.; Buitrago, E.M.; Caraballo, M.Á.; Yñiguez, R. Economic impact of Rugulopteryx okamurae (Dictyotales, Ochrophyta) along the Andalusian coastline: The case of Tarifa, Spain. Wetl. Ecol. Manag. 2024, 32, 19–32. [Google Scholar] [CrossRef]
- Laamraoui, M.R.; Mghili, B.; Roca, M.; Chaieb, O.; Ostalé-Valriberas, E.; Martín-Zorrillae, A.; Sabino-Lorenzo, A.; Aarab, S. Rapid invasion and expansion of the invasive macroalgae Rugulopteryx okamurae in the Mediterranean and Atlantic: A 10-year review. Mar. Pollut. Bull. 2024, 209, 117194. [Google Scholar] [CrossRef]
- Barcellos, L.; Pham, C.K.; Menezes, G.; Bettencourt, R.; Rocha, N.; Carvalho, M.; Felgueiras, H.P. A Concise Review on the Potential Applications of Rugulopteryx okamurae Macroalgae. Mar. Drugs 2023, 21, 40. [Google Scholar] [CrossRef]
- Sarangi, N.V.; Prakasan, M.V.S.; Rajkumar, R.; Srinivasan, S. Assessment of phytochemicals from marine algae Ulva fasciata and Dictyota dichotoma with antioxidant and antimicrobial potential. Appl. Chem. Eng. 2023, 6, 1–10. [Google Scholar] [CrossRef]
- Sari, A.P.; Nurdin, G.M.; Manguntungi, B.; Mustopa, A.Z. Potential of Red, Brown, and Green Macroalgae from Dato Beach, Majene, Indonesia as Natural Food Preservative. Philipp. J. Sci. 2023, 152, 1483–1493. [Google Scholar] [CrossRef]
- Sahayaraj, K.; Asharaja, A.C.; Rajesh, S.; Martin Rathi, J.A. Qualitative and quantitative profiles of secondary metabolites of chosen Chlorophyta and Ochrophyta from Gulf of Mannor. Cah. Biol. Mar. 2014, 55, 69–76. [Google Scholar]
- Bahrun, B.; Soekamto, N.; Firdaus, F. In vitro and in silico analysis for antibacterial activitiy of various extracts of Gracilaria salicornia (Rhodophyta) from Selayar Islands, Indonesia. Egypt. J. Chem. 2021, 64, 7203–7212. [Google Scholar] [CrossRef]
- Salunke, M.; Wakure, B.; Wakte, P. High-resolution liquid chromatography mass spectrometry (HR-LCMS) and 1H NMR analysis of methanol extracts from marine seaweed Gracilaria edulis. Nat. Prod. Res. 2024, 38, 1441–1444. [Google Scholar] [CrossRef]
- Wirawan, I.G.P.; Dewi, N.K.E.S.; Sasadara, M.M.V.; Sunyamurthi, I.G.N.A.; Jawi, I.M.; Wijaya, I.N.; Darmawati, I.A.P.; Suada, I.K.; Krisnandika, A.A.K. Phytochemical analysis and molecular identification of green macroalgae Caulerpa spp. from Bali, Indonesia. Molecules 2022, 27, 4879. [Google Scholar] [CrossRef]
- Sasikala, C.; Geetha Ramani, D. Comparative study on antimicrobial activity of seaweeds. Asian J. Pharm. Clin. Res. 2017, 10, 384–386. [Google Scholar] [CrossRef][Green Version]
- Mitra, R.; Ghosh, S.; Mukherjee, G.; Chowdhury, A.A. Secondary Metabolites: Treasure Trove for Future Medicine. In Reference Series in Phytochemistry; Springer: Cham, Switzerland, 2025; Volume Part F362, pp. 57–101. [Google Scholar][Green Version]
- Belhadj, R.N.A.; Mellinas, C.; Jiménez, A.; Bordehore, C.; Garrigós, M.C. Invasive Seaweed Rugulopteryx okamurae: A Potential Source of Bioactive Compounds with Antioxidant Activity. Antioxidants 2024, 13, 1298. [Google Scholar] [CrossRef] [PubMed]
- Cheong, K.L.; Chen, W.; Wang, M.; Zhong, S.; Veeraperumal, S. Therapeutic Prospects of Undaria pinnatifida Polysaccharides: Extraction, Purification, and Functional Activity. Mar. Drugs 2025, 23, 163. [Google Scholar] [CrossRef]
- Cui, M.; Wu, J.; Wang, S.; Shu, H.; Zhang, M.; Liu, K.; Liu, K. Characterization and anti-inflammatory effects of sulfated polysaccharide from the red seaweed Gelidium pacificum Okamura. Int. J. Biol. Macromol. 2019, 129, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, L.; Shang, N.; Wu, K.; Liao, W. Recent Advances in the Structure, Extraction, and Biological Activity of Sargassum fusiforme Polysaccharides. Mar. Drugs 2025, 23, 98. [Google Scholar] [CrossRef]
- Agustín, R.-V.; Luis Alberto, F.-G.; Ana, B.; Luis Isidoro, R.-G.; Belén, D.A. Rugulopteryx okamurae: Assessment of its potential as a source of monosaccharides for obtaining bio-products. Chem. Eng. J. 2023, 468, 143578. [Google Scholar] [CrossRef]
- Romero-Vargas, A.; Fdez-Güelfo, L.A.; Blandino, A.; Díaz, M.J.; Díaz, A.B. Rugulopteryx okamurae: Effect of hydrothermal acid pretreatment on the saccharification process. Bioresour. Technol. 2023, 388, 129721. [Google Scholar] [CrossRef]
- Ferreira-Anta, T.; Flórez-Fernández, N.; Torres, M.D.; Mazón, J.; Dominguez, H. Microwave-Assisted Hydrothermal Processing of Rugulopteryx okamurae. Mar. Drugs 2023, 21, 319. [Google Scholar] [CrossRef]
- Paulo, C.; Matos, J.; Afonso, C.; Cardoso, C. Overcoming Extraction Hurdles and Assessing Biological Activity in a Major Invasive Seaweed Species in Europe, Rugulopteryx okamurae. Mar. Drugs 2025, 23, 141. [Google Scholar] [CrossRef]
- Agabo-García, C.; Romero-García, L.I.; Álvarez-Gallego, C.J.; Blandino, A. Valorisation of the invasive alga Rugulopteryx okamurae through the production of monomeric sugars. Appl. Microbiol. Biotechnol. 2023, 107, 1971–1982. [Google Scholar] [CrossRef] [PubMed]
- Vieira, C.B.; Sousa, J.R.; Do Vale, D.A.; Guimarães, C.P.; de Sousa, K.C.; Mattos, A.L.A.; Silva, A.L.C.; de Sá Moreira Souza Filho, M.; Souza, B.W.S. Edible films based on sulfated polysaccharides from the seaweed Gracilaria birdiae: Physicochemical, optical and mechanical properties. Carbohydr. Res. 2025, 552, 109473. [Google Scholar] [CrossRef] [PubMed]
- Donati, I.; Christensen, B.E. Alginate-metal cation interactions: Macromolecular approach. Carbohydr. Polym. 2023, 321, 121280. [Google Scholar] [CrossRef]
- Santana, I.; Felix, M.; Bengoechea, C. Feasibility of Invasive Brown Seaweed Rugulopteryx okamurae as Source of Alginate: Characterization of Products and Evaluation of Derived Gels. Polymers 2024, 16, 702. [Google Scholar] [CrossRef]
- Gade, J.V.; Gupta, N.; Pandey, A.; Rawat, R.; Jain, B. Polysaccharides from seaweeds: Extraction and application. In Marine Molecules from Algae and Cyanobacteria: Extraction, Purification, Toxicology and Applications; Elsevier: Amsterdam, The Netherlands, 2024; pp. 153–167. [Google Scholar]
- Venkatesan, J.; Lowe, B.; Anil, S.; Manivasagan, P.; Kheraif, A.A.A.; Kang, K.H.; Kim, S.K. Seaweed polysaccharides and their potential biomedical applications. Starch/Staerke 2015, 67, 381–390. [Google Scholar] [CrossRef]
- Imbs, T.I.; Ermakova, S.P.; Malyarenko, O.S.; Isakov, V.V.; Zvyagintseva, T.N. Structural elucidation of polysaccharide fractions from the brown alga Coccophora langsdorfii and in vitro investigation of their anticancer activity. Carbohydr. Polym. 2016, 135, 162–168. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Ushakova, N.A.; Preobrazhenskaya, M.E.; Bilan, M.I.; Tsvetkova, E.A.; Krylov, V.B.; Anisimova, N.A.; Kiselevskiy, M.V.; Krukovskaya, N.V.; Li, C.; et al. Fucoidans as a platform for new anticoagulant drugs discovery. J. Macromol. Sci. Part A Pure Appl. Chem. 2014, 86, 1365–1375. [Google Scholar] [CrossRef]
- Ermakova, S.; Men’shova, R.; Vishchuk, O.; Kim, S.M.; Um, B.H.; Isakov, V.; Zvyagintseva, T. Water-soluble polysaccharides from the brown alga Eisenia bicyclis: Structural characteristics and antitumor activity. Algal Res. 2013, 2, 51–58. [Google Scholar] [CrossRef]
- Zvyagintseva, T.N.; Usoltseva, R.V.; Shevchenko, N.M.; Anastyuk, S.D.; Isakov, V.V.; Zvyagintsev, N.V.; Krupnova, T.N.; Zadorozhny, P.A.; Ermakova, S.P. Composition of polysaccharides and radiosensitizing activity of native and sulfated laminarans from the Tauya basicrassa Kloczc. et Krupn. Carbohydr. Polym. 2020, 250, 116921. [Google Scholar] [CrossRef] [PubMed]
- Bilan, M.I.; Usov, A.I. Structural analysis of fucoidans. Nat. Prod. Commun. 2008, 3, 1639–1648. [Google Scholar] [CrossRef]
- Lim, S.J.; Wan Aida, W.M. Extraction of Sulfated Polysaccharides (Fucoidan) From Brown Seaweed. In Seaweed Polysaccharides: Isolation, Biological and Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2017; pp. 27–46. [Google Scholar]
- Rivero-Pino, F.; Gonzalez-de la Rosa, T.; Torrecillas-Lopez, M.; Barrera-Chamorro, L.; del Rio-Vazquez, J.L.; Marquez-Paradas, E.; Fernandez-Prior, A.; Garcia-Vaquero, M.; Garcia-Gomez, J.C.; Montserrat-de la Paz, S.; et al. Characterization of Rugulopteryx okamurae algae: A source of bioactive peptides, omega-3 fatty acids, and volatile compounds. Food Chem. 2025, 473, 143084. [Google Scholar] [CrossRef]
- Ravi, B.; Wells, R. A series of new diterpenes from the brown alga Dilophus marginatus (Dictyotaceae). Aust. J. Chem. 1982, 35, 129–144. [Google Scholar] [CrossRef]
- El Madany, M.; Hassoun, M.; Belmehdi, O.; Sakar, E.H.; Asraoui, F.; Mghili, B.; El Aamri, F.; El Mtili, N. Invasive Biomass Algae Valorization: Rugulopteryx okamurae as a Sustainable Source of Natural Antioxidants. Egypt. J. Aquat. Biol. Fish. 2023, 27, 267–283. [Google Scholar] [CrossRef]
- Cebrian-Lloret, V.; Cartan-Moya, S.; Martinez-Sanz, M.; Gomez-Cortes, P.; Calvo, M.V.; Lopez-Rubio, A.; Martinez-Abad, A. Characterization of the invasive macroalgae Rugulopteryx okamurae for potential biomass valorisation. Food Chem. 2024, 440, 138241. [Google Scholar] [CrossRef]
- De la Lama-Calvente, D.; Fernández-Rodríguez, M.J.; Garrido-Fernández, A.; Borja, R. Process optimization of the extraction of reducing sugars and total phenolic compounds from the invasive alga Rugulopteryx okamurae by response surface methodology (RSM). Algal Res. 2024, 80, 103500. [Google Scholar] [CrossRef]
- Bhuyan, D.J.; Basu, A. Phenolic compounds potential health benefits and toxicity. In Utilisation of Bioactive Compounds from Agricultural and Food Production Waste; CRC Press: Boca Raton, FL, USA, 2017; pp. 27–59. [Google Scholar]
- Tariq, A.; Ahmed, A. Plant Phenolics Production: A Strategy for Biotic Stress Management. In Plant Phenolics in Biotic Stress Management; Springer: Singapore, 2024; pp. 441–454. [Google Scholar]
- Hamid, S.; Yatoo, A.M.; Mir, M.Y.; Ali, S.; Mohamed, H.I. Historical Perspective of Plant Phenolics. In Plant Phenolics in Abiotic Stress Management; Springer: Singapore, 2023; pp. 1–22. [Google Scholar]
- Miyashita, K.; Mikami, N.; Hosokawa, M. Chemical and nutritional characteristics of brown seaweed lipids: A review. J. Funct. Foods 2013, 5, 1507–1517. [Google Scholar] [CrossRef]
- Santos, F.; Monteiro, J.P.; Duarte, D.; Melo, T.; Lopes, D.; da Costa, E.; Domingues, M.R. Unraveling the Lipidome and Antioxidant Activity of Native Bifurcaria bifurcata and Invasive Sargassum muticum Seaweeds: A Lipid Perspective on How Systemic Intrusion May Present an Opportunity. Antioxidants 2020, 9, 642. [Google Scholar] [CrossRef] [PubMed]
- Wielgosz-Collin, G.; Kendel, M.; Couzinet-Mossion, A. Lipids, fatty acids, glycolipids, and phospholipids. In Seaweed in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2016; pp. 185–221. [Google Scholar] [CrossRef]
- Ahmed, N.; Ahmed, K. Chemical and different nutritional characteristics of brown seaweed lipids. Adv. Sci. Technol. Eng. Syst. J. 2016, 1, 23–25. [Google Scholar] [CrossRef][Green Version]
- Terasaki, M.; Hirose, A.; Narayan, B.; Baba, Y.; Kawagoe, C.; Yasui, H.; Saga, N.; Hosokawa, M.; Miyashita, K. Evaluation of recoverable functional lipid components of several brown seaweeds (phaeophyta) from japan with special reference to fucoxanthin and fucosterol contents. J. Phycol. 2009, 45, 974–980. [Google Scholar] [CrossRef]
- Córdoba-Granados, J.J.; Jiménez-Hierro, M.J.; Zuasti, E.; Ochoa-Hueso, R.; Puertas, B.; Zarraonaindia, I.; Hachero-Cruzado, I.; Cantos-Villar, E. Biochemical characterization and potential valorization of the invasive seaweed Rugulopteryx okamurae. J. Appl. Phycol. 2025, 37, 567–577. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.L.; Carmona-Fernández, M.; Chileh-Chelh, T.; Belarbi, E.H.; Urrestarazu, M.; Loeiro Cunha-Chiamolera, T.P.; Ezzaitouni, M.; Rincón-Cervera, M.Á.; Rodríguez-García, I. Fatty acid profiling in the invasive brown seaweed Rugulopterix okamurae: A usefulness taxonomical tool. Cont. Shelf Res. 2025, 286, 105412. [Google Scholar] [CrossRef]
- Güven, K.C.; Sezik, E.; Kaleağasıoğlu, F.; Erdugan, H.; Coban, B.; Karakaş, E. Volatile oils from marine macroalgae. In Natural Products; Springer: Singapore, 2013; pp. 2883–2912. [Google Scholar] [CrossRef]
- Fan, H.; Nie, A.; Ge, Y.; Weng, L.; Gao, Y.; Chen, F.; Shen, H. Influencing factors of plant habitat, phenophase, and organ on the essential oil composition of Agastache rugosa. Acta Aliment. 2024, 53, 61–70. [Google Scholar] [CrossRef]
- Song, J.-S.; Chae, Y.-A.; Hyun-Choong, O. Variability of the Volatile Composition of Agastache rugosa in South Korea. In Proceedings of the III WOCMAP Congress on Medicinal and Aromatic Plants-Volume 1: Bioprospecting and Ethnopharmacology 675, Chiang Mai, Thailand, 3–7 February 2003; pp. 59–64. [Google Scholar] [CrossRef]
- Hernández, J.; Bracho, I.; Rojas-Fermin, L.B.; Usubillaga, A.; Carmona, J. Chemical composition of the essential oil of Erechtites valerianaefolia from Mérida, Venezuela. Nat. Prod. Commun. 2013, 8, 1934578X1300801036. [Google Scholar] [CrossRef]
- Nopsiri, W.; Chansakaow, S.; Putiyanan, S.; Natakankitkul, S.; Nantachit, K.; Khantawa, B.; Santiarworn, D. Chemical constituents and antibacterial activity of volatile oils of Combretum latifolium Bl. and C. quadrangulare Kurz Leaves. CMUJ Nat. Sci. 2015, 14, 245–256. [Google Scholar] [CrossRef]
- Bak, S.G.; Lim, H.J.; Park, E.J.; Won, Y.S.; Lee, S.W.; Ryu, B.; Ha, H.J.; Cheong, S.H.; Lee, S.J. Ishige okamurae Extract: Diphlorethohydroxycarmalol with Effect of Atopic Dermatitis-Like Skin Inflammation. Rev. Bras. Farmacogn. 2024, 34, 338–349. [Google Scholar] [CrossRef]
- Heo, S.J.; Jeon, Y.J. Evaluation of diphlorethohydroxycarmalol isolated from Ishige okamurae for radical scavenging activity and its protective effect against H2O2-induced cell damage. Process Biochem. 2009, 44, 412–418. [Google Scholar] [CrossRef]
- Matos, M.; Custódio, L.; Reis, C.P. Marine Invasive Algae’s Bioactive Ingredients as a Sustainable Pathway in Cosmetics: The Azores Islands as a Case Study. Mar. Drugs 2024, 22, 575. [Google Scholar] [CrossRef]
- Kim, Y.A.; Kong, C.S.; Um, Y.R.; Lee, J.I.; Nam, T.J.; Seo, Y. Antioxidant efficacy of extracts from a variety of seaweeds in a cellular system. Ocean Sci. J. 2008, 43, 31–37. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, G.H. Evaluation of antioxidant activity of marine algae-extracts from Korea. J. Aquat. Food Prod. Technol. 2015, 24, 227–240. [Google Scholar] [CrossRef]
- Ko, E.Y.; Yoon, W.J.; Lee, H.W.; Heo, S.J.; Ko, Y.H.; Shanura Fernando, I.P.; Cho, K.; Lee, C.H.; Hur, S.P.; Cho, S.H.; et al. Anti-inflammatory effect of supercritical extract and its constituents from Ishige okamurae. EXCLI J. 2016, 15, 434–445. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, L.; Sun, L.; Wang, Z.; Wang, Y.; Wang, Y.; He, X. 3α-Angeloyloxy- ent-kaur-16-en-19-oic Acid Isolated from Wedelia trilobata L. Alleviates Xylene-Induced Mouse Ear Edema and Inhibits NF-κB and MAPK Pathway in LPS-Stimulated Macrophages. J. Nat. Prod. 2020, 83, 3726–3735. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, H.Y.; Mady, M.S.; Atya, H.B.; Ali, S.A.; Elsayed, H.E.; Moharram, F.A. Melaleuca rugulosa (Link) Craven Tannins: Appraisal of anti-inflammatory, radical scavenging activities, and molecular modeling studies. J. Ethnopharmacol. 2022, 298, 115596. [Google Scholar] [CrossRef] [PubMed]
- Lakshanee, L.W.; Kadigamuwa, C.C.; Mapa, M.M.S.T. Antimicrobial properties of seaweeds. In The Role of Seaweeds in Blue Bioeconomy; Bentham Science Publishers: Potomac, MD, USA, 2024; pp. 228–264. [Google Scholar] [CrossRef]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial action of compounds from marine seaweed. Mar. Drugs 2016, 14, 52. [Google Scholar] [CrossRef]
- Amri, S.; Bouslama, L.; Mzoughi, Z.; Nouira, F.; Majdoub, H.; Bouraoui, A. Chemical characterization and evaluation of antiviral activity of two fucoidans extracted from Mediterranean brown seaweeds, Padina pavonica and Dictyopteris membranacea. Lett. Appl. Microbiol. 2025, 78, ovaf002. [Google Scholar] [CrossRef]
- Besednova, N.N.; Kuznetsova, T.A.; Zaporozhets, T.S.; Zvyagintseva, T.N. Brown seaweeds as a source of new pharmaceutical substances with antibacterial action. Antibiot. I Khimioterapiya 2015, 60, 31–41. [Google Scholar]
- Talarico, L.B.; Pujol, C.A.; Zibetti, R.G.M.; Faría, P.C.S.; Noseda, M.D.; Duarte, M.E.R.; Damonte, E.B. The antiviral activity of sulfated polysaccharides against dengue virus is dependent on virus serotype and host cell. Antivir. Res. 2005, 66, 103–110. [Google Scholar] [CrossRef]
- Ashwini, K.; Siva, B.; Poornima, P.; Reddy, S.D.; Sastry, V.G.; Babu, K.S. New cytotoxic spatane diterpenoids from marine alga Stoechospermum marginatum. Fitoterapia 2024, 177, 106071. [Google Scholar] [CrossRef]
- Zenthoefer, M.; Geisen, U.; Hofmann-Peiker, K.; Fuhrmann, M.; Kerber, J.; Kirchhöfer, R.; Hennig, S.; Peipp, M.; Geyer, R.; Piker, L.; et al. Isolation of polyphenols with anticancer activity from the Baltic Sea brown seaweed Fucus vesiculosus using bioassay-guided fractionation. J. Appl. Phycol. 2017, 29, 2021–2037. [Google Scholar] [CrossRef]
- Mary, J.S.; Vinotha, P.; Pradeep, A.M. Screening for in vitro cytotoxic activity of seaweed, sargassum sp. against hep-2 and MCF-7 cancer cell lines. Asian Pac. J. Cancer Prev. 2012, 13, 6073–6076. [Google Scholar] [CrossRef]
- Farias, W.R.L. Comparative medicinal properties of seaweed sulfated polysaccharides. In Seaweed: Ecology, Nutrient Composition and Medicinal Uses; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2011; pp. 209–219. [Google Scholar]
- Li, Y.; McGowan, E.; Chen, S.; Santos, J.; Yin, H.; Lin, Y. Immunopotentiating Activity of Fucoidans and Relevance to Cancer Immunotherapy. Mar. Drugs 2023, 21, 128. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Dwan, C.; Wimmer, B.C.; Ronci, M.; Wilson, R.; Johnson, L.; Caruso, V. Neuroprotective and Anti-Inflammatory Activity of Undaria pinnatifida Fucoidan In Vivo—A Proteomic Investigation. Mar. Drugs 2025, 23, 189. [Google Scholar] [CrossRef]
- Pradhan, B.; Patra, S.; Nayak, R.; Behera, C.; Dash, S.R.; Nayak, S.; Sahu, B.B.; Bhutia, S.K.; Jena, M. Multifunctional role of fucoidan, sulfated polysaccharides in human health and disease: A journey under the sea in pursuit of potent therapeutic agents. Int. J. Biol. Macromol. 2020, 164, 4263–4278. [Google Scholar] [CrossRef] [PubMed]
- Vega, J.; Catalá, T.S.; García-Márquez, J.; Speidel, L.G.; Arijo, S.; Cornelius Kunz, N.; Geisler, C.; Figueroa, F.L. Molecular Diversity and Biochemical Content in Two Invasive Alien Species: Looking for Chemical Similarities and Bioactivities. Mar. Drugs 2023, 21, 5. [Google Scholar] [CrossRef] [PubMed]
- Casal-Porras, I.; Zubía, E.; Brun, F.G. Dilkamural: A novel chemical weapon involved in the invasive capacity of the alga Rugulopteryx okamurae in the Strait of Gibraltar. Estuar. Coast. Shelf Sci. 2021, 257, 107398. [Google Scholar] [CrossRef]
- Yang, F.; Pu, H.Y.; Yaseen, A.; Chen, B.; Li, F.; Gu, Y.C.; Shen, X.F.; Wang, M.K.; Guo, D.L.; Wang, L. Terpenoid and phenolic derivatives from the aerial parts of Elsholtzia rugulosa and their anti-inflammatory activity. Phytochemistry 2021, 181, 112543. [Google Scholar] [CrossRef]
- Nodwell, M.B.; Menz, H.; Kirsch, S.F.; Sieber, S.A. Rugulactone and its Analogues Exert Antibacterial Effects through Multiple Mechanisms Including Inhibition of Thiamine Biosynthesis. ChemBioChem 2012, 13, 1439–1446. [Google Scholar] [CrossRef]
- Bernal-Ibáñez, A.; Castilla-Gavilán, M.; Sumariva, E.G.; Hachero-Cruzado, I. Invasion context matters: Vulnerability of the sea urchin Paracentrotus lividus to ingestion of Rugulopteryx okamurae increases with invasion time. Mar. Pollut. Bull. 2025, 214, 117826. [Google Scholar] [CrossRef] [PubMed]
- Jarauta, I.; Ferreira, V.; Cacho, J.F. Synergic, additive and antagonistic effects between odorants with similar odour properties. Dev. Food Sci. 2006, 43, 205–208. [Google Scholar] [CrossRef]
- Bonomi-Barufi, J.; Figueroa, F.L.; Vega, J.; Huesa, R.; Harb, T.B.; Avilés, A.; Mercado, J.M.; Korbee, N. Physiological responses of the alien macroalga Rugulopteryx okamurae (Phaeophyceae, Heterokontophyta) to changes in nutrients and temperature. J. Phycol. 2025, 61, 44–60. [Google Scholar] [CrossRef] [PubMed]






| Parameter | Conventional Chemical Extraction [8] | Microwave-Assisted Hydrothermal Extraction [38] |
|---|---|---|
| Solvents/Chemicals Used | Formaldehyde, HCl, Na2CO3, Ethanol, Acetone | Distilled Water or Seawater |
| Temperature | 65 °C (alkaline extraction) | 160–180 °C (microwave heating) |
| Time Required | Several hours (overnight soaking + 5 h extraction) | 5–20 min |
| Pre-treatment | Yes—pigment removal with formaldehyde and acid preconditioning | No pre-treatment needed |
| Extraction Mechanism | Solubilization via chemical steps | Dielectric heating with pressurised water |
| Yield (Alginate) | High (purified, sodium alginate) | Up to 3.2% (crude, calcium alginate) |
| Purification Steps | Ethanol precipitation, acetone wash, freeze-drying | Dialysis and freeze-drying |
| Advantages | High-purity product; strong gelling (rich in guluronic acid) | Fast, green, low-chemical process; energy efficient |
| Limitations | Time-consuming; chemical use | Lower alginate yield; needs further purification |
| Best Use Case | Biomaterials require precise gelation and structural properties | Quick, sustainable extraction for biorefinery or bulk applications |
| Polysaccharide | Structure | Properties | References |
|---|---|---|---|
| Alginate | Linear, blockwise arrangement of β-D-mannuronate and α-L-guluronate | Gel-forming, used in tissue engineering and drug delivery | [42,44,45] |
| Fucoidan | Sulfated α-L-fucan, alternating (1→3)- and (1→4)-linked α-L-fucopyranose | Anticoagulant, antitumor, antiviral, structural variability | [46,47,51] |
| Laminaran | β-D-glucan, (1→3)-linked backbone with (1→6) linkages | Antitumor, inhibits cancer cell colony formation | [46,48] |
| Compound | Class | Unique Feature | Biological Activity | References |
|---|---|---|---|---|
| Rugukadiol A (1) | Diterpenoid | Bridged tricyclic undecane system | Inhibits NO production, Nos2, and Il1b expression | [6] |
| Rugukamurals A–C (2–4) | Secospatane Diterpenoids | Oxygenated functions | Anti-inflammatory | [6,7] |
| Ruguloptones A–F (5–10) | Secospatane Diterpenoids | Oxygenated functions | Anti-inflammatory | [6] |
| Okacubols A & B (13,14) | Prenylcubebane Diterpenoids | - | Anti-inflammatory | [7] |
| Okamurol A (16) | Prenylkelsoane Diterpenoid | Kelsoane-type tricyclic nucleus | Anti-inflammatory | [7] |
| Okaspatols A–D (16–19) | Spatanes | Oxygenated | Okaspatols A–D (16–19) | [7] |
| Source | Total Lipid Content (% DW) | Methodology | Notes |
|---|---|---|---|
| Rivero-Pino et al. [52] | 21.29% | Hexane/isopropanol extract | - |
| Cebrián-Lloret et al. [55] | 17.3% | Folch method | Concerns over taxonomic accuracy |
| Córdoba-Granados et al. [65] | 4.3–10.0% | Solid-phase extraction was performed for total lipid analysis | - |
| Guil-Guerrero et al. [66] | 2.4–6.8 g FA/100 g DW | Total fatty acid content via GC-FID/GC-MS; indirectly reflects total lipids | Seasonal and geographical variation |
| Fatty Acids | Rivero-Pino [52] | Cebrián-Lloret et al. [55] | Córdoba-Granados et al. [65] a | Guil-Guerrero et al. [66] b |
|---|---|---|---|---|
| Σ SFA | 65.6 | 61.12 | 37.3–49.6 | 57.3–71.3 |
| 14:0 | 8.74 | 15.15 | 4.3–6.5 | 9.9–17.8 |
| 15:0 | 0.95 | 1.48 | 0.0–0.9 | 0.9–2.4 |
| 16:0 | 40.03 | 31.75 | 24.0–31.5 | 30.4–40.5 |
| 18:0 | 13.37 | 3.13 | 3.1–7.7 | 2.2–5.0 |
| 20:0 | 0.54 | - | 2.5–3.8 | 3.3–6.0 |
| Σ MUFA | 13.3 | 15.26 | 26.4–29.9 | 18.8–27.6 |
| 15:1n−5 | 0.26 | - | - | 0.2–4.6 |
| 16:1n−7 | 3.14 | 1.62 | 1.8–7.1 | 3.0–8.7 |
| 16:1n−5 | 3.5–7.2 | |||
| 18:1n−9 | 8.43 | 12.64 | 12.4–7.5 | 11.1–15.5 |
| 20:1n−9 | 0.62 | - | 0.0–0.2 | 0.1–2.2 |
| Σ n−6 PUFA | 9.0 | 5.65 | 6.5–10.6 | 8.1–15.0 |
| 18:2n−6 trans | 0.1 | - | - | 0.9–2.1 |
| 18:2n−6 cis | 2.38 | 1.68 | 1.8–2.6 | 1.2–2.4 |
| 20:2n−6 | 0.14 | 1.58 | 0.0–0.2 | 0.8–3.9 |
| 20:4n−6 | 5.06 | 2.18 | 1.97–5.9 | 3.0–6.7 |
| Σ n−3 PUFA | 12.1 | 17.98 | 8.2–11.0 | 1.8–6.2 |
| 16:4n−3 | - | 7.82 | - | - |
| 18:3n−3 | 3.41 | 4.09 | 1.1–2.1 | 0.6–2.5 |
| 18:4n−3 | - | 3.17 | 1.0–2.2 | 0.0–0.7 |
| 20:5n−3 | 7.75 | 0.70 | 1.6–2.3 | 0.8–3.2 |
| 22:5n−3 | 0.50 | 1.30 | 0.3–2.9 | - |
| 22:6n−3 | 0.24 | - | 0.0–0.4 | 0.0–1.6 |
| Σ PUFA | 21.1 | 23.62 | 15.1–18.8 | 13.3–20.4 |
| n−6/n−3 | 0.74 | 0.31 | 0.79–0.96 | 2.0–4.5 |
| ARA + EPA | 12.85 | 2.88 | 3.6–8.2 | 4.1–9.3 |
| Peptide | Hydrophobicity | Charge | Amphipathicity | Self-Agg | Disorder (%) | Helix (%) | Bioactivity Potential |
|---|---|---|---|---|---|---|---|
| VGDIARIY | 0.01 | 0.00 | 0.27 | 5–9 | 100 | 22.22 | High solubility, coil–β mix, balanced traits |
| ETGIKVVDL | −0.01 | −1.00 | 0.55 | 4–7 | 100 | 33.33 | Highest β-strand (66.67%), soluble, possible structural bioactivity |
| APILPVVGK | 0.17 | 1.00 | 0.41 | 3–7 | 100 | – | Amphipathic, positively charged (ACE/DPP-IV relevance) |
| TVDAAGKVA | −0.11 | 1.00 | 0.55 | 4–7 | 100 | – | Neutral hydrophobicity, amphipathic, good for receptor binding |
| VLVGGSTRIP | 0.04 | 1.00 | 0.25 | 1–4 | 100 | – | Slightly hydrophobic, basic, low aggregation– stable ligand candidate |
| Extraction Method | Solvent | TPC | Key Phenolic Compounds | Antioxidant Activity | References |
|---|---|---|---|---|---|
| Microwave-Assisted | Ethanol | 2.7 mg GAE/g | Gallic acid, Chlorogenic acid | Elevated (DPPH, ABTS) | [32,39,54] |
| Solvent Extraction | Methanol | 17.67 mg GAE/g | Not specified | Elevated (DPPH, ABTS) | [32,39,54] |
| Ethanol Extraction | 70% Ethanol | 310.7 mg GAE/100 g | Not specified | Elevated (FRAP, ABTS) | [32,39,54] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Cervantes, A.M.; Prates, J.A.M.; Guil-Guerrero, J.L. Overview of Primary and Secondary Metabolites of Rugulopteryx okamurae Seaweed: Assessing Bioactivity, Scalability, and Molecular Mechanisms. Mar. Drugs 2025, 23, 351. https://doi.org/10.3390/md23090351
García-Cervantes AM, Prates JAM, Guil-Guerrero JL. Overview of Primary and Secondary Metabolites of Rugulopteryx okamurae Seaweed: Assessing Bioactivity, Scalability, and Molecular Mechanisms. Marine Drugs. 2025; 23(9):351. https://doi.org/10.3390/md23090351
Chicago/Turabian StyleGarcía-Cervantes, Ana Minerva, José A. M. Prates, and José Luis Guil-Guerrero. 2025. "Overview of Primary and Secondary Metabolites of Rugulopteryx okamurae Seaweed: Assessing Bioactivity, Scalability, and Molecular Mechanisms" Marine Drugs 23, no. 9: 351. https://doi.org/10.3390/md23090351
APA StyleGarcía-Cervantes, A. M., Prates, J. A. M., & Guil-Guerrero, J. L. (2025). Overview of Primary and Secondary Metabolites of Rugulopteryx okamurae Seaweed: Assessing Bioactivity, Scalability, and Molecular Mechanisms. Marine Drugs, 23(9), 351. https://doi.org/10.3390/md23090351








