Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis
Abstract
1. Introduction
2. Results
2.1. Material Characterization
2.2. Characteristics of Alloy Degradation Products
2.3. Administration of Active Compounds to Animals
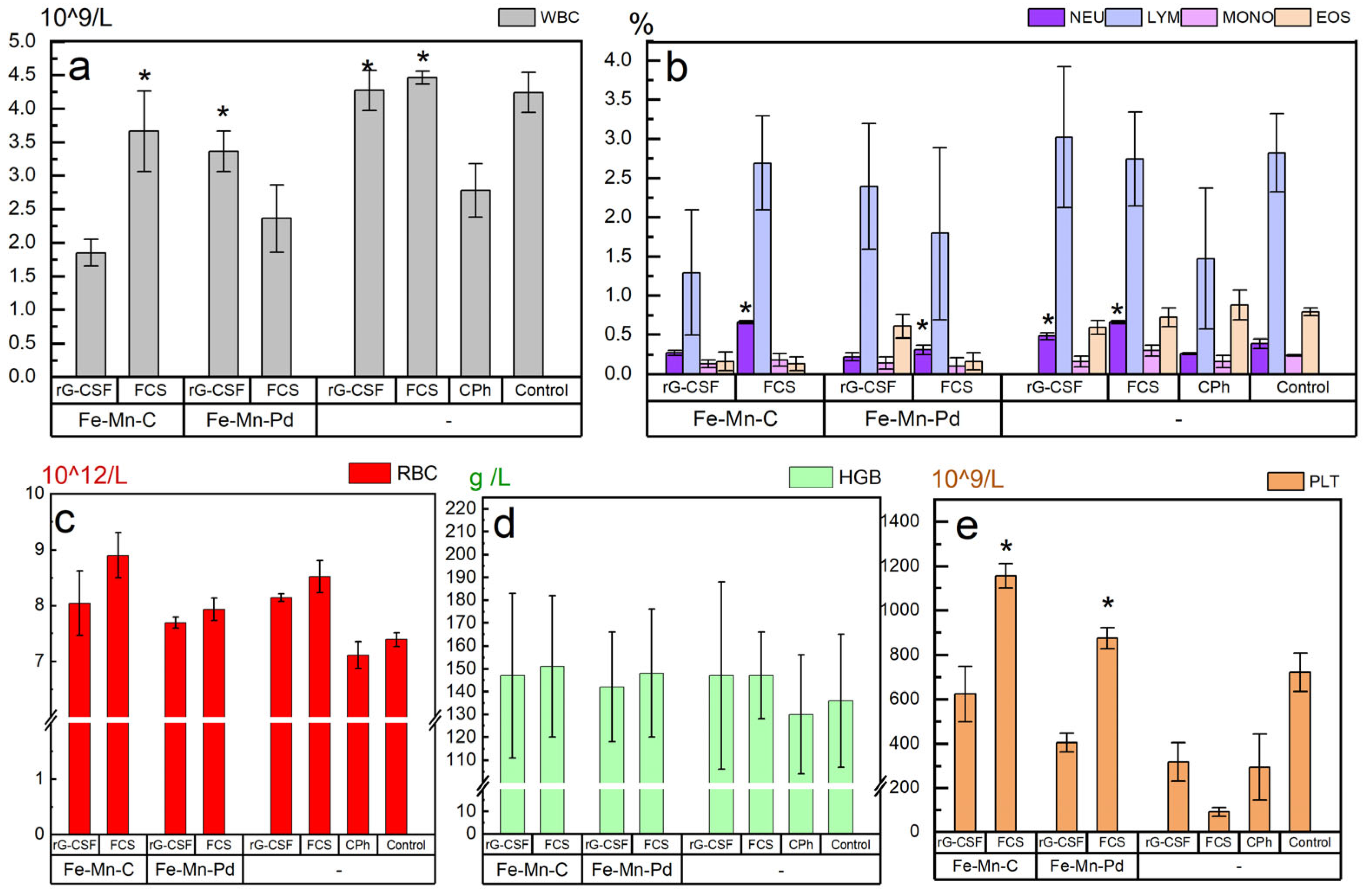
2.4. Hematological Parameters
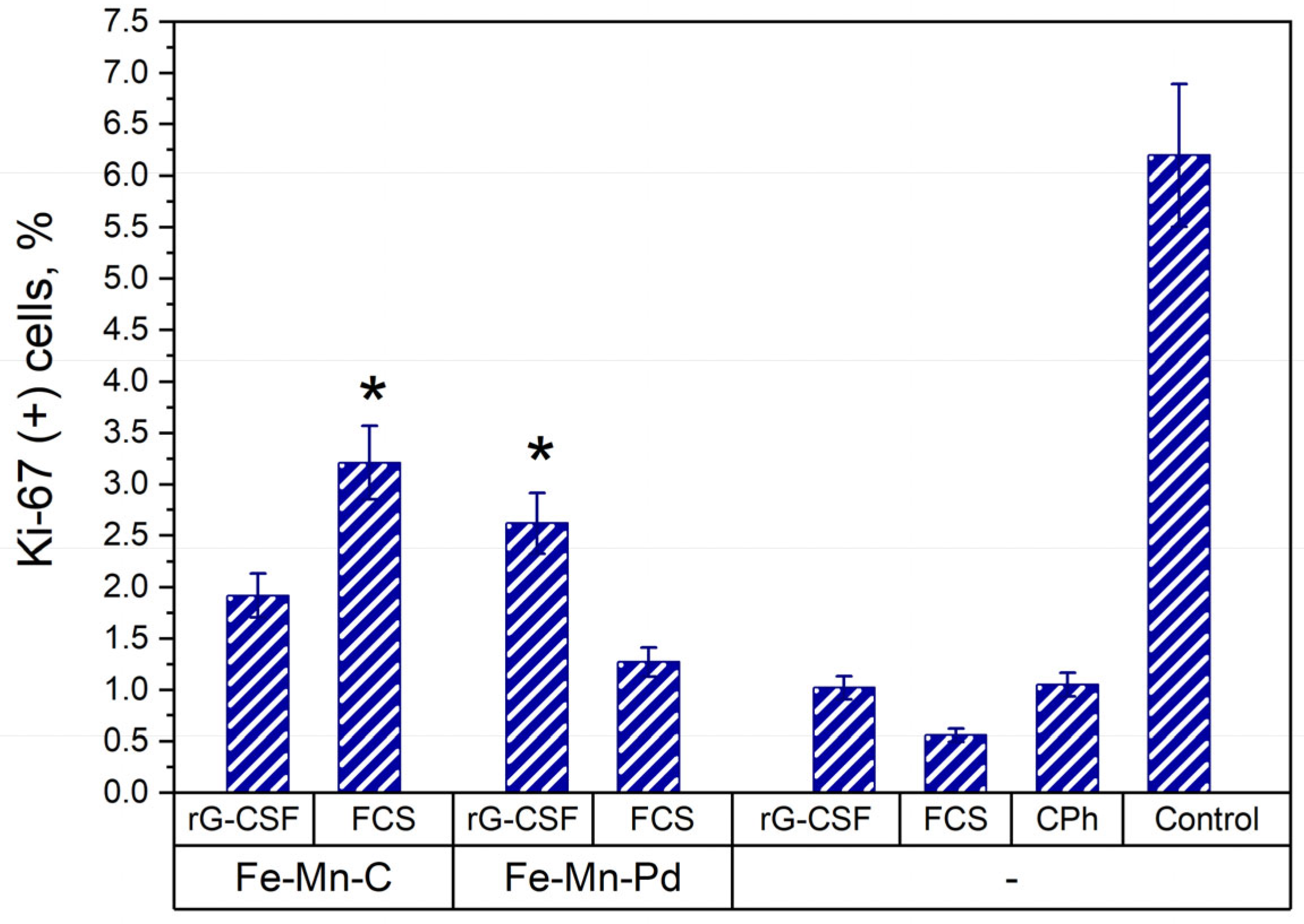
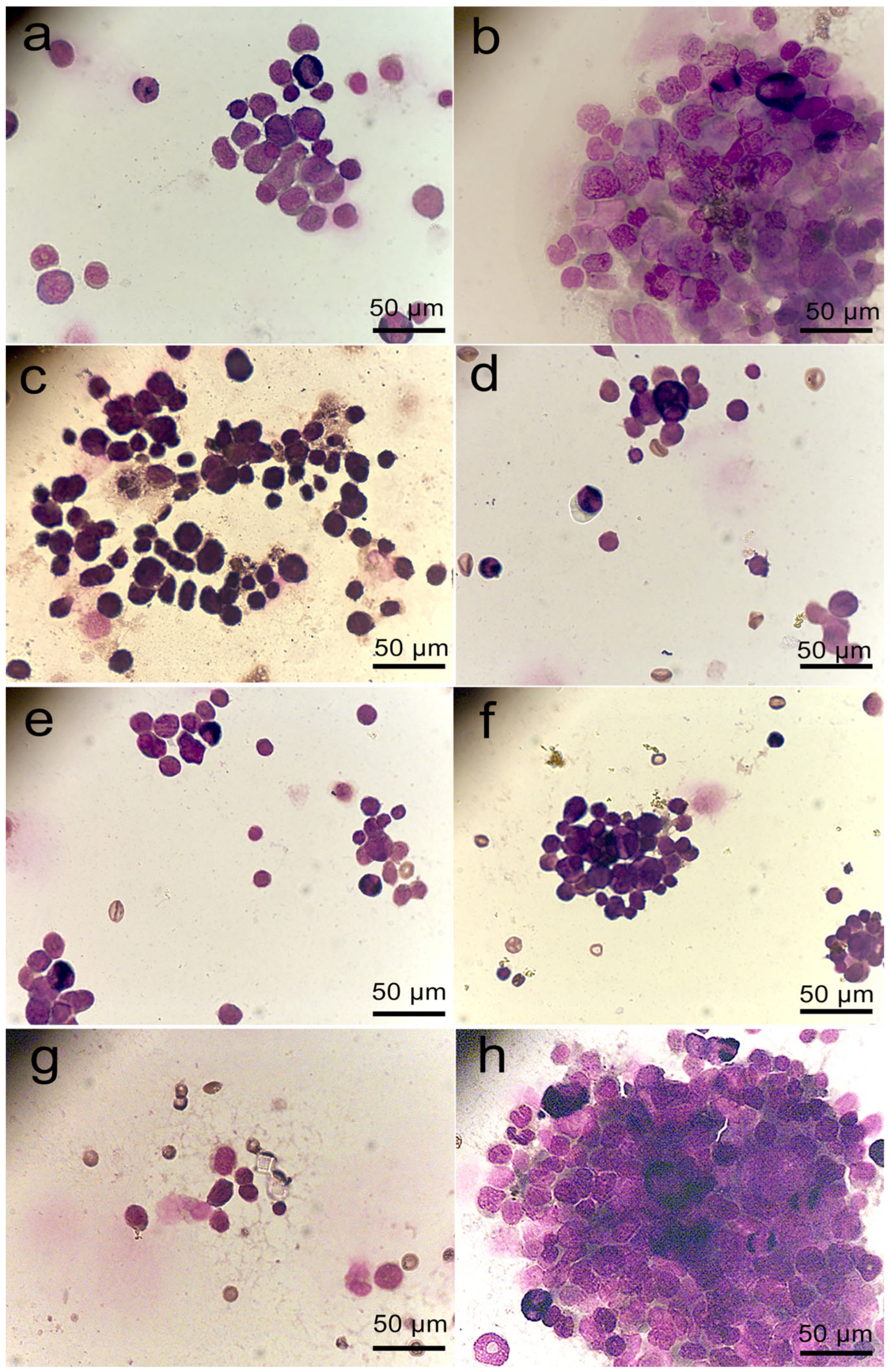
2.5. Proliferative Activity of Cells
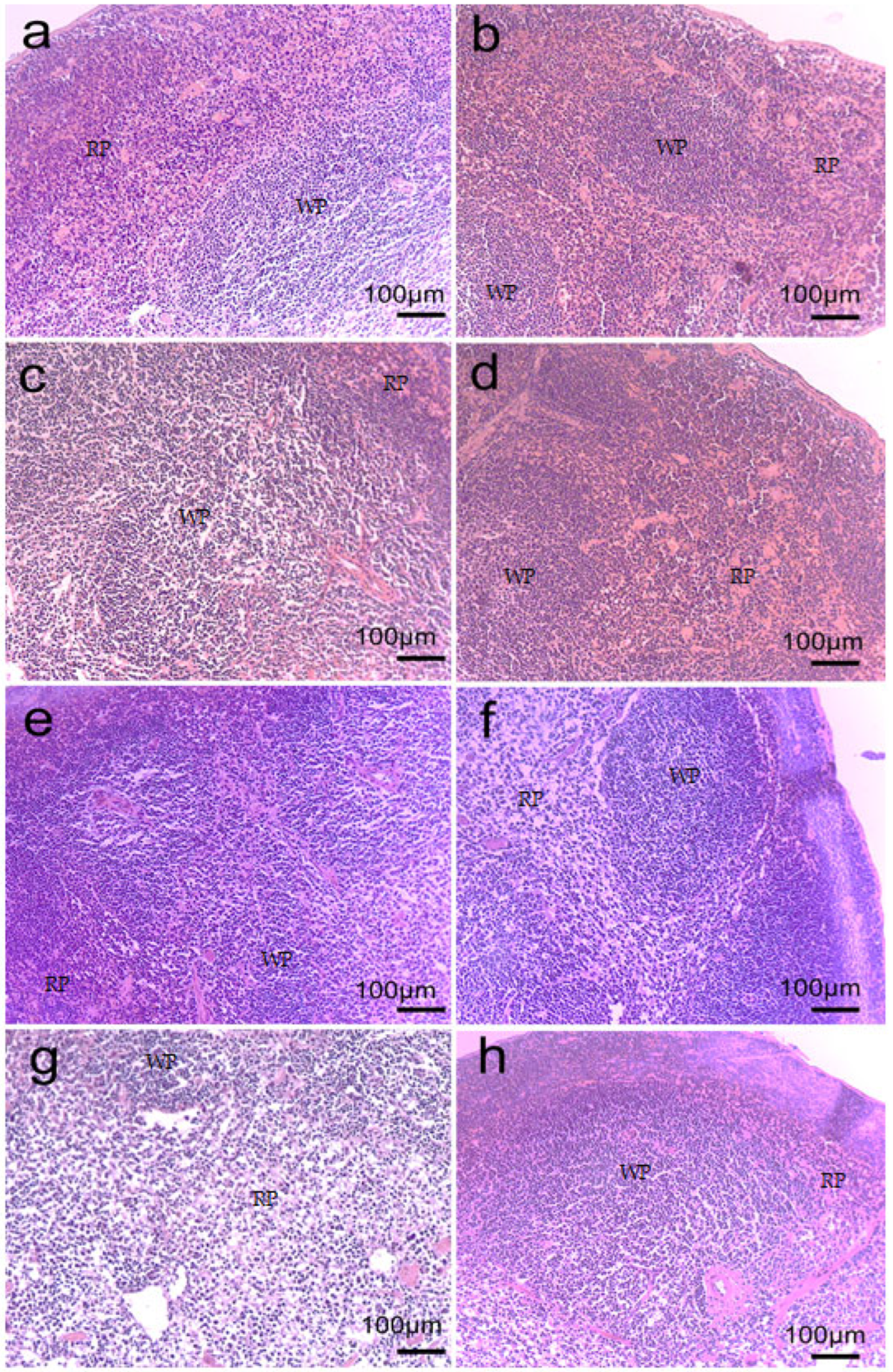
2.6. Spleen Morphology
3. Discussion
4. Materials and Methods
4.1. Sulfated Polysaccharides
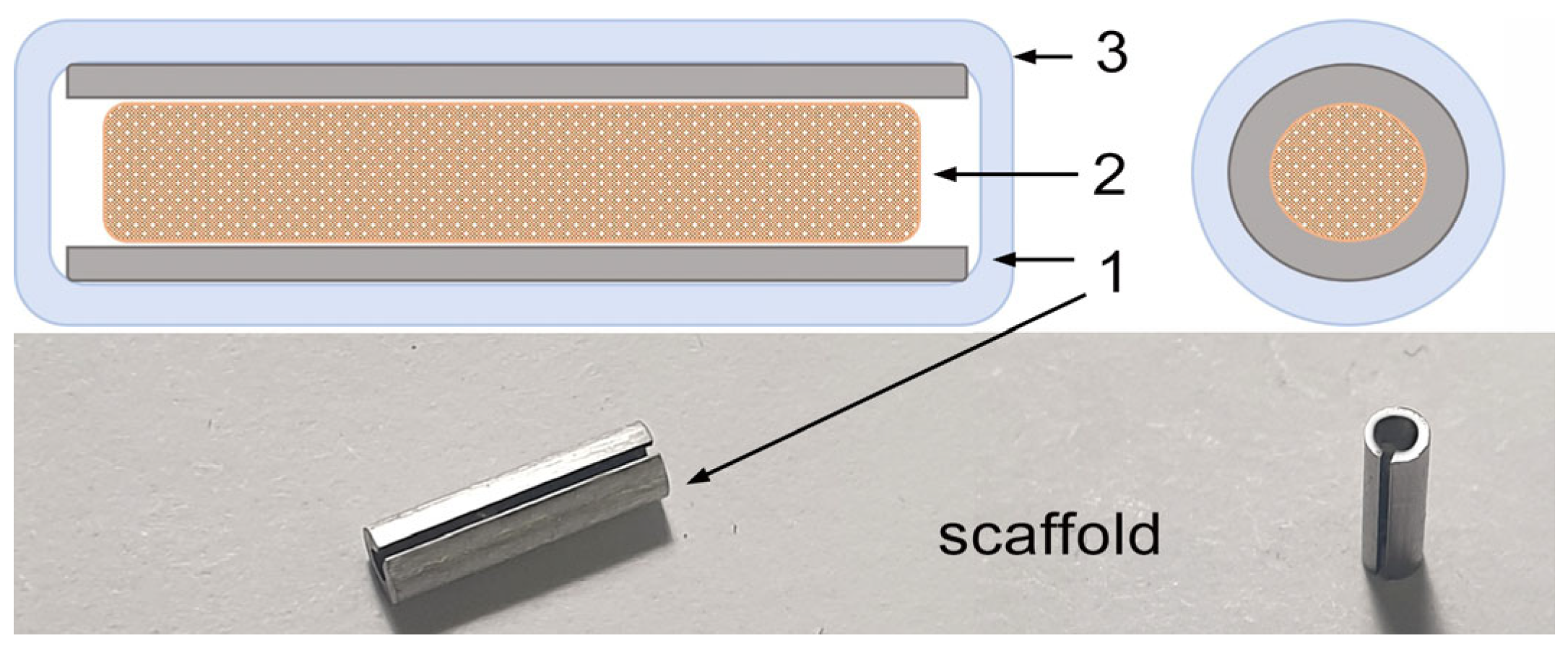
4.2. Scaffolds
4.3. Animal Model
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| rG-CSF | Recombinant human granulocyte colony stimulating factor, filgrastim |
| sTNF-Rs | Soluble tumor necrosis factor receptors |
| GM-CSF | Granulocyte-macrophage colony-stimulating factor, lenograstim |
| WBCs | White blood cells |
| RBCs | Red blood cells |
| HGB | Hemoglobin |
| PLT | Platelets |
| Neu | Neutrophils |
| Lym | Lymphocytes |
| Mono | Monocytes |
| Eos | Eosinophils |
| FCS | Fucosylated chondroitin sulfate |
| CPh | Cyclophosphamide |
| ECAP | Equal channel angular pressing |
| EDTA | Ethylenediaminetetraacetic acid |
| HE | hematoxylin-eosin |
References
- Morstyn, G.; Keech, J.; Sheridan, W.; Campbell, L.; Green, M.; Metcalf, D.; Fox, R.; Souza, L.M.; Alton, N.K. Effect of granulocyte colony stimulating factor on neutropenia induced by cytotoxic chemotherapy. Lancet 1988, 331, 667–672. [Google Scholar] [CrossRef]
- Scotte, F.; Simon, H.; Laplaige, P.; Antoine, E.C.; Spasojevic, C.; Texier, N.; Gouhier, K.; Chouaid, C. Febrile neutropenia prophylaxis, G-CSF physician preferences: Discrete-choice experiment. BMJ Support. Palliat. Care 2021, 14, e003082. [Google Scholar] [CrossRef]
- Straka, C.; Oduncu, F.; Hinke, A.; Einsele, H.; Drexler, E.; Schnabel, B.; Arseniev, L.; Walther, J.; König, A.; Emmerich, B. Responsiveness to G-CSF before leukopenia predicts defense to infection in high-dose chemotherapy recipients. Blood 2004, 104, 1989–1994. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.; Ozer, H.; Stoller, R.; Johnson, D.; Lyman, G.; Tabbara, I.; Kris, M.; Grous, J.; Picozzi, V.; Rausch, G.; et al. Reduction by granulocyte colony-stimulating factor of fever and neutropenia induced by chemotherapy in patients with small-cell lung cancer. N. Engl. J. Med. 1991, 325, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Hartung, T.; Döcke, W.D.; Gantner, F.; Krieger, G.; Sauer, A.; Stevens, P.; Volk, H.D.; Wendel, A. Effect of granulocyte colony-stimulating factor treatment on ex vivo blood cytokine response in human volunteers. Blood 1995, 85, 2482–2489. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, S.; Liu, J.; Liang, B.; Wang, X.; Wang, H.; Li, W.; Tong, Q.; Yi, J.; Zhao, L.; et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. eBioMedicine 2020, 55, 102763. [Google Scholar] [CrossRef]
- Kotto-Kome, A.C.; Fox, S.E.; Lu, W.; Yang, B.B.; Christensen, R.D.; Calhoun, D.A. Evidence that the granulocyte colony-stimulating factor (G-CSF) receptor plays a role in the pharmacokinetics of G-CSF and PegG-CSF using a G-CSF-R KO model. Pharmacol. Res. 2004, 50, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Bagrova, S.G. Granulocyte colony-stimulating factors in the prevention of febrile neutropenia. Eff. Farmakoter. 2015, 31, 6–15. (In Russian) [Google Scholar]
- Ribas, A.; Albanell, J.; Bellmunt, J.; Solé-Calvo, L.A.; Bermejo, B.; Gallardo, E.; Vidal, R.; Vera, R.; Eres, N.; Carulla, J.; et al. Five-day course of granulocyte colony-stimulating factor in patients with prolonged neutropenia after adjuvant chemotherapy for breast cancer is a safe and cost-effective schedule to maintain dose-intensity. J. Clin. Oncol. 1996, 14, 1573–1580. [Google Scholar] [CrossRef]
- Bronchud, M.H.; Howell, A.; Crowther, D.; Hopwood, P.; Souza, L.; Dexter, T.M. The use of granulocyte colony-stimulating factor to increase the intensity of treatment with doxorubicin in patients with advanced breast and ovarian cancer. Br. J. Cancer 1989, 60, 121–125. [Google Scholar] [CrossRef]
- Snegovoy, A.V.; Kagonia, L.M.; Kononenko, I.B.; Larionova, V.B.; Lunin, V.V.; Manyuk, L.V. Practical recommendations for the administration of colony-stimulating factors to prevent the development of febrile neutropenia in cancer patients. Malig. Tumors 2015, 4, 342–349. [Google Scholar]
- Molineux, G. The design and development of pegfilgrastim (PEG-rmetHuG-CSF, Neulasta). Curr. Pharm. Des. 2004, 10, 1235–1244. [Google Scholar] [CrossRef]
- Crawford, J.; Herndon, D.; Gmitter, K.; Weiss, J. The impact of myelosuppression on quality of life of patients treated with chemotherapy. Future Oncol. 2024, 20, 1515–1530. [Google Scholar] [CrossRef]
- Schiffer, C.A.; Bohlke, K.; Delaney, M.; Hume, H.; Magdalinski, A.J.; McCullough, J.J.; Omel, J.L.; Rainey, J.M.; Rebulla, P.; Rowley, S.D.; et al. Platelet transfusion for patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J. Clin. Oncol. 2018, 36, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Bohlius, J.; Bohlke, K.; Castelli, R.; Djulbegovic, B.; Lustberg, M.B.; Martino, M.; Mountzios, G.; Peswani, N.; Porter, L.; Tanaka, T.N.; et al. Management of cancer-associated anemia with erythropoiesis-stimulating agents: ASCO/ASH clinical practice guideline update. J. Clin. Oncol. 2019, 37, 1336–1351. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Ma, J.J.; Lu, C. Venous thromboembolism risk and erythropoiesis-stimulating agents for the treatment of cancer-associated anemia: A meta-analysis. Tumour Biol. 2014, 35, 603–613. [Google Scholar] [CrossRef] [PubMed]
- Kiselevskiy, M.V.; Anisimova, N.Y.; Ustyuzhanina, N.E.; Vinnitskiy, D.Z.; Tokatly, A.I.; Reshetnikova, V.V.; Chikileva, I.O.; Shubina, I.Z.; Kirgizov, K.I.; Nifantiev, N.E. Perspectives for the Use of Fucoidans in Clinical Oncology. Int. J. Mol. Sci. 2022, 23, 11821. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Gerbst, A.G.; Ushakova, N.A.; Tsvetkova, E.A.; Dmitrenok, A.S.; Nifantiev, N.E.; Usov, A.I. Anticoagulant and antithrombotic activities of modified xylofucan sulfate from the brown alga Punctaria plantaginea. Carbohydr. Polym. 2016, 136, 826–833. [Google Scholar] [CrossRef]
- Anisimova, N.; Ustyuzhanina, N.; Bilan, M.; Donenko, F.; Usov, A.; Kiselevskiy, M.; Nifantiev, N. Fucoidan and Fucosylated Chondroitin Sulfate Stimulate Hematopoiesis in Cyclophosphamide-Induced Mice. Mar. Drugs 2017, 15, 301. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Anisimova, N.Y.; Bilan, M.I.; Donenko, F.V.; Morozevich, G.E.; Yashunskiy, D.V.; Usov, A.I.; Siminyan, N.G.; Kirgisov, K.I.; Varfolomeeva, S.R.; et al. Chondroitin Sulfate and Fucosylated Chondroitin Sulfate as Stimulators of Hematopoiesis in Cyclophosphamide-Induced Mice. Pharmaceuticals 2021, 14, 1074. [Google Scholar] [CrossRef]
- Bilan, M.I.; Anisimova, N.Y.; Tokatly, A.I.; Nikogosova, S.P.; Vinnitskiy, D.Z.; Ustyuzhanina, N.E.; Dmitrenok, A.S.; Tsvetkova, E.A.; Kiselevskiy, M.V.; Nifantiev, N.E.; et al. Glycosaminoglycans from the Starfish Lethasterias fusca: Structures and Influence on Hematopoiesis. Mar. Drugs 2023, 21, 205. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Anisimova, N.Y.; Nikogosova, S.P.; Dmitrenok, A.S.; Tsvetkova, E.A.; Panina, E.G.; Sanamyan, N.P.; Avilov, S.A.; Stonik, V.A.; et al. Fucosylated Chondroitin Sulfates with Rare Disaccharide Branches from the Sea Cucumbers Psolus peronii and Holothuria nobilis: Structures and Influence on Hematopoiesis. Pharmaceuticals 2023, 16, 1673. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Anisimova, N.Y.; Dmitrenok, A.S.; Tsvetkova, E.A.; Kiselevskiy, M.V.; Nifantiev, N.E.; Usov, A.I. Depolymerization of a fucosylated chondroitin sulfate from Cucumaria japonica: Structure and activity of the product. Carbohydr. Polym. 2022, 281, 119072. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Borodina, E.Y.; Stonik, V.A.; Nifantiev, N.E.; Usov, A.I. Two fucosylated chondroitin sulfates from the sea cucumber Eupentacta fraudatrix. Carbohydr. Polym. 2017, 164, 8–12. [Google Scholar] [CrossRef]
- Drynda, A.; Hassel, T.; Bach, F.W.; Peuster, M. In vitro and in vivo corrosion properties of new iron–manganese alloys designed for cardiovascular applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H.; Purnama, A.; Dube, D.; Couet, J.; Mantovani, D. Fe–Mn alloys for metallic biodegradable stents: Degradation and cell viability studies. Acta Biomater. 2010, 6, 1852–1860. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Qin, L.; Qi, H.; Zhang, D.; Zhang, G.; Gao, R.; Qiu, H.; Xia, Y.; Cao, P.; Wang, X. Long-term in vivo corrosion behavior, biocompatibility and bioresorption mechanism of a bioresorbable nitrided iron scaffold. Acta Biomater. 2017, 54, 454–468. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Moszner, F.; Fischerauer, S.; Fiedler, M.; Martinelli, E.; Eichler, J.; Witte, F.; Willbold, E.; Schinhammer, M.; Meischel, M.; et al. Biodegradable Fe-based alloys for use in osteosynthesis: Outcome of an in vivo study after 52 weeks. Acta Biomater. 2014, 10, 3346–3353. [Google Scholar] [CrossRef]
- Rybalchenko, O.; Anisimova, N.; Martynenko, N.; Rybalchenko, G.; Kiselevskiy, M.; Tabachkova, N.; Shchetinin, I.; Raab, A.; Dobatkin, S. Structure Optimization of a Fe–Mn–Pd Alloy by Equal-Channel Angular Pressing for Biomedical Use. Materials 2023, 16, 45. [Google Scholar] [CrossRef]
- Rybalchenko, O.V.; Anisimova, N.Y.; Kiselevsky, M.V.; Rybalchenko, G.V.; Martynenko, N.S.; Bochvar, N.R.; Tabachkova, N.Y.; Shchetinin, I.V.; Shibaeva, T.V.; Konushkin, S.V.; et al. Effect of equal-channel angular pressing on structure and properties of Fe-Mn-C alloys for biomedical applications. Mater. Today Commun. 2022, 30, 103048. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, C.O.; Torres-García, S.E.; Olvera-Sandoval, C.; Ramírez-Castillo, F.Y.; Muro, A.L.; Avelar-Gonzalez, F.J.; Guerrero-Barrera, A.L. Cell culture: History, development and prospects. Int. J. Curr. Res. Aca. Rev. 2014, 2, 188–200. [Google Scholar]
- Panopoulos, A.D.; Watowich, S.S. Granulocyte colony-stimulating factor: Molecular mechanisms of action during steady state and ‘emergency’ hematopoiesis. Cytokine 2008, 42, 277–288. [Google Scholar] [CrossRef]
- Nogueira-Pedro, A.; Dos Santos, G.G.; Oliveira, D.C.; Hastreiter, A.A.; Fock, R.A. Erythropoiesis in vertebrates: From ontogeny to clinical relevance. Front. Biosci. 2016, 8, 100–112. [Google Scholar] [CrossRef]
- Oliveira, D.C.; Nogueira-Pedro, A.; Santos, E.W.; Hastreiter, A.; Silva, G.B.; Borelli, P.; Fock, R.A. A review of select minerals influencing the haematopoietic process. Nutr. Res. Rev. 2018, 31, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Callens, C.; Coulon, S.; Naudin, J.; Boldt, D.H. Targeting iron homeostasis induces cellular differentiation and synergizes with differentiating agents in acute myeloid leukemia. J. Exp. Med. 2010, 207, 731–750. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, O.; Kalidas, M.; Baltathakis, I.; Boldt, D.H. Expression of multiple genes regulating cell cycle and apoptosis in differentiating hematopoietic cells is dependent on iron. Exp. Hematol. 2001, 29, 1060–1069. [Google Scholar] [CrossRef] [PubMed]
- Seligman, P.A.; Kovar, J.; Gelfand, E.W. Lymphocyte proliferation is controlled by both iron availability and regulation of iron uptake pathways. Pathobiology 1992, 60, 19–26. [Google Scholar] [CrossRef]
- Katz, N.; Rader, D.J. Manganese homeostasis: From rare single-gene disorders to complex phenotypes and diseases. J. Clin. Investig. 2019, 129, 5082–5085. [Google Scholar] [CrossRef]
- Case, A.J.; Madsen, J.M.; Motto, D.G.; Meyerholz, D.K.; Domann, F.E. Manganese superoxide dismutase depletion in murine hematopoietic stem cells perturbs iron homeostasis, globin switching, and epigenetic control in erythrocyte precursorcells. Free Radic. Biol. Med. 2013, 56, 17–27. [Google Scholar] [CrossRef]
- Wai, K.M.; Sawada, K.; Kumagai, M.; Itai, K.; Tokuda, I.; Murashita, K.; Nakaji, S.; Ihara, K. Relationship between Selected Trace Elements and Hematological Parameters among Japanese Community Dwellers. Nutrients 2020, 12, 1615. [Google Scholar] [CrossRef]
- Petrarca, C.; Clemente, E.; Di Giampaolo, L.; Mariani-Costantini, R.; Leopold, K.; Schindl, R.; Lotti, L.V.; Mangifesta, R.; Sabbioni, E.; Niu, Q.; et al. Palladium nanoparticles induce disturbances in cell cycle entry and progression of peripheral blood mononuclear cells: Paramount role of ions. J. Immunol. Res. 2014, 2014, 295092. [Google Scholar] [CrossRef]
- Kielhorn, J.; Melber, C.; Keller, D.; Mangelsdorf, I. Palladium-a review of exposure and effects to human health. Int. J. Hyg. Environ. Health 2002, 205, 417–432. [Google Scholar] [CrossRef]
- Rybalchenko, O.; Anisimova, N.; Martynenko, N.; Rybalchenko, G.; Belyakov, A.; Shchetinin, I.; Lukyanova, E.; Chernogorova, O.; Raab, A.; Pashintseva, N.; et al. Biocompatibility and Degradation of Fe-Mn-5Si Alloy after Equal-Channel Angular Pressing: In Vitro and In Vivo Study. Appl. Sci. 2023, 13, 9628. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Dmitrenok, A.S.; Shashkov, A.S.; Kusaykin, M.I.; Stonik, V.A.; Nifantiev, N.E.; Usov, A.I. Structural characterization and anticoagulant properties of sulfated polysaccharides of the sea cucumber Cucumaria japonica. Glycobiology 2016, 26, 449–459. [Google Scholar] [CrossRef]
- State Standard 53434-2009; National Standard of the Russian Federation. Principles of Good Laboratory Practice: Paris, France, 2010. Available online: https://docs.cntd.ru/document/1200075972 (accessed on 9 July 2025).



| № | Alloys | Treatment | σUTS [MPa] 1 | σYS [MPa] 2 | ε [%] 3 | CR [mm/y] 4 | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | Fe–Mn–C | ECAP at 450 °C (N = 6) | 1515 | 1327 | 12.5 | 0.63 | [30] |
| 2 | Fe–Mn–Pd | ECAP at 450 °C (N = 4) | 1669 | 1577 | 4 | 0.97 | [29] |
| Alloy | Mn | Pd | Si | Cu | C | P | S | Fe |
|---|---|---|---|---|---|---|---|---|
| Fe–Mn-C | 26.9 | - | 0.496 | 0.576 | 0.213 | 0.015 | 0.007 | Bal. |
| Fe–Mn-Pd | 22.20 | 0.88 | 0.78 | 0.38 | 0.01 | 0.013 | 0.005 | Bal. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anisimova, N.Y.; Rybalchenko, O.V.; Martynenko, N.S.; Rybalchenko, G.V.; Lukyanova, E.A.; Bilan, M.I.; Usov, A.I.; Kiselevskiy, M.V.; Nifantiev, N.E. Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis. Mar. Drugs 2025, 23, 344. https://doi.org/10.3390/md23090344
Anisimova NY, Rybalchenko OV, Martynenko NS, Rybalchenko GV, Lukyanova EA, Bilan MI, Usov AI, Kiselevskiy MV, Nifantiev NE. Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis. Marine Drugs. 2025; 23(9):344. https://doi.org/10.3390/md23090344
Chicago/Turabian StyleAnisimova, Natalia Y., Olga V. Rybalchenko, Natalia S. Martynenko, Georgy V. Rybalchenko, Elena A. Lukyanova, Maria I. Bilan, Anatolii I. Usov, Mikhail V. Kiselevskiy, and Nikolay E. Nifantiev. 2025. "Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis" Marine Drugs 23, no. 9: 344. https://doi.org/10.3390/md23090344
APA StyleAnisimova, N. Y., Rybalchenko, O. V., Martynenko, N. S., Rybalchenko, G. V., Lukyanova, E. A., Bilan, M. I., Usov, A. I., Kiselevskiy, M. V., & Nifantiev, N. E. (2025). Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis. Marine Drugs, 23(9), 344. https://doi.org/10.3390/md23090344








