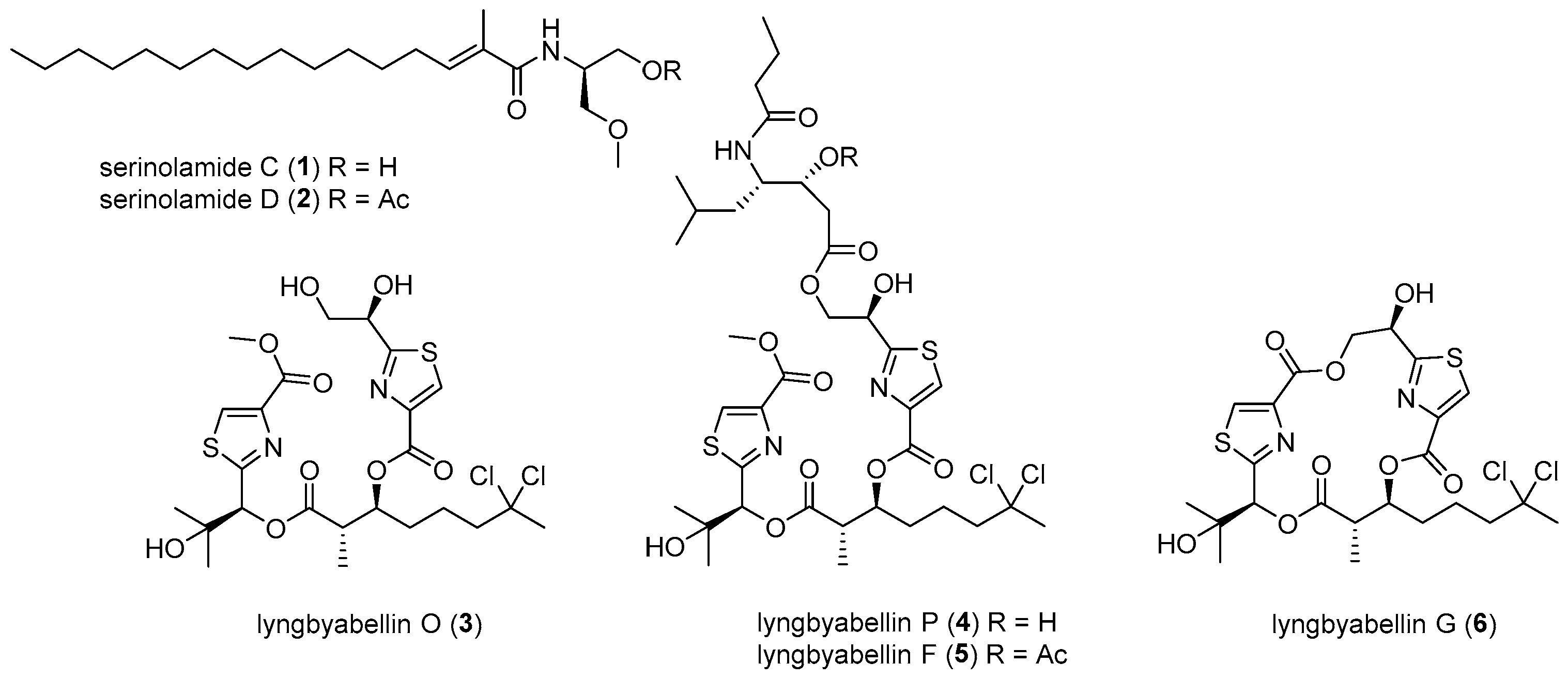
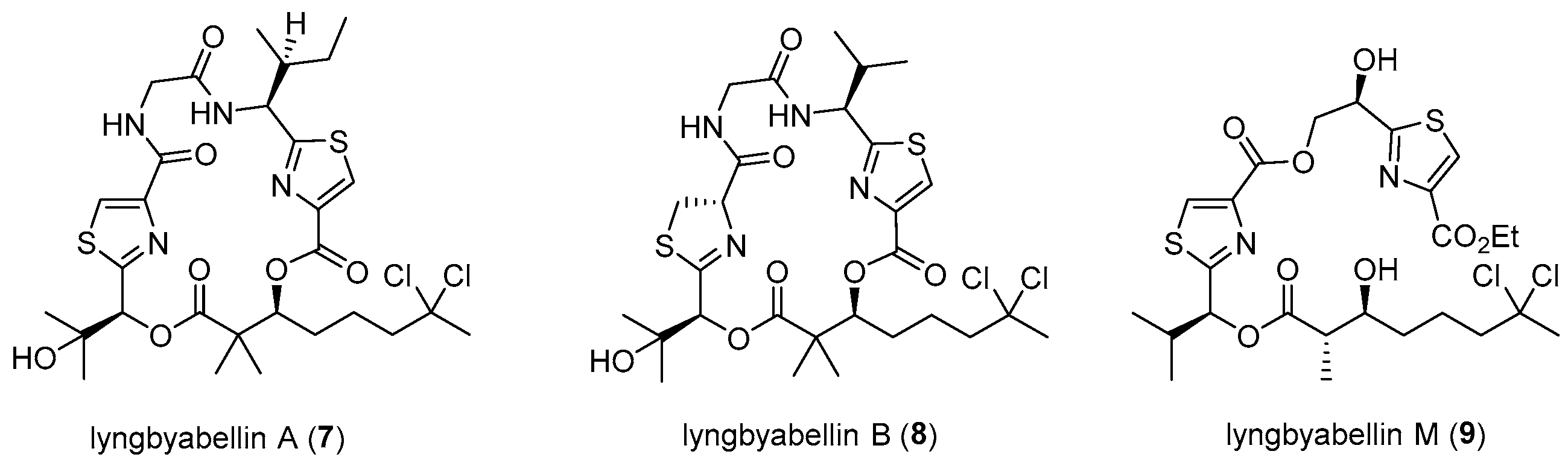
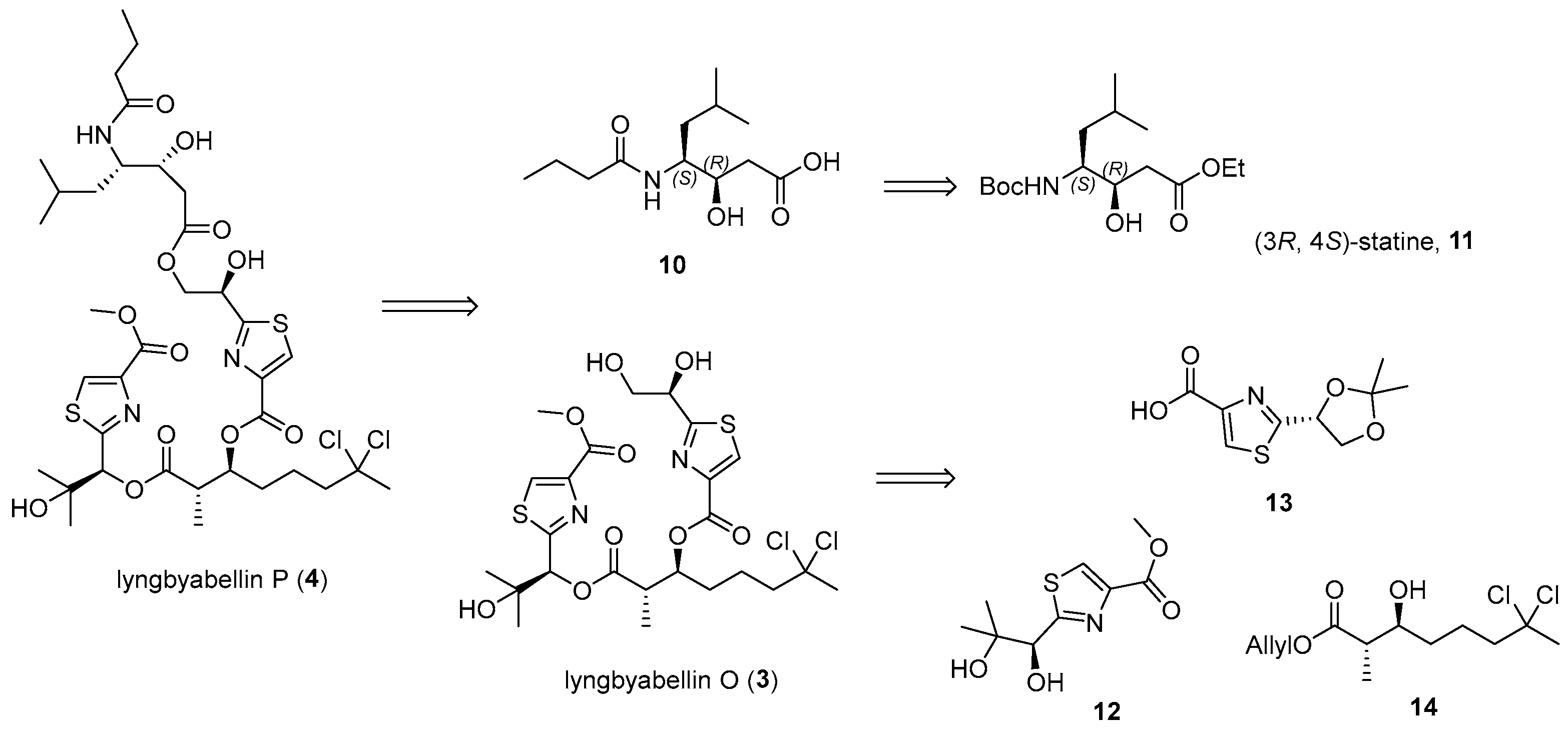
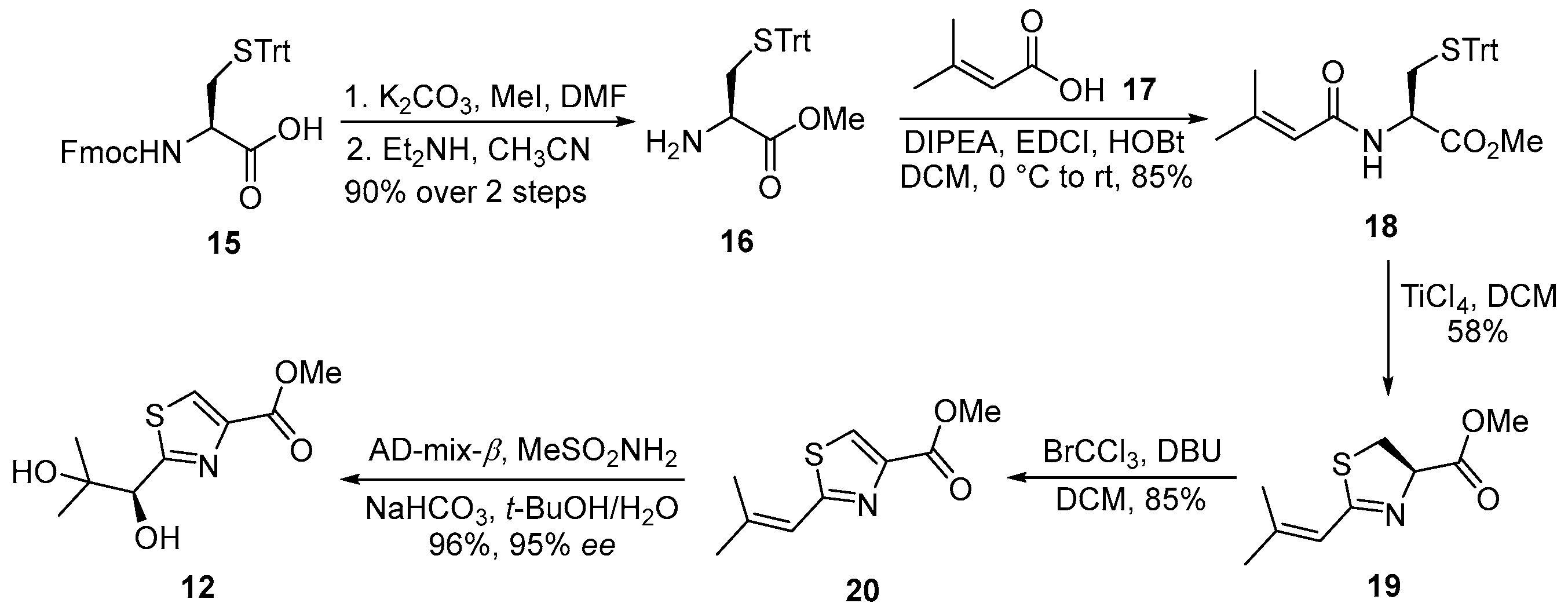
3.2. General Experimental Procedures
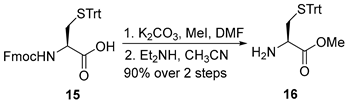
3.2.1. Methyl S-Trityl-L-cysteinate (16)
![Marinedrugs 23 00340 i001 Marinedrugs 23 00340 i001]()
To a stirred solution of N-(((9H-fluoren-9-yl)methoxy)carbonyl)-S-trityl-D-cysteine (15) (5.0 g, 8.5 mmol, 1.0 equiv.) in DMF (100 mL, 0.085 M) at ambient temperature was added K2CO3 (1.4 g, 10.2 mmol, 1.2 equiv.), followed by methyl iodide (MeI; 1.1 mL, 17 mmol, 2.0 equiv.). The reaction mixture was stirred at room temperature for 3 h, after which the insoluble materials were removed by filtration through a Celite® pad, and the pad was washed with ethyl acetate (3 × 100 mL). The combined organic phases were concentrated under reduced pressure to yield a colorless oil, which was used directly in the subsequent step without further purification.
The crude residue was dissolved in acetonitrile (100 mL, 0.085 M), and diethylamine (Et2NH; 1.1 mL, 10.2 mmol, 1.2 equiv.) was added at room temperature. The resulting solution was stirred for 1 h and then concentrated under reduced pressure. The crude product was purified by flash chromatography on silica gel (hexanes/EtOAc = 3:1) to afford compound 16 as a colorless oil (2.9 g, 90% yield over two steps). TLC: Rf = 0.3 (hexanes/EtOAc = 2/1), UV and PMA stain. = +24.5 (c 0.8, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 7.66–6.90 (m, 15H), 3.66 (s, 3H), 3.21 (dd, J = 7.8, 4.8 Hz, 1H), 2.60 (dd, J = 12.5, 4.8 Hz, 1H), 2.48 (dd, J = 12.5, 7.8 Hz, 1H). 13C-NMR (100 MHz, CDCl3) δ 174.2, 144.5, 129.6, 127.9, 126.8, 66.9, 53.8, 52.2, 36.9.
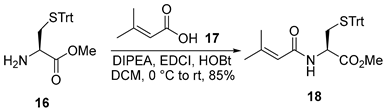
3.2.2. Methyl N-(3-Methylbut-2-enoyl)-S-trityl-L-cysteinate (18)
![Marinedrugs 23 00340 i002 Marinedrugs 23 00340 i002]()
To a solution of compound 16 (500 mg, 1.3 mmol, 1.0 equiv.) in anhydrous dichloromethane (DCM, 20 mL, 0.065 M) at 0 °C was added 3-methylbut-2-enoic acid (17, 130 mg, 1.3 mmol, 1.0 equiv.), followed by N,N-diisopropylethylamine (DIPEA, 1.4 mL, 7.8 mmol, 6.0 equiv.), 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI·HCl, 498 mg, 2.6 mmol, 2.0 equiv.), and 1-hydroxybenzotriazole (HOBt, 270 mg, 2.0 mmol, 1.5 equiv.). The reaction mixture was stirred at ambient temperature for 9 h. Upon completion, the reaction mixture was concentrated under reduced pressure, and the resulting residue was dissolved in ethyl acetate (EtOAc, 50 mL). The organic phase was washed with 4% aqueous citric acid solution, and the aqueous layer was extracted with EtOAc (3 × 30 mL). The combined organic extracts were washed sequentially with saturated aqueous sodium bicarbonate solution (30 mL) and brine (30 mL), dried over anhydrous sodium sulfate (Na2SO4), filtered, and concentrated under reduced pressure. The crude product was purified by flash column chromatography on silica gel (hexanes/EtOAc = 5:1) to afford compound 18 (508 mg, 85%) as a colorless oil. TLC: Rf= 0.3 (hexanes/EtOAc = 4/1), UV and PMA stain. = +4.6 (c 0.9, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 7.57–7.03 (m, 15H), 5.96 (d, J = 7.8 Hz, 1H), 5.70–5.33 (m, 1H), 4.73 (dt, J = 7.9, 5.2 Hz, 1H), 3.76 (s, 3H), 2.74 (dd, J = 5.2, 1.1 Hz, 2H), 2.20 (d, J = 1.3 Hz, 3H), 1.92 (d, J = 1.4 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 171.2, 166.2, 152.4, 144.3, 129.5, 128.0, 126.8, 117.8, 66.8, 52.6, 50.8, 33.9, 27.2, 19.9. HRMS (ESI) calculated for C28H29NNaO3S+ [M + Na]+ 482.1766, found 482.1762.
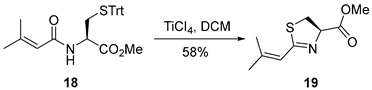
3.2.3. Methyl (R)-2-(2-Methylprop-1-en-1-yl)-4,5-dihydrothiazole-4-carboxylate (19)
![Marinedrugs 23 00340 i003 Marinedrugs 23 00340 i003]()
To a solution of compound 18 (400 mg, 0.87 mmol, 1.0 equiv.) in anhydrous dichloromethane (DCM, 10 mL, 0.087 M) at 0 °C under an argon atmosphere was added titanium tetrachloride (2.6 mL, 2.6 mmol, 3.0 equiv., 1.0 M in DCM). The reaction mixture was subsequently allowed to warm to room temperature and stirred for 12 h. Upon completion, the reaction was quenched with saturated aqueous sodium bicarbonate (5 mL), and the resulting aqueous phase was extracted with dichloromethane (3 × 20 mL). The combined organic extracts were washed sequentially with water (10 mL) and brine (10 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude residue was purified by flash column chromatography on silica gel (hexanes/EtOAc = 8:1), affording compound 19 (100 mg, 58%) as a colorless oil. TLC: Rf = 0.6 (hexanes/EtOAc = 8/1), UV and PMA stain. = +21.1 (c 1.1, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 6.28–5.85 (m, 1H), 5.10 (t, J = 9.0 Hz, 1H), 3.79 (s, 3H), 3.66–3.37 (m, 2H), 2.07 (d, J = 1.3 Hz, 3H), 1.88 (d, J = 1.3 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 171.6, 168.2, 148.5, 118.9, 77.3, 52.7, 35.3, 27.4, 20.7. HRMS (ESI) calculated for C9H13NNaO2S+ [M + Na]+ 222.0565, found 222.0560.
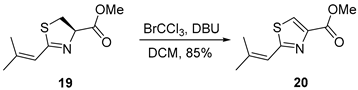
3.2.4. Methyl 2-(2-Methylprop-1-en-1-yl)thiazole-4-carboxylate (20)
![Marinedrugs 23 00340 i004 Marinedrugs 23 00340 i004]()
To a stirred solution of compound 19 (360 mg, 1.8 mmol, 1.0 equiv.) in anhydrous dichloromethane (DCM, 20 mL, 0.090 M) at 0 °C, bromotrichloromethane (BrCCl3, 0.22 mL, 2.2 mmol, 1.2 equiv.) and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU, 0.33 mL, 2.2 mmol, 1.2 equiv.) were added sequentially. The reaction mixture was gradually allowed to warm to ambient temperature and stirred for 12 h. Upon completion, the reaction was quenched with saturated aqueous ammonium chloride solution (8 mL). The resulting aqueous layer was extracted with ethyl acetate (2 × 30 mL), and the combined organic extracts were washed successively with water (30 mL) and brine (30 mL), dried over anhydrous sodium sulfate (Na2SO4), filtered, and concentrated under reduced pressure. The crude product was purified by flash column chromatography on silica gel (hexanes/EtOAc = 10:1) to afford alcohol 20 (301 mg, 85%) as a colorless oil. TLC: Rf = 0.4 (hexanes/EtOAc = 8/1), UV and PMA stain. 1H-NMR (400 MHz, CDCl3) δ 8.08 (d, J = 0.8 Hz, 1H), 6.76–5.97 (m, 1H), 3.92 (s, 3H), 2.21–2.04 (m, 3H), 1.98 (d, J = 1.4 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 166.2, 162.1, 146.2, 144.3, 126.3, 119.4, 52.4, 27.7, 20.8.
3.2.5. Methyl (S)-2-(1,2-Dihydroxy-2-methylpropyl)thiazole-4-carboxylate (12)
![Marinedrugs 23 00340 i005 Marinedrugs 23 00340 i005]()
To a solution of compound 20 (500 mg, 2.5 mmol, 1.0 equiv.) in a 1:1 mixture of tert-butanol and water (20 mL each, 0.063 M) at 0 °C were sequentially added methanesulfonamide (713 mg, 7.5 mmol, 3.0 equiv.), sodium bicarbonate (1.9 g, 23 mmol, 9.0 equiv.), and AD-mix-β (12 g, 15 mmol, 6.0 equiv.). The reaction mixture was stirred at 0 °C for 48 h, after which it was quenched with saturated aqueous sodium sulfite solution (30 mL). The resulting aqueous phase was extracted with ethyl acetate (3 × 100 mL), and the combined organic extracts were washed with water (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude residue was purified by flash column chromatography on silica gel (hexanes/EtOAc = 5:1) to afford acid 12 as a colorless oil (555 mg, 96% yield, 95% ee). TLC: Rf = 0.2 (hexanes/EtOAc = 1/1), UV and PMA stain. = −9.7 (c 1.1, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 8.04 (s, 1H), 5.21 (d, J = 5.2 Hz, 1H), 4.77 (d, J = 5.1 Hz, 1H), 3.85 (s, 1H), 3.79 (s, 3H), 1.19 (s, 3H), 1.05 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ 174.0, 161.9, 145.5, 128.3, 77.3, 73.0, 52.3, 25.3, 24.8. HRMS (ESI) calculated for C9H13NNaO4S+ [M + Na]+ 254.0463, found 254.0454.
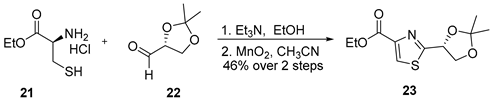
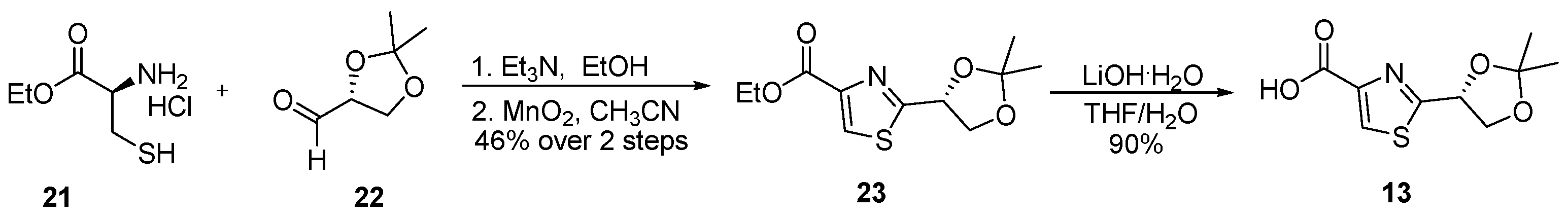
3.2.6. Ethyl (R)-2-(2,2-Dimethyl-1,3-dioxolan-4-yl)thiazole-4-carboxylate (23)
![Marinedrugs 23 00340 i006 Marinedrugs 23 00340 i006]()
To a stirred solution of L-cysteine ethyl ester hydrochloride (21) (5.0 g, 27 mmol, 1.0 equiv.) and triethylamine (4.2 mL, 30 mmol, 1.1 equiv.) in ethanol (250 mL, 0.11 M) at ambient temperature was added a solution of (R)-(+)-glyceraldehyde acetonide (22) (3.5 g, 27 mmol, 1.0 equiv.) in ethanol (10 mL). The reaction mixture was stirred at room temperature for 5 h, after which it was concentrated under reduced pressure. The resulting residue was dissolved in diethyl ether (200 mL), and the organic phase was successively washed with water (30 mL) and brine (30 mL), dried over anhydrous Na2SO4, filtered, and concentrated in vacuo. The crude product was carried forward to the next step without further purification.
The crude intermediate was dissolved in acetonitrile (250 mL, 0.11 M) and treated with manganese dioxide (47 g, 540 mmol, 20 equiv.) at room temperature. The reaction mixture was heated to 60 °C and stirred for 48 h. Upon completion, the suspension was filtered through a pad of Celite, and the filtrate was concentrated. The crude residue was purified by flash column chromatography on silica gel (hexanes/EtOAc = 3:1) to afford compound 23 as an orange oil (3.2 g, 46% yield over two steps). TLC: Rf = 0.3 (hexanes/EtOAc = 2/1), UV and PMA stain. = +15 (c 1.0, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 8.13 (s, 1H), 5.42 (ddd, J = 6.8, 4.9, 1.8 Hz, 1H), 4.48–4.36 (m, 3H), 4.09 (ddd, J = 9.0, 5.0, 1.7 Hz, 1H), 1.58 (s, 3H), 1.46 (s, 3H), 1.39 (td, J = 7.2, 1.8 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 172.5, 160.3, 146.5, 126.4, 110.2, 74.4, 69.4, 60.5, 25.4, 24.1, 13.4. HRMS (ESI) calculated for C11H15NNaO4S+ [M + Na]+ 280.0619, found 280.0612.
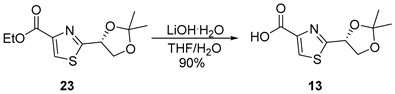
3.2.7. (R)-2-(2,2-Dimethyl-1,3-dioxolan-4-yl)thiazole-4-carboxylic acid (13)
![Marinedrugs 23 00340 i007 Marinedrugs 23 00340 i007]()
To a solution of compound 23 (2.0 g, 7.8 mmol, 1.0 equiv.) in THF/H2O (30 mL/15 mL, 0.17 M), LiOH·H2O (671 mg, 16 mmol, 2.0 equiv.) was added at 0 °C. The reaction mixture was stirred at ambient temperature for 2 h, after which it was quenched with 1 M HCl to adjust the pH to 2–3. The resulting mixture was extracted with EtOAc (3 × 100 mL), and the combined organic extracts were sequentially washed with water (50 mL) and brine (50 mL), dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The crude residue was purified by flash column chromatography on silica gel (DCM/MeOH = 10:1), affording acid 13 (1.6 g, 90%) as a colorless oil. TLC: Rf = 0.3 (DCM/MeOH = 10/1), UV and PMA stain. = +41.1 (c 2.0, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 8.27 (s, 1H), 5.45 (dd, J = 6.8, 5.0 Hz, 1H), 4.48 (dd, J = 8.9, 6.8 Hz, 1H), 4.11 (dd, J = 8.8, 4.9 Hz, 1H), 1.59 (s, 3H), 1.47 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ 174.4, 164.9, 146.6, 129.3, 111.5, 75.4, 70.5, 26.6, 25.2. HRMS (ESI) calculated for C9H11NNaO4S+ [M + Na]+ 252.0306, found 252.0300.
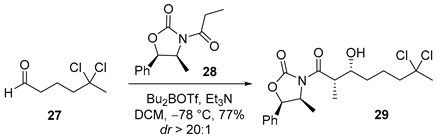
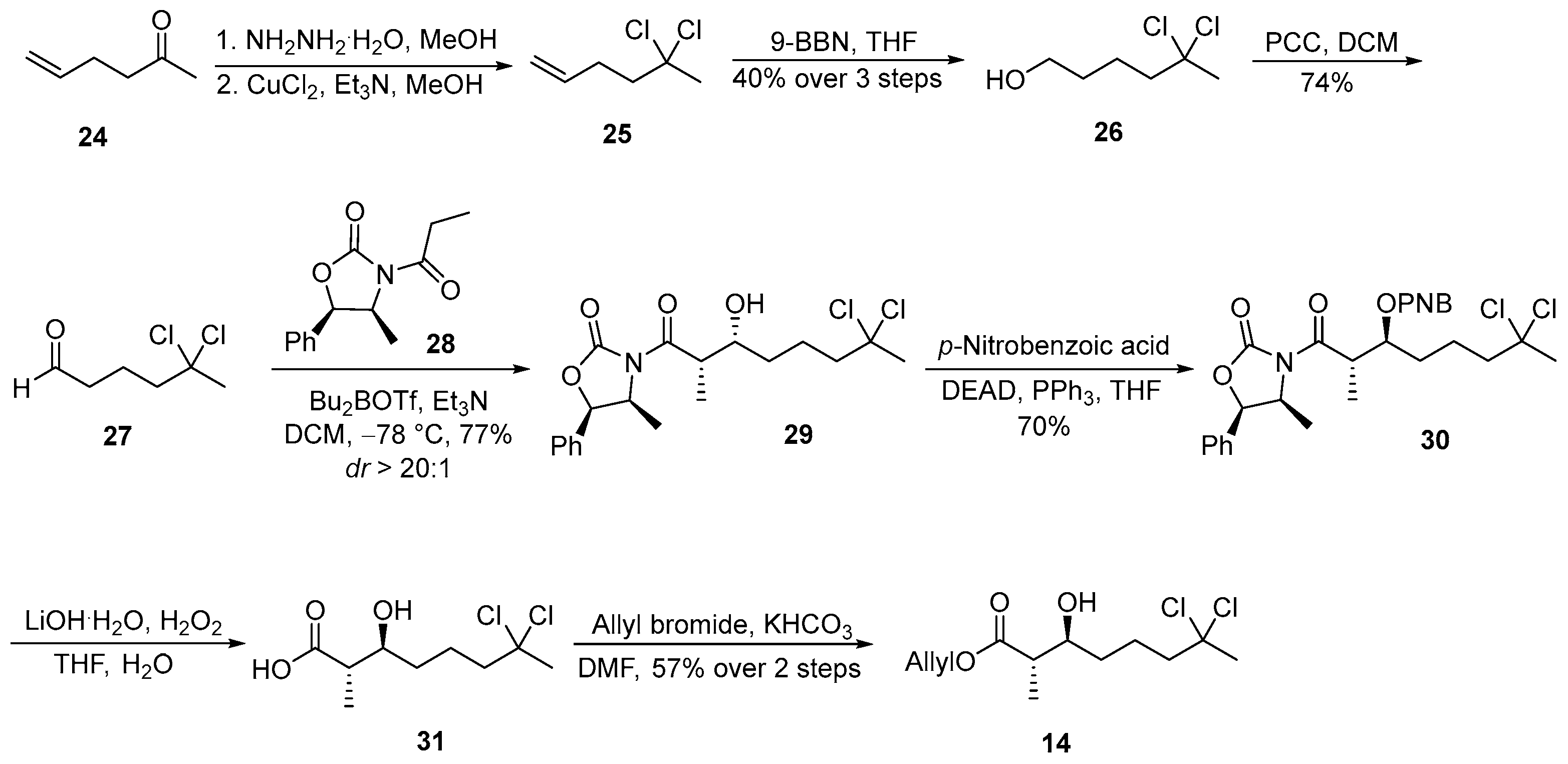
3.2.8. (4S,5R)-3-((2S,3R)-7,7-Dichloro-3-hydroxy-2-methyloctanoyl)-4-methyl-5-phenyloxazolidin-2-one (29)
![Marinedrugs 23 00340 i008 Marinedrugs 23 00340 i008]()
To a solution of oxazolidinone 28 (500 mg, 2.1 mmol, 1.5 equiv.) in anhydrous dichloromethane (DCM, 10 mL, 0.14 M) at −78 °C was added dibutylboron trifluoromethanesulfonate (2.2 mL, 2.2 mmol, 1.6 equiv., 1.0 M in DCM), followed by triethylamine (Et3N, 0.43 mL, 3.1 mmol, 2.2 equiv.). The reaction mixture was maintained at −78 °C for 30 min and then gradually warmed to 0 °C and stirred for an additional 1 h. After re-cooling the mixture to −78 °C, a solution of aldehyde 27 (235 mg, 1.4 mmol, 1.0 equiv.) in DCM (2.0 mL) was added dropwise. The resulting mixture was stirred at −78 °C for 30 min and then allowed to warm to 0 °C and stirred for a further 1.5 h. The reaction was quenched at 0 °C by the addition of 0.5 M phosphate buffer (pH 7.0, 5.0 mL), followed by methanol (12 mL) and 30% aqueous hydrogen peroxide (2.0 mL). The resulting mixture was stirred at 0 °C for 1 h, after which it was concentrated under reduced pressure. The aqueous residue was extracted with ethyl acetate (3 × 50 mL), and the combined organic extracts were washed sequentially with water (30 mL) and brine (30 mL), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo. The crude product was purified by flash column chromatography on silica gel (hexanes/EtOAc = 8:1) to afford acid 29 (441 mg, 77%) as a colorless oil, with a diastereomeric ratio (dr) of >20:1. TLC: Rf = 0.3 (hexanes/EtOAc = 3/1), UV and PMA stain. = −7.48 (c 1.0, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 7.48–7.28 (m, 5H), 5.69 (d, J = 7.3 Hz, 1H), 4.80 (p, J = 6.7 Hz, 1H), 4.00 (ddd, J = 9.0, 3.8, 2.7 Hz, 1H), 3.79 (qd, J = 7.1, 2.7 Hz, 1H), 2.37–2.19 (m, 2H), 2.15 (s, 3H), 1.91 (ddtd, J = 17.8, 9.8, 8.0, 7.3, 5.0 Hz, 1H), 1.74 (dddd, J = 15.2, 9.3, 6.1, 2.4 Hz, 1H), 1.62 (dtd, J = 14.5, 9.3, 5.0 Hz, 1H), 1.53–1.44 (m, 1H), 1.31–1.17 (m, 4H), 0.89 (d, J = 6.6 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 177.2, 152.6, 133.1, 128.9, 128.8, 125.6, 90.6, 79.0, 71.3, 54.8, 49.6, 42.4, 37.3, 33.1, 22.5, 14.4, 10.4. HRMS (ESI) calculated for C19H25Cl2NNaO4+ [M + Na]+ 424.1058, found 424.1062.
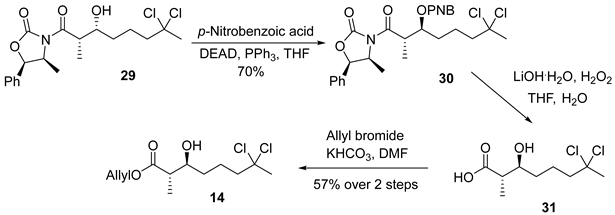
3.2.9. Allyl (2S,3S)-7,7-Dichloro-3-hydroxy-2-methyloctanoate (14)
![Marinedrugs 23 00340 i009 Marinedrugs 23 00340 i009]()
To a solution of compound 29 (400 mg, 1.0 mmol, 1.0 equiv.), triphenylphosphine (2.6 g, 10 mmol, 10 equiv.), and p-nitrobenzoic acid (1.0 g, 6.0 mmol, 6.0 equiv.) in anhydrous THF (20 mL, 0.05 M), a solution of diethyl azodicarboxylate (DEAD; 1.6 mL, 10 mmol, 10 equiv.) was added dropwise at room temperature. The reaction mixture was stirred for 3 h at ambient temperature, after which the solvent was removed under reduced pressure. The resulting residue was purified by flash column chromatography on silica gel (hexanes/EtOAc = 5:1) to afford ester 30 (385 mg, 70%) as a colorless oil. TLC: Rf = 0.5 (hexanes/EtOAc = 3/1), UV and PMA stain.
To a solution of ester 30 (385 mg, 0.70 mmol, 1.0 equiv.) in THF/H2O (4 mL/1 mL, 0.14 M) at 0 °C were added 30% aqueous hydrogen peroxide (0.32 mL, 2.8 mmol, 4.0 equiv.) and lithium hydroxide monohydrate (59 mg, 1.4 mmol, 2.0 equiv.). The reaction mixture was stirred at 0 °C for 2 h, followed by the addition of aqueous sodium thiosulfate (3 mL, 0.75 M). The resulting mixture was extracted with DCM (3 × 3 mL). The combined aqueous layers were acidified to approximately pH 1 using 6 M HCl and subsequently extracted with DCM (3 × 30 mL). The combined organic extracts were washed with water (30 mL), brine (30 mL), dried over anhydrous Na2SO4, filtered, and concentrated in vacuo to yield the crude acid, which was used directly in the next step without further purification.
To a solution of the crude product in DMF (5 mL, 0.14 M) at 0 °C were added KHCO3 (280 mg, 2.8 mmol, 4.0 equiv.) and allyl bromide (0.24 mL, 2.8 mmol, 4.0 equiv.). The reaction mixture was allowed to warm to room temperature and stirred for 8 h. Upon completion, the reaction mixture was diluted with diethyl ether (30 mL), and the organic phase was sequentially washed with 1 M KHSO4 (10 mL), water (10 mL), and brine (10 mL). The organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. Purification by flash column chromatography on silica gel (hexanes/EtOAc = 3:1) afforded compound 14 (113 mg, 57% over 2 steps) as a colorless oil. TLC: Rf = 0.3 (hexanes/EtOAc = 3/2), UV and PMA stain. = −3.3 (c 0.75, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 5.92 (dddd, J = 17.6, 10.0, 6.1, 5.3 Hz, 1H), 5.59–5.13 (m, 2H), 4.62 (d, J = 5.7 Hz, 1H), 3.71 (ddd, J = 9.4, 6.3, 3.6 Hz, 1H), 2.57 (p, J = 7.0 Hz, 1H), 2.23 (dt, J = 10.1, 5.7 Hz, 2H), 2.15 (s, 3H), 1.90 (dtd, J = 12.6, 10.3, 5.1 Hz, 1H), 1.82–1.69 (m, 1H), 1.65–1.54 (m, 1H), 1.49 (ddd, J = 14.2, 9.4, 5.3 Hz, 1H), 1.24 (d, J = 7.2 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 175.7, 132.0, 118.8, 90.7, 73.2, 65.4, 49.7, 45.4, 37.4, 34.0, 22.1, 14.5. HRMS (ESI) calculated for C12H20Cl2NaO3+ [M + Na]+ 305.0687, found 305.0681.
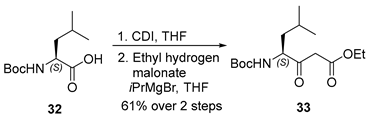
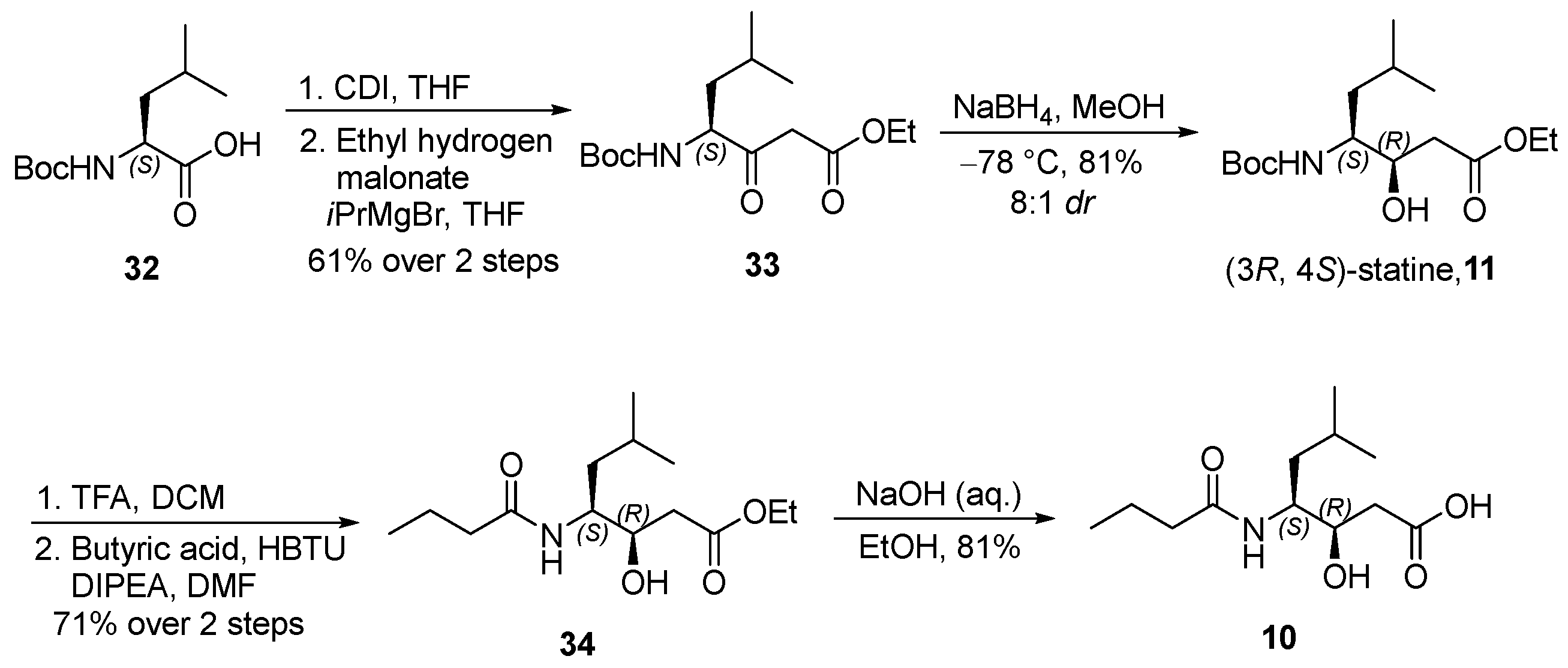
3.2.10. Ethyl (S)-4-((tert-Butoxycarbonyl)amino)-6-methyl-3-oxoheptanoate (33)
![Marinedrugs 23 00340 i010 Marinedrugs 23 00340 i010]()
To a solution of tert-butoxycarbonyl-L-leucine (32) (2.3 g, 10.0 mmol, 1.0 equiv.) in anhydrous tetrahydrofuran (THF, 50 mL, 0.20 M) under an argon atmosphere was added N,N′-carbonyldiimidazole (CDI, 1.8 g, 11.0 mmol, 1.1 equiv.) at 0 °C. The resulting mixture was gradually warmed to ambient temperature and stirred for 3.5 h to generate the corresponding acyl imidazole intermediate.
Separately, a 2.2 M solution of isopropylmagnesium bromide in diethyl ether (18.2 mL, 40.0 mmol, 4.0 equiv.) was added dropwise to a solution of ethyl hydrogen malonate (2.4 mL, 20.0 mmol, 2.0 equiv.) maintained at −10 °C. The reaction mixture was subsequently allowed to warm to room temperature and stirred for 1.5 h to generate magnesium enolate. The resulting enolate solution was cooled in an ice bath, and the previously prepared imidazolide solution was added dropwise at −10 °C. The combined reaction mixture was stirred at −10 °C for 30 min and then at ambient temperature for 24 h. The reaction was quenched by the addition of 10% aqueous citric acid (10 mL), followed by further acidification to pH 3 using an additional portion of 10% aqueous citric acid (30 mL). The aqueous layer was extracted with ethyl acetate (3 × 100 mL), and the combined organic extracts were washed successively with water (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude product was purified by flash column chromatography on silica gel (hexanes/ethyl acetate = 3:1) to afford β-ketoester 33 (1.8 g, 61% yield over two steps) as a colorless oil. TLC: Rf = 0.3 (hexanes/EtOAc = 5/1), UV and PMA stain. = −43.2 (c 4.1, MeOH). 1H-NMR (400 MHz, CDCl3) δ 5.07–4.89 (m, 1H), 4.43–4.26 (m, 1H), 4.15 (qd, J = 7.2, 1.9 Hz, 2H), 3.50 (t, J = 14.2 Hz, 2H), 1.67 (dd, J = 12.5, 5.5 Hz, 1H), 1.63–1.51 (m, 1H), 1.40 (s, 9H), 1.37–1.30 (m, 1H), 1.24 (td, J = 7.1, 2.0 Hz, 3H), 1.04–0.80 (m, 6H). 13C-NMR (100 MHz, CDCl3) δ 203.1, 167.1, 155.6, 80.1, 61.5, 58.3, 46.4, 39.9, 28.4, 24.9, 23.3, 21.6, 14.2. HRMS (ESI) calculated for C15H27NNaO5+ [M + Na]+ 324.1787, found 324.1776.
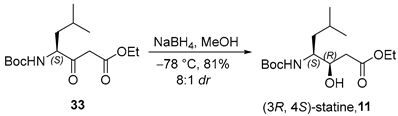
3.2.11. Ethyl (3R,4S)-4-((tert-Butoxycarbonyl)amino)-3-hydroxy-6-methylheptanoate (11)
![Marinedrugs 23 00340 i011 Marinedrugs 23 00340 i011]()
To a solution of β-ketoester 33 (800 mg, 2.7 mmol, 1.0 equiv.) in anhydrous methanol (25 mL, 0.11 M) under an argon atmosphere was added sodium borohydride (204 mg, 5.4 mmol, 2.0 equiv.) at −78 °C. The reaction mixture was stirred at −78 °C for 1 h, quenched with 10% aqueous citric acid (10 mL), and further acidified to pH 2 with an additional portion of 10% aqueous citric acid (20 mL). The resulting aqueous layer was extracted with ethyl acetate (3 × 50 mL), and the combined organic extracts were washed successively with water (50 mL) and brine (50 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude product was purified by flash column chromatography on silica gel (hexanes/EtOAc = 2:1) to afford alcohol 11 (663 mg, 81%, 8:1 dr) as a colorless oil. TLC: Rf = 0.3 (hexanes/EtOAc = 3/1), PMA stain. = −39.2 (c 2.6, MeOH). 1H-NMR (400 MHz, CDCl3) δ 4.71 (d, J = 9.1 Hz, 1H), 4.11 (q, J = 7.1 Hz, 2H), 3.98–3.84 (m, 1H), 3.71–3.26 (m, 1H), 2.41 (d, J = 7.9 Hz, 2H), 1.61 (dt, J = 13.6, 6.7 Hz, 1H), 1.38 (s, 9H), 1.28 (dd, J = 8.4, 5.9 Hz, 2H), 1.21 (t, J = 7.1 Hz, 3H), 0.86 (dd, J = 9.2, 6.6 Hz, 6H). 13C-NMR (100 MHz, CDCl3) δ 172.8, 156.2, 79.5, 71.4, 60.8, 52.8, 38.9, 38.3, 28.4, 24.8, 23.7, 21.6, 14.2. HRMS (ESI) calculated for C15H29NNaO5+ [M + Na]+ 326.1943, found 326.1923.
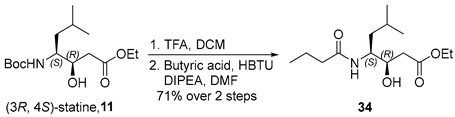
3.2.12. Ethyl (3R,4S)-4-Butyramido-3-hydroxy-6-methylheptanoate (34)
![Marinedrugs 23 00340 i012 Marinedrugs 23 00340 i012]()
To a solution of compound 11 (300 mg, 0.99 mmol, 1.0 equiv.) in anhydrous dichloromethane (DCM, 5 mL, 0.20 M) was added trifluoroacetic acid (TFA, 2.3 mL, 30 mmol, 30 equiv.) dropwise at 0 °C. The reaction mixture was allowed to warm to ambient temperature and stirred for 2 h. Upon completion, the reaction mixture was concentrated under reduced pressure to afford an oily residue, which was carried forward to the next step without further purification.
The resulting crude residue was dissolved in N,N-dimethylformamide (DMF, 5 mL, 0.20 M) and cooled to 0 °C. Butyric acid (0.10 mL, 1.1 mmol, 1.1 equiv.) was then added, followed by N,N-diisopropylethylamine (DIPEA, 0.70 mL, 4.0 mmol, 4.0 equiv.) and O-(benzotriazol-1-yl)-N,N,N′,N′-tetramethyluronium hexafluorophosphate (HBTU, 759 mg, 2.0 mmol, 2.0 equiv.). The reaction mixture was stirred at room temperature for 9 h and subsequently concentrated in vacuo to yield a crude solid.
The crude residue was dissolved in ethyl acetate (30 mL) and washed with 4% aqueous citric acid (5 mL). The aqueous phase was extracted with ethyl acetate (3 × 20 mL), and the combined organic layers were successively washed with saturated aqueous sodium bicarbonate (15 mL) and brine (15 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude product was purified by flash column chromatography on silica gel (hexanes/EtOAc = 1:1) to afford compound 34 (192 mg, 71% over two steps) as a white solid. TLC: Rf = 0.3 (hexanes/EtOAc = 1/1), UV and PMA stain. = −22.7 (c 1.0, MeOH). 1H-NMR (400 MHz, CDCl3) δ 5.86 (d, J = 8.6 Hz, 1H), 4.12 (q, J = 7.2 Hz, 2H), 4.04–3.94 (m, 2H), 3.87–3.76 (m, 1H), 2.42 (d, J = 6.1 Hz, 2H), 2.14 (dd, J = 8.1, 6.8 Hz, 2H), 1.73–1.51 (m, 3H), 1.38 (ddd, J = 14.7, 10.7, 4.2 Hz, 1H), 1.28 (dd, J = 10.1, 3.5 Hz, 1H), 1.23 (t, J = 7.1 Hz, 3H), 0.99–0.75 (m, 9H). 13C-NMR (100 MHz, CDCl3) δ 173.7, 172.8, 71.3, 60.9, 51.6, 38.8, 38.3, 24.9, 23.7, 21.6, 19.3, 14.2, 13.8. HRMS (ESI) calculated for C14H27NNaO4+ [M + Na]+ 296.1838, found 296.1830.
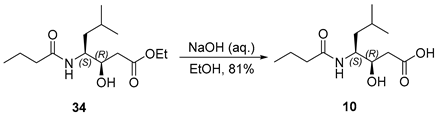
3.2.13. (3R,4S)-4-Butyramido-3-hydroxy-6-methylheptanoic acid (10)
![Marinedrugs 23 00340 i013 Marinedrugs 23 00340 i013]()
To a solution of compound 34 (200 mg, 0.73 mmol, 1.0 equiv.) in ethanol (3 mL, 0.24 M), 1 N sodium hydroxide solution (3 mL) was added dropwise. The resulting mixture was stirred at ambient temperature for 2 h. Subsequently, the ethanol was removed under reduced pressure, and the aqueous residue was diluted with water (20 mL) and cooled to 0 °C. The solution was then acidified to pH 2–3 by the careful addition of 1 N hydrochloric acid. The aqueous phase was extracted with ethyl acetate (3 × 20 mL), and the combined organic extracts were washed with brine (15 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The crude product was purified by flash chromatography on silica gel using a dichloromethane/methanol solvent system (10:1) to afford compound 10 (145 mg, 81%) as a colorless oil. TLC: Rf = 0.3 (DCM/MeOH = 10/1), UV and PMA stain. = −25.2 (c 3.3, MeOH). 1H-NMR (400 MHz, CDCl3) δ 6.47 (d, J = 8.5 Hz, 1H), 4.02 (t, J = 7.1 Hz, 2H), 2.46 (d, J = 6.6 Hz, 2H), 2.20 (t, J = 7.4 Hz, 2H), 1.61 (qd, J = 11.7, 11.3, 4.0 Hz, 3H), 1.48–1.36 (m, 1H), 1.35–1.26 (m, 1H), 0.92 (t, J = 7.2 Hz, 6H), 0.87 (d, J = 6.5 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 175.8, 175.0, 71.3, 52.0, 38.6, 38.3, 25.0, 23.7, 21.6, 19.4, 13.8. HRMS (ESI) calculated for C12H23NNaO4+ [M + Na]+ 268.1525, found 268.1515.
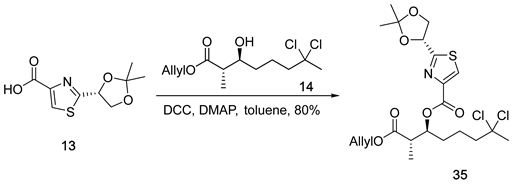
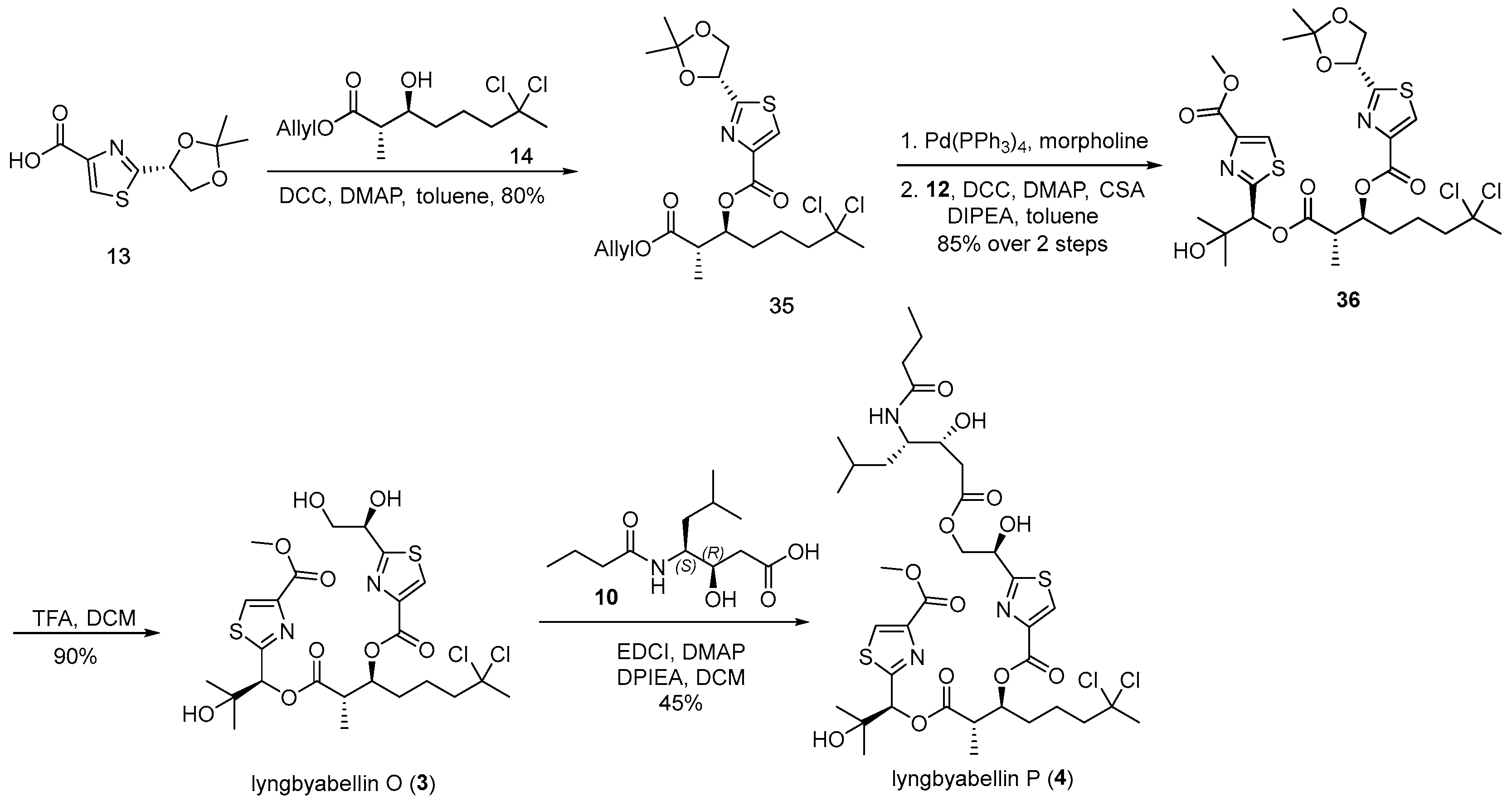
3.2.14. (2S,3S)-1-(Allyloxy)-7,7-dichloro-2-methyl-1-oxooctan-3-yl 2-((R)-2,2-di methyl- 1,3-dioxolan-4-yl)thiazole-4-carboxylate (35)
![Marinedrugs 23 00340 i014 Marinedrugs 23 00340 i014]()
A solution of acid 13 (133 mg, 0.58 mmol, 1.1 equiv.) and alcohol 14 (150 mg, 0.53 mmol, 1.0 equiv.) in anhydrous toluene (5 mL, 0.11 M) was prepared. To this solution, dicyclohexylcarbodiimide (DCC; 132 mg, 0.64 mmol, 1.2 equiv.) and 4-dimethylaminopyridine (DMAP; 78 mg, 0.64 mmol, 1.2 equiv.) were added at 0 °C. The reaction mixture was then stirred at ambient temperature for 6 h. Upon completion, the reaction was concentrated under reduced pressure to afford a crude residue. Purification of the residue by flash chromatography on silica gel, using hexanes/ethyl acetate (10:1) as the eluent, furnished ester 35 (209 mg, 80%) as a colorless oil. TLC: Rf = 0.3 (hexanes/EtOAc = 3/1), UV and PMA stain. = +11.4 (c 1.2, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 8.01 (s, 1H), 5.75 (ddt, J = 17.2, 10.4, 5.8 Hz, 1H), 5.29 (dd, J = 6.8, 4.9 Hz, 2H), 5.17 (dq, J = 17.2, 1.6 Hz, 1H), 5.06 (dq, J = 10.4, 1.3 Hz, 1H), 4.45 (dq, J = 5.9, 1.3 Hz, 2H), 4.34 (dd, J = 8.8, 6.8 Hz, 1H), 3.99 (dd, J = 8.8, 4.9 Hz, 1H), 2.86 (p, J = 7.0 Hz, 1H), 2.18 (ddd, J = 15.1, 6.6, 2.9 Hz, 1H), 2.12–2.03 (m, 1H), 1.99 (s, 3H), 1.78–1.58 (m, 4H), 1.46 (s, 3H), 1.34 (s, 3H), 1.13 (d, J = 7.1 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 173.5, 172.6, 160.2, 146.7, 131.8, 127.7, 118.4, 111.0, 90.1, 75.2, 74.9, 70.2, 65.2, 49.1, 43.1, 37.1, 30.4, 26.3, 24.9, 21.2, 12.8. HRMS (ESI) calculated for C21H29Cl2NNaO6S+ [M + Na]+ 516.0990, found 516.0990.
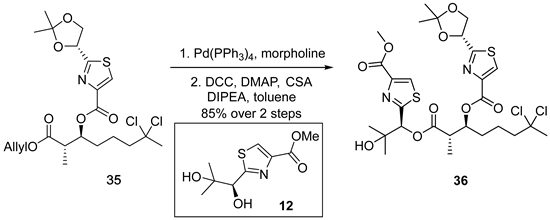
3.2.15. (2S,3S)-7,7-Dichloro-1-((S)-2-hydroxy-1-(4-(methoxycarbonyl)thiazol-2-yl)-2- methylpropoxy)-2-methyl-1-oxooctan-3-yl 2-((R)-2,2-dimethyl-1,3-dioxo lan-4-yl) thiazole-4-carboxylate (36)
![Marinedrugs 23 00340 i015 Marinedrugs 23 00340 i015]()
A solution of compound 35 (200 mg, 0.41 mmol, 1.0 equiv.) in anhydrous tetrahydrofuran (5 mL, 0.082 M) was treated with tetrakis(triphenylphosphine)palladium(0) (Pd(PPh3)4) (47 mg, 0.041 mmol, 0.1 equiv.) and morpholine (0.36 mL, 4.1 mmol, 10 equiv.) at ambient temperature. The reaction mixture was stirred for 15 min before being diluted with diethyl ether (30 mL). The resulting organic layer was washed sequentially with 1 M potassium bisulfate aqueous solution (5 mL) and brine (5 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to yield an oily residue. This residue was carried forward without further purification.
Subsequently, the crude acid thus obtained and alcohol 12 (104 mg, 0.45 mmol, 1.1 equiv.) were dissolved in anhydrous toluene (5 mL, 0.082 M). To this solution, N,N-diisopropylethylamine (DIPEA) (37 µL, 0.21 mmol, 0.5 equiv.), 4-dimethylaminopyridine (DMAP) (26 mg, 0.21 mmol, 0.5 equiv.), camphorsulfonic acid (CSA) (28 mg, 0.12 mmol, 0.3 equiv.), and dicyclohexylcarbodiimide (DCC) (136 mg, 0.66 mmol, 1.6 equiv.) were added at 0 °C. The reaction mixture was stirred at room temperature for 6 h and subsequently concentrated under reduced pressure to afford a crude residue. Purification by flash column chromatography on silica gel, employing dichloromethane/methanol (100:1, v/v) as eluent, furnished ester 36 (233 mg, 85% yield over two steps) as a colorless oil. TLC: Rf = 0.4 (DCM/MeOH = 30/1), UV and PMA stain. = −13.9 (c 1.0, CHCl3). 1H-NMR (400 MHz, CDCl3) δ 8.15 (s, 1H), 8.14 (s, 1H), 6.11 (s, 1H), 5.41 (dd, J = 6.8, 4.8 Hz, 1H), 5.39–5.33 (m, 1H), 4.45 (dd, J = 8.9, 6.8 Hz, 1H), 4.11 (s, 1H), 4.05 (dd, J = 8.9, 4.9 Hz, 1H), 3.90 (s, 3H), 2.97 (qd, J = 7.1, 4.3 Hz, 1H), 2.23 (dt, J = 14.6, 7.3 Hz, 1H), 2.20–2.08 (m, 1H), 2.08 (s, 3H), 1.77 (tdd, J = 18.0, 7.4, 4.6 Hz, 4H), 1.56 (s, 3H), 1.42 (s, 3H), 1.33 (s, 3H), 1.24 (d, J = 7.1 Hz, 3H), 1.15 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ 174.9, 170.9, 167.5, 161.7, 160.9, 146.7, 146.1, 128.6, 128.5, 111.3, 90.1, 78.6, 75.2, 75.0, 71.8, 70.5, 52.5, 49.1, 43.7, 37.4, 31.5, 26.8, 26.5, 25.5, 25.0, 21.7, 13.6. HRMS (ESI) calculated for C27H36Cl2N2NaO9S2+ [M + Na]+ 689.1137, found 689.1430.
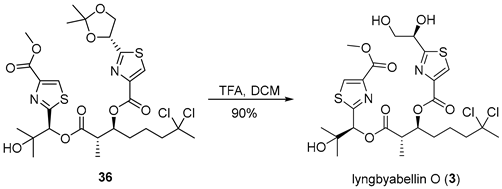
3.2.16. Lyngbyabellin O (3)
![Marinedrugs 23 00340 i016 Marinedrugs 23 00340 i016]()
A solution of compound 36 (30 mg, 0.045 mmol, 1.0 equiv.) in dichloromethane (3 mL, 0.015 M) was cooled to 0 °C, and trifluoroacetic acid (0.11 mL, 1.4 mmol, 30 equiv.) was added dropwise. The resulting mixture was stirred at ambient temperature for 2 h, after which the solvent was removed under reduced pressure to yield a solid residue. The crude product was purified by flash chromatography on silica gel using a dichloromethane/methanol solvent system (10:1) to afford lyngbyabellin O (3) (26 mg, 90%) as a white solid. TLC: Rf = 0.5 (DCM/MeOH = 10/1), UV and PMA stain. = −11.0 (c 1.0, MeOH). 1H-NMR (400 MHz, CDCl3) δ 8.21 (s, 1H), 8.16 (s, 1H), 6.14 (s, 1H), 5.41 (dt, J = 7.7, 4.8 Hz, 1H), 5.08 (t, J = 4.8 Hz, 1H), 4.12–3.95 (m, 2H), 3.94 (s, 3H), 3.06–2.93 (m, 1H), 2.33–2.20 (m, 1H), 2.23–2.14 (m, 1H), 2.12 (s, 3H), 1.95–1.71 (m, 4H), 1.40 (s, 3H), 1.27 (d, J = 7.1 Hz, 3H), 1.18 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ 173.5, 171.0, 167.1, 161.7, 160.9, 146.2, 146.0, 129.1, 128.8, 90.1, 78.3, 75.1, 72.2, 71.8, 66.0, 52.6, 49.1, 43.8, 37.4, 31.6, 26.9, 25.3, 21.6, 13.8. HRMS (ESI) calculated for C24H32Cl2N2NaO9S2+ [M + Na]+ 649.0824, found 649.0821.
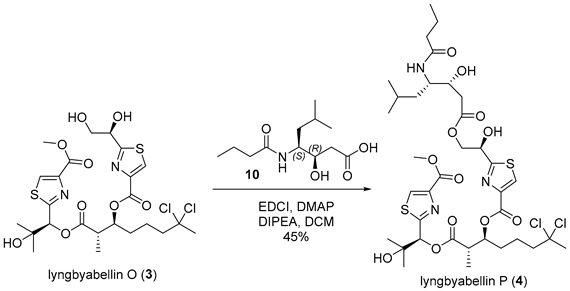
3.2.17. Lyngbyabellin P (4)
![Marinedrugs 23 00340 i017 Marinedrugs 23 00340 i017]()
To a stirred solution of lyngbyabellin O (3) (40 mg, 0.064 mmol, 1.0 equiv.) and acid 10 (47 mg, 0.19 mmol, 3.0 equiv.) in anhydrous dichloromethane (3 mL, 0.021 M) at 0 °C were added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDCI) (36 mg, 0.19 mmol, 3.0 equiv.), N,N-diisopropylethylamine (DIPEA) (66 µL, 0.38 mmol, 6.0 equiv.), and 4-dimethylaminopyridine (DMAP) (4.6 mg, 0.038 mmol, 0.6 equiv.). The reaction mixture was subsequently allowed to warm to ambient temperature and stirred for 6 h. Upon completion, the reaction was concentrated under reduced pressure to afford a crude solid residue. The residue was dissolved in ethyl acetate (30 mL), and the resulting solution was quenched with 4% aqueous citric acid (5 mL). The aqueous phase was separated and extracted with ethyl acetate (3 × 20 mL). The combined organic extracts were washed sequentially with saturated sodium bicarbonate solution (15 mL) and brine (15 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. Purification of the crude product by flash chromatography on silica gel, employing a dichloromethane/methanol mixture (10:1, v/v) as the eluent, afforded lyngbyabellin P (4) as a colorless amorphous solid (25 mg, 45% yield). TLC: Rf = 0.3 (DCM/MeOH = 10/1), UV and PMA stain. = −23.9 (c 1.1, MeOH). 1H-NMR (400 MHz, CDCl3) δ 8.20 (s, 1H), 8.16 (s, 1H), 6.12 (s, 1H), 5.72 (s, 1H), 5.64 (d, J = 8.4 Hz, 1H), 5.43 (dt, J = 7.8, 4.6 Hz, 1H), 5.30 (t, J = 5.6 Hz, 1H), 4.56 (d, J = 5.6 Hz, 2H), 4.18–4.07 (m, 1H), 4.07–4.01 (m, 1H), 3.94 (s, 3H), 3.03 (qd, J = 7.3, 5.0 Hz, 1H), 2.56 (d, J = 5.9 Hz, 2H), 2.32–2.13 (m, 1H), 2.23–2.15 (m, 2H), 2.12 (s, 3H), 1.93–1.74 (m, 4H), 1.74–1.55 (m, 2H), 1.63–1.55 (m, 1H), 1.54–1.42 (m, 1H), 1.37 (s, 3H), 1.38–1.29 (m, 1H), 1.29 (d, J = 7.1 Hz, 3H), 1.19 (s, 3H), 0.94 (t, J = 6.7 Hz, 3H), 0.94 (d, J = 6.3 Hz, 3H), 0.89 (d, J = 6.4 Hz, 3H). 13C-NMR (100 MHz, CDCl3) δ 174.3, 173.1, 171.9, 171.1, 167.4, 161.7, 160.9, 146.4, 146.2, 129.0, 128.6, 90.1, 78.5, 75.0, 72.0, 71.7, 69.9, 67.8, 52.5, 51.6, 49.1, 43.7, 38.8, 38.6, 37.6, 37.4, 31.3, 26.6, 25.4, 24.9, 23.6, 21.6, 21.5, 19.2, 13.7, 13.5. HRMS (ESI) calculated for C36H53Cl2N3NaO12S2+ [M + Na]+ 876.2345, found 876.2339.