Effect of Fucoidan on the Mitochondrial Membrane Potential (ΔΨm) of Leukocytes from Patients with Active COVID-19 and Subjects That Recovered from SARS-CoV-2 Infection
Abstract
:1. Introduction
2. Results
2.1. Phase 1
2.1.1. Characteristics of the Study Population
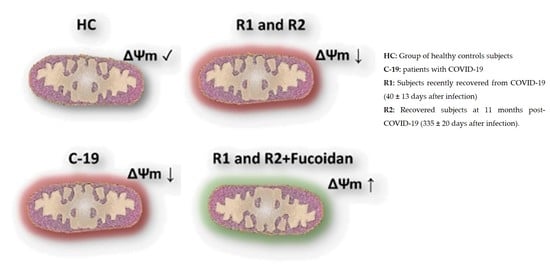
2.1.2. Ex-Vivo Fucoidan Treatment in HPBMCs of HC, C-19, and R1 Subjects
2.2. Phase 2
2.2.1. Characteristics of the Study Population
2.2.2. Ex-Vivo Induction of ΔΨm Loss in HPBMC
2.2.3. Ex-Vivo Treatment with Fucoidan on HPBMCs with CCCP-Induced Loss of ΔΨm from H and R2 Group Subjects
3. Discussion
4. Materials and Methods
4.1. Samples and Data Collection
4.2. Phase 1
4.2.1. Study Design and Participants
4.2.2. HPBMCs Isolation
4.2.3. Ex-Vivo Fucoidan Treatment in HPBMCs from HC, C-19, and R1 Group Subjects
4.2.4. Mitochondrial Membrane Potential Determination (ΔΨm)
4.3. Phase 2
4.3.1. Study Design and Participants
4.3.2. Induction of ΔΨm Loss Ex-Vivo in HPBMC
4.3.3. Ex-Vivo Treatment with Fucoidan in HPBMCs with Loss of ΔΨm Induced by CCCP of H Subjects and R2 Group
4.3.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ortegón-Aznar, I.; Ávila-Mosqueda, S.V. Arribazón de sargazo en la península de Yucatán: ¿Problema local, regional o mundial? Bioagrociencias 2020, 13, 28–37. [Google Scholar]
- Berteau, O.; Mulloy, B. Sulfated fucans, fresh perspectives: Structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 2003, 13, 29R–40R. [Google Scholar] [CrossRef] [Green Version]
- Nava Jiménez, I.A.; Sánchez Hernández, H. El sargazo del mar Caribe mexicano. Ciencia 2020, 71, 58–61. [Google Scholar]
- Rupérez, P.; Ahrazem, O.; Leal, J.A. Potential antioxidant capacity of sulfated polysaccharides from the edible marine brown seaweed Fucus vesiculosus. J. Agric. Food Chem. 2002, 50, 840–845. [Google Scholar] [CrossRef]
- Chen, H.Y.; Huang, T.C.; Lin, L.C.; Shieh, T.M.; Wu, C.H.; Wang, K.L.; Hong, Y.H.; Hsia, S.M. Fucoidan inhibits the proliferation of leiomyoma cells and decreases extracellular matrix-associated protein expression. Cell. Physiol. Biochem. 2018, 45, 1970–1986. [Google Scholar] [CrossRef] [PubMed]
- Vishchuk, O.S.; Sun, H.; Wang, Z.; Ermakova, S.P.; Xiao, J.J.; Lu, T.; Xue, P.P.; Zvyagintseva, T.N.; Xiong, H.; Shao, C.; et al. PDZ-binding kinase/T-LAK cell-originated protein kinase is a target of the fucoidan from brown alga Fucus evanescens in the prevention of EGF-induced neoplastic cell transformation and colon cancer growth. Oncotarget 2016, 7, 18763–18773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, M.D.; Yao, C.J.; Chow, J.M.; Chang, C.L.; Hwang, P.A.; Chuang, S.E.; Whang-Peng, J.; Lai, G.M. Fucoidan elevates MicroRNA-29b to regulate DNMT3B-MTSS1 axis and inhibit EMT in human hepatocellular carcinoma cells. Mar. Drugs 2015, 13, 6099–6116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, P.A.; Hung, Y.L.; Chien, S.Y. Inhibitory activity of Sargassum hemiphyllum sulfated polysaccharide in arachidonic acid-induced animal models of inflammation. J. Food Drug Anal. 2015, 23, 49–56. [Google Scholar] [CrossRef] [Green Version]
- Cho, T.M.; Kim, W.J.; Moon, S.K. AKT signaling is involved in fucoidan-induced inhibition of growth and migration of human bladder cancer cells. Food Chem. Toxicol. 2014, 64, 344–352. [Google Scholar] [CrossRef]
- Cho, M.; Lee, D.J.; Kim, J.K.; You, S. Molecular characterization and immunomodulatory activity of sulfated fucans from Agarum cribrosum. Carbohydr. Polym. 2014, 113, 507–514. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L.; Tinari, N.; Morozevich, G.E.; Berman, A.E.; Bilan, M.I.; et al. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pereira, L.; Critchley, A.T. The COVID 19 novel coronavirus pandemic 2020: Seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times? J. Appl. Phycol. 2020, 32, 1875–1877. [Google Scholar] [CrossRef]
- Sansone, C.; Brunet, C.; Noonan, D.M.; Albini, A. Marine algal antioxidants as potential vectors for controlling viral diseases. Antioxidants 2020, 9, 392. [Google Scholar] [CrossRef]
- Shi, Q.; Wang, A.; Lu, Z.; Qin, C.; Hu, J.; Yin, J. Overview on the antiviral activities and mechanisms of marine polysaccharides from seaweeds. Carbohyd. Res. 2017, 453, 1–9. [Google Scholar] [CrossRef]
- Song, S.; Peng, H.; Wang, Q.; Liu, Z.; Dong, X.; Wen, C.; Ai, C.; Zhang, Y.; Wang, Z.; Zhu, B. Inhibitory activities of marine sulfated polysaccharides against SARS-CoV-2. Food Funct. 2020, 11, 7415–7420. [Google Scholar] [CrossRef] [PubMed]
- Boo, H.J.; Hong, J.Y.; Kim, S.C.; Kang, J.I.; Kim, M.K.; Kim, E.J.; Hyun, J.W.; Koh, S.; Yoo, E.S.; Kwon, J.M.; et al. The anticancer effect of fucoidan in PC-3 prostate cancer cells. Mar. Drugs 2013, 11, 2982–2999. [Google Scholar] [CrossRef] [Green Version]
- Wei, C.M.; Xiao, Q.; Kuang, X.Y.; Zhang, T.; Yang, Z.S.; Wang, L. Fucoidan inhibits proliferation of the SKM-1 acute myeloid leukaemia cell line via the activation of apoptotic pathways and production of reactive oxygen species. Mol. Med. Rep. 2015, 12, 6649–6655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.Q.; Riby, J.E.; Conde, L.; Grizzle, W.E.; Cui, X.Q.; Skibola, C.F. A Fucus vesiculosus extract inhibits estrogen receptor activation and induces cell death in female cancer cell lines. BMC Complement. Altern. Med. 2016, 16, 151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Weelden, G.; Bobiński, M.; Okła, K.; Van Weelden, W.J.; Romano, A.; Pijnenborg, J. Fucoidan structure and activity in relation to anti-cancer mechanisms. Mar Drugs 2019, 17, 32. [Google Scholar] [CrossRef] [Green Version]
- Bae, H.; Lee, J.Y.; Yang, C.; Song, G.; Lim, W. Fucoidan derived from Fucus vesiculosus inhibits the development of human ovarian cancer via the disturbance of calcium homeostasis, endoplasmic reticulum stress, and angiogenesis. Mar. Drugs 2020, 18, 45. [Google Scholar] [CrossRef] [Green Version]
- Díaz-Resendiz, K.J.G.; Toledo-Ibarra, G.A.; Ruiz-Manzano, R.; Girón Pérez, D.A.; Covantes-Rosales, C.E.; Benitez-Trinidad, A.B.; Girón-Pérez, M.I. Ex vivo treatment with fucoidan of mononuclear cells from SARS-CoV-2 infected patients. Int. J. Environ. Health Res. 2021, 31, 1–19. [Google Scholar] [CrossRef]
- Moore, J.B.; June, C.H. Cytokine release syndrome in severe COVID-19. Science 2020, 368, 473–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saleh, J.; Peyssonnaux, C.; Singh, K.K.; Edeas, M. Mitochondria and microbiota dysfunction in COVID-19 pathogenesis. Mitochondrion 2020, 54, 1–7. [Google Scholar] [CrossRef]
- Phua, J.; Weng, L.; Ling, L.; Egi, M.; Lim, C.M.; Divatia, J.V.; Shrestha, B.R.; Arabi, Y.M.; Ng, J.; Gomersall, C.D.; et al. Asian Critical Care Clinical Trials Group. Intensive care management of coronavirus disease 2019 (COVID-19): Challenges and recommendations. Lancet Respir. Med. 2020, 8, 506–517. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Kernan, K.F.; Carcillo, J.A. Hyperferritinemia and inflammation. Int. Immunol. 2017, 29, 401–409. [Google Scholar] [CrossRef]
- Alarcón-Rodríguez, J.; Fernández-Velilla, M.; Ureña-Vacas, A.; Martín-Pinacho, J.J.; Rigual-Bobillo, J.A.; Jaureguízar-Oriol, A.; Gorospe-Sarasúa, L. Manejo y seguimiento radiológico del paciente post-COVID-19. Radiología 2021, 63, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Hernando, J.E.C. Seguimiento de los pacientes con secuelas no respiratorias de la COVID-19. FMC Form. Med. Contin. Aten. Primaria 2021, 28, 81–89. [Google Scholar] [CrossRef]
- Wang, F.; Kream, R.M.; Stefano, G.B. Long-term respiratory and neurological sequelae of COVID-19. Med. Sci. Mon. Int. Med. J. Exp. Clin. Res. 2020, 26, e928996-1. [Google Scholar] [CrossRef]
- Buonsenso, D.; Di Giuda, D.; Sigfrid, L.; Pizzuto, D.A.; Di Sante, G.; De Rose, C.; Lazzareschi, I.; Sali, M.; Baldi, F.; Pia, D.; et al. Evidence of lung perfusion defects and ongoing inflammation in an adolescent with post-acute sequelae of SARS-CoV-2 infection. Lancet Child Adolesc. Health 2021, 5, 677–680. [Google Scholar] [CrossRef]
- Raman, B.; Cassar, M.P.; Tunnicliffe, E.M.; Filippini, N.; Griffanti, L.; Alfaro-Almagro, F.; Okell, T.; Sheerin, F.; Xie, C.; Mahmod, M.; et al. Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine 2021, 31, 100683. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Sinclair, J.E.; Sadeghirad, H.; Fraser, J.F.; Short, K.R.; Kulasinghe, A. Cardiovascular disease in SARS-CoV-2 infection. Clin. Transl. Immunol. 2021, 10, e1343. [Google Scholar] [CrossRef]
- Ciolac, D.; Racila, R.; Duarte, C.; Vasilieva, M.; Manea, D.; Gorincioi, N.; Condrea, A.; Crivorucica, I.; Zota, E.; Efremova, D.; et al. Clinical and radiological deterioration in a case of Creutzfeldt–Jakob disease following SARS-CoV-2 infection: Hints to Accelerated Age-Dependent Neurodegeneration. Biomedicines 2021, 9, 1730. [Google Scholar] [CrossRef]
- Idrees, D.; Kumar, V. SARS-CoV-2 spike protein interactions with amyloidogenic proteins: Potential clues to neurodegeneration. Biochem. Biophys. Res. Commun. 2021, 554, 94–98. [Google Scholar] [CrossRef]
- Singh, K.K.; Chaubey, G.; Chen, J.Y.; Suravajhala, P. Decoding SARS-CoV-2 hijacking of host mitochondria in COVID-19 pathogenesis. Am. J. Physiol. Cell Physiol. 2020, 319, C258–C267. [Google Scholar] [CrossRef] [PubMed]
- Melser, S.; Lavie, J.; Bénard, G. Mitochondrial degradation and energy metabolism. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2015, 1853, 2812–2821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blake, R.; Trounce, I.A. Mitochondrial dysfunction and complications associated with diabetes. Biochim. Biophys. Acta Gen. Subj. 2014, 1840, 1404–1412. [Google Scholar] [CrossRef]
- Mohlke, K.L.; Jackson, A.U.; Scott, L.J.; Peck, E.C.; Suh, Y.D.; Chines, P.S.; Watanabe, R.M.; Buchanan, T.A.; Conneely, K.N.; Erdos, M.R.; et al. Mitochondrial polymorphisms and susceptibility to type 2 diabetes related traits in Finns. Hum. Genet. 2005, 118, 245–254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, T.T.; Yang, F.Y.; Liu, C.; Cao, X.; Lu, J.; Zhang, X.L.; Yuan, M.X.; Chen, C.; Yang, J.K. Angiotensin-converting enzyme 2 regulates mitochondrial function in pancreatic β-cells. Biochem. Biophys. Res. Commun. 2018, 495, 860–866. [Google Scholar] [CrossRef]
- Valero, T. Editorial (thematic issue: Mitochondrial biogenesis: Pharmacological approaches). Curr. Pharm. Des. 2014, 20, 5507–5509. [Google Scholar] [CrossRef]
- Nicoletti, V.; Palermo, G.; Del Prete, E.; Mancuso, M.; Ceravolo, R. Understanding the Multiple role of mitochondria in Parkinson’s disease and related disorders: Lesson from genetics and protein–interaction network. Front. Cell Dev. Biol. 2021, 9, 493. [Google Scholar] [CrossRef]
- Castora, F.J. Mitochondrial Function and Abnormalities Implicated in the Pathogenesis of ASD. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 92, 83–108. [Google Scholar] [CrossRef] [PubMed]
- Ajaz, S.; McPhail, M.J.; Singh, K.K.; Mujib, S.; Trovato, F.M.; Napoli, S.; Agarwal, K. Mitochondrial metabolic manipulation by SARS-CoV-2 in peripheral blood mononuclear cells of patients with COVID-19. Am. J. Physiol. Cell Physiol. 2021, 320, C57–C65. [Google Scholar] [CrossRef] [PubMed]
- Gibellini, L.; De Biasi, S.; Paolini, A.; Borella, R.; Boraldi, F.; Mattioli, M.; Tartaro, D.; Fidanza, L.; Caro-Maldonado, A.; Meschiari, M.; et al. Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia. EMBO Mol. Med. 2020, 12, e13001. [Google Scholar] [CrossRef]
- Baqi, H.R.; Farag, H.A.M.; El Bilbeisi, A.H.H.; Askandar, R.H.; El Afifi, A.M. Oxidative stress and its association with COVID-19: A narrative review. KJAR 2020, 5, 97–105. [Google Scholar] [CrossRef]
- Chernyak, B.V.; Popova, E.N.; Prikhodko, A.S.; Grebenchikov, O.A.; Zinovkina, L.A.; Zinovkin, R.A. COVID-19 and oxidative stress. Biochemistry 2020, 85, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- De las Heras, N.; Martín Giménez, V.M.; Ferder, L.; Manucha, W.; Lahera, V. Implications of oxidative stress and potential role of mitochondrial dysfunction in COVID-19: Therapeutic effects of vitamin D. Antioxidants 2020, 9, 897. [Google Scholar] [CrossRef] [PubMed]
- Cortese-Krott, M.M.; Koning, A.; Kuhnle, G.G.C.; Nagy, P.; Bianco, C.L.; Pasch, A.; Wink, D.A.; Fukuto, J.M.; Jackson, A.A.; Van Goor, H.; et al. The reactive species interactome: Evolutionary emergence, biological significance, and opportunities for redox metabolomics and personalized medicine. Antioxid. Redox Signal 2017, 27, 684–712. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, S.F.; Agili, F.A. Antiviral sulphated polysaccharide from brown algae Padina pavonia characterization and structure elucidation. Int. J. Chemtech Res. 2013, 5, 1469–1476. [Google Scholar]
- Niedzwiecki, A.; Rath, M. Scientific basis of micronutrient applications as an effective, safe and affordable global public health strategy to help to control the coronavirus pandemic. J. Cell. Med. Nat. Health 2021, 15, 1–5. [Google Scholar]
- Sen, I.K.; Chakraborty, I.; Mandal, A.K.; Bhanja, S.K.; Patra, S.; Maity, P. A review on antiviral and immunomodulatory polysaccharides from Indian medicinal plants, which may be beneficial to COVID-19 infected patients. Int. J. Biol. 2021, 181, 462–470. [Google Scholar] [CrossRef]
- Thuy, T.T.T.; Ly, B.M.; Van, T.T.T.; Quang, N.V.; Tu, H.C.; Zheng, Y.; Seguin-Devaux, C.; Mi, B.; Ai, U. Anti-HIV activity of fucoidans from three brown seaweed species. Carbohydr. Polym. 2015, 115, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Yim, J.H.; Kim, S.Y.; Kim, H.S.; Lee, W.G.; Kim, S.J.; Kang, P.S.; Lee, C.K. In vitro inhibition of influenza, a virus infection by marine microalga-derived sulfated polysaccharide p-KG03. Antivir. Res. 2012, 93, 253–259. [Google Scholar] [CrossRef]
- Elizondo-Gonzalez, R.; Cruz-Suarez, L.E.; Ricque-Marie, D.; Mendoza-Gamboa, E.; Rodriguez-Padilla, C.; Trejo-Avila, L.M. In vitro characterization of the antiviral activity of fucoidan from Cladosiphon okamuranus against Newcastle disease virus. Virol. J. 2012, 9, 307. [Google Scholar] [CrossRef] [Green Version]
- Trejo-Avila, L.M.; Morales-Martínez, M.E.; Ricque-Marie, D.; Cruz-Suarez, L.D.; Zapata-Benavides, P.; Morán-Santibañez, K.; Rodríguez-Padilla, C. In vitro anti-canine distemper virus activity of fucoidan extracted from the brown alga Cladosiphon okamuranus. Virus Dis. 2014, 25, 474–480. [Google Scholar] [CrossRef] [Green Version]
- Moran-Santibanez, K.; Cruz-Suarez, L.E.; Ricque-Marie, D.; Robledo, D.; Freile-Pelegrin, Y.; Pena-Hernandez, M.A.; Rodriguez-Padilla, C.; Trejo-Avila, L.M. Synergistic effects of sulfated polysaccharides from Mexican seaweeds against measles virus. BioMed Res. Int. 2016, 2016, 8502123. [Google Scholar] [CrossRef] [Green Version]
- Colona-Vallejos, E.H.; Alzamora-Gonzales, L.; Chávez Pérez, J.; Apumayta Suárez, E.V.; Chang Avila, I. Incremento de la viabilidad, producción de especies reactivas de oxígeno, IL-1 y TNF-α en células mononucleares de sangre periférica humana trata-das con fucoidan de Lessonia trabeculata. Rev. Peru. Biol. 2019, 26, 291–300. [Google Scholar] [CrossRef]
- Fitton, J.H.; Stringer, D.N.; Karpiniec, S.S. Therapies from fucoidan: An update. Mar. Drugs 2015, 13, 5920–5946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitton, J.H. Therapies from fucoidan; multifunctional marine polymers. Mar. Drugs 2011, 9, 1731–1760. [Google Scholar] [CrossRef]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.; Song, Y.; Wang, Q.; Hu, Y.; He, Y.; Ren, D.; Wu, L.; Liu, S.; Cong, H.; Zhou, H. In vitro and in vivo immunomodulatory effects of fucoidan compound agents. Int. J. Biol. Macromol. 2019, 127, 48–56. [Google Scholar] [CrossRef]
- Apostolova, E.; Lukova, P.; Baldzhieva, A.; Katsarov, P.; Nikolova, M.; Iliev, I.; Peychev, L.; Trica, B.; Oancea, F.; Delattre, C.; et al. Immunomodulatory and anti-inflammatory effects of fucoidan: A review. Polymers 2020, 12, 2338. [Google Scholar] [CrossRef]
- Borazjani, N.J.; Tabarsa, M.; You, S.; Rezaei, M. Improved immunomodulatory and antioxidant properties of unrefined fucoidans from Sargassum angustifolium by hydrolysis. J. Food Sci. Technol. 2017, 54, 4016–4025. [Google Scholar] [CrossRef]
- Marudhupandi, T.; Kumar, T.T.; Senthil, S.L.; Devi, K.N. In vitro antioxidant properties of fucoidan fractions from Sargassum tenerrimum. Pak. J. Biol. Sci. 2014, 17, 402–407. [Google Scholar] [CrossRef]
- Li, Q.; Jiang, S.; Shi, W.; Qi, X.; Song, W.; Mou, J.; Yang, J. Structure characterization, antioxidant and immunoregulatory properties of a novel fucoidan from the sea cucumber Stichopus chloronotus. Carbohydr. Polym. 2020, 231, 115767. [Google Scholar] [CrossRef] [PubMed]
- Ashayerizadeh, O.; Dastar, B.; Pourashouri, P. Study of antioxidant and antibacterial activities of depolymerized fucoidans extracted from Sargassum tenerrimum. Int. J. Biol. Macromol. 2020, 151, 1259–1266. [Google Scholar] [CrossRef]
- Fitton, J.H.; Park, A.Y.; Karpiniec, S.S.; Stringer, D.N. Fucoidan and lung function: Value in viral infection. Mar. Drugs 2021, 19, 4. [Google Scholar] [CrossRef]
- Han, Y.S.; Lee, J.H.; Lee, S.H. Fucoidan suppresses mitochondrial dysfunction and cell death against 1-methyl-4-phenylpyridinum-induced neuronal cytotoxicity via regulation of PGC-1α expression. Mar. Drugs 2019, 17, 518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, G.; Myers, R.; Li, Y.; Chen, Y.; Shen, X.; Fenyk-Melody, J.; Wu, M.; Ventre, J.; Doebber, T.; Fujii, N.; et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Investig. 2001, 108, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Zong, H.; Ren, J.M.; Young, L.H.; Pypaert, M.; Mu, J.; Birnbaum, M.J.; Shulman, G.I. AMP kinase is required for mitochondrial biogenesis in skeletal muscle in response to chronic energy deprivation. Proc. Natl. Acad. Sci. USA 2002, 99, 15983–15987. [Google Scholar] [CrossRef] [Green Version]
- Reznick, R.M.; Shulman, G.I. The role of AMP-activated protein kinase in mitochondrial biogenesis. J. Physiol. 2006, 574, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Bachstetter, A.D.; Van Eldik, L.J. The p38 map kinase family as regulators of proinflammatory cytokine production in degenerative diseases of the CNS. Aging Dis. 2010, 1, 199–211. [Google Scholar] [PubMed]
- Kwak, J.Y. Fucoidan as a marine anticancer agent in preclinical development. Mar. Drugs 2014, 12, 851–870. [Google Scholar] [CrossRef]
- Ortiz-Lazareno, P.C.; Bravo-Cuellar, A.; Lerma-Díaz, J.M.; Jave-Suárez, L.F.; Aguilar-Lemarroy, A.; Domínguez-Rodríguez, J.R.; González-Ramella, O.; De Célis, R.; Gómez-Lomelí, P.; Hernández-Flores, G. Sensitization of U937 leukemia cells to doxorubicin by the MG132 proteasome inhibitor induces an increase in apoptosis by suppressing NF-kappa B and mitochondrial membrane potential loss. Cancer Cell Int. 2014, 14, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Subject | n | Female | Male | Age | SAR-CoV-2 Detection (qRT-PCR Result) |
|---|---|---|---|---|---|
| Phase 1 | |||||
| HC | 24 | 16 | 8 | 40 (23–70) | − |
| C-19 | 31 | 18 | 13 | 38 (18–65) | + |
| R1 | 21 | 12 | 9 | 40 (26–64) | − |
| Phase 2 | |||||
| H | 19 | 11 | 8 | 41 (21–69) | − |
| R2 | 19 | 11 | 8 | 41 (24–74) | − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Resendiz, K.J.G.; Covantes-Rosales, C.E.; Benítez-Trinidad, A.B.; Navidad-Murrieta, M.S.; Razura-Carmona, F.F.; Carrillo-Cruz, C.D.; Frias-Delgadillo, E.J.; Pérez-Díaz, D.A.; Díaz-Benavides, M.V.; Zambrano-Soria, M.; et al. Effect of Fucoidan on the Mitochondrial Membrane Potential (ΔΨm) of Leukocytes from Patients with Active COVID-19 and Subjects That Recovered from SARS-CoV-2 Infection. Mar. Drugs 2022, 20, 99. https://doi.org/10.3390/md20020099
Díaz-Resendiz KJG, Covantes-Rosales CE, Benítez-Trinidad AB, Navidad-Murrieta MS, Razura-Carmona FF, Carrillo-Cruz CD, Frias-Delgadillo EJ, Pérez-Díaz DA, Díaz-Benavides MV, Zambrano-Soria M, et al. Effect of Fucoidan on the Mitochondrial Membrane Potential (ΔΨm) of Leukocytes from Patients with Active COVID-19 and Subjects That Recovered from SARS-CoV-2 Infection. Marine Drugs. 2022; 20(2):99. https://doi.org/10.3390/md20020099
Chicago/Turabian StyleDíaz-Resendiz, Karina Janice Guadalupe, Carlos Eduardo Covantes-Rosales, Alma Betsaida Benítez-Trinidad, Migdalia Sarahy Navidad-Murrieta, Francisco Fabian Razura-Carmona, Christian Daniel Carrillo-Cruz, Edwin Jaime Frias-Delgadillo, Daniela Alejandra Pérez-Díaz, Matxil Violeta Díaz-Benavides, Mercedes Zambrano-Soria, and et al. 2022. "Effect of Fucoidan on the Mitochondrial Membrane Potential (ΔΨm) of Leukocytes from Patients with Active COVID-19 and Subjects That Recovered from SARS-CoV-2 Infection" Marine Drugs 20, no. 2: 99. https://doi.org/10.3390/md20020099
APA StyleDíaz-Resendiz, K. J. G., Covantes-Rosales, C. E., Benítez-Trinidad, A. B., Navidad-Murrieta, M. S., Razura-Carmona, F. F., Carrillo-Cruz, C. D., Frias-Delgadillo, E. J., Pérez-Díaz, D. A., Díaz-Benavides, M. V., Zambrano-Soria, M., Ventura-Ramón, G. H., Romero-Castro, A., Alam-Escamilla, D., & Girón-Pérez, M. I. (2022). Effect of Fucoidan on the Mitochondrial Membrane Potential (ΔΨm) of Leukocytes from Patients with Active COVID-19 and Subjects That Recovered from SARS-CoV-2 Infection. Marine Drugs, 20(2), 99. https://doi.org/10.3390/md20020099