Microalgae as Sustainable Bio-Factories of Healthy Lipids: Evaluating Fatty Acid Content and Antioxidant Activity
Abstract
:1. Introduction
2. Results
2.1. Lipid Content in Different Microalgae
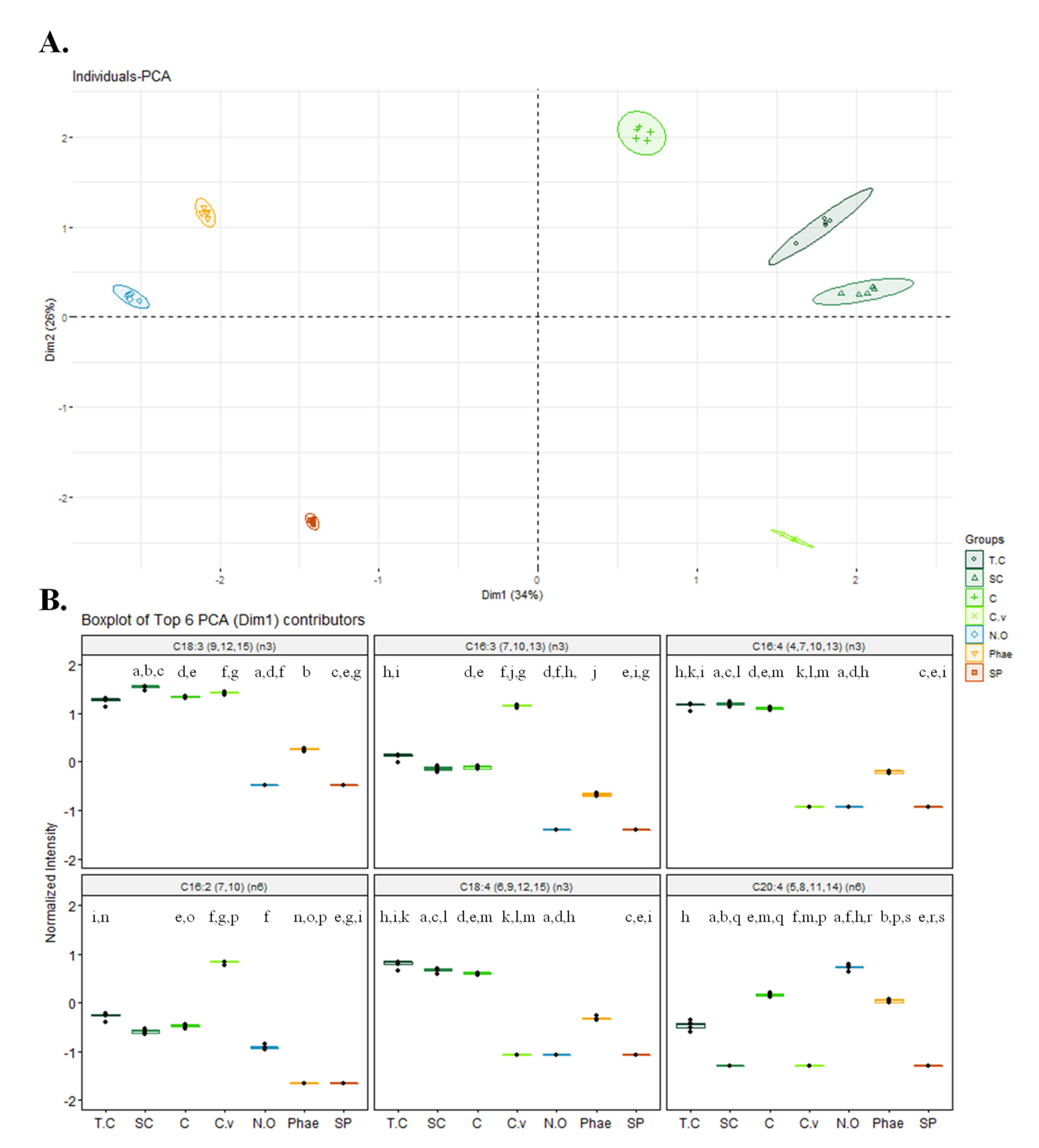
2.2. Fatty Acid Profile of Different Microalgae and Nutritional Indices
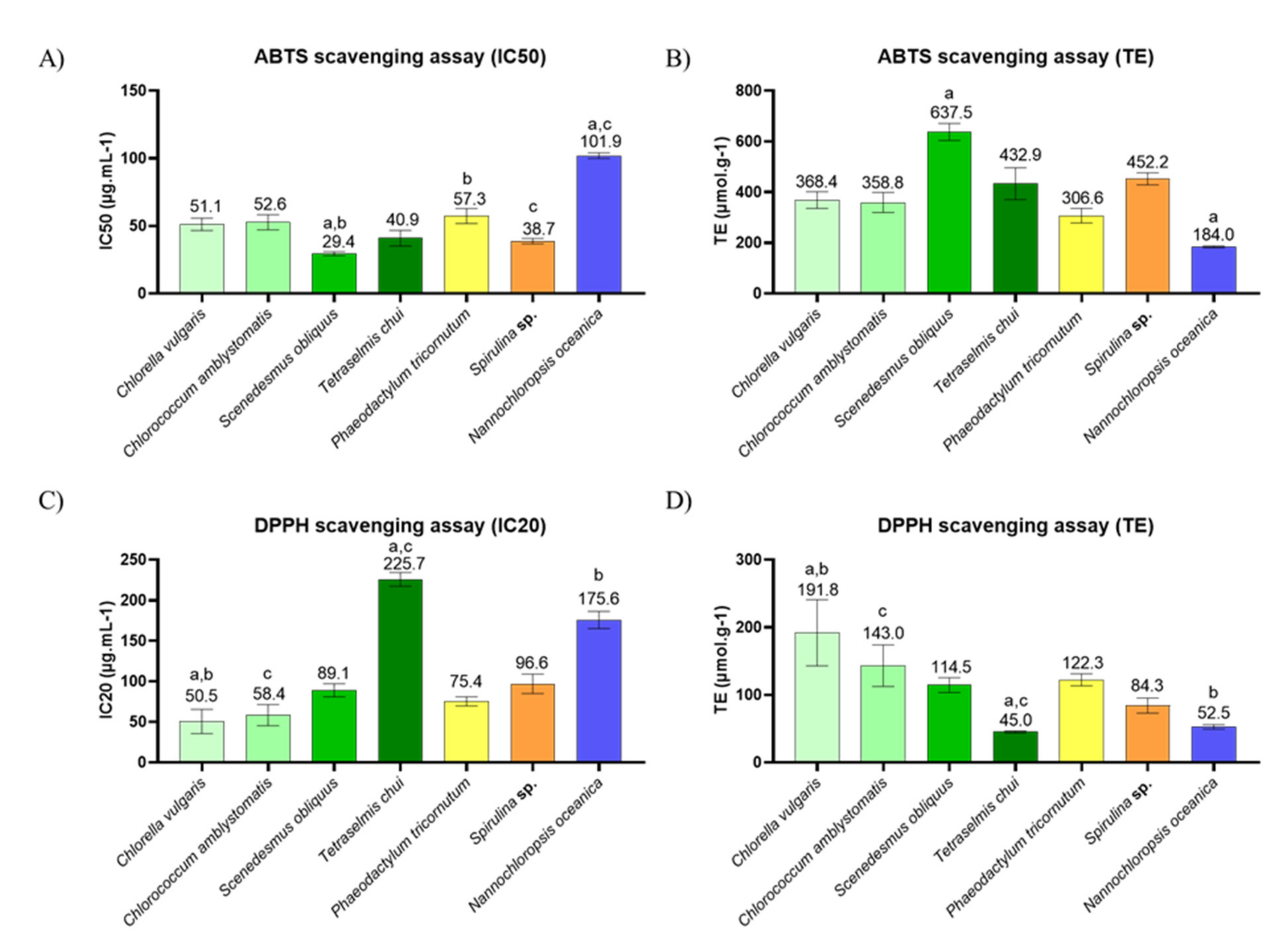
2.3. In Chemico Evaluation of Antioxidant Activity
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Microalgae Material
4.3. Lipid Extraction Procedure
4.4. Analysis of Fatty Acids by Gas Chromatography-Mass Spectrometry (GC-MS)
4.5. Data Analysis
4.6. Statistical Analysis
4.7. DPPH Radical Scavenging Assay
4.8. ABTS Cation Radical Scavenging Assay
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Katiyar, R.; Arora, A. Health promoting functional lipids from microalgae pool: A review. Algal Res. 2020, 46, 101800. [Google Scholar] [CrossRef]
- Maltsev, Y.; Maltseva, K. Fatty acids of microalgae: Diversity and applications. Rev. Environ. Sci. Bio. Technol. 2021, 20, 515–547. [Google Scholar] [CrossRef]
- Adarme-Vega, T.C.; Lim, D.K.Y.; Timmins, M.; Vernen, F.; Felicitas, V.; Schenk, P.M. Microalgal biofactories: A promising approach towards sustainable omega-3 fatty acid production. Microb. Cell Factories 2012, 11, 96. [Google Scholar] [CrossRef] [Green Version]
- Innis, S.M. Omega-3 Fatty Acid Biochemistry: Perspectives from Human Nutrition. Mil. Med. 2014, 179, 82–87. [Google Scholar] [CrossRef] [Green Version]
- Krupanidhi, S.; Sanjeevi, C.B. Omega-3 Fatty Acids for Nutrition and Medicine: Considering Microalgae Oil as a Vegetarian Source of EPA and DHA. Curr. Diabetes Rev. 2007, 3, 198–203. [Google Scholar] [CrossRef]
- Lopes, D.; Melo, T.; Meneses, J.; Abreu, M.H.; Pereira, R.; Domingues, M.R.; Lillebø, A.I.; Calado, R. A New Look for the Red Macroalga Palmaria palmata: A Seafood with Polar Lipids Rich in EPA and with Antioxidant Properties. Mar. Drugs 2019, 17, 533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wysoczański, T.; Sokoła-Wysoczańska, E.; Pękala, J.; Lochyński, S.; Czyż, K.; Bodkowski, R.; Herbinger, G.; Patkowska-Sokoła, B.; Librowski, T. Omega-3 Fatty Acids and their Role in Central Nervous System—A Review. Curr. Med. Chem. 2016, 23, 816–831. [Google Scholar] [CrossRef]
- Innis, S.M. Essential fatty acids in growth and development. Prog. Lipid Res. 1991, 30, 39–103. [Google Scholar] [CrossRef]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef]
- Eckert, G.P.; Lipka, U.; Muller, W.E. Omega-3 fatty acids in neurodegenerative diseases: Focus on mitochondria. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 105–114. [Google Scholar] [CrossRef]
- Thomas, J.; Thomas, C.J.; Radcliffe, J.; Itsiopoulos, C. Omega-3 Fatty Acids in Early Prevention of Inflammatory Neurodegenerative Disease: A Focus on Alzheimer’s Disease. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Li, D. Omega-3 polyunsaturated fatty acids and non-communicable diseases: Meta-analysis based systematic review. Asia Pac. J. Clin. Nutr. 2015, 24, 10–15. [Google Scholar] [PubMed]
- Phillips, C.M.; Chen, L.-W.; Heude, B.; Bernard, J.Y.; Harvey, N.C.; Duijts, L.; Mensink-Bout, S.M.; Polanska, K.; Mancano, G.; Suderman, M.; et al. Dietary Inflammatory Index and Non-Communicable Disease Risk: A Narrative Review. Nutrients 2019, 11, 1873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budreviciute, A.; Damiati, S.; Sabir, D.K.; Onder, K.; Schuller-Goetzburg, P.; Plakys, G.; Katileviciute, A.; Khoja, S.; Kodzius, R. Management and Prevention Strategies for Non-communicable Diseases (NCDs) and Their Risk Factors. Front. Public Health 2020, 8, 1–11. [Google Scholar] [CrossRef]
- Melo, H.M.; Santos, L.; Ferreira, S.T. Diet-Derived Fatty Acids, Brain Inflammation, and Mental Health. Front. Neurosci. 2019, 13, 265. [Google Scholar] [CrossRef] [Green Version]
- GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specifc mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar]
- FAO; WHO. Sustainable and Healhy Diets: Guiding Principles; FAO: Rome, Italy, 2019. [Google Scholar]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Vuppaladadiyam, A.K.; Prinsen, P.; Raheem, A.; Luque, R.; Zhao, M. Sustainability Analysis of Microalgae Production Systems: A Review on Resource with Unexploited High-Value Reserves. Environ. Sci. Technol. 2018, 52, 14031–14049. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 1–21. [Google Scholar] [CrossRef]
- Charles, C.N.; Msagati, T.; Swai, H.; Chacha, M. Microalgae: An alternative natural source of bioavailable omega-3 DHA for promotion of mental health in East Africa. Sci. Afr. 2019, 6, e00187. [Google Scholar] [CrossRef]
- Caporgno, M.P.; Mathys, A. Trends in Microalgae Incorporation into Innovative Food Products with Potential Health Benefits. Front. Nutr. 2018, 5, 58. [Google Scholar] [CrossRef] [PubMed]
- Pina-Pérez, M.; Brück, W.; Brück, T.; Beyrer, M. Microalgae as healthy ingredients for functional foods. In The Role of Alternative and Innovative Food Ingredients and Products in Consumer Wellness; Elsevier: Amsterdam, The Netherlands, 2019; pp. 103–137. [Google Scholar]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for High-Value Products towards Human Health and Nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef]
- Remize, M.; Brunel, Y.; Silva, J.L.; Berthon, J.-Y.; Filaire, E. Microalgae n-3 PUFAs Production and Use in Food and Feed Industries. Mar. Drugs 2021, 19, 113. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.; Bannenberg, G.; Rice, H.B.; Schutt, E.; Mackay, D. Oxidation in EPA- and DHA-rich oils: An overview. Lipid Technol. 2016, 28, 55–59. [Google Scholar] [CrossRef]
- Jerez-Martel, I.; García-Poza, S.; Rodríguez-Martel, G.; Rico, M.; Afonso-Olivares, C.; Pinchetti, J.L.G. Phenolic Profile and Antioxidant Activity of Crude Extracts from Microalgae and Cyanobacteria Strains. J. Food Qual. 2017, 2017, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Li, H.B.; Cheng, K.W.; Wong, C.C.; Fan, K.W.; Chen, F.; Jiang, Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Conde, T.A.; Couto, D.; Melo, T.; Costa, M.; Silva, J.; Domingues, M.R.; Domingues, P. Polar lipidomic profile shows Chlorococcum amblystomatis as a promising source of value-added lipids. Sci. Rep. 2021, 11, 1–23. [Google Scholar] [CrossRef]
- Couto, D.; Melo, T.; Conde, T.A.; Costa, M.; Silva, J.; Domingues, M.R.M.; Domingues, P. Chemoplasticity of the polar lipid profile of the microalgae Chlorella vulgaris grown under heterotrophic and autotrophic conditions. Algal Res. 2020, 53, 102128. [Google Scholar] [CrossRef]
- da Costa, E.; Amaro, H.M.; Melo, T.; Guedes, A.C.; Domingues, M.R. Screening for polar lipids, antioxidant, and anti-inflammatory activities of Gloeothece sp. lipid extracts pursuing new phytochemicals from cyanobacteria. J. Appl. Phycol. 2020, 32, 3015–3030. [Google Scholar] [CrossRef]
- Jeong, E.-Y.; Seo, P.J.; Woo, J.C.; Park, C.-M. Fatty acid profiles and their distribution patterns in microalgae: A comprehensive analysis of more than 2000 strains from the SAG culture collection. BMC Plant Biol. 2011, 11, 1–16. [Google Scholar]
- Peltomaa, E.; Johnson, M.D.; Taipale, S.J. Marine Cryptophytes Are Great Sources of EPA and DHA. Mar. Drugs 2017, 16, 3. [Google Scholar] [CrossRef] [Green Version]
- Peltomaa, E.; Hällfors, H.; Taipale, S.J. Comparison of Diatoms and Dinoflagellates from Different Habitats as Sources of PUFAs. Mar. Drugs 2019, 17, 233. [Google Scholar] [CrossRef] [Green Version]
- Jónasdóttir, S.H. Fatty Acid Profiles and Production in Marine Phytoplankton. Mar. Drugs 2019, 17, 151. [Google Scholar] [CrossRef] [Green Version]
- Taipale, S.; Peltomaa, E.; Salmi, P. Variation in ω-3 and ω-6 Polyunsaturated Fatty Acids Produced by Different Phytoplankton Taxa at Early and Late Growth Phase. Biomolecules 2020, 10, 559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gladyshev, M.I.; Sushchik, N.N. Long-chain Omega-3 Polyunsaturated Fatty Acids in Natural Ecosystems and the Human Diet: Assumptions and Challenges. Biomolecules 2019, 9, 485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamilton, M.L.; Warwick, J.; Terry, A.; Allen, M.J.; Napier, J.A.; Sayanova, O. Towards the Industrial Production of Omega-3 Long Chain Polyunsaturated Fatty Acids from a Genetically Modified Diatom Phaeodactylum tricornutum. PLoS ONE 2015, 10, e0144054. [Google Scholar] [CrossRef] [PubMed]
- El Baky, H.H.A.; El-Baroty, G.S.; Bouaid, A.; Martinez, M.; Aracil, J. Enhancement of lipid accumulation in Scenedesmus obliquus by Optimizing CO2 and Fe3+ levels for biodiesel production. Bioresour. Technol. 2012, 119, 429–432. [Google Scholar] [CrossRef] [PubMed]
- El Baky, H.H.A.; El Baroty, G.S.; Mostafa, E.M. Optimization Growth of Spirulina (Arthrospira) Platensis in Photobioreactor under Varied Nitrogen Concentration for Maximized Biomass, Carotenoids and Lipid Contents. Recent Pat. Food Nutr. Agric. 2018, 11, 40–48. [Google Scholar] [CrossRef]
- Li, Y.; Naghdi, F.G.; Garg, S.; Adarme-Vega, T.C.; Thurecht, K.J.; Ghafor, W.A.; Tannock, S.; Schenk, P.M. A comparative study: The impact of different lipid extraction methods on current microalgal lipid research. Microb. Cell Factories 2014, 13, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Figueiredo, A.; da Costa, E.; Silva, J.; Domingues, M.R.; Domingues, P. The effects of different extraction methods of lipids from Nannochloropsis oceanica on the contents of omega-3 fatty acids. Algal Res. 2019, 41, 101556. [Google Scholar] [CrossRef]
- Wu, Z.; Zhang, Q.; Li, N.; Pu, Y.; Wang, B.; Zhang, T. Comparison of critical methods developed for fatty acid analysis: A review. J. Sep. Sci. 2016, 40, 288–298. [Google Scholar] [CrossRef]
- Araújo, R.; Calderón, F.V.; López, J.S.; Azevedo, I.C.; Bruhn, A.; Fluch, S.; Tasende, M.G.; Ghaderiardakani, F.; Ilmjärv, T.; Laurans, M.; et al. Current Status of the Algae Production Industry in Europe: An Emerging Sector of the Blue Bioeconomy. Front. Mar. Sci. 2021, 7, 1–24. [Google Scholar] [CrossRef]
- Sun, X.M.; Ren, L.J.; Zhao, Q.Y.; Ji, X.J.; Huang, H. Biotechnology for Biofuels Microalgae for the production of lipid and carotenoids: A review with focus on stress regulation and adaptation. Biotechnol. Biofuels 2018, 11, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sajjadi, B.; Chen, W.-Y.; Raman, A.A.A.; Ibrahim, S. Microalgae lipid and biomass for biofuel production: A comprehensive review on lipid enhancement strategies and their effects on fatty acid composition. Renew. Sustain. Energy Rev. 2018, 97, 200–232. [Google Scholar] [CrossRef]
- Banskota, A.H.; Sperker, S.; Stefanova, R.; McGinn, P.J.; O’Leary, S.J.B. Antioxidant properties and lipid composition of selected microalgae. Environ. Boil. Fishes 2018, 31, 309–318. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Phospholipids of Animal and Marine Origin: Structure, Function, and Anti-Inflammatory Properties. Molecules 2017, 22, 1964. [Google Scholar] [CrossRef] [Green Version]
- Tudor, C.; Gherasim, E.C.; Dulf, F.V.; Pintea, A. In vitro bioaccessibility of macular xanthophylls from commercial microalgal powders of Arthrospira platensis and Chlorella pyrenoidosa. Food Sci. Nutr. 2021, 9, 1896–1906. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, J.; Cui, J.; Feng, Y.; Cui, Q. Metabolic profiles of Nannochloropsis oceanica IMET1 under nitrogen-deficiency stress. Bioresour. Technol. 2013, 130, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Sun, B.; She, X.; Zhao, F.; Cao, Y.; Ren, D.; Lu, J. Lipid production and composition of fatty acids in Chlorella vulgaris cultured using different methods: Photoautotrophic, heterotrophic, and pure and mixed conditions. Ann. Microbiol. 2013, 64, 1239–1246. [Google Scholar] [CrossRef]
- de Oliveira, C.Y.B.; Viegas, T.L.; Lopes, R.G.; Cella, H.; Menezes, R.S.; Soares, A.T.; Filho, N.R.A.; Derner, R. A comparison of harvesting and drying methodologies on fatty acids composition of the green microalga Scenedesmus obliquus. Biomass Bioenergy 2020, 132, 105437. [Google Scholar] [CrossRef]
- Matsui, H.; Sasaki, T.; Kobari, T.; Waqalevu, V.; Kikuchi, K.; Ishikawa, M.; Kotani, T. DHA Accumulation in the Polar Lipids of the Euryhaline Copepod Pseudodiaptomus inopinus and Its Transfer to Red Sea Bream Pagrus major Larvae. Front. Mar. Sci. 2021, 8, 1–15. [Google Scholar] [CrossRef]
- Mohammadi, M.; Kazeroni, N.; Baboli, M.J. Fatty acid composition of the marine micro alga Tetraselmis chuii Butcher in response to culture conditions. J. Algal. Biomass Utln. 2015, 6, 49–55. [Google Scholar]
- Olaizola, M.; Grewe, C. Commercial Microalgal Cultivation Systems. In Grand Challenges in Algae Biotechnology; Hallmann, A., Rampelotto, P.H., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 3–34. [Google Scholar] [CrossRef]
- Nguyen, H.T.D.; Ramli, A.; Kee, L.M. A Review on Methods Used in Analysis of Microalgae Lipid Composition. J. Jpn. Inst. Energy 2017, 96, 532–537. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.; Li, Y.; Li, Y.; Ye, D.; Yuan, L.; Sun, Y.; Han, D.; Hu, Q. Solid Matrix-Supported Supercritical CO2 Enhances Extraction of γ-Linolenic Acid from the Cyanobacterium Arthrospira (Spirulina) platensis and Bioactivity Evaluation of the Molecule in Zebrafish. Mar. Drugs 2019, 17, 203. [Google Scholar] [CrossRef] [Green Version]
- de Morais, E.G.; Nunes, I.L.; Druzian, J.I.; de Morais, M.G.; da Rosa, A.P.C.; Costa, J.A.V. Increase in biomass productivity and protein content of Spirulina sp. LEB 18 (Arthrospira) cultivated with crude glycerol. Biomass Convers. Biorefinery 2020, 18, 1–9. [Google Scholar] [CrossRef]
- de Morais, E.G.; Druzian, J.I.; Nunes, I.L.; de Morais, M.G.; Costa, J.A.V. Glycerol increases growth, protein production and alters the fatty acids profile of Spirulina (Arthrospira) sp LEB 18. Process. Biochem. 2019, 76, 40–45. [Google Scholar] [CrossRef]
- Jiang, L.; Sun, J.; Nie, C.; Li, Y.; Jenkins, J.; Pei, H. Filamentous cyanobacteria triples oil production in seawater-based medium supplemented with industrial waste: Monosodium glutamate residue. Biotechnol. Biofuels 2019, 12, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Andrade, B.B.; Cardoso, L.G.; Assis, D.D.J.; Costa, J.A.V.; Druzian, J.I.; Lima, S.T.D.C. Production and characterization of Spirulina sp. LEB 18 cultured in reused Zarrouk’s medium in a raceway-type bioreactor. Bioresour. Technol. 2019, 284, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Roohani, A.M.; Abedian Kenari, A.; Fallahi Kapoorchali, M.; Borani, M.S.; Zoriezahra, S.J.; Smiley, A.H.; Esmaeili, M.; Rombenso, A.N. Effect of spirulina Spirulina platensis as a complementary ingredient to reduce dietary fish meal on the growth performance, whole-body composition, fatty acid and amino acid profiles, and pigmentation of Caspian brown trout (Salmo trutta caspius) juvenil. Aquac. Nutr. 2019, 25, 633–645. [Google Scholar] [CrossRef]
- Vidyashankar, S.; Sireesha, E.; Chauhan, V.S.; Sarada, R. Evaluation of microalgae as vegetarian source of dietary polyunsaturated fatty acids under autotrophic growth conditions. J. Food Sci. Technol. 2015, 52, 7070–7080. [Google Scholar] [CrossRef]
- Perez-Velazquez, M.; Gatlin, D.M.; González-Félix, M.L.; García-Ortega, A.; de Cruz, C.R.; Juárez-Gómez, M.L.; Chen, K. Effect of fishmeal and fish oil replacement by algal meals on biological performance and fatty acid profile of hybrid striped bass (Morone crhysops ♀ × M. saxatilis ♂). Aquaculture 2019, 507, 83–90. [Google Scholar] [CrossRef]
- Matos, Â.P.; Feller, R.; Moecke, E.H.S.; De Oliveira, J.V.; Junior, A.F.; Derner, R.; Sant’Anna, E.S. Chemical Characterization of Six Microalgae with Potential Utility for Food Application. J. Am. Oil Chem. Soc. 2016, 93, 963–972. [Google Scholar] [CrossRef]
- Darwish, R.; Gedi, M.; Eakpetch, P.; Assaye, H.; Zaky, A.; Gray, D. Chlamydomonas reinhardtii Is a Potential Food Supplement with the Capacity to Outperform Chlorella and Spirulina. Appl. Sci. 2020, 10, 6736. [Google Scholar] [CrossRef]
- Oliveira, C.Y.B.; Oliveira, C.D.L.; Prasad, R.; Ong, H.C.; Araujo, E.S.; Shabnam, N.; Gálvez, A.O. A multidisciplinary review of Tetradesmus obliquus: A microalga suitable for large-scale biomass production and emerging environmental applications. Rev. Aquac. 2021, 13, 1594–1618. [Google Scholar] [CrossRef]
- He, M.; Yan, Y.; Pei, F.; Wu, M.; Gebreluel, T.; Zou, S.; Wang, C. Improvement on lipid production by Scenedesmus obliquus triggered by low dose exposure to nanoparticles. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Hassan, M.E.; Mohafrash, S.M.M.; Fallatah, S.A.; El-Sayed, A.E.K.B.; Mossa, A.T.H. Eco-friendly larvicide of Amphora coffeaeformis and Scenedesmus obliquus microalgae extracts against Culex pipiens. J. Appl. Phycol. 2021, 1–11. [Google Scholar]
- Darki, B.Z.; Seyfabadi, J.; Fayazi, S. Effect of nutrients on total lipid content and fatty acids profile of Scenedesmus obliquus. Braz. Arch. Biol. Technol. 2017, 60, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Salama, E.-S.; Kim, H.-C.; Abou-Shanab, R.; Ji, M.-K.; Oh, Y.-K.; Kim, S.-H.; Jeon, B.-H. Biomass, lipid content, and fatty acid composition of freshwater Chlamydomonas mexicana and Scenedesmus obliquus grown under salt stress. Bioprocess Biosyst. Eng. 2013, 36, 827–833. [Google Scholar] [CrossRef]
- Chu, F.-F.; Chu, P.-N.; Shen, X.-F.; Lam, P.K.; Zeng, R.J. Effect of phosphorus on biodiesel production from Scenedesmus obliquus under nitrogen-deficiency stress. Bioresour. Technol. 2014, 152, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.-F.; Hu, H.; Ma, L.-L.; Lam, P.K.S.; Yan, S.-K.; Zhou, S.-B.; Zeng, R.J. FAMEs production from Scenedesmus obliquus in autotrophic, heterotrophic and mixotrophic cultures under different nitrogen conditions. Environ. Sci. Water Res. Technol. 2018, 4, 461–468. [Google Scholar] [CrossRef]
- An, M.; Gao, L.; Zhao, W.; Chen, W.; Li, M. Effects of Nitrogen Forms and Supply Mode on Lipid Production of Microalga Scenedesmus obliquus. Energies 2020, 13, 697. [Google Scholar] [CrossRef] [Green Version]
- Liang, C.; Wang, L.; Zhang, Y.; Zhang, J.; Zhang, X.; Ye, N. The effects of elevated CO2 concentrations on changes in fatty acids and amino acids of three species of microalgae. Phycologia 2020, 59, 208–217. [Google Scholar] [CrossRef]
- Guerra, I.; Pereira, H.; Costa, M.; Silva, J.; Santos, T.; Varela, J.; Mateus, M.; Silva, J. Operation Regimes: A Comparison Based on Nannochloropsis oceanica Biomass and Lipid Productivity. Energies 2021, 14, 1542. [Google Scholar] [CrossRef]
- Osorio, H.; Jara, C.; Fuenzalida, K.; Rey-Jurado, E.; Vásquez, M. High-efficiency nuclear transformation of the microalgae Nannochloropsis oceanica using Tn5 Transposome for the generation of altered lipid accumulation phenotypes. Biotechnol. Biofuels 2019, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.-J. Omega-3 fatty acid obtained from Nannochloropsis oceanica cultures grown under low urea protect against Abeta-induced neural damage. J. Food Sci. Technol. 2015, 52, 2982–2989. [Google Scholar] [CrossRef] [Green Version]
- Bongiovani, N.; Popovich, C.A.; Martínez, A.M.; Constenla, D.; Leonardi, P.I. Biorefinery Approach from Nannochloropsis oceanica CCALA 978: Neutral Lipid and Carotenoid Co-Production Under Nitrate or Phosphate Deprivation. Bioenergy Res. 2019, 13, 518–529. [Google Scholar] [CrossRef]
- Du, Z.-Y.; Alvaro, J.; Hyden, B.; Zienkiewicz, K.; Benning, N.; Zienkiewicz, A.; Bonito, G.; Benning, C. Enhancing oil production and harvest by combining the marine alga Nannochloropsis oceanica and the oleaginous fungus Mortierella elongata. Biotechnol. Biofuels 2018, 11, 174. [Google Scholar] [CrossRef]
- Touliabah, H.; Almutairi, A. Effect of Phytohormones Supplementation under Nitrogen Depletion on Biomass and Lipid Production of Nannochloropsis oceanica for Integrated Application in Nutrition and Biodiesel. Sustainability 2021, 13, 592. [Google Scholar] [CrossRef]
- Pudney, A.; Gandini, C.; Economou, C.K.; Smith, R.; Goddard, P.; Napier, J.A.; Spicer, A.; Sayanova, O. Multifunctionalizing the marine diatom Phaeodactylum tricornutum for sustainable co-production of omega-3 long chain polyunsaturated fatty acids and recombinant phytase. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.L.; Haslam, R.P.; Napier, J.A.; Sayanova, O. Metabolic engineering of Phaeodactylum tricornutum for the enhanced accumulation of omega-3 long chain polyunsaturated fatty acids. Metab. Eng. 2014, 22, 3–9. [Google Scholar] [CrossRef]
- Shen, P.-L.; Wang, H.-T.; Pan, Y.-F.; Meng, Y.-Y.; Wu, P.-C.; Xue, S. Identification of Characteristic Fatty Acids to Quantify Triacylglycerols in Microalgae. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.-H.; Du, L.; Hosokawa, M.; Miyashita, K.; Kokubun, Y.; Arai, H.; Taroda, H. Fatty Acid and Lipid Class Composition of the Microalga Phaeodactylum tricornutum. J. Oleo Sci. 2017, 66, 363–368. [Google Scholar] [CrossRef] [Green Version]
- Siron, R.; Giusti, G.; Berland, B. Changes in the fatty acid composition of Phaeodactylum tricornutum and Dunaliella tertiolecta during growth and under phosphorus deficiency. Mar. Ecol. Prog. Ser. 1989, 55, 95–100. [Google Scholar] [CrossRef]
- Steinrücken, P.; Prestegard, S.K.; de Vree, J.H.; Storesund, J.E.; Pree, B.; Mjøs, S.A.; Erga, S.R. Comparing EPA production and fatty acid profiles of three Phaeodactylum tricornutum strains under western Norwegian climate conditions. Algal Res. 2018, 30, 11–22. [Google Scholar] [CrossRef]
- Domergue, F.; Spiekermann, P.; Lerchl, J.; Beckmann, C.; Kilian, O.; Kroth, P.G.; Boland, W.; Zähringer, U.; Heinz, E. New Insight into Phaeodactylum tricornutum Fatty Acid Metabolism. Cloning and Functional Characterization of Plastidial and Microsomal Δ12-Fatty Acid Desaturases. Plant Physiol. 2003, 131, 1648–1660. [Google Scholar] [CrossRef] [Green Version]
- Cui, Y.; Thomas-Hall, S.R.; Chua, E.T.; Schenk, P.M. Development of High-Level Omega-3 Eicosapentaenoic Acid (EPA) Production from Phaeodactylum tricornutum. J. Phycol. 2021, 57, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The Importance of the Omega-6/Omega-3 Fatty Acid Ratio in Cardiovascular Disease and Other Chronic Diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Šimat, V.; Bogdanović, T.; Poljak, V.; Petričević, S. Changes in fatty acid composition, atherogenic and thrombogenic health lipid indices and lipid stability of bogue (Boops boops Linnaeus, 1758) during storage on ice: Effect of fish farming activities. J. Food Compos. Anal. 2015, 40, 120–125. [Google Scholar] [CrossRef]
- Marques, B.; Lillebø, A.I.; Domingues, M.D.R.M.; Saraiva, J.A.; Calado, R. Effect of High-Pressure Processing (HPP) on the Fatty Acid Profile of Different Sized Ragworms (Hediste diversicolor) Cultured in an Integrated Multi-Trophic Aquaculture (IMTA) System. Molecules 2019, 24, 4503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopes, D.; Melo, T.; Rey, F.; Meneses, J.; Monteiro, F.L.; Helguero, L.A.; Abreu, M.H.; Lillebø, A.I.; Calado, R.; Domingues, M.R. Valuing Bioactive Lipids from Green, Red and Brown Macroalgae from Aquaculture, to Foster Functionality and Biotechnological Applications. Molecules 2020, 25, 3883. [Google Scholar] [CrossRef] [PubMed]
- Łuczyńska, J.; Paszczyk, B.; Nowosad, J.; Łuczyński, M.J. Mercury, Fatty Acids Content and Lipid Quality Indexes in Muscles of Freshwater and Marine Fish on the Polish Market. Risk Assessment of Fish Consumption. Int. J. Environ. Res. Public Health 2017, 14, 1120. [Google Scholar] [CrossRef] [PubMed]
- Magdugo, R.P.; Terme, N.; Lang, M.; Pliego-Cortés, H.; Marty, C.; Hurtado, A.Q.; Bedoux, G.; Bourgougnon, N. An Analysis of the Nutritional and Health Values of Caulerpa racemosa (Forsskål) and Ulva fasciata (Delile)—Two Chlorophyta Collected from the Philippines. Molecules 2020, 25, 2901. [Google Scholar] [CrossRef] [PubMed]
- Rueda, F.M.; Hernández, M.D.; Egea, M.A.; Aguado, F.; García, B.; Martínez, F.J. Differences in tissue fatty acid composition between reared and wild sharpsnout sea bream, Diplodus puntazzo (Cetti, 1777). Br. J. Nutr. 2001, 86, 617–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valfré, F.; Caprino, F.; Turchini, G. The Health Benefit of Seafood. Vet. Res. Commun. 2003, 27, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Puchau, B.; Ochoa, M.C.; Zulet, M. Ángeles; Marti, A.; Martínez, J.A.; Members, G. Dietary total antioxidant capacity and obesity in children and adolescents. Int. J. Food Sci. Nutr. 2010, 61, 713–721. [Google Scholar] [CrossRef] [Green Version]
- Athukorala, Y.; Lee, K.-W.; Park, E.-J.; Heo, M.-S.; Yeo, I.-K.; Lee, Y.-D.; Jeon, Y.-J. Reduction of lipid peroxidation and H2O2-mediated DNA damage by a red alga (Grateloupia filicina) methanolic extract. J. Sci. Food Agric. 2005, 85, 2341–2348. [Google Scholar] [CrossRef]
- Cox, S.; Abu-Ghannam, N. Incorporation of himanthalia elongata seaweed to enhance the phytochemical content of breadsticks using response surface methodology (RSM). Int. Food Res. J. 2013, 20, 1537–1545. [Google Scholar]
- Levasseur, W.; Perré, P.; Pozzobon, V. A review of high value-added molecules production by microalgae in light of the classification. Biotechnol. Adv. 2020, 41, 107545. [Google Scholar] [CrossRef]
- Haoujar, I.; Cacciola, F.; Abrini, J.; Mangraviti, D.; Giuffrida, D.; El Majdoub, Y.O.; Kounnoun, A.; Miceli, N.; Taviano, M.F.; Mondello, L.; et al. The Contribution of Carotenoids, Phenolic Compounds, and Flavonoids to the Antioxidative Properties of Marine Microalgae Isolated from Mediterranean Morocco. Molecules 2019, 24, 4037. [Google Scholar] [CrossRef] [Green Version]
- Da Costa, E.; Silva, J.; Mendonça, S.H.; Abreu, M.H.; Domingues, M.R.R.; Mendonça, S.H.; Abreu, M.H.; Domingues, M.R.R. Lipidomic approaches towards deciphering glycolipids from microalgae as a reservoir of bioactive lipids. Mar. Drugs 2016, 14, 101. [Google Scholar] [CrossRef] [Green Version]
- Oppedisano, F.; Macrì, R.; Gliozzi, M.; Musolino, V.; Carresi, C.; Maiuolo, J.; Bosco, F.; Nucera, S.; Zito, M.C.; Guarnieri, L.; et al. The Anti-Inflammatory and Antioxidant Properties of n-3 PUFAs: Their Role in Cardiovascular Protection. Biomedicines 2020, 8, 306. [Google Scholar] [CrossRef] [PubMed]
- Melo, T.; Marques, S.; Ferreira, I.; Cruz, M.T.; Domingues, P.; Segundo, M.; Domingues, M.R.M. New Insights into the Anti-Inflammatory and Antioxidant Properties of Nitrated Phospholipids. Lipids 2018, 53, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, I.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Kitts, D.D. Antioxidant Property of Coffee Components: Assessment of Methods that Define Mechanisms of Action. Molecules 2014, 19, 19180–19208. [Google Scholar] [CrossRef] [Green Version]
- Gouda, M.; Chen, K.; Li, X.; Liu, Y.; He, Y. Detection of microalgae single-cell antioxidant and electrochemical potentials by gold microelectrode and Raman micro-spectroscopy combined with chemometrics. Sens. Actuators B Chem. 2021, 329, 129229. [Google Scholar] [CrossRef]
- Richard, D.; Kefi, K.; Barbe, U.; Bausero, P.; Visioli, F. Polyunsaturated fatty acids as antioxidants. Pharmacol. Res. 2008, 57, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Meital, L.T.; Windsor, M.T.; Perissiou, M.; Schulze, K.; Magee, R.; Kuballa, A.; Golledge, J.; Bailey, T.G.; Askew, C.D.; Russell, F.D. Omega-3 fatty acids decrease oxidative stress and inflammation in macrophages from patients with small abdominal aortic aneurysm. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Sun, T.; Magnuson, A.D.; Qamar, T.R.; Lei, X.G. Defatted Microalgae-Mediated Enrichment of n–3 Polyunsaturated Fatty Acids in Chicken Muscle Is Not Affected by Dietary Selenium, Vitamin E, or Corn Oil. J. Nutr. 2018, 148, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- El-Fayoumy, E.A.; Shanab, S.M.M.; Gaballa, H.S.; Tantawy, M.A.; Shalaby, E.A. Evaluation of antioxidant and anticancer activity of crude extract and different fractions of Chlorella vulgaris axenic culture grown under various concentrations of copper ions. BMC Complement. Med. Ther. 2021, 21, 1–16. [Google Scholar] [CrossRef]
- Custódio, L.; Justo, T.; Silvestre, L.; Barradas, A.; Duarte, C.V.; Pereira, H.; Barreira, L.; Rauter, A.P.; Albericio, F.; Varela, J. Microalgae of different phyla display antioxidant, metal chelating and acetylcholinesterase inhibitory activities. Food Chem. 2012, 131, 134–140. [Google Scholar] [CrossRef]
- Gangadhar, K.N.; Pereira, H.; Rodrigues, M.J.; Custódio, L.; Barreira, L.; Malcata, F.X.; Varela, J. Microalgae-based unsaponifiable matter as source of natural antioxidants and metal chelators to enhance the value of wet Tetraselmis chuii biomass. Open Chem. 2016, 14, 299–307. [Google Scholar] [CrossRef] [Green Version]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Aued-Pimentel, S.; Lago, J.H.G.; Chaves, M.H.; Kumagai, E.E. Evaluation of a methylation procedure to determine cyclopropenoids fatty acids from Sterculia striata St. Hil. Et Nauds seed oil. J. Chromatogr. A 2004, 1054, 235–239. [Google Scholar] [CrossRef] [PubMed]
- LIPID MAPS® Lipidomics Gateway [Internet]. Available online: https://lipidmaps.org/resources/lipidweb/index.php?page=ms/methesters/me-arch/index.htm (accessed on 4 June 2021).
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- RStudio Team. RStudio: Integrated Development Environment for R; RStudio Team: Boston, MA, USA, 2016. [Google Scholar]
- Xia, J.; Sinelnikov, I.V.; Han, B.; Wishart, D.S. MetaboAnalyst 3.0—making metabolomics more meaningful. Nucleic Acids Res. 2015, 43, W251–W257. [Google Scholar] [CrossRef] [Green Version]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 36249. [Google Scholar] [CrossRef] [Green Version]
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses. CRAN R-Project, Package Version 1.0.7; R Packag Version. 2020. Available online: https://cran.r-project.org/web/packages/factoextra/index.html (accessed on 14 May 2021).
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S.; Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef] [Green Version]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2–Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Kolde, R. Pheatmap: Pretty Heatmaps. CRAN R-Project, R Package Version 1.0.12. 2019. Available online: https://cran.r-project.org/web/packages/pheatmap/index.html (accessed on 14 May 2021).
- Wickham, H.; François, R.; Henry, L.; Müller, K. dplyr: A Grammar of Data Manipulation, R package version; Media; GitHub, Inc.: San Francisco, CA, USA, 2019. [Google Scholar]
- Wickham, H.; Henry, L. Tidyr: Easily Tidy Data with “Spread()” and “Gather()” Functions. R Packag Version 080. 2018. Available online: https//CRANR-project.org/package=tidyr (accessed on 22 April 2021).
- Magalhães, L.M.; Segundo, M.A.; Reis, S.; Lima, J.L.F.C. Automatic method for determination of total antioxidant capacity using 2,2-diphenyl-1-picrylhydrazyl assay. Anal. Chim. Acta 2006, 558, 310–318. [Google Scholar] [CrossRef]
- Magalhães, L.M.; Barreiros, L.; Maia, M.; Reis, S.; Segundo, M. Rapid assessment of endpoint antioxidant capacity of red wines through microchemical methods using a kinetic matching approach. Talanta 2012, 97, 473–483. [Google Scholar] [CrossRef] [PubMed]

| FA | Chlorophyta | Bacillariophyta | Cyanobacteria | Ochrophyta | |||
|---|---|---|---|---|---|---|---|
| Chlorella vulgaris | Chlorococcum amblystomatis | Scenedesmus obliquus | Tetraselmis chui | Phaeodactylum tricornutum | Spirulina sp. | Nannochloropsis oceanica | |
| C12:0 | 0.2 ± 0.0 | ||||||
| C14:0 | 0.3 ± 0.0 | 1.4 ± 0.2 | 0.5 ± 0.1 | 0.5 ± 0.1 | 4.2 ± 0.3 | 0.1 ± 0.0 | 3.5 ± 0.1 |
| C15:0 | 0.3 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.3 ± 0.0 | 0.2 ± 0.0 | |
| C16:0 | 21.5 ± 1.3 | 14.9 ± 2.2 | 24.2 ± 2.1 | 14.7 ± 0.9 | 38.6 ± 0.4 | 22.6 ± 2.0 | |
| C16:1 | 2.9 ± 0.2 | ||||||
| C16:1 ∆11 (ω-5) | 0.5 ± 0.0 | 2.7 ± 0.2 | 0.4 ± 0.1 | 1.3 ± 0.1 | 0.7 ± 0.1 | ||
| C16:1 ∆9 (ω-7) | 2.4 ± 0.2 | 8.9 ± 0.5 | 1.3 ± 0.1 | 1.1 ± 0.1 | 16.4 ± 0.3 | 5.1 ± 0.1 | 21.6 ± 1.3 |
| C16:1 ∆7 (ω-9) | 0.9 ± 0.1 | 1.2 ± 0.1 | 0.7 ± 0.1 | 0.2 ± 0.0 | 1.6 ± 0.1 | 0.1 ± 0.0 | |
| C16:2 | 0.8 ± 0.0 | ||||||
| C16:2 ∆9,12 (ω-4) | 5.1 ± 0.1 | 0.2 ± 0.0 | |||||
| C16:2 ∆7,10 (ω-6) | 6.7 ± 0.4 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.5 ± 0.1 | 0.1 ± 0.0 | ||
| C16:3 ∆7,10,13 (ω-3) | 14.2 ± 0.8 | 0.8 ± 0.0 | 0.7 ± 0.1 | 1.3 ± 0.2 | 0.2 ± 0.0 | ||
| C16:3 ∆6,9,12 (ω-4) | 4.7 ± 0.1 | ||||||
| C16:3 ∆4,7,10 (ω-6) | 0.7 ± 0.0 | 0.2 ± 0.0 | 0.4 ± 0.0 | ||||
| C16:4 ∆6,9,12,15 (ω-1) | 7.6 ± 0.2 | ||||||
| C16:4 ∆4,7,10,13 (ω-3) | 12.7 ± 0.6 | 15.5 ± 1.4 | 14.5 ± 1.8 | 0.6 ± 0.0 | |||
| C17:0 | 0.7 ± 0.0 | 0.2 ± 0.0 | 0.3 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 | 0.1 ± 0.0 |
| C17:1 | 0.5 ± 0.0 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 | 0.4 ± 0.0 | ||
| C18:0 | 6.5 ± 2.2 | 4.0 ± 2.1 | 9.7 ± 3.9 | 8.8 ± 6.3 | 3.7 ± 1.4 | 5.4 ± 2.1 | 6.3 ± 3.2 |
| C18:1 ∆11 (ω-7) | 2.3 ± 0.1 | 4.4 ± 0.2 | 1.8 ± 0.1 | 4.8 ± 0.6 | 2.6 ± 0.1 | 1.4 ± 0.1 | 0.5 ± 0.1 |
| C18:1 ∆9 (ω-9) | 4.1 ± 0.2 | 2.2 ± 0.1 | 4.3 ± 0.6 | 7.0 ± 0.9 | 1.3 ± 0.1 | 2.6 ± 0.1 | 4.1 ± 0.3 |
| C18:2 ∆9,12 (ω-6) | 17.6 ± 0.9 | 2.9 ± 0.2 | 3.5 ± 0.3 | 3.6 ± 0.5 | 3.1 ± 0.1 | 21.4 ± 0.8 | 3.2 ± 0.3 |
| C18:3 ∆9,12,15 (ω-3) | 26.3 ± 1.4 | 21.9 ± 0.9 | 35.0 ± 2.7 | 18.3 ± 2.4 | 1.8 ± 0.1 | ||
| C18:3 ∆6,9,12 (ω-6) | 1.3 ± 0.1 | 0.4 ± 0.1 | 2.3 ± 0.3 | 0.2 ± 0.0 | 23.3 ± 0.8 | 0.2 ± 0.0 | |
| C18:4 ∆6,9,12,15 (ω-3) | 4.0 ± 0.2 | 4.7 ± 0.4 | 6.3 ± 0.9 | 0.5 ± 0.0 | |||
| C20:1 ∆11 | 0.6 ± 0.1 | ||||||
| C20:3 ∆5,11,14 (ω-6) | 0.3 ± 0.1 | ||||||
| C20:4 ∆5,11,14,17 (ω-3) | 0.3 ± 0.0 | ||||||
| C20:4 ∆5,8,11,14 (ω-6) | 1.5 ± 0.1 | 0.3 ± 0.1 | 1.1 ± 0.1 | 5.3 ± 0.6 | |||
| C20:5 ∆5,8,11,14,17 (ω-3) | 10.7 ± 0.6 | 4.2 ± 0.6 | 27.3 ± 1.5 | 30.8 ± 2.4 | |||
| C22:6 ∆4,7,10,13,16,19 (ω-3) | 0.6 ± 0.1 | ||||||
| C24:0 | 0.9 ± 0.1 | ||||||
| Indicators | Chlorophyta | Bacillariophyta | Cyanobacteria | Ochrophyta | |||
|---|---|---|---|---|---|---|---|
| Chlorella vulgaris | Chlorococcum amblystomatis | Scenedesmus obliquus | Tetraselmis chui | Phaeodactylum tricornutum | Spirulina sp. | Nannochloropsis oceanica | |
| ∑ SFA | 24.9 ± 4.1 | 27.3 ± 3.3 | 25.5 ± 5.7 | 33.6 ± 8.4 | 23.8 ± 2.4 | 44.2 ± 1.8 | 33.0 ± 5.1 |
| ∑ MUFA | 10.3 ± 0.6 | 16.1 ± 0.8 | 14.3 ± 1.1 | 14.5 ± 1.8 | 21.9 ± 0.5 | 10.9 ± 0.3 | 27.4 ± 1.8 |
| ∑ PUFA | 64.6 ± 3.5 | 56.7 ± 2.6 | 60.1 ± 4.8 | 51.8 ± 6.6 | 54.3 ± 2.0 | 44.8 ± 1.5 | 39.7 ± 3.4 |
| ∑ PUFA ω-6 | 24.3 ± 1.4 | 6.7 ± 0.4 | 4.2 ± 0.4 | 7.3 ± 1.0 | 4.8 ± 0.3 | 44.6 ± 1.5 | 8.9 ± 0.9 |
| ∑ PUFA ω-3 | 40.5 ± 2.2 | 50.0 ± 2.2 | 55.9 ± 4.5 | 44.6 ± 5.7 | 31.3 ± 1.7 | -- | 30.8 ± 2.4 |
| ω-6/ω-3 ratio | 0.6 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 | -- | 0.3 ± 0.0 |
| AI | 0.2 ± 0.0 a,b,c | 0.4 ± 0.0 d | 0.2 ± 0.1 e,f,g | 0.4 ± 0.1 | 0.5 ± 0.1 a,f | 0.7 ± 0.0 b,d,g | 0.6 ± 0.1 c,e |
| TI | 0.2 ± 0.0 b | 0.2 ± 0.0 h,i | 0.1 ± 0.0 e,g | 0.2 ± 0.1 | 0.2 ± 0.2 | 1.6 ± 0.1 b,g,h | 0.3 ± 0.1 e,i |
| (h/H) | 2.8 ± 0.4 b,j | 1.7 ± 0.2 | 2.9 ± 0.5 g,k | 1.4 ± 0.3 j,k | 1.8 ± 0.2 l | 0.6 ± 0.0 b,g,l | 1.7 ± 0.2 |
| Microalgae | Temperature (°C) | pH |
|---|---|---|
| Chlorella vulgaris | <25 | 7.5–8.5 |
| Chlorococcum amblystomatis | <28 | 7.5–8.5 |
| Scenedesmus obliquus | <25 | 7.5–8.5 |
| Tetraselmis chui | <25 | 7.5–8.5 |
| Phaeodactylum tricornutum | <23 | 7.5–8.5 |
| Nannochloropsis oceanica | <25 | 7.5–8.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conde, T.A.; Neves, B.F.; Couto, D.; Melo, T.; Neves, B.; Costa, M.; Silva, J.; Domingues, P.; Domingues, M.R. Microalgae as Sustainable Bio-Factories of Healthy Lipids: Evaluating Fatty Acid Content and Antioxidant Activity. Mar. Drugs 2021, 19, 357. https://doi.org/10.3390/md19070357
Conde TA, Neves BF, Couto D, Melo T, Neves B, Costa M, Silva J, Domingues P, Domingues MR. Microalgae as Sustainable Bio-Factories of Healthy Lipids: Evaluating Fatty Acid Content and Antioxidant Activity. Marine Drugs. 2021; 19(7):357. https://doi.org/10.3390/md19070357
Chicago/Turabian StyleConde, Tiago A., Bruna F. Neves, Daniela Couto, Tânia Melo, Bruno Neves, Margarida Costa, Joana Silva, Pedro Domingues, and M. Rosário Domingues. 2021. "Microalgae as Sustainable Bio-Factories of Healthy Lipids: Evaluating Fatty Acid Content and Antioxidant Activity" Marine Drugs 19, no. 7: 357. https://doi.org/10.3390/md19070357
APA StyleConde, T. A., Neves, B. F., Couto, D., Melo, T., Neves, B., Costa, M., Silva, J., Domingues, P., & Domingues, M. R. (2021). Microalgae as Sustainable Bio-Factories of Healthy Lipids: Evaluating Fatty Acid Content and Antioxidant Activity. Marine Drugs, 19(7), 357. https://doi.org/10.3390/md19070357










