The Application of Seaweed Polysaccharides and Their Derived Products with Potential for the Treatment of Alzheimer’s Disease
Abstract
1. Introduction
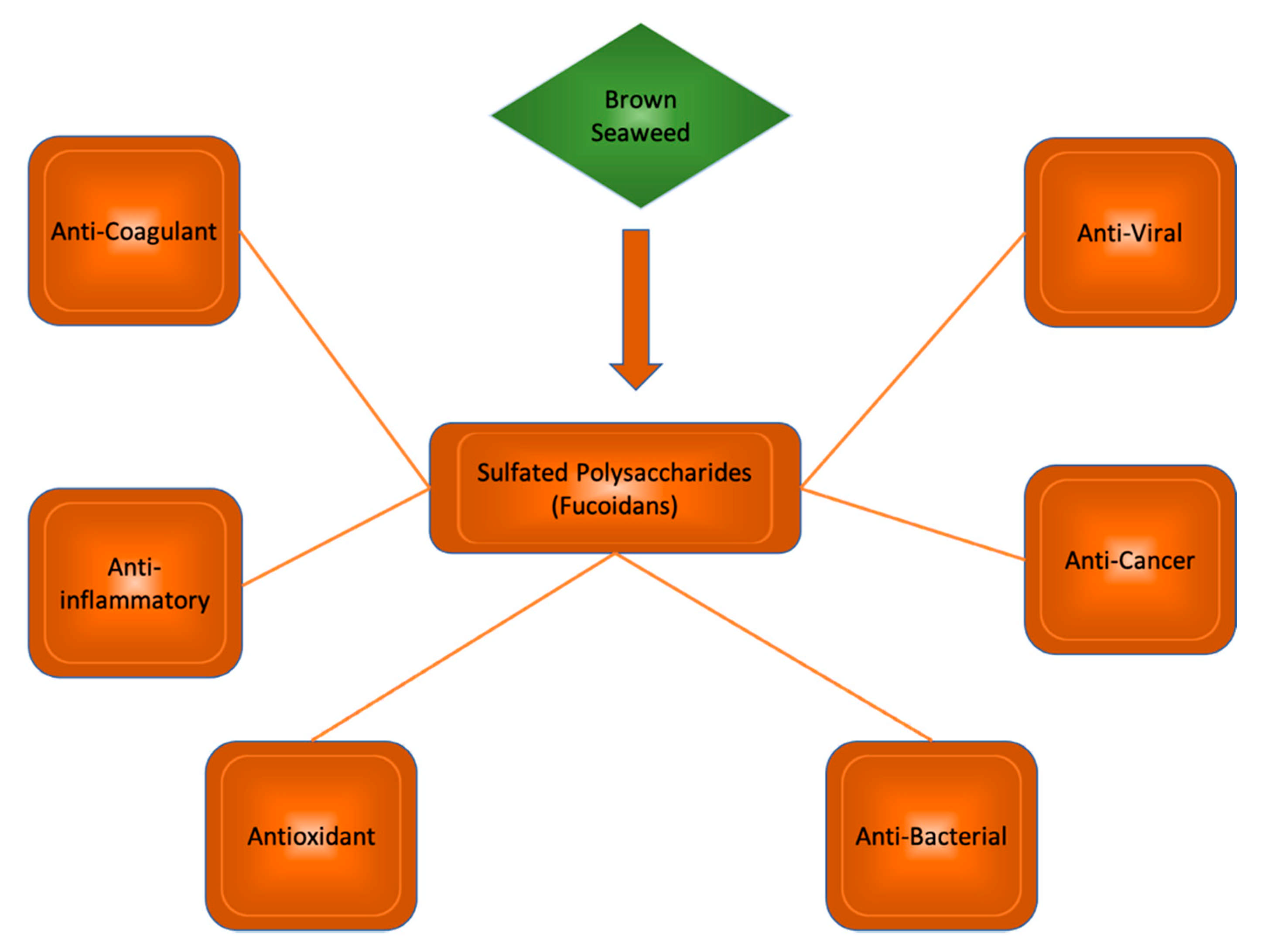
2. Application of Polysaccharides from Brown Algae in Treating AD
2.1. Phaeophyta
2.2. Findings of Recent Studies
3. Application of Polysaccharides from Green Algae in Treating AD
3.1. Chlorophyta
3.2. Findings of Recent Studies
4. Application of Polysaccharides from Red Algae in Treating AD
4.1. Rhodophyta
4.2. Findings Findings of Recent Studies
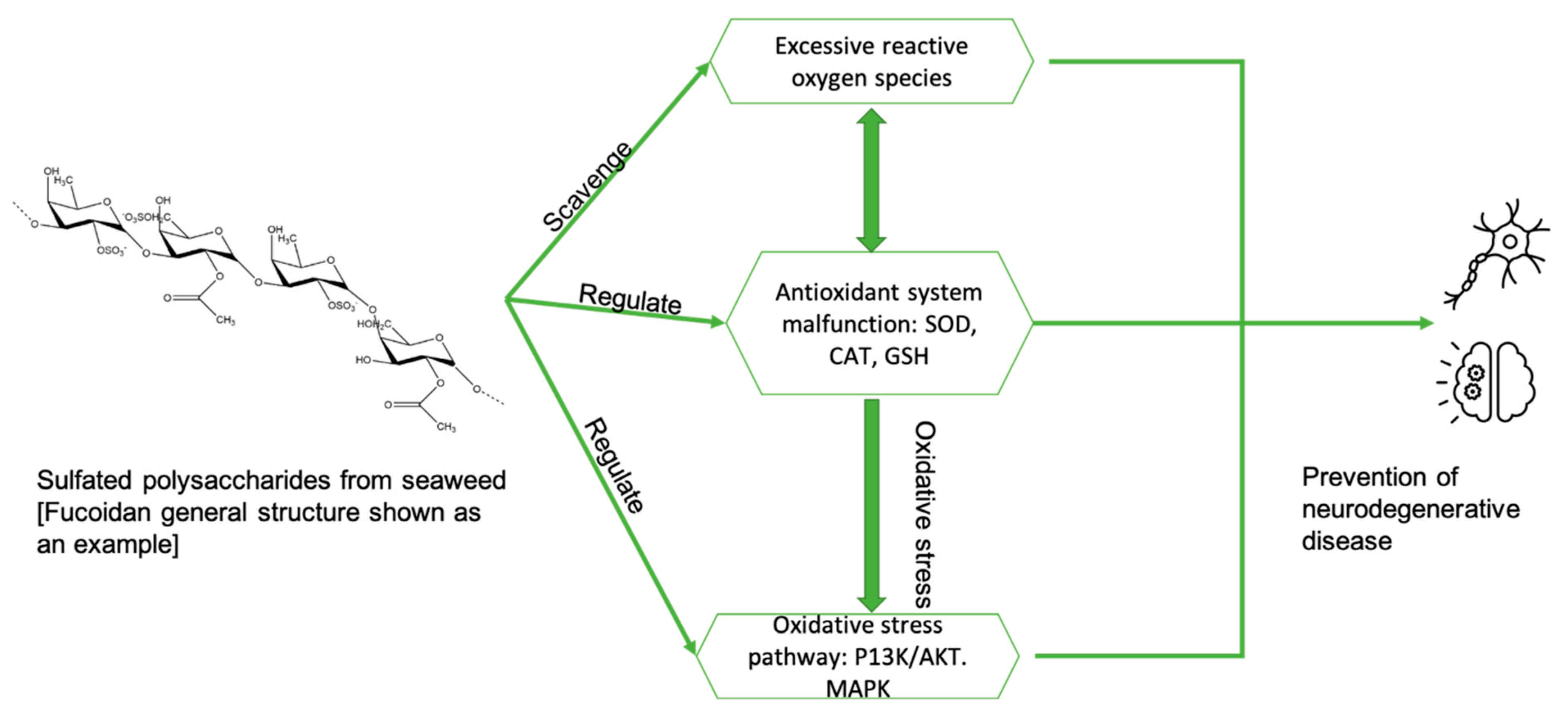
5. The Mechanism of Polysaccharides from Seaweeds for the Treatment of AD
6. Challenges and Opportunities
Author Contributions
Funding
Conflicts of Interest
References
- Lin, M.T.; Beal, M.F. Mitochondrial Dysfunction and Oxidative Stress in Neurodegenerative Diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef]
- Huang, C.Y.; Kuo, C.H.; Chen, P.W. Compressional-Puffing Pretreatment Enhances Neuroprotective Effects of Fucoidans from the Brown Seaweed Sargassum Hemiphyllum on 6-Hydroxydopamine-Induced Apoptosis in SH-SY5Y Cells. Molecules 2018, 23, 78. [Google Scholar] [CrossRef]
- Manigandan, V.; Karthik, R.; Saravanan, R. Marine Carbohydrate Based Therapeutics for Alzheimer Disease-Mini Review. J. Neurol. Neurosci. 2015, 6. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Olaniran, A.O.; Okoh, A.I. Macroalgae as a Valuable Source of Naturally Occurring Bioactive Compounds for the Treatment of Alzheimer’s Disease. Mar. Drugs 2019, 17, 609. [Google Scholar] [CrossRef]
- Bertsch, M.; Franchi, B.; Meschini, V.; Tesi, M.C.; Tosin, A. A Sensitivity Analysis of a Mathematical Model for the Synergistic Interplay of Amyloid Beta and Tau on the Dynamics of Alzheimer’s Disease. Brain Multiphys. 2021, 2, 100020. [Google Scholar] [CrossRef]
- Barbalace, M.C.; Malaguti, M.; Giusti, L.; Lucacchini, A.; Hrelia, S.; Angeloni, C. Anti-Inflammatory Activities of Marine Algae in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 61. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Wu, Y.; Jiang, G.; Feng, L.; Wang, Z.; Yuan, G.; Tong, H. Sulfated Polysaccharides from Rhodiola Sachalinensis Reduce D-Gal-Induced Oxidative Stress in NIH 3T3 Cells. Int. J. Biol. Macromol. 2019, 140, 288–293. [Google Scholar] [CrossRef]
- Silva, J.; Alves, C.; Pinteus, S.; Mendes, S.; Pedrosa, R. Neuroprotective Effects of Seaweeds against 6-Hydroxidopamine-Induced Cell Death on an in Vitro Human Neuroblastoma Model. BMC Complement. Altern. Med. 2018, 18. [Google Scholar] [CrossRef] [PubMed]
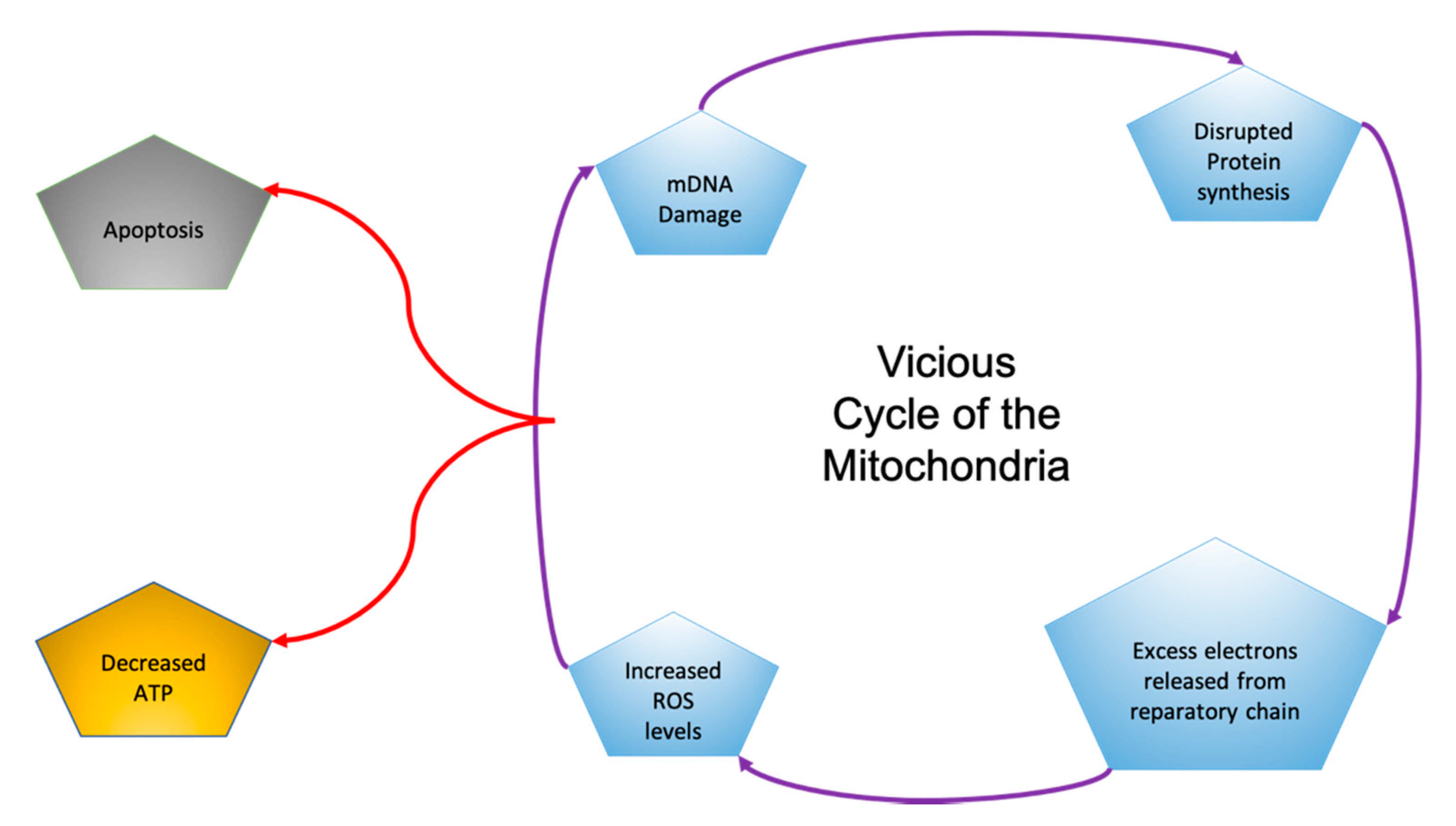
- Elfawy, H.A.; Das, B. Crosstalk between Mitochondrial Dysfunction, Oxidative Stress, and Age Related Neurodegenerative Disease: Etiologies and Therapeutic Strategies. Life Sci. 2019, 218, 165–184. [Google Scholar] [CrossRef] [PubMed]
- Alghazwi, M.; Charoensiddhi, S.; Smid, S.; Zhang, W. Impact of Ecklonia Radiata Extracts on the Neuroprotective Activities against Amyloid Beta (Aβ1-42) Toxicity and Aggregation. J. Funct. Foods 2020, 68. [Google Scholar] [CrossRef]
- Heo, J.-H.; Lee, S.-T.; Oh, M.-J.; Park, H.-J.; Shim, J.-Y.; Chu, K.; Kim, M.-H. Improvement of Cognitive Deficit in Alzheimer’s Disease Patients by Long Term Treatment with Korean Red Ginseng. J. Ginseng Res. 2011, 35, 457–461. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, Y.; Zhu, J.; Liang, H.; He, X.; Qian, J.; Lin, H.; Tao, Y.; Zhu, K. Quantitative Assessment of Hippocampal Tau Pathology in AD and PART. J. Mol. Neurosci. 2020, 70, 1808–1811. [Google Scholar] [CrossRef] [PubMed]
- Park, S.K.; Kang, J.Y.; Kim, J.M.; Park, S.H.; Kwon, B.S.; Kim, G.H.; Heo, H.J. Protective Effect of Fucoidan Extract from Ecklonia Cava on Hydrogen Peroxide-Induced Neurotoxicity. J. Microbiol. Biotechnol. 2018, 28, 40–49. [Google Scholar] [CrossRef]
- Park, S.K.; Kang, J.Y.; Kim, J.M.; Yoo, S.K.; Han, H.J.; Chung, D.H.; Kim, D.O.; Kim, G.H.; Heo, H.J. Fucoidan-Rich Substances from Ecklonia Cava Improve Trimethyltin-Induced Cognitive Dysfunction via Down-Regulation of Amyloid β Production/Tau Hyperphosphorylation. Mar. Drugs 2019, 17, 591. [Google Scholar] [CrossRef]
- Collins, K.G.; Fitzgerald, G.F.; Stanton, C.; Ross, R.P. Looking beyond the Terrestrial: The Potential of Seaweed Derived Bioactives to Treat Non-Communicable Diseases. Mar. Drugs 2016, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, A.R.; Tiwari, U.; Rajauria, G. Seaweed Nutraceuticals and Their Therapeutic Role in Disease Prevention. Food Sci. Hum. Wellness 2019, 8, 252–263. [Google Scholar] [CrossRef]
- Freile-Pelegrín, Y.; Tasdemir, D. Seaweeds to the Rescue of Forgotten Diseases: A Review. Bot. Mar. 2019, 62, 211–226. [Google Scholar] [CrossRef]
- Jutur, P.P.; Nesamma, A.A.; Shaikh, K.M. Algae-Derived Marine Oligosaccharides and Their Biological Applications. Front. Mar. Sci. 2016, 3, 83. [Google Scholar] [CrossRef]
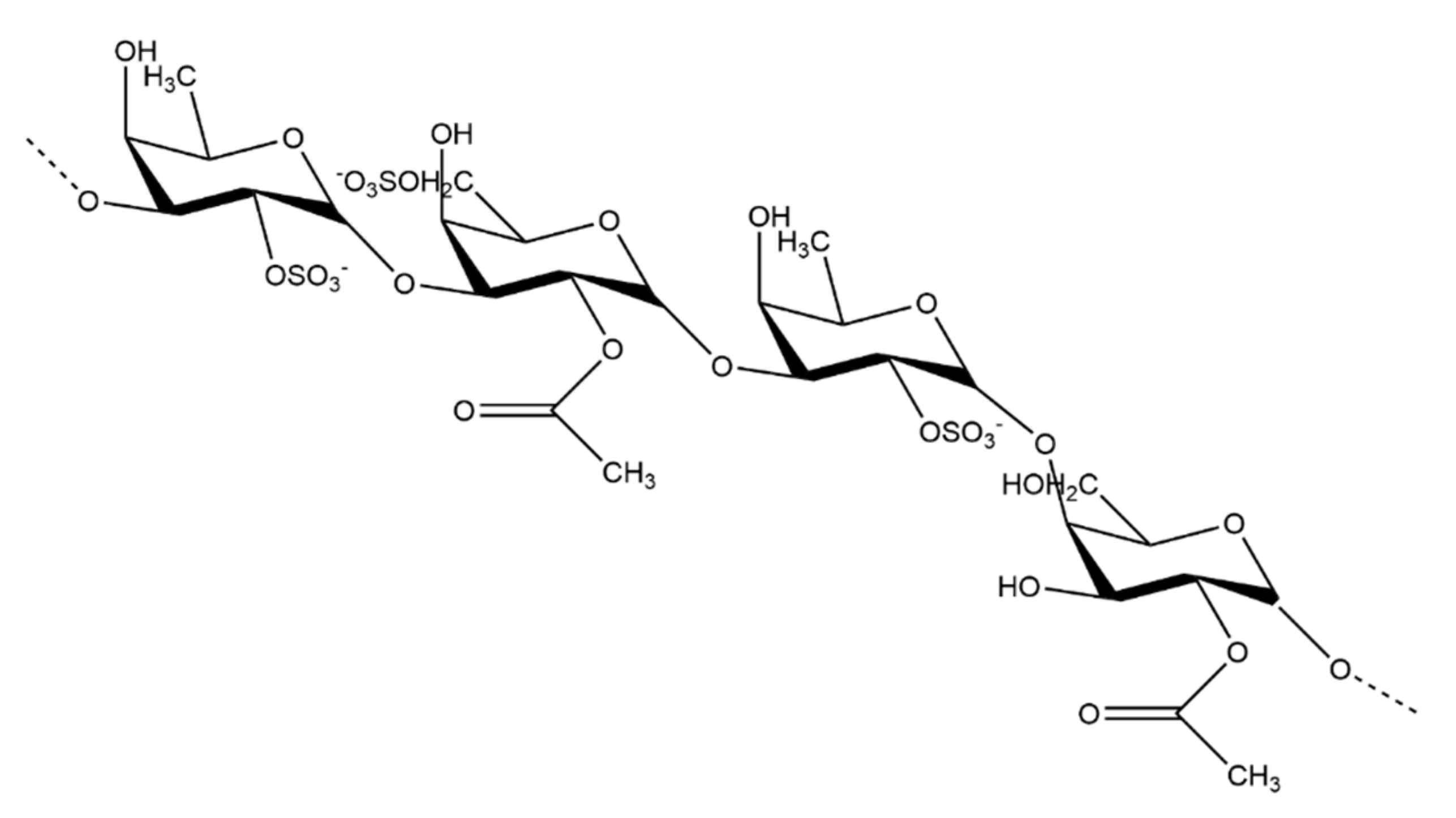
- Ehrig, K.; Alban, S. Sulfated Galactofucan from the Brown Alga Saccharina Latissima-Variability of Yield, Structural Composition and Bioactivity. Mar. Drugs 2015, 13, 76–101. [Google Scholar] [CrossRef] [PubMed]
- Bogie, J.; Hoeks, C.; Schepers, M.; Tiane, A.; Cuypers, A.; Leijten, F.; Chintapakorn, Y.; Suttiyut, T.; Pornpakakul, S.; Struik, D.; et al. Dietary Sargassum Fusiforme Improves Memory and Reduces Amyloid Plaque Load in an Alzheimer’s Disease Mouse Model. Sci. Rep. 2019, 9, 4908. [Google Scholar] [CrossRef]
- Sanjeewa, K.K.A.; Kang, N.; Ahn, G.; Jee, Y.; Kim, Y.T.; Jeon, Y.J. Bioactive Potentials of Sulfated Polysaccharides Isolated from Brown Seaweed Sargassum Spp in Related to Human Health Applications: A Review. Food Hydrocoll. 2018, 81, 200–208. [Google Scholar] [CrossRef]
- Zhong, Q.; Wei, B.; Wang, S.; Ke, S.; Chen, J.; Zhang, H.; Wang, H. The Antioxidant Activity of Polysaccharides Derived from Marine Organisms: An Overview. Mar. Drugs 2019, 17, 674. [Google Scholar] [CrossRef]
- Gao, Y.; Li, C.; Yin, J.; Shen, J.; Wang, H.; Wu, Y.; Jin, H. Fucoidan, a sulfated polysaccharide from brown algae, improves cognitive impairment induced by infusion of Aβ peptide in rats. Environ. Toxicol. Pharmacol. 2012, 33, 304–311. [Google Scholar] [CrossRef]
- Rizki, I.F.; Sinurat, E.; Fajriah, S.; Saefudin, E. Antioxidant Activity of Sulfated Polysaccharide Extract from Green Seaweed (Caulerpa Lentillifera) Makassar, Indonesia. Key Eng. Mater. 2020, 840, 214–220. [Google Scholar] [CrossRef]
- Barzkar, N.; Jahromi, S.T.; Poorsaheli, H.B.; Vianello, F. Metabolites from Marine Microorganisms, Micro, and Macroalgae: Immense Scope for Pharmacology. Mar. Drugs 2019, 17, 464. [Google Scholar] [CrossRef]
- Olasehinde, T.A.; Mabinya, L.V.; Olaniran, A.O.; Okoh, A.I. Chemical characterization, antioxidant properties, cholinesterase inhibitory and anti-amyloidogenic activities of sulfated polysaccharides from some seaweeds. Bioactive. Carbohydr. Diet. Fibre 2019, 18, 100182. [Google Scholar] [CrossRef]
- Déléris, P.; Nazih, H.; Bard, J.M. Seaweeds in Human Health. In Seaweed in Health and Disease Prevention; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 319–367. ISBN 978-0-12-802793-6. [Google Scholar]
- Liu, J.; Yang, S.; Li, X.; Yan, Q.; Reaney, M.J.T.; Jiang, Z. Alginate Oligosaccharides: Production, Biological Activities, and Potential Applications. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1859–1881. [Google Scholar] [CrossRef] [PubMed]
- Tanna, B.; Mishra, A. Nutraceutical Potential of Seaweed Polysaccharides: Structure, Bioactivity, Safety, and Toxicity. Compr. Rev. Food Sci. Food Saf. 2019, 18, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.-J.; Xu, F.-Q.; Li, Y.-H.; Li, J.; Liu, X.; Wang, X.-F.; Hu, L.-G.; An, Y. Alginate Oligosaccharide Alleviates Myocardial Reperfusion Injury by Inhibiting Nitrative and Oxidative Stress and Endoplasmic Reticulum Stress-Mediated Apoptosis. Drug Des. Devel. Ther. 2017, 11, 2387–2397. [Google Scholar] [CrossRef] [PubMed]
- Olasehinde, T.A.; Mabinya, L.V.; Olaniran, A.O.; Okoh, A.I. Chemical Characterization of Sulfated Polysaccharides from Gracilaria Gracilis and Ulva Lactuca and Their Radical Scavenging, Metal Chelating, and Cholinesterase Inhibitory Activities. Int. J. Food Prop. 2019, 22, 100–110. [Google Scholar] [CrossRef]
- Wang, L.; Oh, J.Y.; Je, J.G.; Jayawardena, T.U.; Kim, Y.-S.; Ko, J.Y.; Fu, X.; Jeon, Y.-J. Protective Effects of Sulfated Polysaccharides Isolated from the Enzymatic Digest of Codium Fragile against Hydrogen Peroxide-Induced Oxidative Stress in in Vitro and in Vivo Models. Algal Res. 2020, 48, 101891. [Google Scholar] [CrossRef]
- Ismail, M.M.; Alotaibi, B.S.; EL-Sheekh, M.M. Therapeutic Uses of Red Macroalgae. Molecules 2020, 25, 4411. [Google Scholar] [CrossRef]
- Yao, Z.A.; Xu, L.; Wu, H.G. Immunomodulatory function of κ-carrageenan oligosaccharides acting on LPS-activated microglial cells. Neurochem. Res. 2014, 39, 333–343. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, L.; Li, X. κ-Carrageenan-Derived Pentasaccharide Attenuates Aβ25–35-Induced Apoptosis in SH-SY5Y Cells via Suppression of the JNK Signaling Pathway. Mol. Med. Rep. 2017, 15, 285–290. [Google Scholar] [CrossRef]
- Suganya, A.M.; Sanjivkumar, M.; Chandran, M.N.; Palavesam, A.; Immanuel, G. Pharmacological Importance of Sulphated Polysaccharide Carrageenan from Red Seaweed Kappaphycus Alvarezii in Comparison with Commercial Carrageenan. Biomed. Pharmacother. 2016, 84, 1300–1312. [Google Scholar] [CrossRef] [PubMed]
- Schepers, M.; Martens, N.; Tiane, A.; Vanbrabant, K.; Liu, H.B.; Lütjohann, D.; Mulder, M.; Vanmierlo, T. Edible Seaweed-Derived Constituents: An Undisclosed Source of Neuroprotective Compounds. Neural Regen. Res. 2020, 15, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Mariya Jose, G.; Muraleedhara Kurup, G. In Vitro Antioxidant Properties of Edible Marine Algae Sargassum Swartzii, Ulva Fasciata and Chaetomorpha Antennina of Kerala Coast. J. Pharmacol. Rep. 2016, 4, 100–108. [Google Scholar]
- Li, H.; Ding, F.; Xiao, L.; Shi, R.; Wang, H.; Han, W.; Huang, Z. Food-Derived Antioxidant Polysaccharides and Their Pharmacological Potential in Neurodegenerative Diseases. Nutrients 2017, 9, 778. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Hwang, J.-H.; Kim, K.-J.; Lee, B.-Y. Ecklonia Cava-Derived Polysaccharide Prevent Hydro Peroxide-Induced Oxidative Stress and Neurotoxicity in Human Microglial HMO6 Cells. J. Food Nutr. Res. 2017, 5, 187–190. [Google Scholar] [CrossRef]
- Watson, P.M.D.; Kavanagh, E.; Allenby, G.; Vassey, M. Bioengineered 3D Glial Cell Culture Systems and Applications for Neurodegeneration and Neuroinflammation. SLAS Discov. 2017, 22, 583–601. [Google Scholar] [CrossRef] [PubMed]
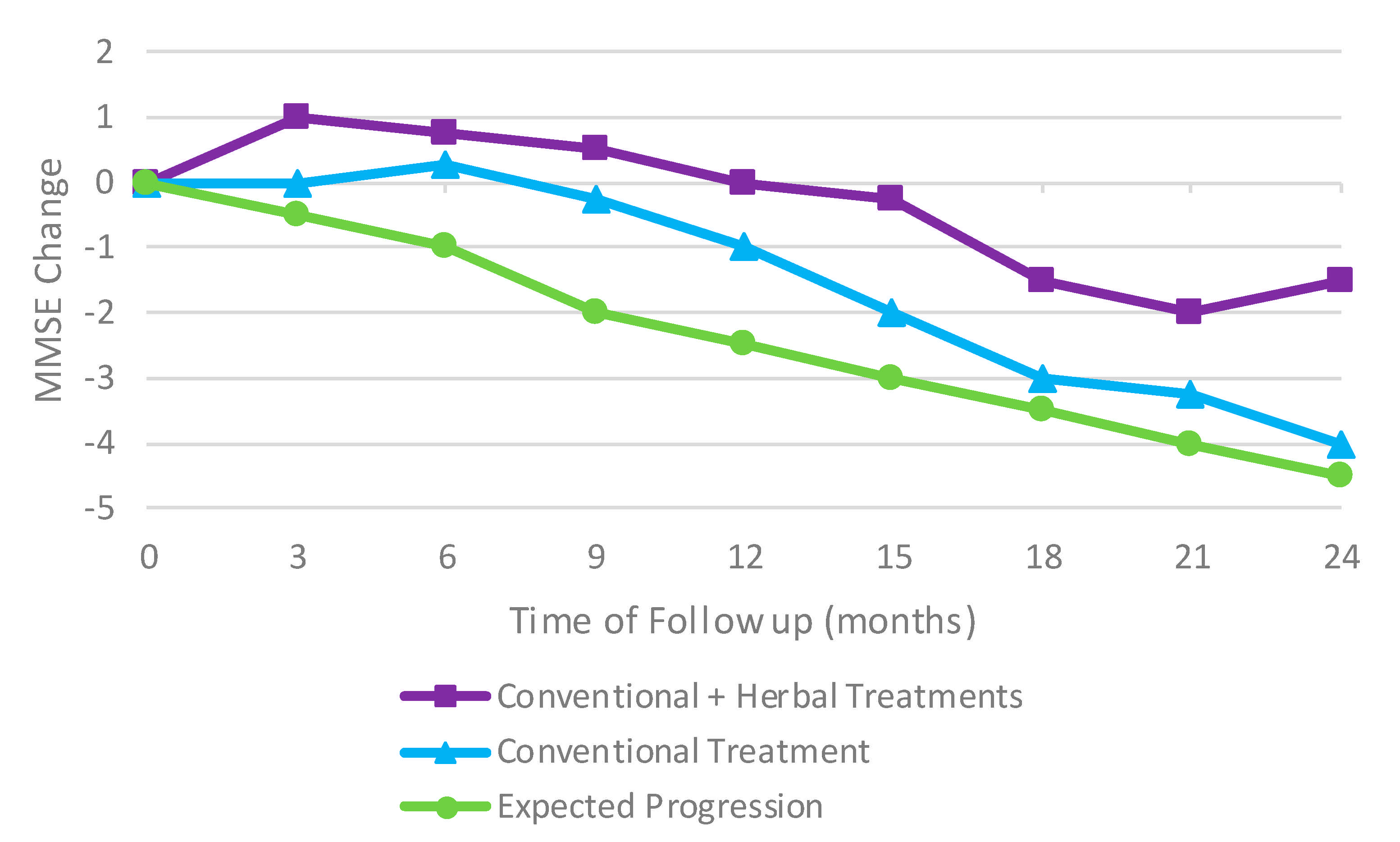
- Shi, J.; Ni, J.; Lu, T.; Zhang, X.; Wei, M.; Li, T.; Liu, W.; Wang, Y.; Shi, Y.; Tian, J. Adding Chinese Herbal Medicine to Conventional Therapy Brings Cognitive Benefits to Patients with Alzheimer’s Disease: A Retrospective Analysis. BMC Complement. Altern. Med. 2017, 17, 533. [Google Scholar] [CrossRef]
- Heo, J.-H.; Lee, S.-T.; Chu, K.; Oh, M.J.; Park, H.-J.; Shim, J.-Y.; Kim, M. Heat-Processed Ginseng Enhances the Cognitive Function in Patients with Moderately Severe Alzheimer’s Disease. Nutr. Neurosci. 2012, 15, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhao, X.; Wang, F.; Lu, Z.; Wang, Y.; Jin, M. Proteomic Study of Sulfated Polysaccharide from Enterobacter Cloacae Z0206 against H2O2-Induced Oxidative Damage in Murine Macrophages. Carbohydr. Polym. 2020, 237, 116147. [Google Scholar] [CrossRef] [PubMed]
- Mohd Sairazi, N.S.; Sirajudeen, K.N.S. Natural Products and Their Bioactive Compounds: Neuroprotective Potentials against Neurodegenerative Diseases. Evid. Based Complement. Alternat. Med. 2020, 2020. [Google Scholar] [CrossRef]
- Cheng, K.K.; Yeung, C.F.; Ho, S.W.; Chow, S.F.; Chow, A.H.L.; Baum, L. Highly Stabilized Curcumin Nanoparticles Tested in an In Vitro Blood–Brain Barrier Model and in Alzheimer’s Disease Tg2576 Mice. AAPS J. 2013, 15, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Grossberg, G.T. Cholinesterase Inhibitors for the Treatment of Alzheimer’s Disease: Getting on and Staying On. Curr. Ther. Res. Clin. Exp. 2003, 64, 216–235. [Google Scholar] [CrossRef]
- Nagarajan, S.; Mathaiyan, M. Emerging Novel Anti HIV Biomolecules from Marine Algae: An Overview. J. Appl. Pharm. Sci. 2015, 5, 153–158. [Google Scholar] [CrossRef]
- Poo, M. New Light on the Horizon of Alzheimer’s Disease. Natl. Sci. Rev. 2020, 7, 831. [Google Scholar] [CrossRef]
- Wang, X.; Sun, G.; Feng, T.; Zhang, J.; Huang, X.; Wang, T.; Xie, Z.; Chu, X.; Yang, J.; Wang, H.; et al. Sodium Oligomannate Therapeutically Remodels Gut Microbiota and Suppresses Gut Bacterial Amino Acids-Shaped Neuroinflammation to Inhibit Alzheimer’s Disease Progression. Cell Res. 2019, 29, 787–803. [Google Scholar] [CrossRef]
| Seaweed Species | Classification | References | Activity Presented in References |
|---|---|---|---|
| Saccharina latissima | Brown | [19] | Anti-Inflammatory |
| Sargassum fusiforme | Brown | [20,21] | Antioxidant, Antiamyloidogenic |
| Ecklonia cava | Brown | [22] | Antioxidant |
| Sargassum fluitans | Brown | [22] | Antioxidant |
| Turbinaria decurrens | Brown | [22] | Antioxidant |
| Laminaria japonica | Brown | [23] | Neuroreparative, Antioxidant |
| Undaria pinnatifida | Brown | [22] | Antioxidant |
| Caulerpa lentillifera | Green | [24] | Antioxidant |
| Ulva lactuca | Green | [25] | Antioxidant |
| Gelidium pristoides | Red | [26] | Antioxidant, Antiamyloidogenic |
| Polysaccharide Sample | Degree of Sulfation (DS) | IC50 Elastase (μg/mL) | ||
|---|---|---|---|---|
| Fraction | F2 | F3 | F2 | F3 |
| B06-SP | 0.28 | 0.82 | 1.87 ± 0.12 | 0.26 ± 0.02 |
| A05-SP | 0.22 | 0.76 | 2.81 ± 0.21 | 0.28 ± 0.01 |
| A09-SP | 0.19 | 0.81 | 3.77 ± 0.16 | 0.21 ± 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bauer, S.; Jin, W.; Zhang, F.; Linhardt, R.J. The Application of Seaweed Polysaccharides and Their Derived Products with Potential for the Treatment of Alzheimer’s Disease. Mar. Drugs 2021, 19, 89. https://doi.org/10.3390/md19020089
Bauer S, Jin W, Zhang F, Linhardt RJ. The Application of Seaweed Polysaccharides and Their Derived Products with Potential for the Treatment of Alzheimer’s Disease. Marine Drugs. 2021; 19(2):89. https://doi.org/10.3390/md19020089
Chicago/Turabian StyleBauer, Sarah, Weihua Jin, Fuming Zhang, and Robert J. Linhardt. 2021. "The Application of Seaweed Polysaccharides and Their Derived Products with Potential for the Treatment of Alzheimer’s Disease" Marine Drugs 19, no. 2: 89. https://doi.org/10.3390/md19020089
APA StyleBauer, S., Jin, W., Zhang, F., & Linhardt, R. J. (2021). The Application of Seaweed Polysaccharides and Their Derived Products with Potential for the Treatment of Alzheimer’s Disease. Marine Drugs, 19(2), 89. https://doi.org/10.3390/md19020089







