Changing Trends in Paralytic Shellfish Poisonings Reflect Increasing Sea Surface Temperatures and Practices of Indigenous and Recreational Harvesters in British Columbia, Canada
Abstract
:1. Introduction
2. Results
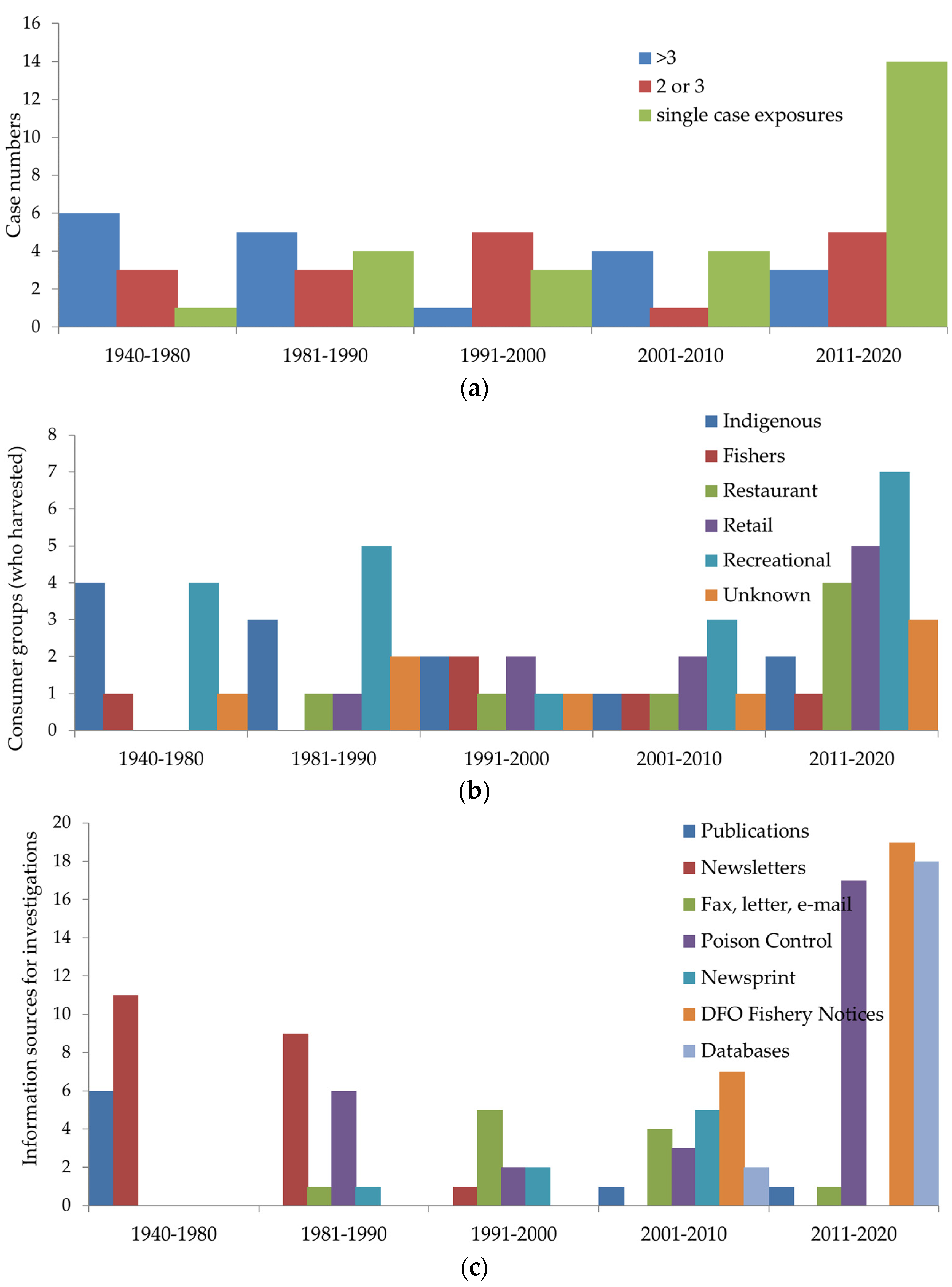
2.1. Investigation Trends
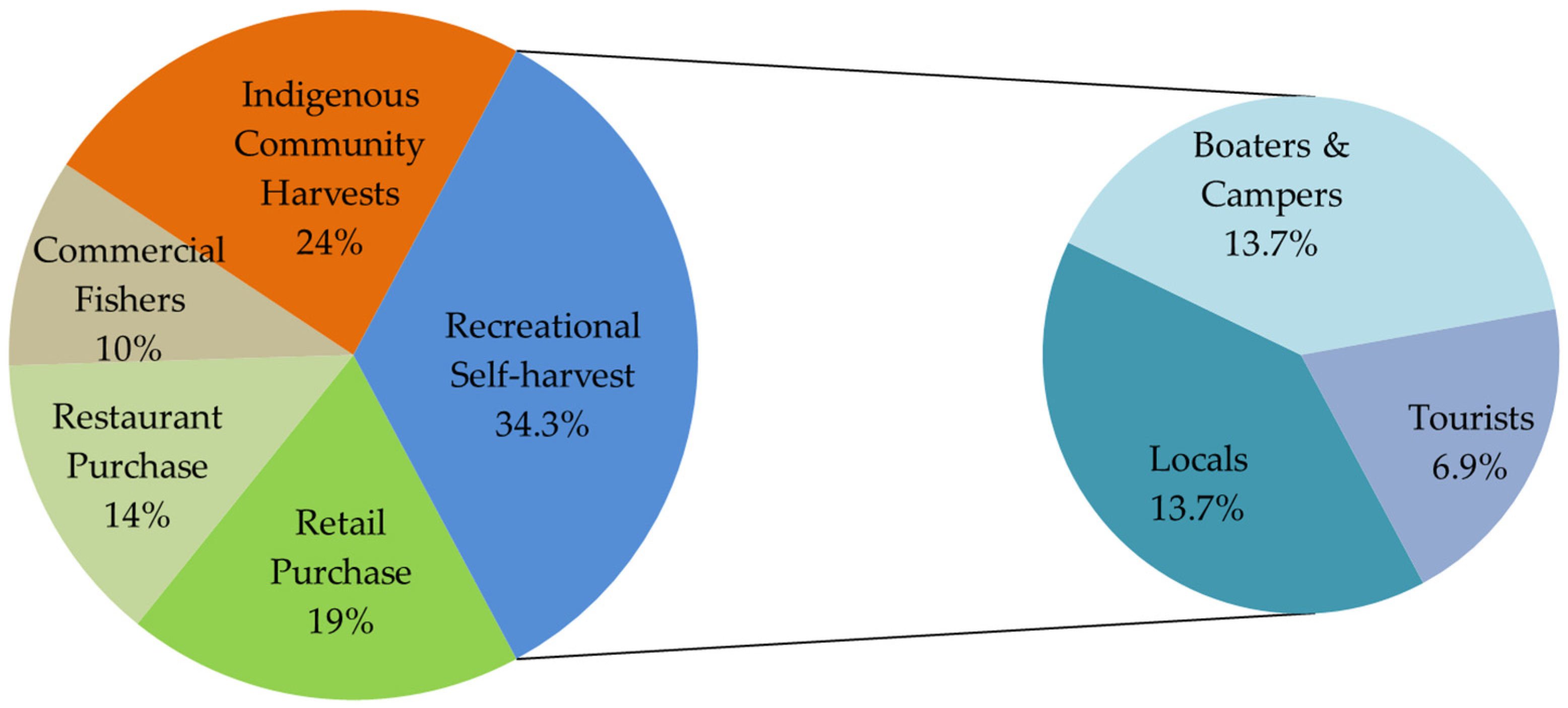
2.2. Case Demographics and Symptoms
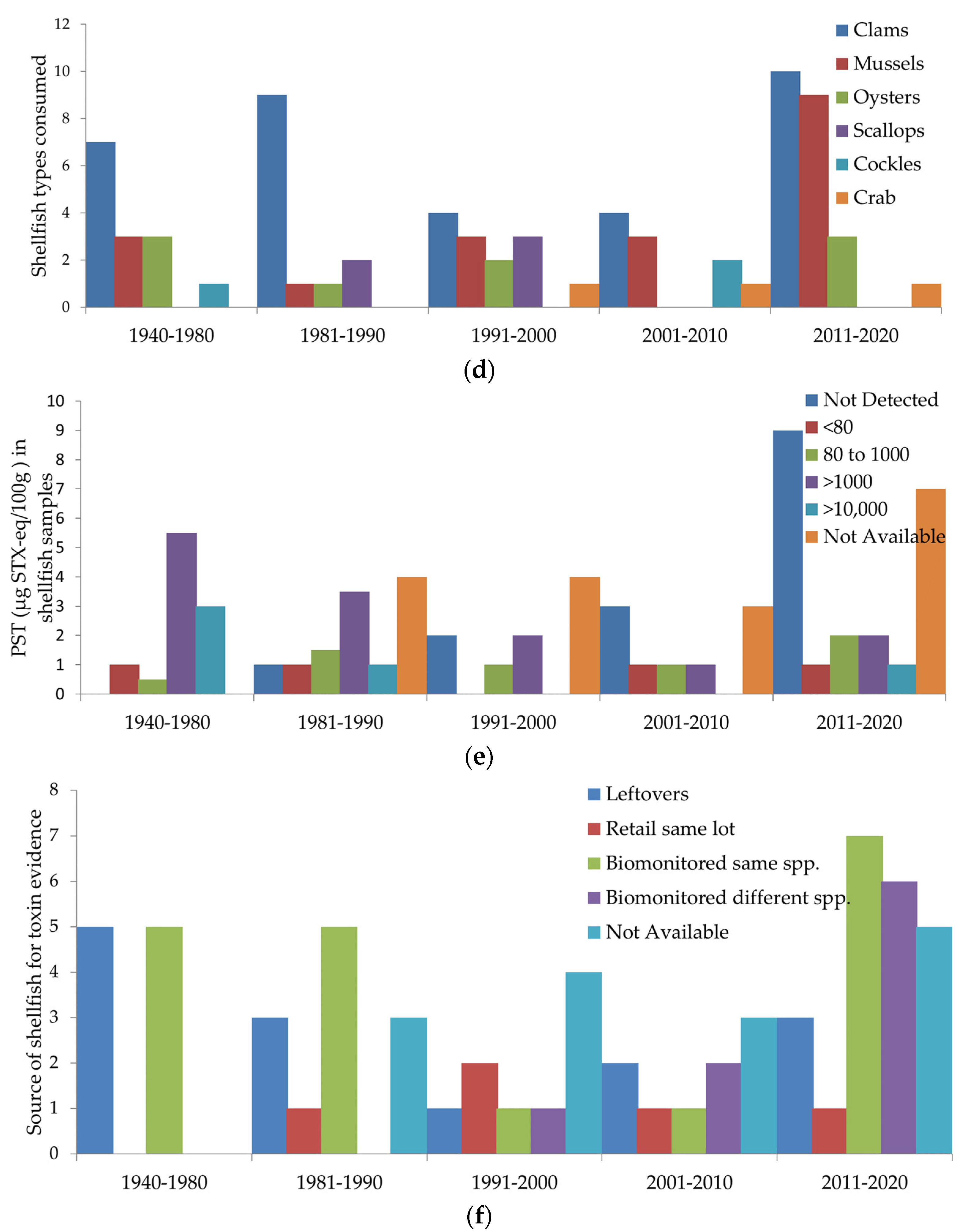
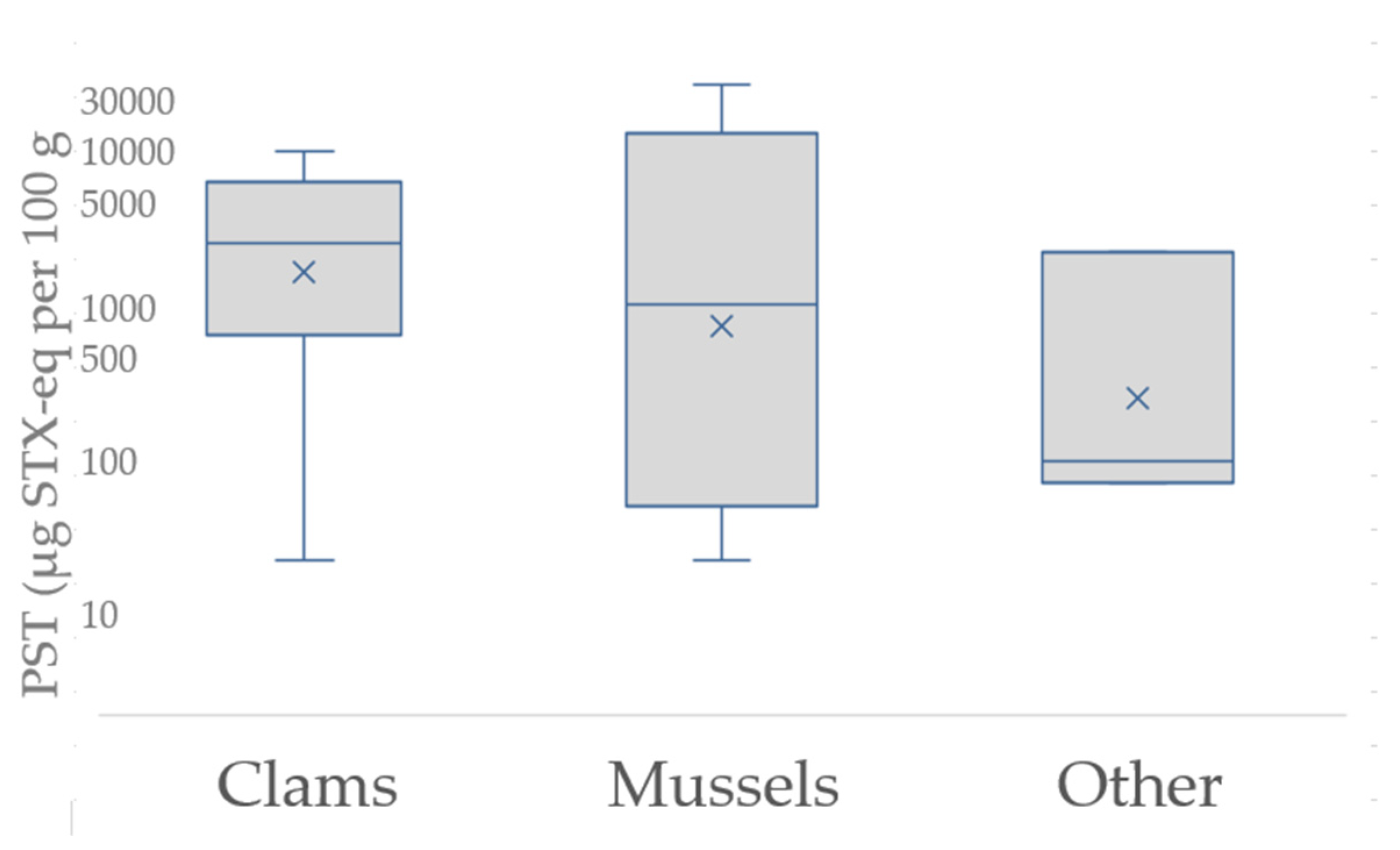
2.3. Food Sources and Toxin Dose
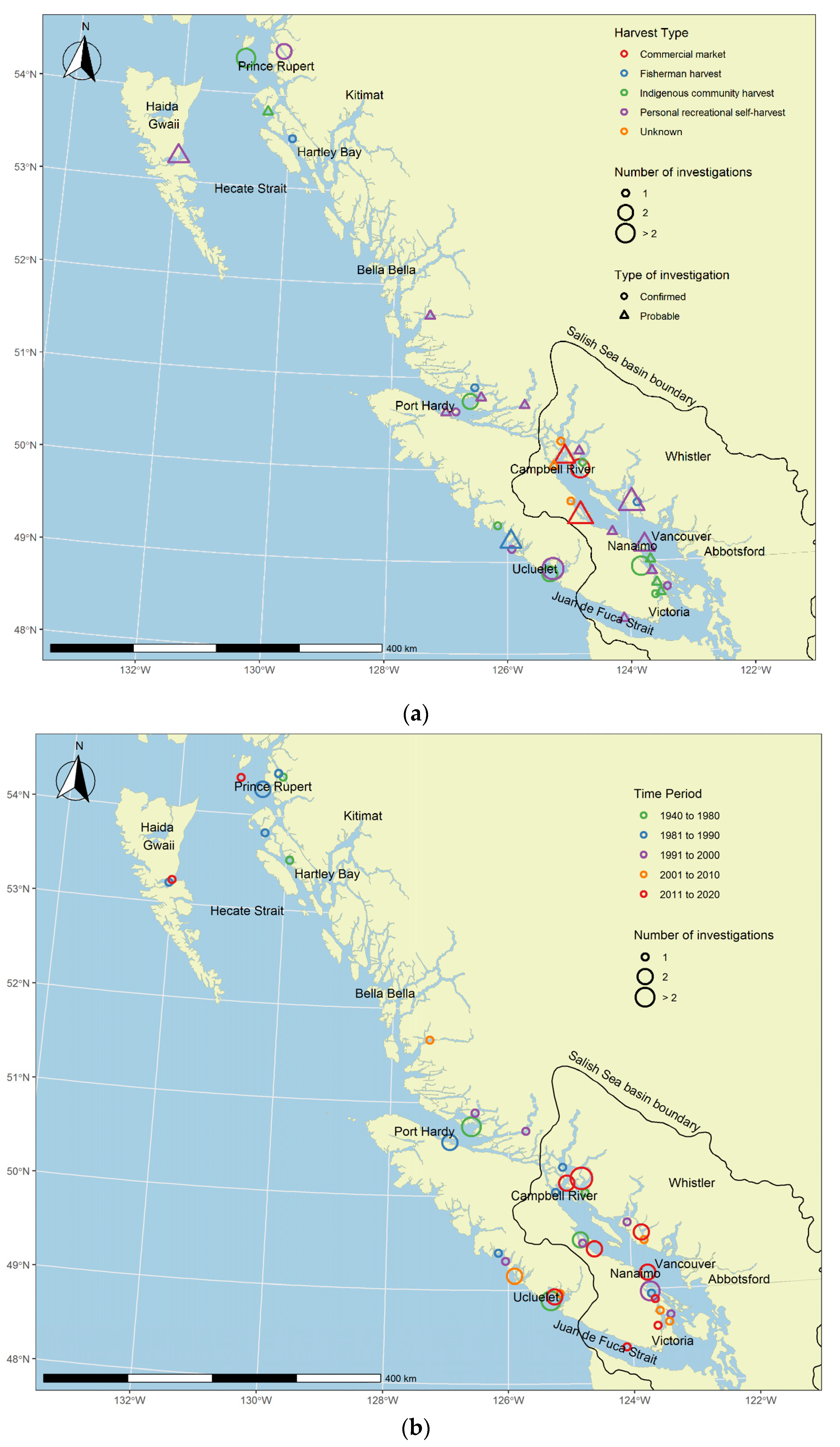
2.4. Harvest Site Locations and Status of PSP Investigations
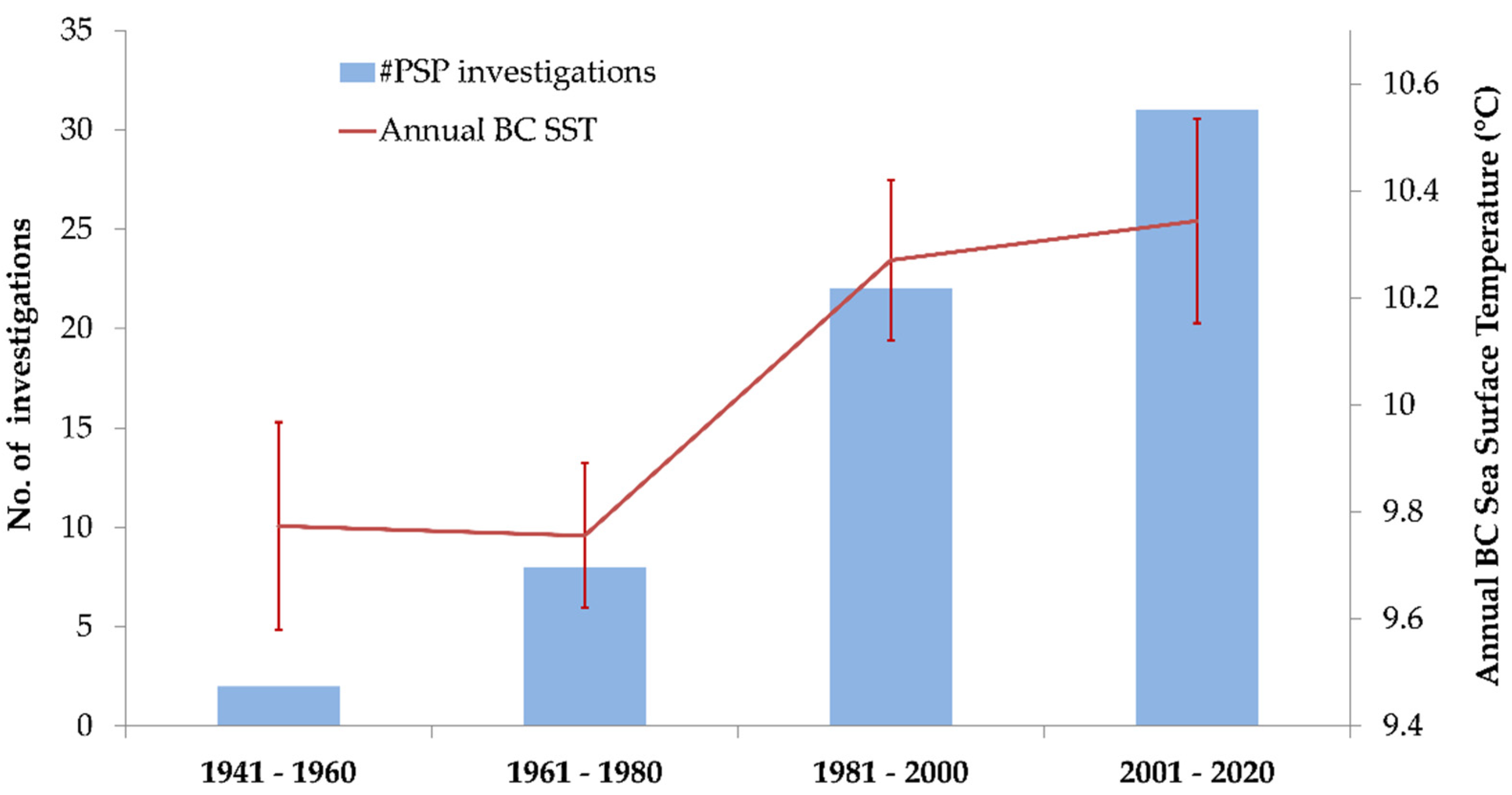
2.5. Seasonal Trends and Temperature
3. Discussion
4. Materials and Methods
4.1. Establishing a Historical Record of Paralytic Shellfish Poisonings in BC
4.2. Definitions for Paralytic Shellfish Poisoning Cases and Investigations
4.3. Temperature and Seasonal Trends
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, D.M.; Kulis, D.M.; Qi, Y.Z.; Zheng, L.; Lu, S.; Lin, Y.T. Paralytic shellfish poisoning in southern China. Toxicon 1996, 34, 579–590. [Google Scholar] [CrossRef]
- Azanza, R.V.; Taylor, F.J. Are Pyrodinium blooms in the Southeast Asian region recurring and spreading? A view at the end of the millennium. Ambio 2001, 30, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Gessner, B.D.; Middaugh, J.P. Paralytic shellfish poisoning in Alaska: A 20-year retrospective analysis. Am. J. Epidemiol. 1995, 141, 766–770. [Google Scholar] [CrossRef] [PubMed]
- La Barbera-Sánchez, A.; Franco Soler, J.; Rojas de Astudillo, L.; Chang-Yen, I. Paralytic shellfish poisoning (PSP) in Margarita Island, Venezuela. Rev. Biol. Trop. 2004, 52 (Suppl. 1), 89–98. [Google Scholar]
- Lewitus, A.J.; Horner, R.A.; Caron, D.A.; Garcia-Mendoza, E.; Hickey, B.M.; Hunter, M.; Huppert, D.D.; Kudela, R.M.; Langlois, G.W.; Largier, J.L.; et al. Harmful algal blooms along the North American west coast region: History, trends, causes, and impacts. Harmful Algae 2012, 19, 133–159. [Google Scholar] [CrossRef] [Green Version]
- Mee, L.D.; Espinosa, M.; Diaz, G. Paralytic shellfish poisoning with a Gymnodinium catenatum red tide on the Pacific coast of Mexico. Mar. Environ. Res. 1986, 19, 77–92. [Google Scholar] [CrossRef]
- Van Dolah, F.M. Marine algal toxins: Origins, health effects, and their increased occurrence. Environ. Health Perspect. 2000, 108 (Suppl. 1), 133–141. [Google Scholar] [CrossRef] [Green Version]
- Popkiss, M.; Horstman, D.; Harpur, D. Paralytic shellfish poisoning. A report of 17 cases in Cape Town. S. Afr. Med. J. 1979, 55, 1017–1023. [Google Scholar]
- Quayle, D.B. Paralytic Shellfish Poisoning in British Columbia, in Bulletin 168; Fisheries Research Board of Canada: Nanaimo, BC, Canada, 1969; p. 68. [Google Scholar]
- European Food Safety Authority. Marine biotoxins in shellfish—Saxitoxin group. EFSA J. 2009, 7, 1019. [Google Scholar] [CrossRef]
- Cusick, K.D.; Sayler, G.S. An overview on the marine neurotoxin, saxitoxin: Genetics, molecular targets, methods of detection and ecological functions. Mar. Drugs 2013, 11, 991–1018. [Google Scholar] [CrossRef] [Green Version]
- Deeds, J.R.; Landsberg, J.H.; Etheridge, S.M.; Pitcher, G.C.; Longan, S.W. Non-traditional vectors for paralytic shellfish poisoning. Mar. Drugs 2008, 6, 308–348. [Google Scholar] [CrossRef]
- Anderson, D.M. Approaches to monitoring, control and management of harmful algal blooms (HABs). Ocean Coast Manag. 2009, 52, 342. [Google Scholar] [CrossRef] [Green Version]
- Berdalet, E.; Fleming, L.E.; Gowen, R.; Davidson, K.; Hess, P.; Backer, L.C.; Moore, S.K.; Hoagland, P.; Enevoldsen, H. Marine harmful algal blooms, human health and wellbeing: Challenges and opportunities in the 21st century. J. Mar. Biol. Assoc. United Kingd. 2016, 96, 61–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lilly, E.; Kulis, D.; Gentien, P.; Anderson, D. Paralytic shellfish poisoning toxins in France linked to a human-introduced strain of Alexandrium catenella from the western Pacific: Evidence from DNAand toxin analysis. J. Plankton. Res. 2002, 24, 443–452. [Google Scholar] [CrossRef] [Green Version]
- Wells, M.L.; Trainer, V.L.; Smayda, T.J.; Karlson, B.S.; Trick, C.G.; Kudela, R.M.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.M.; et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 2015, 49, 68–93, . [Google Scholar] [CrossRef] [PubMed] [Green Version]
- U.S. Environmental Protection Agency. Impacts of Climate Change on the Occurrence of Harmful Algal Blooms; USEPA: Washington, DC, USA, 2013; 3p.
- The Intergovernmental Panel on Climate Change Special Report on the Ocean and Cryosphere in a Changing Climate. Available online: https://www.ipcc.ch/srocc/ (accessed on 10 December 2020).
- James, K.J.; Carey, B.; O’Halloran, J.; van Pelt, F.N.A.M.; Škrabáková, Z. Shellfish toxicity: Human health implications of marine algal toxins. Epidemiol. Infect. 2010, 138, 927–940. [Google Scholar] [CrossRef] [Green Version]
- Scriven, D.R.; DiBacco, C.; Locke, A.; Therriault, T.W. Ballast water management in Canada: A historical perspective and implications for the future. Mar. Policy 2015, 59, 121–133. [Google Scholar] [CrossRef]
- Vilarino, N.; Louzao, M.C.; Abal, P.; Cagide, E.; Carrera, C.; Vieytes, M.R.; Botana, L.M. Human poisoning from marine toxins: Unknowns for optimal consumer protection. Toxins 2018, 10, 324. [Google Scholar] [CrossRef] [Green Version]
- Etheridge, S.M. Paralytic shellfish poisoning: Seafood safety and human health perspectives. Toxicon 2010, 56, 108–122. [Google Scholar] [CrossRef] [Green Version]
- Bates, S.; Beach, D.; Comeau, L.; Haigh, N.; Lewis, N.; Locke, A.; Martin, J.; McCarron, P.; McKenzie, C.; Michel, C.; et al. Marine Harmful Algal Blooms and Phycotoxins of Concern to Canada; Canadian technical report of fisheries and aquatic sciences. No. 3384; Fisheries and Oceans Canada, Gulf Fisheries Centre: Moncton, NB, Canada, 2020.
- Sobel, J.; Painter, J. Illnesses caused by marine toxins. Clin. Infect. Dis. 2005, 41, 1290–1296. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. A review of harmful algal blooms and their apparent global increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef] [Green Version]
- Government of Canada Canadian Shellfish Sanitation Program Manual. Available online: https://www.inspection.gc.ca/food-safety-for-industry/food-specific-requirements-and-guidance/fish/canadian-shellfish-sanitation-program/eng/1527251566006/1527251566942?chap=0 (accessed on 10 December 2020).
- Government of Canada Health Canada’s Maximum Levels for Various Chemical Contaminants in Foods. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/chemical-contaminants/maximum-levels-chemical-contaminants-foods.html (accessed on 10 May 2021).
- Rees, G.; Pond, K.; Kay, D. Safe Management of Shellfish and Harvest Waters; IWA Publishing Alliance House for World Health Organization: London, UK, 2010. [Google Scholar]
- Government of Canada Reported Cases from 1924 to 2018 in Canada—Notifiable Diseases Online. Available online: https://diseases.canada.ca/notifiable/charts?c=pl (accessed on 10 December 2020).
- BC Centre for Disease Control Seafood-Related Illness (excluding Vibrio spp.) Case Report Form. Available online: http://www.bccdc.ca/resource-gallery/Documents/Guidelines%20and%20Forms/Forms/Epid/Enterics/Surveillence_form_shellfish_fish_Feb132014.pdf (accessed on 20 June 2021).
- Office of the Auditor General of British Columbia. An Audit of the Panorama Public Health System; Office of the Auditor General of British Columbia: Victoria, BC, Canada, 2015; 42p.
- WA Geoservices Washington Geospatial Open Data Portal, Salish Sea Basin Boundary. Available online: https://geo.wa.gov/datasets/c5f923f0930b4c3e8314e0636fb96401_0?geometry=-144.196%2C46.468%2C-102.382%2C51.508 (accessed on 12 June 2021).
- Finnis, S.; Krstic, N.; McIntyre, L.; Nelson, T.A.; Henderson, S.B. Spatiotemporal patterns of paralytic shellfish toxins and their relationships with environmental variables in British Columbia, Canada from 2002 to 2012. Environ. Res. 2017, 156, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Knaack, J.S.; Porter, K.A.; Jacob, J.T.; Sullivan, K.; Forester, M.; Wang, R.Y.; Trainer, V.L.; Morton, S.; Eckert, G.; McGahee, E.; et al. Case diagnosis and characterization of suspected paralytic shellfish poisoning in Alaska. Harmful Algae 2016, 57, 45–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinder, S.; Hays, G.; Brooks, C.; Davies, A.; Edwards, M.; Walne, A.; Gravenor, M. Toxic marine microalgae and shellfish poisoning in the British isles: History, review of epidemiology, and future implications. Environ. Health 2011, 10, 54. [Google Scholar] [CrossRef] [Green Version]
- Hurley, W.; Wolterstorff, C.; MacDonald, R.; Schultz, D. Paralytic shellfish poisoning: A case series. West. J. Emerg. Med. 2014, 15, 378–381. [Google Scholar] [CrossRef] [Green Version]
- Trainer, V.L.; Eberhart, B.-T.L.; Wekell, J.C.; Adams, N.G.; Hanson, L.; Cox, F.; Dowell, J. Paralytic shellfish toxins in Puget Sound, Washington state. J. Shellfish Res. 2003, 22, 213–223. [Google Scholar]
- Nishitani, L.; Chew, K. PSP toxins in the Pacific coast states: Monitoring programs and effects on bivalve industries. J. Shellfish Res. 1988, 7, 653–669. [Google Scholar]
- Fleming, L.E.; Broad, K.; Clement, A.; Dewailly, E.; Elmir, S.; Knap, A.; Pomponi, S.A.; Smith, S.; Solo Gabriele, H.; Walsh, P. Oceans and human health: Emerging public health risks in the marine environment. Mar. Pollut. Bull. 2006, 53, 545–560. [Google Scholar] [CrossRef] [Green Version]
- Statistics Canada Focus on Geography Series, 2016 Census. Available online: https://www12.statcan.gc.ca/census-recensement/2016/as-sa/fogs-spg/Facts-PR-Eng.cfm?TOPIC=9&LANG=Eng&GK=PR&GC=59 (accessed on 17 June 2021).
- Trainer, V.L.; Sullivan, K.; Le Eberhart, B.-T.; Shuler, A.; Hignutt, E.; Kiser, J.; Eckert, G.L.; Shumway, S.E.; Morton, S.L. Enhancing shellfish safety in Alaska through monitoring of harmful algae and their toxins. J. Shellfish Res. 2014, 33, 531–540. [Google Scholar] [CrossRef]
- Walton, P. Fish Store Fined $2000: Sea Drift Market Sold Red Tide Clams. Nanaimo Daily News, 2006; p. A3.
- Wilson, V. Shellfish harvester under investigation. Nanaimo Daily News, 2003; p. A.3.
- Van Egmond, H. Marine Biotoxins: In Food and Nutrition Paper; Food and Agricultural Organization of the United Nations (FAO): Rome, Italy, 2004. [Google Scholar]
- Canadian Food Inspection Agency. 20030803-Health Hazard Alert (Paralytic Shellfish Toxins)/Avertissement de Danger Pour lat Sante (Phycotoxines Paralysante); Canadian Food Inspection Agency: Ottawa, ON, Canada, 2003; p. 1.
- Rourke, W.A.; Justason, A.; Martin, J.L.; Murphy, C.J. Shellfish toxin uptake and depuration in multiple Atlantic Canadian molluscan species: Application to selection of sentinel species in monitoring programs. Toxins 2021, 13, 168. [Google Scholar] [CrossRef]
- Hallegraeff, G.M.; Anderson, D.M.; Cembella, A.D. (Eds.) Manual on Harmful Marine Microalgae, 2nd rev ed.; UNESCO: Paris, France, 2003; Volume 11, pp. 649–692. [Google Scholar]
- Trainer, V.L.; Hardy, F.J. Integrative monitoring of marine and freshwater harmful algae in Washington State for public health protection. Toxins 2015, 7, 1206–1234. [Google Scholar] [CrossRef] [PubMed]
- British Columbia Drug and Poison Information Centre (BC DPIC) about the BC Drug and Poison Information Centre. Available online: http://www.dpic.org/content/about-bc-drug-and-poison-information-centre (accessed on 1 April 2021).
- Loppie, S.; Reading, C.; de Leeuw, S. Indigenous Experiences with Racism and Its Impacts; National Collaborating Centre for Indigenous Health: Prince Geoge, BC, Canada, 2014; Available online: https://www.nccih.ca/495/Indigenous_experiences_with_racism_and_its_impacts.nccih?id=131 (accessed on 24 August 2021).
- Wan, V.; McIntyre, L.; Kent, D.; Leong, D.; Henderson, S.B. Near-real-time surveillance of illnesses related to shellfish consumption in British Columbia: Analysis of poison center data. JMIR Public Health Surveill. 2018, 4, e17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- BC Centre for Disease Control Paralytic shellfish poisoning. Available online: http://www.bccdc.ca/health-professionals/clinical-resources/case-definitions/paralytic-shellfish-poisoning (accessed on 10 December 2020).
- McKenzie, C.H.; Bates, S.S.; Martin, J.L.; Haigh, N.; Howland, K.L.; Lewis, N.I.; Locke, A.; Peña, A.; Poulin, M.; Rochon, A.; et al. Three decades of Canadian marine harmful algal events: Phytoplankton and phycotoxins of concern to human and ecosystem health. Harmful Algae 2021, 102, 101852. [Google Scholar] [CrossRef] [PubMed]
- Iwabuchi, B.L.; Gosselin, L.A. Long-term trends and regional variability in extreme temperature and salinity conditions experienced by coastal marine organisms on Vancouver Island, Canada. Bull. Mar. Sci. 2019, 95, 337–354. [Google Scholar] [CrossRef]
- Government of Canada British Columbia Lightstation Sea-Surface Temperature and Salinity Data (Pacific), 1914-Present. Available online: https://open.canada.ca/data/en/dataset/719955f2-bf8e-44f7-bc26-6bd623e82884 (accessed on 1 December 2020).
- Silva, M.; Rey, V.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Botana, A.; Botana, L.M.; Vasconcelos, V. Paralytic shellfish toxins occurrence in non-traditional invertebrate vectors from North Atlantic waters (Azores, Madeira, and Morocco). Toxins 2018, 10, 362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Sex | Age (yrs) | Onset (hrs) | Duration (hrs) | Symptoms | |
|---|---|---|---|---|---|
| Male 56.7% | Average 43.7 | Average 3.2 | Average 38.3 | Perioral tingling 74.1% and perioral numbness 20.6% | |
| Female 43.2% | Median 46 | Median 1.5 | Median 24 | Numbness 68.3% | |
| Range 8–91 | Range 0.08–18 | Range 0.5–252 | Extremities hands, finger, legs, and toes tingling 45.5%, and numbness 19.6% | ||
| Ataxia 42.9% | |||||
| Paralysis 18.0% | |||||
| Abdominal cramping 15.3% | |||||
| Floating, dizziness 14.3% | |||||
| Nausea 13.8% | |||||
| Vomiting 13.8% | |||||
| Weakness 9.5% | |||||
| Diarrhea 7.4% | |||||
| Headache 5.8% | |||||
| Difficulty breathing 4.2% | |||||
| Swelling in lips, tongue, or face 3.2% | |||||
| Dysphagia/dysarthria 2.6% | |||||
| Chest pain or rapid pulse 2.6% | |||||
| Hot, sweaty, and feverish 2.6% | |||||
| Ptosis 2.1% | |||||
| Loss of consciousness 1.6% | |||||
| Summarized from no. of cases | 111 | 61 | 97 | 102 | 189 |
| Self-Harvested | Commercial | |||
|---|---|---|---|---|
| Harvest Area Status | Confirmed | Probable | Confirmed | Probable |
| Open | 7 | 6 | 0 | 8 |
| Closed | 13 | 13 | 2 | 0 |
| Unknown | 2 | 3 | 0 | 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McIntyre, L.; Miller, A.; Kosatsky, T. Changing Trends in Paralytic Shellfish Poisonings Reflect Increasing Sea Surface Temperatures and Practices of Indigenous and Recreational Harvesters in British Columbia, Canada. Mar. Drugs 2021, 19, 568. https://doi.org/10.3390/md19100568
McIntyre L, Miller A, Kosatsky T. Changing Trends in Paralytic Shellfish Poisonings Reflect Increasing Sea Surface Temperatures and Practices of Indigenous and Recreational Harvesters in British Columbia, Canada. Marine Drugs. 2021; 19(10):568. https://doi.org/10.3390/md19100568
Chicago/Turabian StyleMcIntyre, Lorraine, Aroha Miller, and Tom Kosatsky. 2021. "Changing Trends in Paralytic Shellfish Poisonings Reflect Increasing Sea Surface Temperatures and Practices of Indigenous and Recreational Harvesters in British Columbia, Canada" Marine Drugs 19, no. 10: 568. https://doi.org/10.3390/md19100568
APA StyleMcIntyre, L., Miller, A., & Kosatsky, T. (2021). Changing Trends in Paralytic Shellfish Poisonings Reflect Increasing Sea Surface Temperatures and Practices of Indigenous and Recreational Harvesters in British Columbia, Canada. Marine Drugs, 19(10), 568. https://doi.org/10.3390/md19100568






