Exploitation of Filamentous and Picoplanktonic Cyanobacteria for Cosmetic Applications: Potential to Improve Skin Structure and Preserve Dermal Matrix Components
Abstract
1. Introduction
2. Results
2.1. Phytochemical Analysis
2.1.1. Carotenoids
2.1.2. Total Phenolic Content
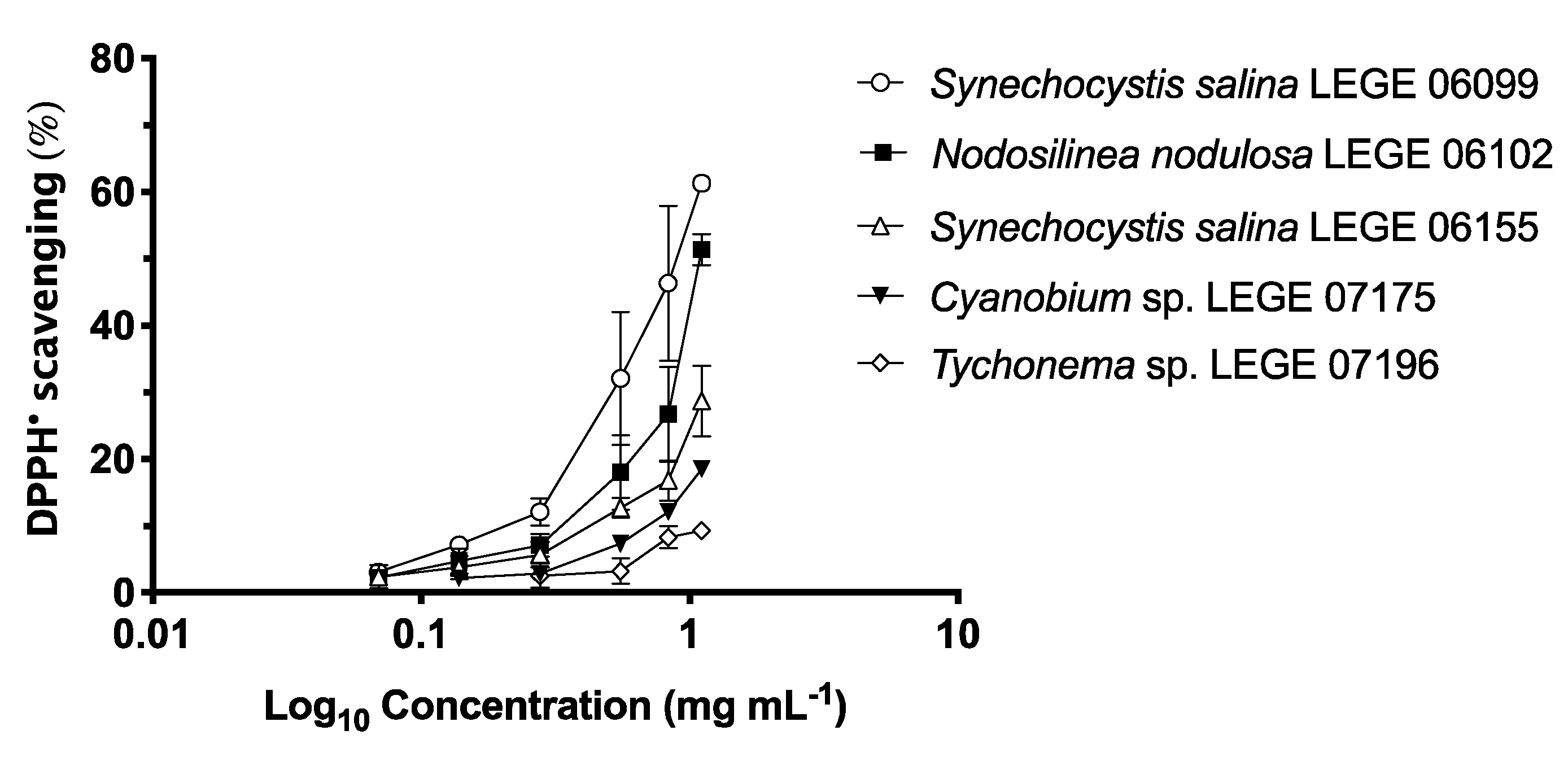
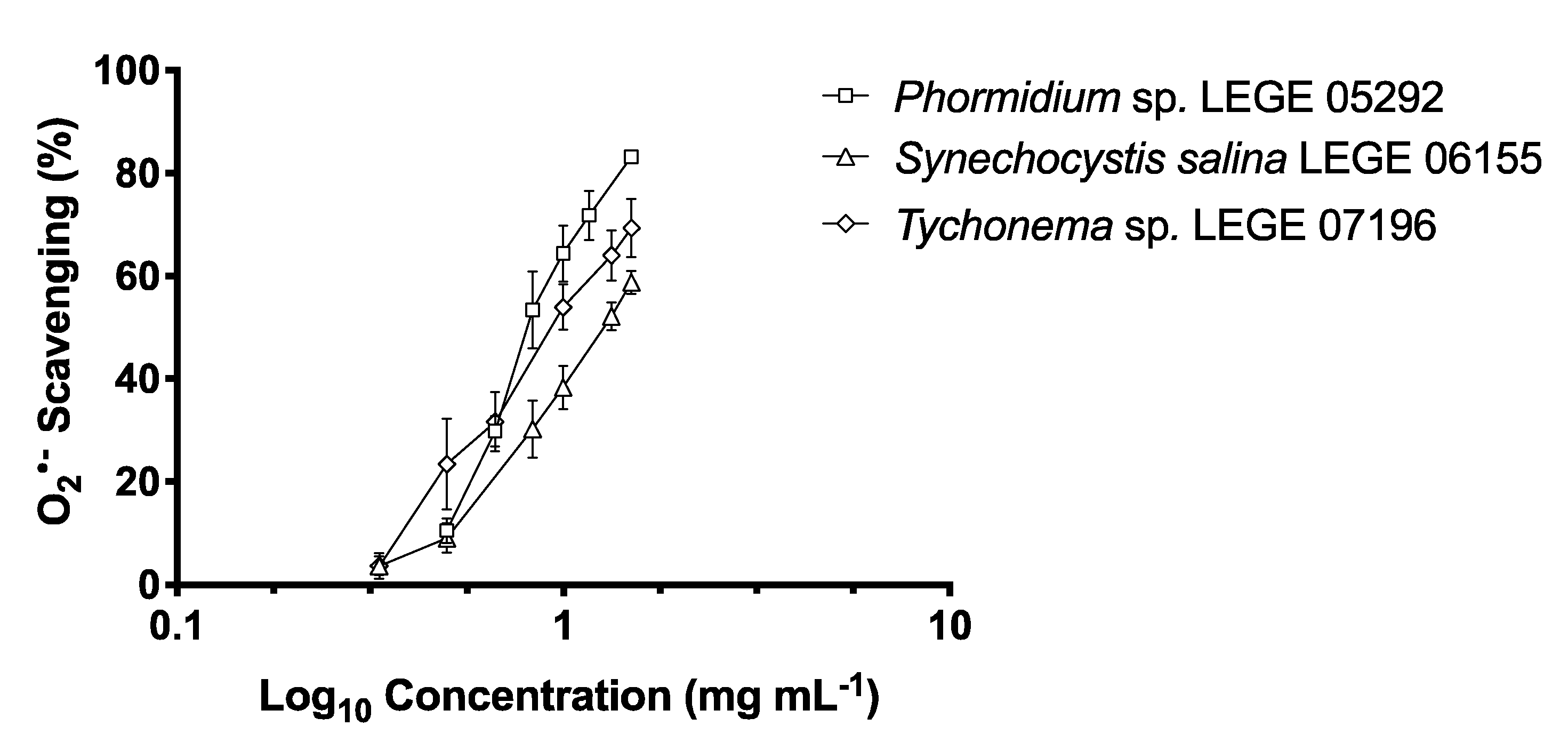
2.2. Antioxidant Activity
2.2.1. DPPH• (2,2-diphenyl-1-picrylhydrazyl) Scavenging Activity
2.2.2. O2•− Scavenging Activity
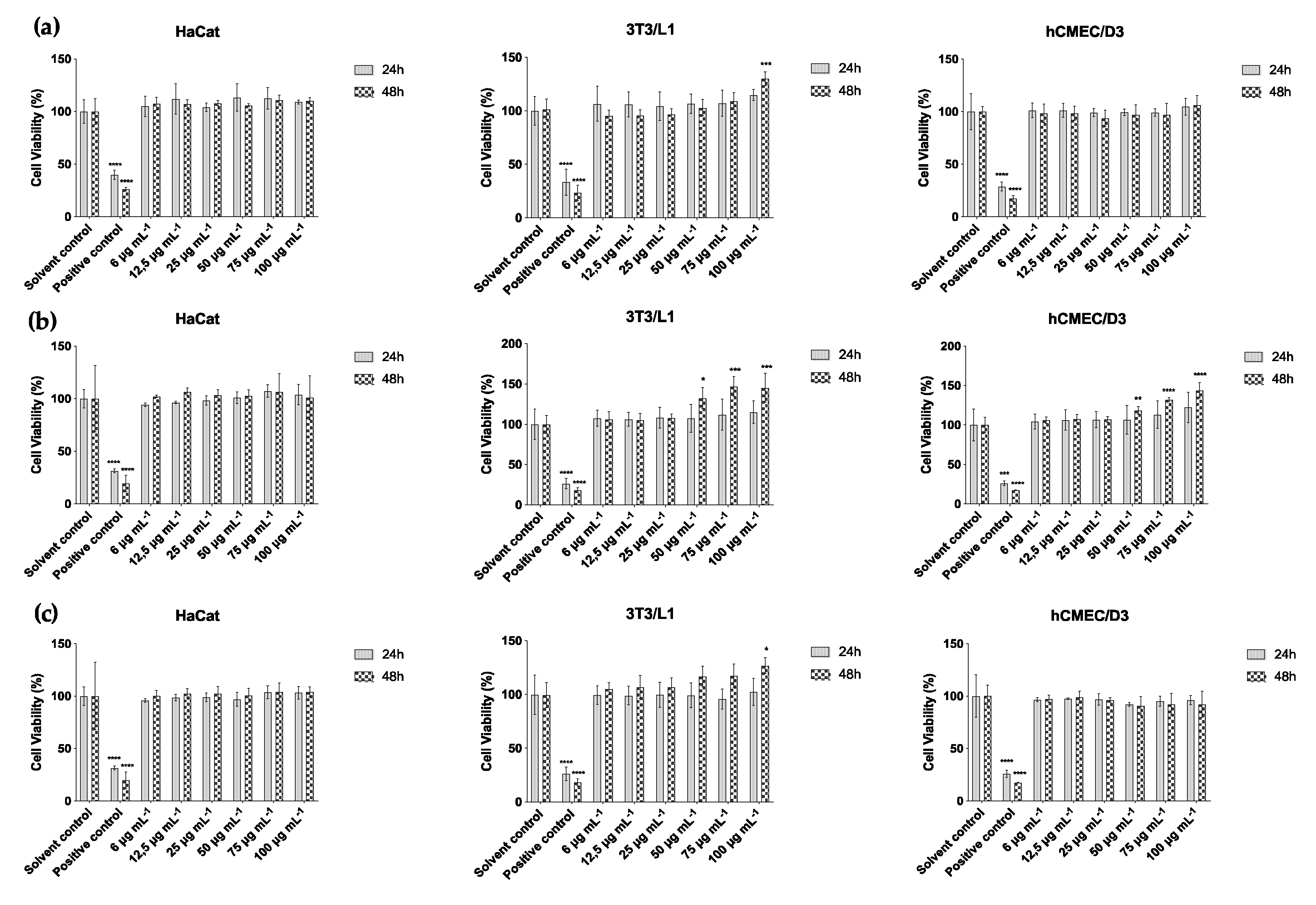
2.3. Cytotoxicity Assay
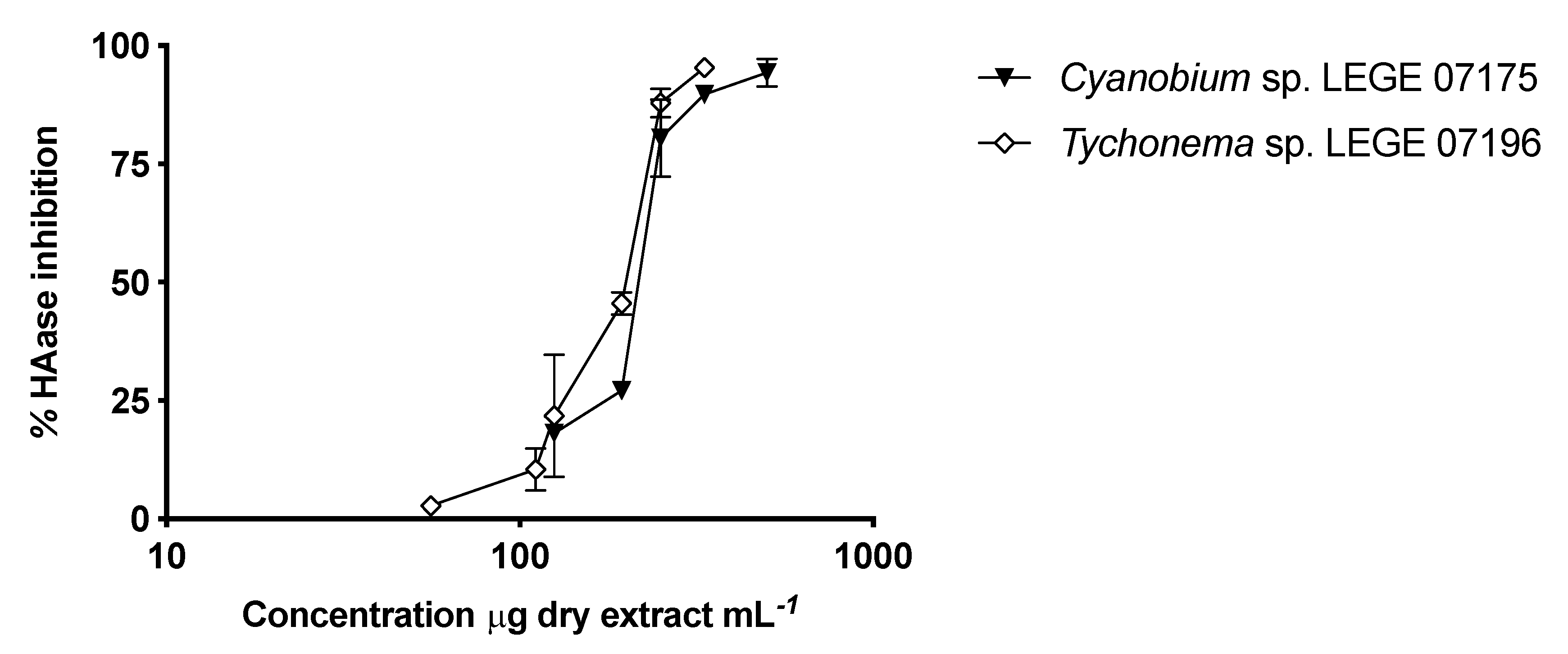
2.4. Hyaluronidase Inhibition
3. Discussion
4. Materials and Methods
4.1. Cyanobacteria Strains
4.2. Cyanobacteria Biomass: Culture and Harvest
4.3. Extract Preparation
4.4. Phytochemical Analysis
4.4.1. Determination of Pigments Profile by HPLC-PDA
4.4.2. Determination of Total Phenolic Content (TPC)
4.5. Antioxidant Assays
4.5.1. DPPH• Scavenging Activity
4.5.2. Superoxide Anion Radical (O2•−) Scavenging Activity
4.6. Cell Culture and Cytotoxicity Analysis
4.6.1. Cell Culture
4.6.2. Cytotoxicity Assay—MTT Assay
4.7. HAase Inhibition Assay
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Avenel-Audran, M.; Dutartre, H.; Goossens, A.; Jeanmougin, M.; Comte, C.; Bernier, C.; Benkalfate, L.; Michel, M.; Ferrier-Lebouedec, M.C.; Vigan, M.; et al. Octocrylene, an Emerging Photoallergen. Arch. Dermatol. 2010, 146, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Brausch, J.M.; Rand, G.M. A review of personal care products in the aquatic environment: Environmental concentrations and toxicity. Chemosphere 2011, 82, 1518–1532. [Google Scholar] [CrossRef] [PubMed]
- Mourelle, M.; Gómez, C.; Legido, J. The Potential Use of Marine Microalgae and Cyanobacteria in Cosmetics and Thalassotherapy. Cosmetics 2017, 4, 46. [Google Scholar] [CrossRef]
- Cavinato, M.; Waltenberger, B.; Baraldo, G.; Grade, C.V.C.; Stuppner, H.; Jansen-Durr, P. Plant extracts and natural compounds used against UVB-induced photoaging. Biogerontology 2017, 18, 499–516. [Google Scholar] [CrossRef]
- Ariede, M.B.; Candido, T.M.; Jacome, A.L.M.; Velasco, M.V.R.; de Carvalho, J.C.M.; Baby, A.R. Cosmetic attributes of algae—A review. Algal Res. 2017, 25, 483–487. [Google Scholar] [CrossRef]
- Morone, J.; Alfeus, A.; Vasconcelos, V.; Martins, R. Revealing the potential of cyanobacteria in cosmetics and cosmeceuticals—A new bioactive approach. Algal Res. 2019, 41, 101541. [Google Scholar] [CrossRef]
- Mazard, S.; Penesyan, A.; Ostrowski, M.; Paulsen, I.T.; Egan, S. Tiny Microbes with a Big Impact: The Role of Cyanobacteria and Their Metabolites in Shaping Our Future. Mar. Drugs 2016, 14, 97. [Google Scholar] [CrossRef]
- Derikvand, P.; Llewellyn, C.A.; Purton, S. Cyanobacterial metabolites as a source of sunscreens and moisturizers: A comparison with current synthetic compounds. Eur. J. Phycol. 2016, 52, 43–56. [Google Scholar] [CrossRef]
- Sathasivam, R.; Ki, J.S. A Review of the Biological Activities of Microalgal Carotenoids and Their Potential Use in Healthcare and Cosmetic Industries. Mar. Drugs 2018, 16, 26. [Google Scholar] [CrossRef]
- Lopes, G.; Clarinha, D.; Vasconcelos, V. Carotenoids from Cyanobacteria: A Biotechnological Approach for the Topical Treatment of Psoriasis. Microorganisms 2020, 8, 302. [Google Scholar] [CrossRef]
- Landau, M.; Fagien, S. Science of Hyaluronic Acid Beyond Filling: Fibroblasts and Their Response to the Extracellular Matrix. Plast. Reconstr. Surg. 2015, 136, 188S–195S. [Google Scholar] [CrossRef]
- Salwowska, N.M.; Bebenek, K.A.; Żądło, D.A.; Wcisło-Dziadecka, D.L. Physiochemical properties and application of hyaluronic acid: A systematic review. J. Cosmet. Dermatol. 2016, 15, 520–526. [Google Scholar] [CrossRef]
- Barlow, R.G.; Cummings, D.G.; Gibb, S.W. Improved resolution of mono- and divinyl chlorophylls a and b and zeaxanthin and lutein in phytoplankton extracts using reverse phase C-8 HPLC. Mar. Ecol. Prog Ser. 1997, 161, 303–307. [Google Scholar] [CrossRef]
- Barbosa, M.; Lopes, G.; Valentao, P.; Ferreres, F.; Gil-Izquierdo, A.; Pereira, D.M.; Andrade, P.B. Edible seaweeds’ phlorotannins in allergy: A natural multi-target approach. Food Chem. 2018, 265, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Langenkamp, E.; Molema, G. Microvascular endothelial cell heterogeneity: General concepts and pharmacological consequences for anti-angiogenic therapy of cancer. Cell Tissue Res. 2009, 335, 205–222. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R.; Deng, Z. Separation procedures for naturally occurring antioxidant phytochemicals. J. Chromatogr. B 2004, 812, 85–99. [Google Scholar] [CrossRef]
- Conde, F.R.; Churio, M.S.; Previtali, C.M. The photoprotector mechanism of mycosporine-like amino acids. Excited-state properties and photostability of porphyra-334 in aqueous solution. J. Photochem. Photobiol. B 2000, 56, 139–144. [Google Scholar] [CrossRef]
- Hashtroudi, M.S.; Shariatmadari, Z.; Riahi, H.; Ghassempour, A. Analysis of Anabaena vaginicola and Nostoc calcicola from Northern Iran, as rich sources of major carotenoids. Food Chem. 2013, 136, 1148–1153. [Google Scholar] [CrossRef]
- Amaro, H.M.; Fernandes, F.; Valentao, P.; Andrade, P.B.; Sousa-Pinto, I.; Malcata, F.X.; Guedes, A.C. Effect of Solvent System on Extractability of Lipidic Components of Scenedesmus obliquus (M2-1) and Gloeothece sp. on Antioxidant Scavenging Capacity Thereof. Mar. Drugs 2015, 13, 6453–6471. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu. Rev. Nutr. 2003, 23, 171–201. [Google Scholar] [CrossRef]
- Lee, E.H.; Faulhaber, D.; Hanson, K.M.; Ding, W.; Peters, S.; Kodali, S.; Granstein, R.D. Dietary lutein reduces ultraviolet radiation-induced inflammation and immunosuppression. J. Investig. Derm. 2004, 122, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M.; Cajigal, C. Lutein in patients with cataracts and age-related macular degeneration: A long-term supplementation study. J. Sci. Food Agric. 2001, 81, 904–909. [Google Scholar] [CrossRef]
- Punginelli, C.; Wilson, A.; Routaboul, J.M.; Kirilovsky, D. Influence of zeaxanthin and echinenone binding on the activity of the orange carotenoid protein. Biochim. Biophys. Acta 2009, 1787, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Kusama, Y.; Inoue, S.; Jimbo, H.; Takaichi, S.; Sonoike, K.; Hihara, Y.; Nishiyama, Y. Zeaxanthin and Echinenone Protect the Repair of Photosystem II from Inhibition by Singlet Oxygen in Synechocystis sp. PCC 6803. Plant. Cell Physiol. 2015, 56, 906–916. [Google Scholar] [CrossRef]
- Patias, L.D.; Fernandes, A.S.; Petry, F.C.; Mercadante, A.Z.; Jacob-Lopes, E.; Zepka, L.Q. Carotenoid profile of three microalgae/cyanobacteria species with peroxyl radical scavenger capacity. Food Res. Int. 2017, 100, 260–266. [Google Scholar] [CrossRef]
- Pagels, F.; Salvaterra, D.; Amaro, H.M.; Lopes, G.; Sousa-Pinto, I.; Vasconcelos, V.; Guedes, A.C. Bioactive potential of Cyanobium sp. pigment-rich extracts. J. Appl. Phycol. 2020, 1–10. [Google Scholar] [CrossRef]
- Pagels, F.; Lopes, G.; Vasconcelos, V.; Guedes, A.C. White and red LEDs as two-phase batch for cyanobacterial pigments production. Bioresour. Technol. 2020, 307, 123105. [Google Scholar] [CrossRef]
- Katsumura, N.; Okuno, M.; Onogi, N.; Moriwaki, H.; Muto, Y.; Kojima, S. Suppression of mouse skin papilloma by canthaxanthin and beta-carotene in vivo: Possibility of the regression of tumorigenesis by carotenoids without conversion to retinoic acid. Nutr. Cancer 1996, 26, 203–208. [Google Scholar] [CrossRef]
- Palozza, P.; Maggiano, N.; Calviello, G.; Lanza, P.; Piccioni, E.; Ranelletti, F.O.; Bartoli, G.M. Canthaxanthin induces apoptosis in human cancer cell lines. Carcinogenesis 1998, 19, 373–376. [Google Scholar] [CrossRef]
- Lanfer-Marquez, U.M.; Barros, R.M.C.; Sinnecker, P. Antioxidant activity of chlorophylls and their derivatives. Food Res. Int. 2005, 38, 885–891. [Google Scholar] [CrossRef]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Park, W.S.; Kim, H.J.; Li, M.; Lim, D.H.; Kim, J.; Kwak, S.S.; Kang, C.M.; Ferruzzi, M.G.; Ahn, M.J. Two Classes of Pigments, Carotenoids and C-Phycocyanin, in Spirulina Powder and Their Antioxidant Activities. Molecules 2018, 23, 2065. [Google Scholar] [CrossRef] [PubMed]
- Kent, M.; Welladsen, H.M.; Mangott, A.; Li, Y. Nutritional evaluation of Australian microalgae as potential human health supplements. PLoS ONE 2015, 10, e0118985. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.P.; Prabha, R.; Verma, S.; Meena, K.K.; Yandigeri, M. Antioxidant properties and polyphenolic content in terrestrial cyanobacteria. 3 Biotech 2017, 7, 134. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cheng, K.; Wong, C.; Fan, K.; Chen, F.; Jiang, Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem. 2007, 102, 771–776. [Google Scholar] [CrossRef]
- Ijaz, S.; Hasnain, S. Antioxidant potential of indigenous cyanobacterial strains in relation with their phenolic and flavonoid contents. Nat. Prod. Res. 2016, 30, 1297–1300. [Google Scholar] [CrossRef]
- Hossain, M.F.; Ratnayake, R.R.; Meerajini, K.; Wasantha Kumara, K.L. Antioxidant properties in some selected cyanobacteria isolated from fresh water bodies of Sri Lanka. Food Sci. Nutr. 2016, 4, 753–758. [Google Scholar] [CrossRef]
- Pumas, C.; Vacharapiyasophon, P.; Peerapornpisal, Y.; Leelapornpisid, P.; Boonchum, W.; Ishii, M.; Khanongnuch, C. Thermostablility of phycobiliproteins and antioxidant activity from four thermotolerant cyanobacteria. Phycol. Res. 2011, 59, 166–174. [Google Scholar] [CrossRef]
- Singh, S.; Singh, R.P. In VitroMethods of Assay of Antioxidants: An Overview. Food Rev. Int. 2008, 24, 392–415. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Babaoglu Aydas, S.; Ozturk, S.; Aslim, B. Phenylalanine ammonia lyase (PAL) enzyme activity and antioxidant properties of some cyanobacteria isolates. Food Chem. 2013, 136, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Cebeci, F.; Sahin-Yesilcubuk, N. The matrix effect of blueberry, oat meal and milk on polyphenols, antioxidant activity and potential bioavailability. Int. J. Food Sci. Nutr. 2014, 65, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Babić, O.; Kovač, D.; Rašeta, M.; Šibul, F.; Svirčev, Z.; Simeunović, J. Evaluation of antioxidant activity and phenolic profile of filamentous terrestrial cyanobacterial strains isolated from forest ecosystem. J. Appl. Phycol. 2016, 28, 2333–2342. [Google Scholar] [CrossRef]
- Parwani, L.; Bhatnagar, M.; Bhatnagar, A.; Sharma, V. Antioxidant and iron-chelating activities of cyanobacterial exopolymers with potential for wound healing. J. Appl. Phycol. 2013, 26, 1473–1482. [Google Scholar] [CrossRef]
- Iglesias-de la Cruz, M.C.; Sanz-Rodríguez, F.; Zamarrón, A.; Reyes, E.; Carrasco, E.; González, S.; Juarranz, A. A secretion of the mollusc Cryptomphalus aspersa promotes proliferation, migration and survival of keratinocytes and dermal fibroblasts in vitro. Int. J. Cosmet. Sci. 2012, 34, 183–189. [Google Scholar] [CrossRef]
- Frantz, C.; Stewart, K.M.; Weaver, V.M. The extracellular matrix at a glance. J. Cell Sci 2010, 123, 4195–4200. [Google Scholar] [CrossRef]
- Puizina-Ivic, N. Skin aging. Acta Derm. Alp. Pannonica Adriat 2008, 17, 47–54. [Google Scholar]
- Bassino, E.; Antoniotti, S.; Gasparri, F.; Munaron, L. Effects of flavonoid derivatives on human microvascular endothelial cells. Nat. Prod. Res. 2016, 30, 2831–2834. [Google Scholar] [CrossRef]
- Naoki, F.; Setsuko, S.; Yuji, Y.; Takenaka, H. Inhibitory effects of microalgae on the activation of hyaluronidase. J. Appl. Phycol. 2001, 13, 489–492. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Koketsu, M. Isolation and analysis of polysaccharide showing high hyaluronidase inhibitory activity in Nostochopsis lobatus MAC0804NAN. J. Biosci. Bioeng. 2016, 121, 345–348. [Google Scholar] [CrossRef]
- Singh, R.; Parihar, P.; Singh, M.; Bajguz, A.; Kumar, J.; Singh, S.; Singh, V.P.; Prasad, S.M. Uncovering Potential Applications of Cyanobacteria and Algal Metabolites in Biology, Agriculture and Medicine: Current Status and Future Prospects. Front. Microbiol. 2017, 8, 515. [Google Scholar] [CrossRef]
- Ramos, V.; Morais, J.; Castelo-Branco, R.; Pinheiro, A.; Martins, J.; Regueiras, A.; Pereira, A.L.; Lopes, V.R.; Frazao, B.; Gomes, D.; et al. Cyanobacterial diversity held in microbial biological resource centers as a biotechnological asset: The case study of the newly established LEGE culture collection. J. Appl. Phycol. 2018, 30, 1437–1451. [Google Scholar] [CrossRef]
- Costa, M.; Garcia, M.; Costa-Rodrigues, J.; Costa, M.S.; Ribeiro, M.J.; Fernandes, M.H.; Barros, P.; Barreiro, A.; Vasconcelos, V.; Martins, R. Exploring bioactive properties of marine cyanobacteria isolated from the Portuguese coast: High potential as a source of anticancer compounds. Mar. Drugs 2013, 12, 98–114. [Google Scholar] [CrossRef]
- Kotai, J. Instructions for Preparation of Modified Nutrients Solution Z8 for Algae. Nor. Inst. Water Res. 1972, 11, 5. [Google Scholar]
- Freitas, S.; Martins, R.; Campos, A.; Azevedo, J.; Osorio, H.; Costa, M.; Barros, P.; Vasconcelos, V.; Urbatzka, R. Insights into the potential of picoplanktonic marine cyanobacteria strains for cancer therapies–Cytotoxic mechanisms against the RKO colon cancer cell line. Toxicon 2016, 119, 140–151. [Google Scholar] [CrossRef]
- Barroso, M.F.; Ramalhosa, M.J.; Alves, R.C.; Dias, A.; Soares, C.M.D.; Oliva-Teles, M.T.; Delerue-Matos, C. Total antioxidant capacity of plant infusions: Assessment using electrochemical DNA-based biosensor and spectrophotometric methods. Food Control. 2016, 68, 153–161. [Google Scholar] [CrossRef]
- Sanchez-Moreno, C.; Larrauri, J.A.; Saura-Calixto, F. A Procedure to Measure the Antiradical Efficiency of Polyphenols. J. Sci. Food Agric. 1998, 76, 270–276. [Google Scholar] [CrossRef]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-diphenyl-1-picrylhydrazyl free radical (DPPH) assay for antioxidant activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef]
- Ferreres, F.; Lopes, G.; Gil-Izquierdo, A.; Andrade, P.B.; Sousa, C.; Mouga, T.; Valentao, P. Phlorotannin extracts from fucales characterized by HPLC-DAD-ESI-MSn: Approaches to hyaluronidase inhibitory capacity and antioxidant properties. Mar. Drugs 2012, 10, 2766–2781. [Google Scholar] [CrossRef] [PubMed]
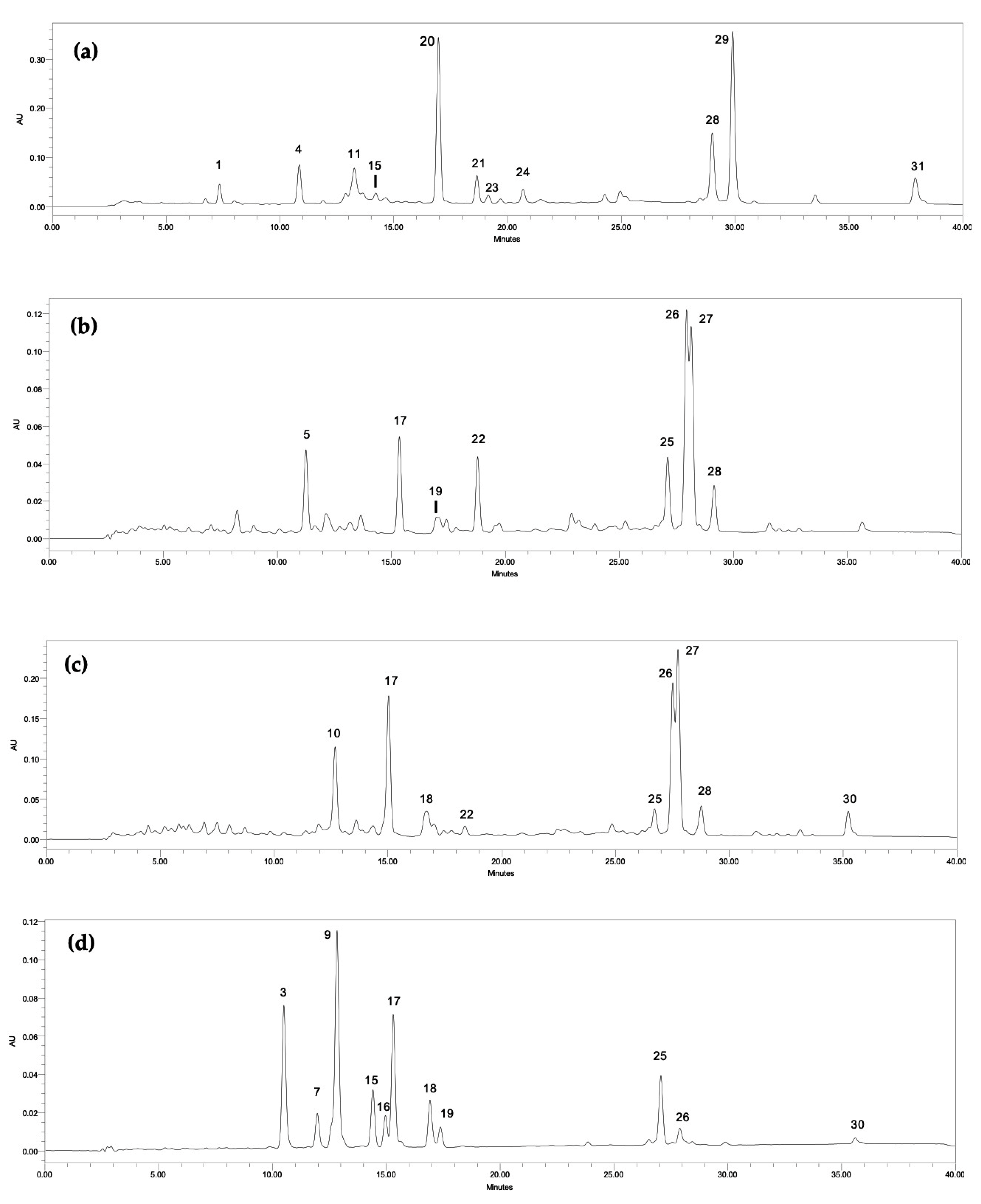
| Peak | Compound | RT (min) | Phormidium sp. LEGE 05292 | Synechocystis salina LEGE 06099 | Nodosilinea nodulosa LEGE 06102 | Cyanobium sp. LEGE 06113 | Synechocystis salina LEGE 06155 | Cyanobium sp. LEGE 07175 | Tychonema sp. LEGE 07196 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Unidentified Chlorophyll | 7.36 | 383.04 ± 44.56 | nd | nd | nd | nd | nd | nd |
| 2 | Unidentified carotenoid | 9.71 | nd | nd | 21.95 ± 0.17 a | nd | nd | nd | nd |
| 3 | β-Carotene oxygenated derivative | 10.35 | nd | nd | 12.58 ± 0.16 c | 20.79 ± 0.14 a | nd | 16.83 ± ≤ 0.08 b | nd |
| 4 | Unidentified carotenoid | 10.85 | 6.73 ± ≤ 0.04 b | 13.26 ± 0.42 a | nd | nd | nd | nd | nd |
| 5 | Unidentified carotenoid | 11.27 | nd | nd | nd | 8.13 ± ≤ 0.07 a | nd | nd | 7.63 ± 0.21 b |
| 6 | Unidentified carotenoid | 11.59 | nd | nd | 13.80 ± ≤ 0.02 | nd | nd | nd | nd |
| 7 | Lutein derivative | 12.02 | nd | nd | nd | 18.02 ± 0.31 a | nd | 14.32 ± ≤ 0.07 b | nd |
| 8 | Unidentified carotenoid | 12.40 | nd | nd | 63.07 ± 0.23 | nd | nd | nd | nd |
| 9 | β-Carotene oxygenated derivative | 12.51 | nd | nd | nd | 38.95 ± 0.41 a | nd | 25.53 ± 0.11 b | nd |
| 10 | Unidentified carotenoid | 12.76 | nd | 118.17 ± 1.04 a | nd | nd | 14.62 ± 0.89 b | nd | nd |
| 11 | Unidentified carotenoid | 13.25 | 8.20 ± 0.54 | nd | nd | nd | nd | nd | nd |
| 12 | Unidentified carotenoid | 13.67 | nd | 17.01 ± 0.15 a | 12.29 ± 0.14 b | nd | nd | nd | nd |
| 13 | Lutein derivative | 13.91 | nd | nd | 43.73 ± 0.84 a | 29.59 ± 0.16 b | nd | nd | nd |
| 14 | Unidentified carotenoid | 14.44 | nd | 39.18 ± 1.43 a | 12.05 ± 0.30 b | nd | nd | nd | nd |
| 15 | Lutein derivative | 14.72 | 8.47 ± 0.58 b | nd | nd | nd | nd | 21.54 ± 0.29 a | nd |
| 16 | Lutein derivative | 15.01 | nd | nd | nd | nd | nd | 12.66 ± 0.65 | nd |
| 17 | Zeaxanthin | 15.36 | nd | 49.82 ± 1.36 a | 39.41 ± 0.06 b | 25.93 ± 0.22 c | 19.93 ± 0.16 d | 16.31 ± 0.23 e | 7.97 ± ≤ 0.06 f |
| 18 | Lutein | 16.33 | nd | 79.08 ± 0.44 a | 50.25 ± 0.80 b | 23.38 ± 0.20 c | 18.94 ± ≤ 0.06 d | 19.91 ± 0.55 d | |
| 19 | Lutein derivative | 16.78 | nd | 19.02 ± ≤ 0.07 a | 15.26 ± 0.31 b | 8.39 ± 0.18 d | 9.70 ± ≤ 0.08 c | 7.54 ± ≤ 0.06 e | |
| 20 | β-Carotene oxygenated derivative | 16.97 | 23.48 ± 0.28 | nd | nd | nd | nd | nd | nd |
| 21 | Lutein derivative | 18.66 | 14.83 ± 0.11 | nd | nd | nd | nd | nd | nd |
| 22 | Canthaxanthin | 18.81 | nd | nd | nd | nd | 9.96 ± ≤ 0.07 b | nd | 37.30 ± 0.68 a |
| 23 | Lutein derivative | 19.16 | 5.35 ± 0.13 | nd | nd | nd | nd | nd | nd |
| 24 | Unidentified carotenoid | 20.70 | 3.55 ± ≤ 0.03 | nd | nd | nd | nd | nd | nd |
| 25 | Chlorophyll a derivative | 26.73 | nd | 634.71 ± 3.18 e | 1425.40 ± 6.25 b | 1796.97 ± 6.44 a | 557.64 ± 0.95 f | 1050.51± 12.98 c | 742.71 ± 17.82 d |
| 26 | Echinenone | 27.54 | nd | 48.37 ± 0.45 c | 46.27 ± 0.53 d | nd | 76.02 ± 0.70 a | 9.48 ± 0.17 e | 58.25 ± 0.35 b |
| 27 | Chlorophyll a derivative | 28.17 | nd | nd | nd | nd | 3588.41 ± 74.03 a | nd | 1826.25 ± 57.00 b |
| 28 | Chlorophyll-a | 29.01 | 1741.99 ± 68.68 a | nd | nd | nd | 616.85 ± 4.04 b | nd | 456.56 ± 3.29 c |
| 29 | Echinenone derivative | 29.91 | 105.81 ± 0.87 | nd | nd | nd | nd | nd | nd |
| 30 | β-Carotene | 35.06 | nd | 40.76 ± ≤ 0.09 a | 15.66 ± 0.21 c | 22.96 ± 0.93 b | 8.06 ± 0.21 d | nd | |
| 31 | β-Carotene derivative | 37.94 | 39.46 ± 0.94 | nd | nd | nd | nd | nd | nd |
| Total carotenoids | 215.88 ± 4.64 c | 383.89 ± 3.54 a | 371.43 ± 22.12 b | 188.84 ± 0.44 d | 162.43 ± 1.29 e | 154.33 ±1.68 d,e | 118.69 ± 1.07 f | ||
| Total chlorophylls | 2125.04 ± 65.01 c | 634.71 ± 3.18 g | 1425.40 ± 6.25 e | 1796.97 ± 6.44 d | 4762.90 ± 73.72 a | 1050.51 ± 12.98 f | 3025.52 ± 46.73 b | ||
| Strains | mg GAE g−1 |
|---|---|
| Phormidium sp. LEGE 05292 | 1.52 ± 0.03 b |
| Synechocystis salina LEGE 06099 | 2.45 ± 0.13 a |
| Nodosilinea nodulosa LEGE 06102 | 1.23 ± 0,00 b,c,d |
| Cyanobium sp. LEGE 06113 | 1.41 ± 0.05 b,c |
| Synechocystis salina LEGE 06155 | 1.18 ± 0.05 c,d,e |
| Cyanobium sp. LEGE 07175 | 1.09 ± 0.14 d,e |
| Tychonema sp. LEGE 07196 | 1.07 ± 0.04 d,e |
| Strains | DPPH• (μg mL−1) | O2•− (μg mL−1) | ||
|---|---|---|---|---|
| IC25 | IC50 | IC25 | IC50 | |
| Phormidium sp. LEGE 05292 | nd | nd | 626.54 ± 0.02 a,b | 822.70 ± 0.06 b |
| Synechocystis salina LEGE 06099 | 481.96 ± 0.09 b | 863.82 ± 0.17 | nd | nd |
| Nodosilinea nodulosa LEGE 06102 | 764.14 ± 0.16 a,b | 1077.59 ± 0.03 | nd | nd |
| Cyanobium sp. LEGE 06113 | nd | nd | nd | nd |
| Synechocystis salina LEGE 06155 | 929.76 ± 0.12 a | nd | 756.42 ± 0.74 a | 1275.86 ± 0.07 a |
| Cyanobium sp. LEGE 07175 | nd | nd | nd | nd |
| Tychonema sp. LEGE 07196 | nd | nd | 555.54 ± 0.09 b | 924.21 ± 0.07 b |
| Standards | Calibration Curve | r2 | LOD (µg/mL) a | LOQ (µg/mL) b |
|---|---|---|---|---|
| Zeaxanthin | y = 1040515632x − 285181 | 0.9981 | 0.0072 | 0.0239 |
| Lutein | y = 273528935x − 49102 | 0.9988 | 0.0921 | 0.3070 |
| Canthaxanthin | y = 12144399x − 12640 | 0.9992 | 0.3251 | 1.0840 |
| Echinenone | y = 227303816x − 19677 | 0.9998 | 0.0935 | 0.3312 |
| Chlorophyll-a | y = 6636898x − 6835 | 0.9993 | 0.5143 | 1.7140 |
| β-Carotene | y = 140882609x − 43144 | 0.9988 | 0.0323 | 0.1080 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morone, J.; Lopes, G.; Preto, M.; Vasconcelos, V.; Martins, R. Exploitation of Filamentous and Picoplanktonic Cyanobacteria for Cosmetic Applications: Potential to Improve Skin Structure and Preserve Dermal Matrix Components. Mar. Drugs 2020, 18, 486. https://doi.org/10.3390/md18090486
Morone J, Lopes G, Preto M, Vasconcelos V, Martins R. Exploitation of Filamentous and Picoplanktonic Cyanobacteria for Cosmetic Applications: Potential to Improve Skin Structure and Preserve Dermal Matrix Components. Marine Drugs. 2020; 18(9):486. https://doi.org/10.3390/md18090486
Chicago/Turabian StyleMorone, Janaína, Graciliana Lopes, Marco Preto, Vítor Vasconcelos, and Rosário Martins. 2020. "Exploitation of Filamentous and Picoplanktonic Cyanobacteria for Cosmetic Applications: Potential to Improve Skin Structure and Preserve Dermal Matrix Components" Marine Drugs 18, no. 9: 486. https://doi.org/10.3390/md18090486
APA StyleMorone, J., Lopes, G., Preto, M., Vasconcelos, V., & Martins, R. (2020). Exploitation of Filamentous and Picoplanktonic Cyanobacteria for Cosmetic Applications: Potential to Improve Skin Structure and Preserve Dermal Matrix Components. Marine Drugs, 18(9), 486. https://doi.org/10.3390/md18090486









