The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo
Abstract
1. Introduction
2. Results
2.1. The Cytotoxicity and In Vitro Antiviral Activity of Fucoidans against DNA and RNA Viruses
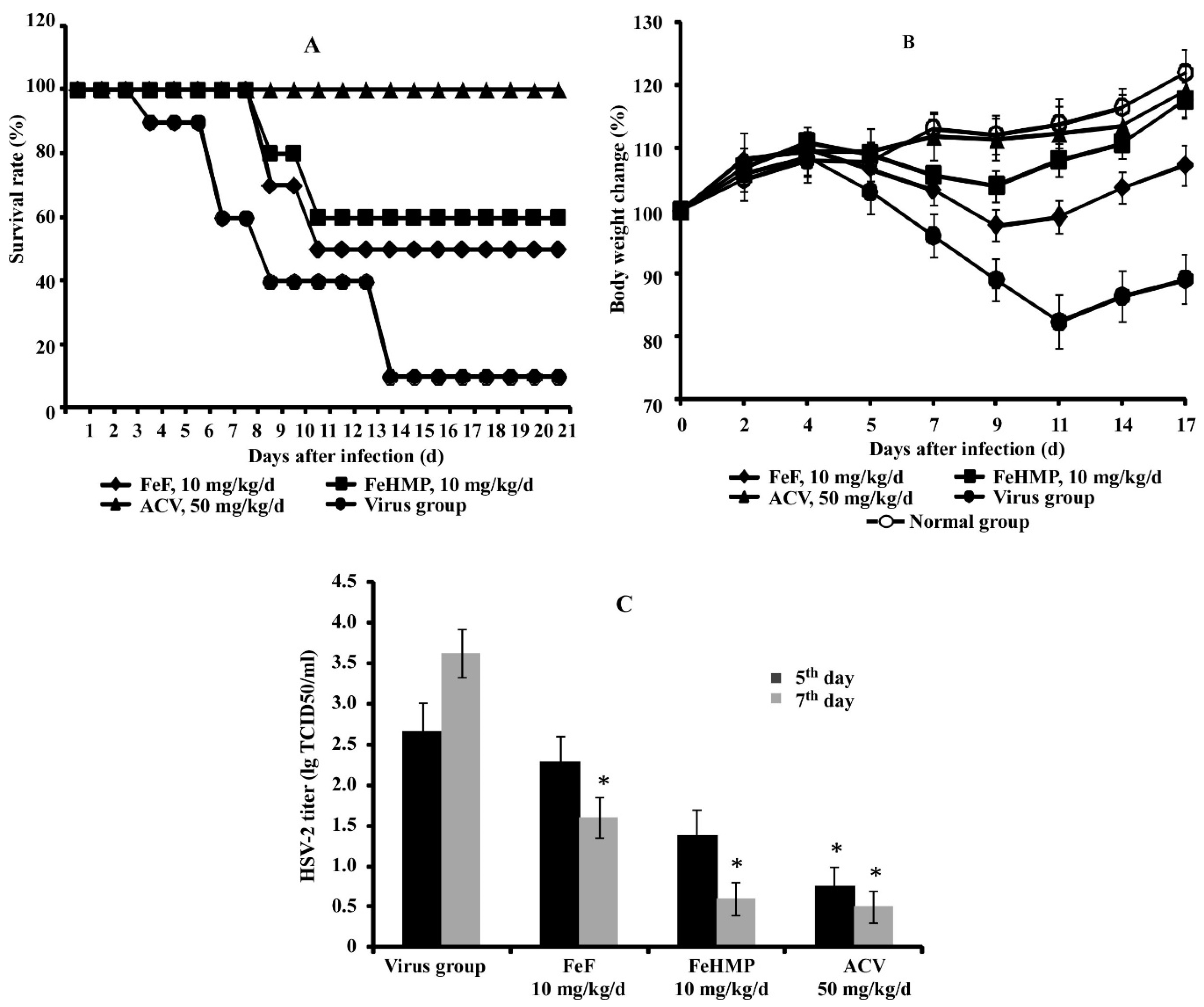
2.2. In Vivo Efficacy of Fucoidans against HSV-2 Infection in a Mouse Vaginitis Model
3. Discussion
4. Materials and Methods
4.1. Viruses and Cell Cultures
4.2. Animals
4.3. Studied Compounds
4.4. Cytotoxicity Assay of the Fucoidans
4.5. Antiviral Activity Assay of Fucoidans In Vitro
4.6. Antiviral Activity Assay of Fucoidans against HSV-2 In Vivo
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Usov, A.I.; Bilan, M.I. Fucoidans-sulfated polysaccharides of brown algae. Success Chem. 2009, 78, 846–862. [Google Scholar] [CrossRef]
- Rabanal, M.; Ponce, N.M.A.; Navarro, D.A.; Gómez, R.M.; Stortz, C.A. The system of fucoidans from the brown seaweed Dictyota dichotoma: Chemical analysis and antiviral activity. Carbohydr. Polym. 2014, 101, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xing, M.; Cao, Q.; Ji, A.; Liang, H.; Song, S. Biological Activities of Fucoidan and the Factors Mediating Its Therapeutic Effects: A Review of Recent Studies. Mar. Drugs 2019, 17, 183. [Google Scholar] [CrossRef]
- Sanina, N. Vaccine Adjuvants Derived from Marine Organisms. Biomolecules 2019, 9, 340. [Google Scholar] [CrossRef]
- Cumashi, A.; Ushakova, N.A.; Preobrazhenskaya, M.E.; D’Incecco, A.; Piccoli, A.; Totani, L. A comparative study of the anti-inflammatory, anticoagulant, antiangiogenic, and antiadhesive activities of nine different fucoidans from brown seaweeds. Glycobiology 2007, 17, 541–552. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.J.; Zhao, R.X. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671. [Google Scholar] [CrossRef]
- Imbs, T.I.; Krasovskaya, N.P.; Ermakova, S.P.; Makarieva, T.N.; Shevchenko, N.M.; Zvyagintseva, T.N. Comparative study of chemical composition and antitumor activity of aqueous-ethanol extracts of brown algae Laminaria cichorioides, Costaria costata, and Fucus evanescens. Russ. J. Mar. Biol. 2009, 35, 164–170. [Google Scholar] [CrossRef]
- Ale, M.T.; Mikkelsen, J.D.; Meyer, A.S. Important determinants for fucoidan bioactivity: A critical review of structure-function relations and extraction methods for fucose-containing sulfated polysaccharides from brown seaweeds. Mar. Drugs 2011, 9, 2106–2130. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Faustova, N.M.; Obluchinskaya, E.D.; Kosman, V.M.; Vuorela, H.; Makarov, V.G. Pharmacokinetic and tissue distribution of fucoidan from Fucus vesiculosus after oral administration to rats. Mar. Drugs 2018, 16, 132. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Obluchinskaya, E.D.; Vuorela, H. The pharmacokinetics of fucoidan after topical application to rats. Mar. Drugs 2019, 17, 687. [Google Scholar] [CrossRef] [PubMed]
- Sagawa, T.I.H.; Kato, I. Fucoidan as functional foodstuff. Structure and biological potency. Japan J. Phycol. (Sorui) 2003, 51, 19–25. [Google Scholar]
- Sezer, A.D.; Akbuğa, J. Fucosphere - new microsphere carriers for peptide and protein delivery: Preparation and in vitro characterization. J. Microencapsul. 2006, 23, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Manivasagan, P.; Hoang, G.; Santha Moorthy, M.; Mondal, S.; Minh Doan, V.H.; Kim, H.; Vy Phan, T.T.; Nguyen, T.P.; Oh, J. Chitosan/fucoidan multilayer coating of gold nanorods as highly efficient near-infrared photothermal agents for cancer therapy. Carbohydr. Polym. 2019, 211, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M. Pilot clinical study to evaluate the anticoagulant activity of fucoidan. Blood Coagul. Fibrinolysis. 2009, 20, 607–610. [Google Scholar] [CrossRef]
- Fitton, J.H.; Stringer, D.N.; Park, A.Y.; Karpiniec, S.S. Therapies from fucoidan: New developments. Mar. Drugs 2019, 17, 571. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kusaykin, M.I.; Kurilenko, V.V.; Zakharenko, A.M.; Isakov, V.V.; Zaporozhets, T.S. Hydrolysis of fucoidan by fucoidanase isolated from the marine bacterium, Formosa algae. Mar. Drugs 2013, 11, 2413–2430. [Google Scholar] [CrossRef]
- Kusaykin, M.I.; Silchenko, A.S.; Zakharenko, A.M.; Zvyagintseva, T.N. Fucoidanases. Glycobiology 2016, 26, 3–12. [Google Scholar]
- Silchenko, A.S.; Rasin, A.B.; Kusaykin, M.I.; Malyarenko, O.S.; Shevchenko, N.M.; Zueva, A.O.; Kalinovsky, A.I.; Zvyagintseva, T.N.; Ermakova, S.P. Modification of native fucoidan from Fucus evanescens by recombinant fucoidanase from marine bacteria Formosa algae. Carbohydr. Polym. 2018, 193, 189–195. [Google Scholar] [CrossRef]
- Zvyagintseva, T.N.; Shevchenko, N.M.; Nazarova, I.V.; Scobun, A.S.; Luk’yanov, P.A.; Elyakova, L.A. Water-soluble polysaccharides of some far-eastern brown seaweeds. Distribution, structure, and their dependence on the developmental conditions. J. Exp. Mar. Biol. Ecol. 2003, 294, 1–13. [Google Scholar] [CrossRef]
- Anastyuk, S.D.; Shevchenko, N.M.; Dmitrenok, P.S.; Zvyagintseva, T.N. Structural similarities of fucoidans from brown algae Silvetia babingtonii and Fucus evanescens, determined by tandem MALDI-TOF mass spectrometry. Carbohydr. Res. 2012, 358, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Menshova, R.V.; Shevchenko, N.M.; Imbs, T.I.; Zvyagintseva, T.N.; Malyarenko, O.S.; Zaporoshets, T.S.; Besednova, N.N.; Ermakova, S.P. Fucoidans from brown alga Fucus evanescens: Structure and biological activity. Front Mar. Sci. 2016, 3, 129. [Google Scholar] [CrossRef]
- Makarenkova, I.D.; Krylova, N.V.; Leonova, G.N.; Besednova, N.N.; Zvyagintseva, T.N.; Shevchenko, N.M. The protective effect of fucoidan from brown alga Laminaria japonica in experimental tick-borne encephalitis. Pac. Med. J. 2009, 3, 89–92. [Google Scholar]
- Hayashi, K.; Nakano, T.; Hashimoto, M.; Kanekiyo, K.; Hayashi, T. Defensive effects of a fucoidan from brown alga Undaria pinnatifida against herpes simplex virus infection. Int. Immunopharmacol. 2008, 8, 109–116. [Google Scholar] [CrossRef]
- Jung-Bum, L.; Hayashi, K.; Hashimoto, M.; Nakano, T.; Hayashi, T. Novel antiviral fucoidan from sporophyll of Undaria pinnatifida (Mekabu). Chem. Pharm. Bull. 2004, 52, 1091–1094. [Google Scholar]
- Harden, E.A.; Falshaw, R.; Carnachan, S.M.; Kern, E.R.; Prichard, M.N. Virucidal activity of polysaccharide extracts from four algal species against herpes simplex virus. Antiviral. Res. 2009, 83, 282–289. [Google Scholar] [CrossRef]
- Hidari, K.I.; Takahashi, N.; Arihara, M.; Nagaoka, M.; Morita, K.; Suzuki, T. Structure and anti-dengue virus activity of sulfated polysaccharide from a marine alga. Biochem. Biophys. Res. Commun. 2008, 376, 91–95. [Google Scholar] [CrossRef]
- Elizondo-Gonzalez, R.; Cruz-Suarez, L.E.; Ricque-Marie, D.; Mendoza-Gamboa, E.; Rodriguez-Padilla, C.; Trejo-Avila, L.M. In vitro characterization of the antiviral activity of fucoidan from Cladosiphon okamuranus against Newcastle Disease Virus. Virol. J. 2012, 9, 307–316. [Google Scholar] [CrossRef]
- Wang, W.; Wu, J.; Zhang, X.; Hao, C.; Zhao, X.; Jiao, G.; Shan, X.; Tai, W.; Yu, G. Inhibition of influenza A virus infection by fucoidan targeting viral neuraminidase and cellular EGFR pathway. Sci. Rep. 2017, 7, 40760. [Google Scholar] [CrossRef]
- Sanniyasi, E.; Venkatasubramanian, G.; Anbalagan, M.M.; Raj, P.P.; Gopal, R.K. In vitro anti-HIV-1 activity of the bioactive compound extracted and purified from two different marine macroalgae (seaweeds) (Dictyota bartayesiana J.V. Lamouroux and Turbinaria decurrens Bory). Sci. Rep. 2019, 9, 12185. [Google Scholar] [CrossRef]
- Yang, C.; Chung, D.; Shin, I.S.; Lee, H.; Kim, J.; Lee, Y.; You, S. Effects of molecular weight and hydrolysis conditions on anticancer activity of fucoidans from sporophyll of Undaria pinnatifida. Int. J. Biol. Macromol. 2008, 43, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Zayed, A.; Ulber, R. Fucoidan production: Approval key challenges and opportunities. Carbohydr. Polym. 2019, 211, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Preeprame, S.; Lee, J.B.; Hayashi, K.; Sankawa, U. A novel antivirally active fucan sulfate derived from an edible brown alga, Sargassum horneri. Chem. Pharm. Bull. 2001, 49, 484–485. [Google Scholar] [CrossRef] [PubMed]
- Hahn, T.; Ulber, R.; Lang, S.; Muffler, K. Novel procedures for the extraction of fucoidan from brown algae. Process Biochem. 2012, 47, 1691–1698. [Google Scholar] [CrossRef]
- Mandal, P.; Mateu, C.G.; Chattopadhyay, K.; Pujol, C.A.; Damonte, E.B.; Ray, B. Structural features and antiviral activity of sulphated fucans from the brown seaweed Cystoseira indica. Antivir. Chem. Chemother. 2007, 18, 153–162. [Google Scholar] [CrossRef]
- Trejo-Avila, L.M.; Morales-Martinez, M.E.; Ricque-Marie, D.; Cruz-Suarez, L.E.; Zapata-Benavides, P.; Moran-Santibanez, K.; Rodrıguez-Padilla, C. In vitro anti-canine distemper virus activity of fucoidan extracted from the brown alga Cladosiphon okamuranus. Virus Dis. 2014, 25, 474–480. [Google Scholar] [CrossRef]
- Damonte, E.B.; Matulewicz, M.C.; Cerezo, A.S. Sulfated seaweed polysaccharides as antiviral agents. Curr. Med. Chem. 2004, 11, 2399–2419. [Google Scholar] [CrossRef]
- Ghosh, T.; Chattopadhyay, K.; Marschall, M.; Karmakar, P.; Mandal, P.; Ray, B. Focus on antivirally active sulfated polysaccharides: From structure–activity analysis to clinical evaluation. Glycobiology 2009, 19, 2–15. [Google Scholar] [CrossRef]
- Prokofjeva, M.M.; Imbs, T.I.; Shevchenko, N.M.; Spirin, P.V.; Horn, S.; Fehse, B.; Zvyagintseva, T.N.; Prassolov, V.S. Fucoidans as potential inhibitors of HIV-1. Mar. Drugs 2013, 11, 3000–3014. [Google Scholar] [CrossRef]
- Chiu, Y.H.; Chan, Y.L.; Tsai, L.W.; Li, T.L.; Wu, C.J. Prevention of human enterovirus 71 infection by kappa carrageenan. Antivir. Res. 2012, 95, 128–134. [Google Scholar] [CrossRef]
- Yue, Y.; Li, Z.; Li, P.; Song, N.; Li, B.; Lin, W.; Liu, S. Antiviral activity of a polysaccharide from Laminaria japonica against enterovirus 71. Biomed. Pharmacother. 2017, 96, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, W.; Hao, C.; Yunjia, Y.; Qin, L.; He, M.; Mao, W. Antiviral activity against enterovirus 71 of sulfated rhamnan isolated from the green alga Monostroma latissimum. Carbohydr. Polym. 2018, 200, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Thuy, T.T.T.; Ly, B.M.; Van, T.T.T.; Quang, N.V.; Tu, H.C.; Zheng, Y.; Seguin-Devaux, C.; Mi, B.; Ai, U. Anti-HIV activity of fucoidans from three brown seaweed species. Carbohydr. Polym. 2015, 115, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Besednova, N.N.; Zvyagintseva, T.N.; Kuznetsova, T.A.; Makarenkova, I.D.; Smolina, T.P.; Fedyanina, L.N.; Kryzhanovsky, S.P.; Zaporozhets, T.S. Marine algae metabolites as promising therapeutics for the prevention and treatment of HIV/AIDS. Metabolites 2019, 9, 87. [Google Scholar] [CrossRef] [PubMed]
- Luthuli, S.; Wu, S.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic effects of fucoidan: A review on recent studies. Mar. Drugs 2019, 17, 487. [Google Scholar] [CrossRef]
- Wang, W.; Wang, S.X.; Guan, H.S. The Antiviral activities and mechanisms of marine polysaccharides: An overview. Mar. Drugs 2012, 10, 2795–2816. [Google Scholar] [CrossRef]
- Paskaleva, E.E.; Lin, X.; Li, W.; Cotter, R.; Klein, M.T.; Roberge, E.; Yu, E.K.; Clark, B.; Veille, J.C.; Liu, Y.; et al. Inhibition of highly productive HIV-1 infection in T cells, primary human macrophages, microglia, and astrocytes by Sargassum fusiforme. AIDS Res. Ther. 2006, 3, 15. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, X.; Tang, Y.; Mao, J. Composition, isolation, purification and biological activities of Sargassum fusiforme polysaccharides: A review. Carbohydr. Polym. 2020, 228, 115381. [Google Scholar] [CrossRef]
- Lehtinen, M.; Koskela, P.; Jellum, E.; Bloigu, A.; Anttila, T.; Hallmans, G.; Luukkaala, T.; Thoresen, S.; Youngman, L.; Dillner, J. Herpes simplex virus and risk of cervical cancer: A longitudinal, nested case-control study in the Nordic countries. Am. J. Epidemiol. 2002, 156, 687–692. [Google Scholar] [CrossRef]
- Corey, L.; Wald, A.; Celum, C.L.; Quinn, T.C. The effects of herpes simplex virus-2 on HIV-1 acquisition and transmission: A review of two overlapping epidemics. J. Acquir. Immune Defic. Syndr. 2004, 35, 435–445. [Google Scholar] [CrossRef]
- Freeman, E.E.; Weiss, H.A.; Glynn, J.R.; Cross, P.L.; Whitworth, J.A.; Hayes, R.J. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS (London England) 2006, 20, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Carlucci, M.J.; Scolaro, L.A.; Noseda, M.D.; Cerezo, A.S.; Damonte, E.B. Protective effect of a natural carrageenan on genital herpes simplex virus infection in mice. Antivirial Res. 2004, 64, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Cardozo, F.T.G.S.; Larsen, I.V.; Carballo, E.V.; Jose, G.; Stern, R.A.; Brummel, R.C.; Camelini, C.M.; Rossi, M.J.; Simões, C.M.O.; Brandt, C.R. In Vivo anti-herpes simplex virus activity of a sulfated derivative of Agaricus brasiliensis mycelial polysaccharide. Antimicrob. Agents Chemother. 2013, 57, 2541–2549. [Google Scholar] [CrossRef] [PubMed]
- Kusaykin, M.I.; Chizhov, A.O.; Grachev, A.A.; Alekseeva, S.A.; Bakunina, I.Y.; Nedashkovskaya, O.I.; Sova, V.V.; Zvyagintseva, T.N. A comparative study of specificity of fucoidanases from marine microorganisms and invertebrates. J. Appl. Phycol. 2006, 18, 369–373. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Weislow, O.S.; Kiser, R.; Fine, D.L.; Bader, J.; Shoemaker, R.H.; Boyd, M.R. New soluble-formazan assay for HIV-1 cytopathic effects: Application to high-flux screening of synthetic and natural products for AIDS-antiviral activity. J. Natl. Cancer Inst. 1989, 81, 577–586. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent’s endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Astani, A.; Schnitzler, P. Antiviral activity of monoterpenes beta-pinene and limonene against herpes simplex virus in vitro. Iran J. Microbiol. 2014, 6, 149–155. [Google Scholar]
| Viruses | Compounds | Pretreatment of Virus | Pretreatment of Cells | Simultaneous Treatment | Treatment of Infected Cells | ||||
|---|---|---|---|---|---|---|---|---|---|
| IC50 (μg/mL) | SI | IC50 (μg/mL) | SI | IC50 (μg/mL) | SI | IC50 (μg/mL) | SI | ||
| HSV-1 | FeF | 106 ± 13 | 19 ± 2 | 53 ± 7 | 38 ± 4 | 59 ± 8 | 34 ± 4 | 80 ± 9 | 25 ± 3 |
| FeHMP | 127 ± 15 | 16 ± 2 | 100 ± 15 * | 20 ± 2 * | 95 ± 12 * | 21 ± 2 * | 100 ± 13 | 20 ± 2 | |
| Acyclovir | NA | NA | 2.1 ± 0.3 | ˃950 | 0.1 ± 0.01 | ˃20.000 | |||
| HSV-2 | FeF | 95 ± 10 | 21 ± 2 | 45 ± 6 | 44 ± 5 | 50 ± 7 | 40 ± 5 | 65 ± 8 | 31 ± 4 |
| FeHMP | 110 ± 13 | 18 ± 2 | 77 ± 8 * | 26 ± 3 * | 80 ± 10 * | 25 ± 3 * | 85 ± 11 | 24 ± 3 | |
| Acyclovir | NA | NA | 1.6 ± 0.2 | ˃1200 | 0.1 ± 0.01 | ˃20.000 | |||
| ECHO-1 | FeF | 710 ± 80 | 2.8 ± 0.2 | 105 ± 12 | 19 ± 2 | 90 ± 12 | 22 ± 2 | 110 ± 13 | 18 ± 1 |
| FeHMP | 580 ± 65 | 3.4 ± 0.2 | 83 ± 10 | 24 ± 2 | 85 ± 11 | 24 ± 2 | 93 ± 11 | 21 ± 2 | |
| Ribavirin | NA | NA | ˃500 | ≤1.5 | ˃500 | ≤1.5 | |||
| HIV-1 | FeF | 25 ± 3 | 8 ± 1 | 25 ± 3 | 8 ± 1 | 25 ± 3 | 8 ± 1 | 25 ± 3 | 8 ± 1 |
| FeHMP | 25 ± 3 | 8 ± 1 | 25 ± 3 | 8 ± 1 | 50 ± 6 | 4 ± 0.5 | 25 ± 3 | 8 ± 1 | |
| Retrovir | 5 ± 0.6 | 10 ± 1 | NA | NA | 1.3 ± 0.2 | 38 ± 4 | 2.5 ± 0.3 | 20 ± 3 | |
| Epivir | 1.5 ± 0.2 | 33 ± 4 | NA | NA | 1.5 ± 0.2 | 33 ± 4 | 1.5 ± 0.2 | 33 ± 4 | |
| Groups | Survivors/Total | Protection Index (%) | Average Survival Time (d) |
|---|---|---|---|
| FeF (5 mg/kg/d) | 2/10 | 11.1 | 11.0 ± 2.1 |
| FeF (10 mg/kg/d) | 5/10 * | 44.4 | 14.1 ± 1.4 * |
| FeHMP (5 mg/kg/d) | 3/10 | 22.2 | 13.4 ± 1.8 |
| FeHMP (10 mg/kg/d) | 6/10 * | 55.6 | 16.0 ± 1.3 * |
| Acyclovir (50 mg/kg/d) | 10/10 * | 100 | 21.0 ± 0.5 * |
| Virus group | 1/10 | - | 9.7 ± 2.6 |
| Normal group | 10/10 | 100 | ˃21 |
| Polysaccharide | Molecular Weight, kDa | OSO3− % | Monosaccharide Composition | ||
|---|---|---|---|---|---|
| Fuc | Gal | Xyl | |||
| FeF | 160 | 28 | 0.9 | 0.1 | 0 |
| FeHMP | 50.8 | 40 | 1.0 | 0 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krylova, N.V.; Ermakova, S.P.; Lavrov, V.F.; Leneva, I.A.; Kompanets, G.G.; Iunikhina, O.V.; Nosik, M.N.; Ebralidze, L.K.; Falynskova, I.N.; Silchenko, A.S.; et al. The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo. Mar. Drugs 2020, 18, 224. https://doi.org/10.3390/md18040224
Krylova NV, Ermakova SP, Lavrov VF, Leneva IA, Kompanets GG, Iunikhina OV, Nosik MN, Ebralidze LK, Falynskova IN, Silchenko AS, et al. The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo. Marine Drugs. 2020; 18(4):224. https://doi.org/10.3390/md18040224
Chicago/Turabian StyleKrylova, Natalya V., Svetlana P. Ermakova, Vyacheslav F. Lavrov, Irina A. Leneva, Galina G. Kompanets, Olga V. Iunikhina, Marina N. Nosik, Linna K. Ebralidze, Irina N. Falynskova, Artem S. Silchenko, and et al. 2020. "The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo" Marine Drugs 18, no. 4: 224. https://doi.org/10.3390/md18040224
APA StyleKrylova, N. V., Ermakova, S. P., Lavrov, V. F., Leneva, I. A., Kompanets, G. G., Iunikhina, O. V., Nosik, M. N., Ebralidze, L. K., Falynskova, I. N., Silchenko, A. S., & Zaporozhets, T. S. (2020). The Comparative Analysis of Antiviral Activity of Native and Modified Fucoidans from Brown Algae Fucus evanescens In Vitro and In Vivo. Marine Drugs, 18(4), 224. https://doi.org/10.3390/md18040224





