Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications
Abstract
1. Introduction
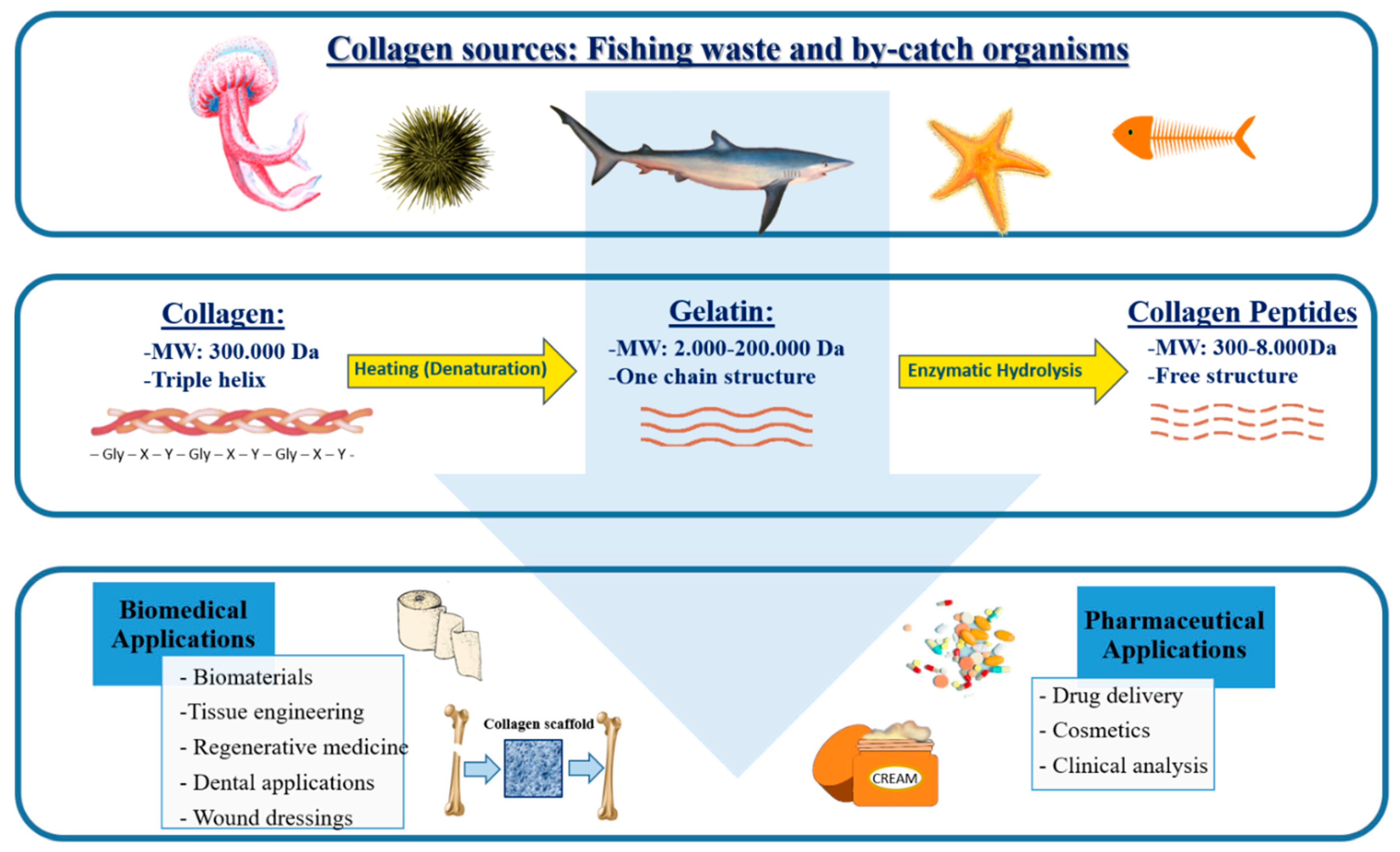
2. Sustainable Marine Sources of Collagen
3. Collagen Structure
4. Extraction of Marine Collagen
5. Marine Collagen Proceeding
5.1. Dry Process
5.2. Extrusion-Based Processes
6. Biomedical and Pharmaceutical Applications of Collagen
6.1. Collagen and other Collagen-Derived Peptide Bioactivities from Sustainable Marine Sources
6.1.1. Fish Waste
6.1.2. Echinoderms
6.1.3. Jellyfish
7. Other Potential Applications of Marine Collagen: Food Additives and Packaging
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Muller, W.E. The origin of metazoan complexity: Porifera as integrated animals. Integr. Comp. Biol. 2003, 43, 3–10. [Google Scholar] [CrossRef]
- Hashim, P.; Ridzwan, M.; Bakar, J.; Hashim, D. Collagen in food and beverage industries. Int. Food Res. J. 2015, 22, 1–8. [Google Scholar]
- Karsdal, M. Biochemistry of Collagens, Laminins and Elastin: Structure, Function and Biomarkers; Karsdal, M., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 127–129. [Google Scholar]
- Wahyudi, H.; Reynolds, A.A.; Li, Y.; Owen, S.C.; Yu, S.M. Targeting collagen for diagnostic imaging and therapeutic delivery. J. Control. Release 2016, 240, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Lv, Y. Application of collagen scaffold in tissue engineering: Recent advances and new perspectives. Polymers 2016, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lakes, R.S. Biomaterials: An Introduction; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Bama, P.; Vijayalakshimi, M.; Jayasimman, R.; Kalaichelvan, P.; Deccaraman, M.; Sankaranarayanan, S. Extraction of collagen from cat fish (Tachysurus maculatus) by pepsin digestion and preparation and characterization of collagen chitosan sheet. Int. J. Pharm. Pharm. Sci. 2010, 2, 133–137. [Google Scholar]
- Hackett, C.; Grim, B.; Stonawski, M.; Skirbekk, V.; Potančoková, M.; Abel, G. The Global Religious Landscape; Pew Research Center: Washington, DC, USA, 2012. [Google Scholar]
- Avila Rodríguez, M.I.; Rodriguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef]
- Felician, F.F.; Xia, C.; Qi, W.; Xu, H. Collagen from Marine Biological Sources and Medical Applications. Chem. Biodivers. 2018, 15, e1700557. [Google Scholar] [CrossRef]
- Langasco, R.; Cadeddu, B.; Formato, M.; Lepedda, A.J.; Cossu, M.; Giunchedi, P.; Pronzato, R.; Rassu, G.; Manconi, R.; Gavini, E. Natural collagenic skeleton of marine sponges in pharmaceutics: Innovative biomaterial for topical drug delivery. Mater. Sci. Eng. C 2017, 70, 710–720. [Google Scholar] [CrossRef]
- Lim, Y.-S.; Ok, Y.-J.; Hwang, S.-Y.; Kwak, J.-Y.; Yoon, S. Marine Collagen as A Promising Biomaterial for Biomedical Applications. Mar. Drugs 2019, 17, 467. [Google Scholar] [CrossRef]
- Parisi, J.; Fernandes, K.; Avanzi, I.; Dorileo, B.; Santana, A.; Andrade, A.; Gabbai-Armelin, P.; Fortulan, C.; Trichês, E.; Granito, R. Incorporation of collagen from marine sponges (spongin) into hydroxyapatite samples: Characterization and in vitro biological evaluation. Mar. Biotechnol. 2019, 21, 30–37. [Google Scholar] [CrossRef]
- Rahman, M.A. Collagen of extracellular matrix from marine invertebrates and its medical applications. Mar. Drugs 2019, 17, 118. [Google Scholar] [CrossRef] [PubMed]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M.J. Characterisation of acid-soluble collagen from skin and bone of bigeye snapper (Priacanthus tayenus). Food Chem. 2005, 89, 363–372. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, W.; Li, G. Isolation and characterisation of collagens from the skin of largefin longbarbel catfish (Mystus macropterus). Food Chem. 2009, 115, 826–831. [Google Scholar] [CrossRef]
- Khan, S.; Zhong-Ji, Q.; Bomi, R.; Kim, S.J. Isolation and biochemical characterization of collagens from seaweed pipefish, Syngnathus schlegeli. Biotechnol. Bioproc. E 2009, 14, 436–442. [Google Scholar] [CrossRef]
- Senaratne, L.; Park, P.-J.; Kim, S.-K. Isolation and characterization of collagen from brown backed toadfish (Lagocephalus gloveri) skin. Bioresour. Technol. 2006, 97, 191–197. [Google Scholar] [CrossRef]
- Nagai, T.; Araki, Y.; Suzuki, N. Collagen of the skin of ocellate puffer fish (Takifugu rubripes). Food Chem. 2002, 78, 173–177. [Google Scholar] [CrossRef]
- Minh Thuyle, T.; Okazaki, E.; Osako, K. Isolation and characterization of acid-soluble collagen from the scales of marine fishes from Japan and Vietnam. Food Chem. 2014, 149, 264–270. [Google Scholar] [CrossRef]
- Kołodziejska, I.; Sikorski, Z.E.; Niecikowska, C. Parameters affecting the isolation of collagen from squid (Illex argentinus) skins. Food Chem. 1999, 66, 153–157. [Google Scholar] [CrossRef]
- Shanmugam, V.; Ramasamy, P.; Subhapradha, N.; Sudharsan, S.; Seedevi, P.; Moovendhan, M.K.J.; Shanmugam, A.; Srinivasan, A. Extraction, structural and physical characterization of type I collagen from the outer skin of Sepiella inermis (Orbigny, 1848). Afr. J. Biotechnol. 2012, 11, 14326–14337. [Google Scholar] [CrossRef]
- Kittiphattanabawon, P.; Benjakul, S.; Visessanguan, W.; Shahidi, F.J. Isolation and characterization of collagen from the cartilages of brownbanded bamboo shark (Chiloscyllium punctatum) and blacktip shark (Carcharhinus limbatus). LWT-Food Sci. Technol. 2010, 43, 792–800. [Google Scholar] [CrossRef]
- Benedetto, C.D.; Barbaglio, A.; Martinello, T.; Alongi, V.; Fassini, D.; Cullora, E.; Patruno, M.; Bonasoro, F.; Barbosa, M.A.; Carnevali, M.D.; et al. Production, characterization and biocompatibility of marine collagen matrices from an alternative and sustainable source: The sea urchin Paracentrotus lividus. Mar. Drugs 2014, 12, 4912–4933. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Suzuki, N. Partial characterization of collagen from purple sea urchin (Anthocidaris crassispina) test. Int. J. Food Sci. Technol. 2000, 35, 497–501. [Google Scholar] [CrossRef]
- Qi, H.; Li, N.; Zhao, X.; Xu, Z.; Qi, L. Physicochemical Properties and the Radical Scavenging Capacities of Pepsin-Solubilized Collagen from the Body Wall of Starfish (Asterina pectinifera). J. Aquat. Food Prod. Technol. 2017, 26, 376–389. [Google Scholar] [CrossRef]
- Addad, S.; Exposito, J.-Y.; Faye, C.; Ricard-Blum, S.; Lethias, C. Isolation, characterization and biological evaluation of jellyfish collagen for use in biomedical applications. Mar. Drugs 2011, 9, 967–983. [Google Scholar] [CrossRef]
- Barzideh, Z.; Latiff, A.; Gan, C.-Y.; Benjakul, S.; Karim, A. Isolation and characterisation of collagen from the ribbon jellyfish (Chrysaora sp.). Int. J. Food Sci. Technol. 2013, 49, 1490–1499. [Google Scholar] [CrossRef]
- Calejo, M.; Morais, Z.; Fernandes, A. Isolation and biochemical characterisation of a novel collagen from Catostylus tagi. J. Biomater. Sci. Polym. Ed. 2009, 20, 2073–2087. [Google Scholar] [CrossRef]
- Nagai, T.; Worawattanamateekul, W.; Suzuki, N.; Nakamura, T.; Ito, T.; Fujiki, K.; Nakao, M.; Yano, T. Isolation and characterization of collagen from rhizostomous jellyfish (Rhopilema asamushi). Food Chem. 2000, 70, 205–208. [Google Scholar] [CrossRef]
- Cheng, X.; Shao, Z.; Li, C.; Yu, L.; Raja, M.A.; Liu, C. Isolation, Characterization and Evaluation of Collagen from Jellyfish Rhopilema esculentum Kishinouye for Use in Hemostatic Applications. PLoS ONE 2017, 12, e0169731. [Google Scholar] [CrossRef]
- Nagai, T.; Ogawa, T.; Nakamura, T.; Ito, T.; Nakagawa, H.; Fujiki, K.; Nakao, M.; Yano, T. Collagen of edible jellyfish exumbrella. J. Sci. Food Agric. 1999, 79, 855–858. [Google Scholar] [CrossRef]
- Miura, S.; Kimura, S. Jellyfish mesogloea collagen. Characterization of molecules as alpha 1 alpha 2 alpha 3 heterotrimers. J. Biol. Chem. 1985, 260, 15352–15356. [Google Scholar]
- Chinh, N.T.; Manh, V.Q.; Trung, V.Q.; Lam, T.D.; Huynh, M.D.; Tung, N.Q.; Trinh, N.D.; Hoang, T. Characterization of Collagen Derived From Tropical Freshwater Carp Fish Scale Wastes and Its Amino Acid Sequence. Nat. Prod. Commun. 2019, 14, 1934578X19866288. [Google Scholar] [CrossRef]
- Mahboob, S. Isolation and characterization of collagen from fish waste material-skin, scales and fins of Catla catla and Cirrhinus mrigala. J. Food Sci. Technol. 2015, 52, 4296–4305. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, N.; Shakila, R.J.; Sukumar, D.; Jeyasekaran, G. Skin, bone and muscle collagen extraction from the trash fish, leather jacket (Odonus niger) and their characterization. J. Food Sci. Technol. 2013, 50, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Raman, M.; Gopakumar, K. Fish Collagen and its Applications in Food and Pharmaceutical Industry: A Review. EC Nutr. 2018, 13, 752–767. [Google Scholar]
- Silvipriya, K.; Kumar, K.K.; Kumar, B.D.; John, A.; Lakshmanan, P.J. Fish processing waste: A promising source of type-i collagen. Curr. Trends Biotechnol. Pharm. 2016, 10, 374–383. [Google Scholar]
- Sotelo, C.G.; Comesaña, M.B.; Ariza, P.R.; Pérez-Martín, R. Characterization of collagen from different discarded fish species of the West coast of the Iberian Peninsula. J. Aquat. Food Prod. Technol. 2016, 25, 388–399. [Google Scholar] [CrossRef]
- Susanti, E.; Lutfiana, N.; Retnosari, R. Screening of Proteolytic Bacteria from tauco Surabaya based on pathogenicity and selectivity of its protease on milky fish (Chanos chanos) scales for healthy and halal collagen production. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; p. 012044. [Google Scholar]
- Mo, W.Y.; Man, Y.B.; Wong, M.H. Use of food waste, fish waste and food processing waste for China’s aquaculture industry: Needs and challenge. Sci. Total Environ. 2018, 613, 635–643. [Google Scholar] [CrossRef]
- Arnesen, J.A.; Gildberg, A. Extraction and characterisation of gelatine from Atlantic salmon (Salmo salar) skin. Bioresour. Technol. 2007, 98, 53–57. [Google Scholar] [CrossRef]
- Tsagarakis, K.; Palialexis, A.; Vassilopoulou, V.J. Mediterranean fishery discards: Review of the existing knowledge. ICES J. Mar. Sci. 2014, 71, 1219–1234. [Google Scholar] [CrossRef]
- Condon, R.H.; Duarte, C.M.; Pitt, K.A.; Robinson, K.L.; Lucas, C.H.; Sutherland, K.R.; Mianzan, H.W.; Bogeberg, M.; Purcell, J.E.; Decker, M.B. Recurrent jellyfish blooms are a consequence of global oscillations. Proc. Natl. Acad. Sci. USA 2013, 110, 1000–1005. [Google Scholar] [CrossRef]
- Boero, F. Review of jellyfish blooms in the Mediterranean and Black Sea. FAO Fish. Rep. 2013, 92, 64. [Google Scholar]
- Brotz, L.; Pauly, D. Jellyfish populations in the Mediterranean Sea. Acta Adriat 2012, 53, 213–231. [Google Scholar]
- Omori, M.; Nakano, E. Jellyfish fisheries in southeast Asia. Hydrobiologia 2001, 451, 19–26. [Google Scholar] [CrossRef]
- Purcell, J.E.; Uye, S.-I.; Lo, W.-T. Anthropogenic causes of jellyfish blooms and their direct consequences for humans: A review. Mar. Ecol. Prog. Ser. 2007, 350, 153–174. [Google Scholar] [CrossRef]
- Khong, N.M.; Yusoff, F.M.; Jamilah, B.; Basri, M.; Maznah, I.; Chan, K.W.; Armania, N.; Nishikawa, J. Improved collagen extraction from jellyfish (Acromitus hardenbergi) with increased physical-induced solubilization processes. Food Chem. 2018, 251, 41–50. [Google Scholar] [CrossRef]
- Widdowson, J.P.; Picton, A.J.; Vince, V.; Wright, C.J.; Mearns-Spragg, A. In vivo comparison of jellyfish and bovine collagen sponges as prototype medical devices. J. Biomed. Mater. Res. 2018, 106, 1524–1533. [Google Scholar] [CrossRef]
- Merquiol, L.; Romano, G.; Ianora, A.; D’Ambra, I. Biotechnological Applications of Scyphomedusae. Mar. Drugs 2019, 17, 604. [Google Scholar] [CrossRef]
- Halper, J.; Kjaer, M. Progress in Heritable Soft Connective Tissue Diseases; Springer: Berlin/Heidelberg, Germany, 2014; Volume 802. [Google Scholar]
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200. [Google Scholar] [CrossRef]
- Szpak, P. Fish bone chemistry and ultrastructure: Implications for taphonomy and stable isotope analysis. J. Archaeol. Sci. 2011, 38, 3358–3372. [Google Scholar] [CrossRef]
- Lacroix, M.; Cooksey, K. Edible films and coatings from animal origin proteins. In Innovations in Food Packaging; Elsevier: Amsterdam, The Netherlands, 2005; pp. 301–317. [Google Scholar]
- Mariod, A.A.; Fadul, H. Gelatin, source, extraction and industrial applications. Acta Sci. Pol. Technol. Aliment. 2013, 12, 135–147. [Google Scholar]
- Kariduraganavar, M.Y.; Kittur, A.A.; Kamble, R.R. Polymer synthesis and processing. In Natural and Synthetic Biomedical Polymers; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1–31. [Google Scholar]
- Nasri, M. Bioactive Peptides from Fish Collagen Byproducts: A Review. In Byprod. Agric. Fish: Adding Value for Food, Feed, Pharma, Fuels; Wiley online library: Hoboken, NJ, USA, 2019; pp. 309–333. [Google Scholar]
- Nasri, M. Protein hydrolysates and biopeptides: Production, biological activities, and applications in foods and health benefits: A review. In Advances in Food and Nutrition Research; Elsevier: Amsterdam, The Netherlands, 2017; Volume 81, pp. 109–159. [Google Scholar]
- Nagai, T. Characterization of acid-soluble collagen from skins of surf smelt (Hypomesus pretiosus japonicus Brevoort). Food Nutr. Sci. 2010, 1, 59. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N. Isolation of collagen from fish waste material—Skin, bone and fins. Food Chem. 2000, 68, 277–281. [Google Scholar] [CrossRef]
- Jongjareonrak, A.; Benjakul, S.; Visessanguan, W.; Nagai, T.; Tanaka, M. Isolation and characterisation of acid and pepsin-solubilised collagens from the skin of Brownstripe red snapper (Lutjanus vitta). Food Chem. 2005, 93, 475–484. [Google Scholar] [CrossRef]
- Sadowska, M.; Kołodziejska, I.; Niecikowska, C. Isolation of collagen from the skins of Baltic cod (Gadus morhua). Food Chem. 2003, 81, 257–262. [Google Scholar] [CrossRef]
- Duan, R.; Zhang, J.; Du, X.; Yao, X.; Konno, K. Properties of collagen from skin, scale and bone of carp (Cyprinus carpio). Food Chem. 2009, 112, 702–706. [Google Scholar] [CrossRef]
- Żelechowska, E.; Sadowska, M.; Turk, M. Isolation and some properties of collagen from the backbone of Baltic cod (Gadus morhua). Food Hydrocoll. 2010, 24, 325–329. [Google Scholar] [CrossRef]
- Yusoff, F.M.; Bakar, J.; Basri, M.; Ismail, M.; Khong, N.M.H. A method for extracting collagen from aquatic animals, collagen and products containing it. WO2015012682A3, 2013. [Google Scholar]
- Zhang, Y.; Liu, W.; Li, G.; Shi, B.; Miao, Y.; Wu, X. Isolation and partial characterization of pepsin-soluble collagen from the skin of grass carp (Ctenopharyngodon idella). Food Chem. 2007, 103, 906–912. [Google Scholar] [CrossRef]
- Cao, H.; Xu, S.Y. Purification and characterization of type II collagen from chick sternal cartilage. Food Chem. 2008, 108, 439–445. [Google Scholar] [CrossRef]
- Lin, Y.; Liu, D. Effects of pepsin digestion at different temperatures and times on properties of telopeptide-poor collagen from bird feet. Food Chem. 2006, 94, 621–625. [Google Scholar] [CrossRef]
- Schmidt, M.; Dornelles, R.; Mello, R.; Kubota, E.; Mazutti, M.; Kempka, A.; Demiate, I. Collagen extraction process. Int. Food Res. J. 2016, 23, 913–922. [Google Scholar]
- Silva, T.H.; Moreira-Silva, J.; Marques, A.L.; Domingues, A.; Bayon, Y.; Reis, R.L. Marine origin collagens and its potential applications. Mar. Drugs 2014, 12, 5881–5901. [Google Scholar] [CrossRef] [PubMed]
- Song, E.; Yeon Kim, S.; Chun, T.; Byun, H.J.; Lee, Y.M. Collagen scaffolds derived from a marine source and their biocompatibility. Biomaterials 2006, 27, 2951–2961. [Google Scholar] [CrossRef] [PubMed]
- Di Benedetto, C. Progenitor Cells and Regenerative Potential in Echinoderms: An In Vivo and In Vitro Approach. Ph.D. Thesis, University of Milan, Milan, Italy, 2010. [Google Scholar]
- Barbaglio, A.; Tricarico, S.; Ribeiro, A.; Ribeiro, C.; Sugni, M.; Di Benedetto, C.; Wilkie, I.; Barbosa, M.; Bonasoro, F.; Carnevali, M.C. The mechanically adaptive connective tissue of echinoderms: Its potential for bio-innovation in applied technology and ecology. Mar. Environ. Res. 2012, 76, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, C.; Leggio, L.; Leone, R.; di benedetto, C.; Guidetti, L.; Coccè, V.; Ascagni, M.; Bonasoro, F.; La Porta, C.; Candia, D.; et al. Marine-derived collagen biomaterials from echinoderm connective tissues. Mar. Environ. Res. 2016, 128, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Scott, I.; Yamauchi, M.; Sricholpech, M. Lysine post-translational modifications of collagen. Essays Biochem. 2012, 52, 113–133. [Google Scholar] [CrossRef]
- Muyonga, J.; Cole, C.; Duodu, K. Extraction and physico-chemical characterisation of Nile perch (Lates niloticus) skin and bone gelatin. Food Hydrocoll. 2004, 18, 581–592. [Google Scholar] [CrossRef]
- Finch, C. Chemical modification and some cross-linking reactions of water-soluble polymers. In Chemistry and Technology of Water-Soluble Polymers; Springer: Berlin/Heidelberg, Germany, 1983; pp. 81–111. [Google Scholar]
- Fratzl, P.; Misof, K.; Zizak, I.; Rapp, G.; Amenitsch, H.; Bernstorff, S. Fibrillar structure and mechanical properties of collagen. J. Struct. Biol. 1998, 122, 119–122. [Google Scholar] [CrossRef]
- Ottani, V.; Martini, D.; Franchi, M.; Ruggeri, A.; Raspanti, M. Hierarchical structures in fibrillar collagens. Micron 2002, 33, 587–596. [Google Scholar] [CrossRef]
- Ottani, V.; Raspanti, M.; Ruggeri, A. Collagen structure and functional implications. Micron 2001, 32, 251–260. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.; Pérez-Mateos, M.; Gómez-Estaca, J.; López-Caballero, E.; Giménez, B.; Montero, P. Fish gelatin: A renewable material for developing active biodegradable films. Trends Food Sci. Technol. 2009, 20, 3–16. [Google Scholar] [CrossRef]
- Sothornvit, R.; Krochta, J.M. Plasticizer effect on mechanical properties of β-lactoglobulin films. J. Food Eng. 2001, 50, 149–155. [Google Scholar] [CrossRef]
- Sothornvit, R.; Krochta, J.M. Plasticizers in edible films and coatings. In Innovations in Food Packaging; Elsevier: Amsterdam, The Netherlands, 2005; pp. 403–433. [Google Scholar]
- Di Gioia, L.; Guilbert, S. Corn protein-based thermoplastic resins: Effect of some polar and amphiphilic plasticizers. J. Agric. Food Chem. 1999, 47, 1254–1261. [Google Scholar] [CrossRef]
- Pommet, M.; Redl, A.; Guilbert, S.; Morel, M.-H.J. Intrinsic influence of various plasticizers on functional properties and reactivity of wheat gluten thermoplastic materials. J. Cereal Sci. 2005, 42, 81–91. [Google Scholar] [CrossRef]
- Zhang, J.; Mungara, P.; Jane, J.-L. Mechanical and thermal properties of extruded soy protein sheets. Polym. Degrad. 2001, 42, 2569–2578. [Google Scholar] [CrossRef]
- Hernandez-Izquierdo, V.; Krochta, J. Thermoplastic processing of proteins for film formation—A review. J. Food Sci. 2008, 73, R30–R39. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Q.; Rempel, C.J. Processing and characteristics of canola protein-based biodegradable packaging: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 475–485. [Google Scholar] [CrossRef]
- Wang, Y.; Padua, G.W.J. Tensile properties of extruded zein sheets and extrusion blown films. Macromol. Mater. Eng. 2003, 288, 886–893. [Google Scholar] [CrossRef]
- Oliviero, M.; Di Maio, E.; Iannace, S. Effect of molecular structure on film blowing ability of thermoplastic zein. J. Appl. Polym. Sci. 2010, 115, 277–287. [Google Scholar] [CrossRef]
- Salerno, A.; Oliviero, M.; Di Maio, E.; Iannace, S. Thermoplastic foams from zein and gelatin. Int. Polym. Process. 2007, 22, 480–488. [Google Scholar] [CrossRef]
- Reddy, M.; Mohanty, A.K.; Misra, M. Thermoplastics from soy protein: A review on processing, blends and composites. J. Biobased Mater. Bioenergy 2010, 4, 298–316. [Google Scholar] [CrossRef]
- Wang, X.-L.; Yang, K.-K.; Wang, Y.-Z. Properties of starch blends with biodegradable polymers. J. Macromol. Sci. 2003, 43, 385–409. [Google Scholar] [CrossRef]
- Oliviero, M.; Sorrentino, L.; Cafiero, L.; Galzerano, B.; Sorrentino, A.; Iannace, S. Foaming behavior of bio-based blends based on thermoplastic gelatin and poly (butylene succinate). J. Appl. Polym. Sci. 2015, 132, 42704. [Google Scholar] [CrossRef]
- Salerno, A.; Di Maio, E.; Iannace, S.; Netti, P. Tailoring the pore structure of PCL scaffolds for tissue engineering prepared via gas foaming of multi-phase blends. J. Porous Mater. 2012, 19, 181–188. [Google Scholar] [CrossRef]
- Salerno, A.; Guarnieri, D.; Iannone, M.; Zeppetelli, S.; Di Maio, E.; Iannace, S.; Netti, P. Engineered μ-bimodal poly (ε-caprolactone) porous scaffold for enhanced hMSC colonization and proliferation. Acta Biomater. 2009, 5, 1082–1093. [Google Scholar] [CrossRef]
- Salerno, A.; Oliviero, M.; Di Maio, E.; Iannace, S.; Netti, P. Design and preparation of μ-bimodal porous scaffold for tissue engineering. J. Appl. Polym. Sci. 2007, 106, 3335–3342. [Google Scholar] [CrossRef]
- Jeong, S.I.; Kim, S.Y.; Cho, S.K.; Chong, M.S.; Kim, K.S.; Kim, H.; Lee, S.B.; Lee, Y.M. Tissue-engineered vascular grafts composed of marine collagen and PLGA fibers using pulsatile perfusion bioreactors. Biomaterials 2007, 28, 1115–1122. [Google Scholar] [CrossRef]
- Zhang, D.; Wu, X.; Chen, J.; Lin, K. The development of collagen based composite scaffolds for bone regeneration. Bioact. Mater. 2018, 3, 129–138. [Google Scholar] [CrossRef]
- Hayashi, Y.; Ikeda, T.; Yamada, S.; Koyama, Z.; Yanagiguchi, K. The application of fish collagen to dental and hard tissue regenerative medicine. In Seafood Processing By-Products; Springer: Berlin/Heidelberg, Germany, 2014; pp. 455–462. [Google Scholar]
- Divya, P.; Nandakumar, K. Local drug delivery—Periocol in periodontics. Trends Biomater. Artif. Organ. 2006, 19, 74–80. [Google Scholar]
- Panwar, M.; Gupta, S. Local drug delivery with tetracycline fiber: An alternative to surgical periodontal therapy. Med. J. Armed Forces India 2009, 65, 244–246. [Google Scholar] [CrossRef]
- Mahesh, L.; Kurtzman, G.; Shukla, S. Regeneration in Periodontics: Collagen-A Review of Its Properties and Applications in Dentistry. Compend. Contin. Educ. Dent. 2015, 36, 358–363. [Google Scholar]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Sibilla, S.; Godfrey, M.; Brewer, S.; Budh-Raja, A.; Genovese, L. An overview of the beneficial effects of hydrolysed collagen as a nutraceutical on skin properties: Scientific background and clinical studies. Open Nutraceuticals J. 2015, 8, 29–42. [Google Scholar] [CrossRef]
- Zhuang, Y.; Hou, H.; Zhao, X.; Zhang, Z.; Li, B. Effects of collagen and collagen hydrolysate from jellyfish (Rhopilema esculentum) on mice skin photoaging induced by UV irradiation. J. Food Sci. 2009, 74, H183–H188. [Google Scholar] [CrossRef]
- Alves, A.; Marques, A.; Martins, E.; Silva, T.; Reis, R.L. Cosmetic Potential of Marine Fish Skin Collagen. Cosmetics 2017, 4, 39. [Google Scholar] [CrossRef]
- Chen, X.L.; Peng, M.; Li, J.; Tang, B.L.; Shao, X.; Zhao, F.; Liu, C.; Zhang, X.Y.; Li, P.Y.; Shi, M.; et al. Preparation and functional evaluation of collagen oligopeptide-rich hydrolysate from fish skin with the serine collagenolytic protease from Pseudoalteromonas sp. SM9913. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Abuine, R.; Rathnayake, A.U.; Byun, H.-G. Biological activity of peptides purified from fish skin hydrolysates. Fish. Aquat. Sci. 2019, 22, 10. [Google Scholar] [CrossRef]
- Xu, L.; Dong, W.; Zhao, J.; Xu, Y. Effect of Marine Collagen Peptides on Physiological and Neurobehavioral Development of Male Rats with Perinatal Asphyxia. Mar. Drugs 2015, 13, 3653–3671. [Google Scholar] [CrossRef]
- Hoyer, B.; Bernhardt, A.; Heinemann, S.; Stachel, I.; Meyer, M.; Gelinsky, M. Biomimetically mineralized salmon collagen scaffolds for application in bone tissue engineering. Biomacromolecules 2012, 13, 1059–1066. [Google Scholar] [CrossRef]
- Lee, E.J.; Hur, J.; Ham, S.A.; Jo, Y.; Lee, S.; Choi, M.J.; Seo, H.G. Fish collagen peptide inhibits the adipogenic differentiation of preadipocytes and ameliorates obesity in high fat diet-fed mice. Int. J. Biol. Macromol. 2017, 104, 281–286. [Google Scholar] [CrossRef]
- Kumar, L.V.; Shakila, R.J.; Jeyasekaran, G. In vitro Anti-Cancer, Anti-Diabetic, Anti-Inflammation and Wound Healing Properties of Collagen Peptides Derived from Unicorn Leatherjacket (Aluterus Monoceros) at Different Hydrolysis. Turk. J. Fish. Aquat. Sci. 2019, 19, 551–560. [Google Scholar]
- Limpisophon, K.; Iguchi, H.; Tanaka, M.; Suzuki, T.; Okazaki, E.; Saito, T.; Takahashi, K.; Osako, K. Cryoprotective effect of gelatin hydrolysate from shark skin on denaturation of frozen surimi compared with that from bovine skin. Fish. Sci. 2015, 81, 383–392. [Google Scholar] [CrossRef]
- Jeevithan, E.; Bao, B.; Bu, Y.; Zhou, Y.; Zhao, Q.; Wu, W. Type II collagen and gelatin from silvertip shark (Carcharhinus albimarginatus) cartilage: Isolation, purification, physicochemical and antioxidant properties. Mar. Drugs 2014, 12, 3852–3873. [Google Scholar] [CrossRef] [PubMed]
- Ben Slimane, E.; Sadok, S. Collagen from Cartilaginous Fish By-Products for a Potential Application in Bioactive Film Composite. Mar. Drugs 2018, 16, 211. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Han, X.; Li, Y. Effect of marine collagen peptides on long bone development in growing rats. J. Sci. Food Agric. 2010, 90, 1485–1491. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, A.; Osaki, T.; Matahira, Y.; Tsuka, T.; Imagawa, T.; Okamoto, Y.; Minami, S. Evaluation of the chondroprotective effects of glucosamine and fish collagen peptide on a rabbit ACLT model using serum biomarkers. J. Vet. Med. Sci. 2012, 75, 421–429. [Google Scholar] [CrossRef]
- Zhou, T.; Sui, B.; Mo, X.; Sun, J. Multifunctional and biomimetic fish collagen/bioactive glass nanofibers: Fabrication, antibacterial activity and inducing skin regeneration in vitro and in vivo. Int. J. Nanomed. 2017, 12, 3495. [Google Scholar] [CrossRef]
- Tang, J.; Saito, T. Biocompatibility of novel type I collagen purified from tilapia fish scale: An in vitro comparative study. BioMed Res. Int. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Cao, H.; Chen, M.-M.; Liu, Y.; Liu, Y.-Y.; Huang, Y.-Q.; Wang, J.-H.; Chen, J.-D.; Zhang, Q.-Q. Fish collagen-based scaffold containing PLGA microspheres for controlled growth factor delivery in skin tissue engineering. Colloids Surf. B Biointerfaces 2015, 136, 1098–1106. [Google Scholar] [CrossRef]
- Barbaglio, A.; Tricarico, S.; di benedetto, C.; Fassini, D.; Lima, A.; Ribeiro, A.; Ribeiro, C.; Sugni, M.; Bonasoro, F.; Wilkie, I.; et al. The smart connective tissue of echinoderms: A materializing promise for biotech applications. Cah. Biol. Mar. 2013, 54, 713–720. [Google Scholar]
- Felician, F.F.; Yu, R.H.; Li, M.Z.; Li, C.J.; Chen, H.Q.; Jiang, Y.; Tang, T.; Qi, W.Y.; Xu, H.M. The wound healing potential of collagen peptides derived from the jellyfish Rhopilema esculentum. Chin. J. Traumatol. 2019, 22, 12–20. [Google Scholar] [CrossRef]
- Hoyer, B.; Bernhardt, A.; Lode, A.; Heinemann, S.; Sewing, J.; Klinger, M.; Notbohm, H.; Gelinsky, M. Jellyfish collagen scaffolds for cartilage tissue engineering. Acta Biomater. 2014, 10, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Pustlauk, W.; Paul, B.; Gelinsky, M.; Bernhardt, A. Jellyfish collagen and alginate: Combined marine materials for superior chondrogenesis of hMSC. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 64, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Rastian, Z.; Pütz, S.; Wang, Y.; Kumar, S.; Fleissner, F.; Weidner, T.; Parekh, S. Type I Collagen from Jellyfish Catostylus mosaicus for Biomaterial Applications. ACS Biomater. Sci. Eng. 2018, 4, 2115–2125. [Google Scholar] [CrossRef]
- Bermueller, C.; Schwarz, S.; Elsaesser, A.F.; Sewing, J.; Baur, N.; von Bomhard, A.; Scheithauer, M.; Notbohm, H.; Rotter, N. Marine collagen scaffolds for nasal cartilage repair: Prevention of nasal septal perforations in a new orthotopic rat model using tissue engineering techniques. Tissue Eng. Part. A 2013, 19, 2201–2214. [Google Scholar] [CrossRef]
- Keller, L.; Pugliano, M. Combined Jellyfish Collagen Type II, Human Stem Cells and Tgf-β3 as a Therapeutic Implant for Cartilage Repair. J. Stem Cell Res. Ther. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Sugahara, T.; Ueno, M.; Goto, Y.; Shiraishi, R.; Doi, M.; Akiyama, K.; Yamauchi, S. Immunostimulation effect of jellyfish collagen. Biosci. Biotechnol. Biochem. 2006, 70, 2131–2137. [Google Scholar] [CrossRef]
- Mackman, N. Triggers, targets and treatments for thrombosis. Nature 2008, 451, 914–918. [Google Scholar] [CrossRef]
- Zhuang, Y.; Sun, L.; Li, B. Production of the Angiotensin-I-Converting Enzyme (ACE)-Inhibitory Peptide from Hydrolysates of Jellyfish (Rhopilema esculentum) Collagen. Food Bioprocess Technol. 2010, 5, 1–8. [Google Scholar] [CrossRef]
- Derkus, B.; Arslan, Y.; Bayraç, T.; Kantarcıoğlu, İ.; Emregul, K.; Emregül, E. Development of a novel aptasensor using jellyfish collagen as matrix and thrombin detection in blood samples obtained from patients with various neurodisease. Sens. Actuators B Chem. 2016, 228, 725–736. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, M.; Shi, Y.; Qiao, R.; Tang, W.; Sun, Z. Production of the angiotensin I converting enzyme inhibitory peptides and isolation of four novel peptides from jellyfish (Rhopilema esculentum) protein hydrolysate. J. Sci. Food Agric. 2016, 96, 3240–3248. [Google Scholar] [CrossRef]
- Zhuang, Y.; Sun, L.; Zhang, Y.; Liu, G. Antihypertensive effect of long-term oral administration of jellyfish (Rhopilema esculentum) collagen peptides on renovascular hypertension. Mar. Drugs 2012, 10, 417–426. [Google Scholar] [CrossRef] [PubMed]
- De Domenico, S.; De Rinaldis, G.; Paulmery, M.; Piraino, S.; Leone, A. Barrel Jellyfish (Rhizostoma pulmo) as Source of Antioxidant Peptides. Mar. Drugs 2019, 17, 134. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.-F.; Li, Y.-Y.; Xu, J.-J.; Su, X.-R.; Gao, X.; Yue, F.-P. Study on effect of jellyfish collagen hydrolysate on anti-fatigue and anti-oxidation. Food Hydrocoll. 2011, 25, 1350–1353. [Google Scholar] [CrossRef]
- Yu, H.; Liu, X.; Xing, R.; Liu, S.; Li, C.; Li, P. Radical scavenging activity of protein from tentacles of jellyfish Rhopilema esculentum. Bioorganic Med. Chem. Lett. 2005, 15, 2659–2664. [Google Scholar] [CrossRef]
- Zhuang, Y.; Sun, L.; Zhao, X.; Wang, J.; Hou, H.; Li, B. Antioxidant and melanogenesis-inhibitory activities of collagen peptide from jellyfish (Rhopilema esculentum). J. Sci. Food Agric. 2009, 89, 1722–1727. [Google Scholar] [CrossRef]
- Zhuang, Y.; Sun, L. Anti-Melanogenic Activities of Collagen Peptides from Jellyfish (Stomolophus meleagris). Adv. Mater. Res. 2011, 343–344, 505–512. [Google Scholar] [CrossRef]
- Neklyudov, A.D. Nutritive fibers of animal origin: Collagen and its fractions as essential components of new and useful food products. Appl. Biochem. Microbiol. 2003, 39, 229–238. [Google Scholar] [CrossRef]
- Bourtoom, T. Edible films and coatings: Characteristics and properties. Int. Food Res. J. 2008, 15, 237–248. [Google Scholar]
- Gennadios, A.; Hanna, M.A.; Kurth, L.B. Application of edible coatings on meats, poultry and seafoods: A review. LWT-Food Sci. Technol. 1997, 30, 337–350. [Google Scholar] [CrossRef]
- Fang, J.; Fowler, P.; Escrig, C.; Gonzalez, R.; Costa, J.; Chamudis, L. Development of biodegradable laminate films derived from naturally occurring carbohydrate polymers. Carbohydr. Polym. 2005, 60, 39–42. [Google Scholar] [CrossRef]
- Martucci, J.F.; Ruseckaite, R.A. Biodegradation of three-layer laminate films based on gelatin under indoor soil conditions. Polym. Degrad. Stab. 2009, 94, 1307–1313. [Google Scholar] [CrossRef]
- Martucci, J.; Ruseckaite, R. Three-layer sheets based on gelatin and poly (lactic acid), part 1: Preparation and properties. J. Appl. Polym. Sci. 2010, 118, 3102–3110. [Google Scholar] [CrossRef]
- Ikura, K.; Kometani, T.; Sasaki, R.; Chiba, H. Crosslinking of Soybean 7S and 11S Proteins by Transglutaminase. Agric. Biol. Chem. 1980, 44, 2979–2984. [Google Scholar]
- Peña, C.; Martin, M.D.; Retegi, A.; de la Caba, K.; Ruseckaite, R.; Mondragon, I. COMATCOMP09 (V International Conference on Science and Technology of Composite Materials, Extended Abstracts. Donostia-San Sebastián 2009. [Google Scholar]
- Ramos, M.; Valdes, A.; Beltran, A.; Garrigós, M.C. Gelatin-based films and coatings for food packaging applications. Coatings 2016, 6, 41. [Google Scholar] [CrossRef]
- Bower, C.; Avena-Bustillos, R.; Olsen, C.; McHugh, T.; Bechtel, P. Characterization of fish-skin gelatin gels and films containing the antimicrobial enzyme lysozyme. J. Food Sci. 2006, 71, M141–M145. [Google Scholar] [CrossRef]
- Yanwong, S.; Threepopnatkul, P. Effect of peppermint and citronella essential oils on properties of fish skin gelatin edible films. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2015; p. 012064. [Google Scholar]
- Gómez-Guillén, M.; Giménez, B.; López-Caballero, M.A.; Montero, M. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocoll. 2011, 25, 1813–1827. [Google Scholar] [CrossRef]
- Wu, J.; Liu, H.; Ge, S.; Wang, S.; Qin, Z.; Chen, L.; Zheng, Q.; Liu, Q.; Zhang, Q. The preparation, characterization, antimicrobial stability and in vitro release evaluation of fish gelatin films incorporated with cinnamon essential oil nanoliposomes. Food Hydrocoll. 2015, 43, 427–435. [Google Scholar] [CrossRef]
- Lauritano, C.; Ianora, A. Grand challenges in marine biotechnology: Overview of recent EU-funded projects. In Grand Challenges in Marine Biotechnology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 425–449. [Google Scholar]
- Jaspars, M.; De Pascale, D.; Andersen, J.H.; Reyes, F.; Crawford, A.D.; Ianora, A. The marine biodiscovery pipeline and ocean medicines of tomorrow. J. Mar. Biol. Assoc. U. K. 2016, 96, 151–158. [Google Scholar] [CrossRef]
- Lauritano, C.; Ferrante, M.I.; Rogato, A. Marine natural products from microalgae: An-omics overview. Mar. Drugs 2019, 17, 269. [Google Scholar] [CrossRef]


| Species | Tissue | Collagen Content | Reference |
|---|---|---|---|
| Pisces | |||
| Priacanthus tayenus (Richardson, 1846) | bone | 1.6 | [15] |
| skin | 10.9* | [15] | |
| Hemibragus macropterus (Bleeker, 1870) | skin | 28.0* | [16] |
| Syngnathus schlegeli (Kaup, 1856) | skin | 33.2* | [17] |
| Lagocephalus gloveri (Abe and Tabeta, 1983) | skin | 54.3* | [18] |
| Takifugu rubripes (Temminck and Schlegel, 1850) | skin | 44.7* | [19] |
| Saurida spp. | scales | 0.79 | [20] |
| Trachurus japonicas (Temminck and Schlegel, 1844) | scales | 1.5 | [20] |
| Mugil cephalis (Linnaeus, 1758) | scales | 0.4 | [20] |
| Cheilopogon melanurus (Valenciennes, 1847) | scales | 0.7 | [20] |
| Dentex tumifrons (Temminck and Schlegel, 1843) | scales | 0.9 | [20] |
| Mollusca | |||
| Illex argentinus (Castellanos, 1960) | skin | 53 | [21] |
| Sepiella inermis (Van Hasselt, 1835) | skin | 16.2* | [22] |
| Elasmobranchii | |||
| Chiloscyllium punctatum (Müller and Henle, 1838) | cartilage | 9.59* | [23] |
| Carcharhinus limbatus (Müller and Henle, 1839) | cartilage | 10.3* | [23] |
| Echinoidea | |||
| Paracentrotus lividus (Lamarck, 1816) | whole | 7* | [24] |
| Anthocidaris crassispina (Agassiz, 1864) | whole | 35* | [25] |
| Asteroidea | |||
| Patiria pectinifera (Muller and Troschel, 1842) | body wall | 6.1 | [26] |
| Scyphomedusae | |||
| Aurelia aurita (Linnaeus, 1758) | whole | 0.01 | [27] |
| Chrysaora sp. | bell | 9–19 | [28] |
| Pelagia noctiluca (Forsskål, 1775) | whole | 0.07 | [27] |
| Catostylus tagi (Haeckel, 1869) | bell | 2.7* | [29] |
| bell | 4.5 | [27] | |
| Cotylorhiza tuberculate (Macri, 1778) | oral arms | 19.4 | [27] |
| bell | < 10* | [27] | |
| bell | 8.3–31.5 | [27] | |
| Rhizostoma pulmo (Macri, 1778) | oral arms | 26–90 | [27] |
| bell | < 10* | [27] | |
| Rhopilema asamushi (Uchida, 1927) | - | 35.2* | [30] |
| Rhopilema esculentum (Kishinouye, 1891) | mesoglea | 0.28* | [31] |
| Stomolophus meleagris (Agassiz, 1860) | mesoglea | 46.4* | [32] |
| Nemopilema nomurai (Kishinouye, 1922) | mesoglea | 2.2* | [33] |
| Collagen Type | Tissue |
|---|---|
| I | Bone, skin, tendon, ligaments, cornea |
| II | Cartilage, vitreous body, nucleus pulposus |
| III | Skin, vessel walls, reticular fibers of lungs, liver, spleen |
| IV | Basement membranes |
| VI | Cornea (often associated with type I collagen) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coppola, D.; Oliviero, M.; Vitale, G.A.; Lauritano, C.; D’Ambra, I.; Iannace, S.; de Pascale, D. Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications. Mar. Drugs 2020, 18, 214. https://doi.org/10.3390/md18040214
Coppola D, Oliviero M, Vitale GA, Lauritano C, D’Ambra I, Iannace S, de Pascale D. Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications. Marine Drugs. 2020; 18(4):214. https://doi.org/10.3390/md18040214
Chicago/Turabian StyleCoppola, Daniela, Maria Oliviero, Giovanni Andrea Vitale, Chiara Lauritano, Isabella D’Ambra, Salvatore Iannace, and Donatella de Pascale. 2020. "Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications" Marine Drugs 18, no. 4: 214. https://doi.org/10.3390/md18040214
APA StyleCoppola, D., Oliviero, M., Vitale, G. A., Lauritano, C., D’Ambra, I., Iannace, S., & de Pascale, D. (2020). Marine Collagen from Alternative and Sustainable Sources: Extraction, Processing and Applications. Marine Drugs, 18(4), 214. https://doi.org/10.3390/md18040214










