Anti-Fouling Effects of Saponin-Containing Crude Extracts from Tropical Indo-Pacific Sea Cucumbers
Abstract
1. Introduction
2. Results
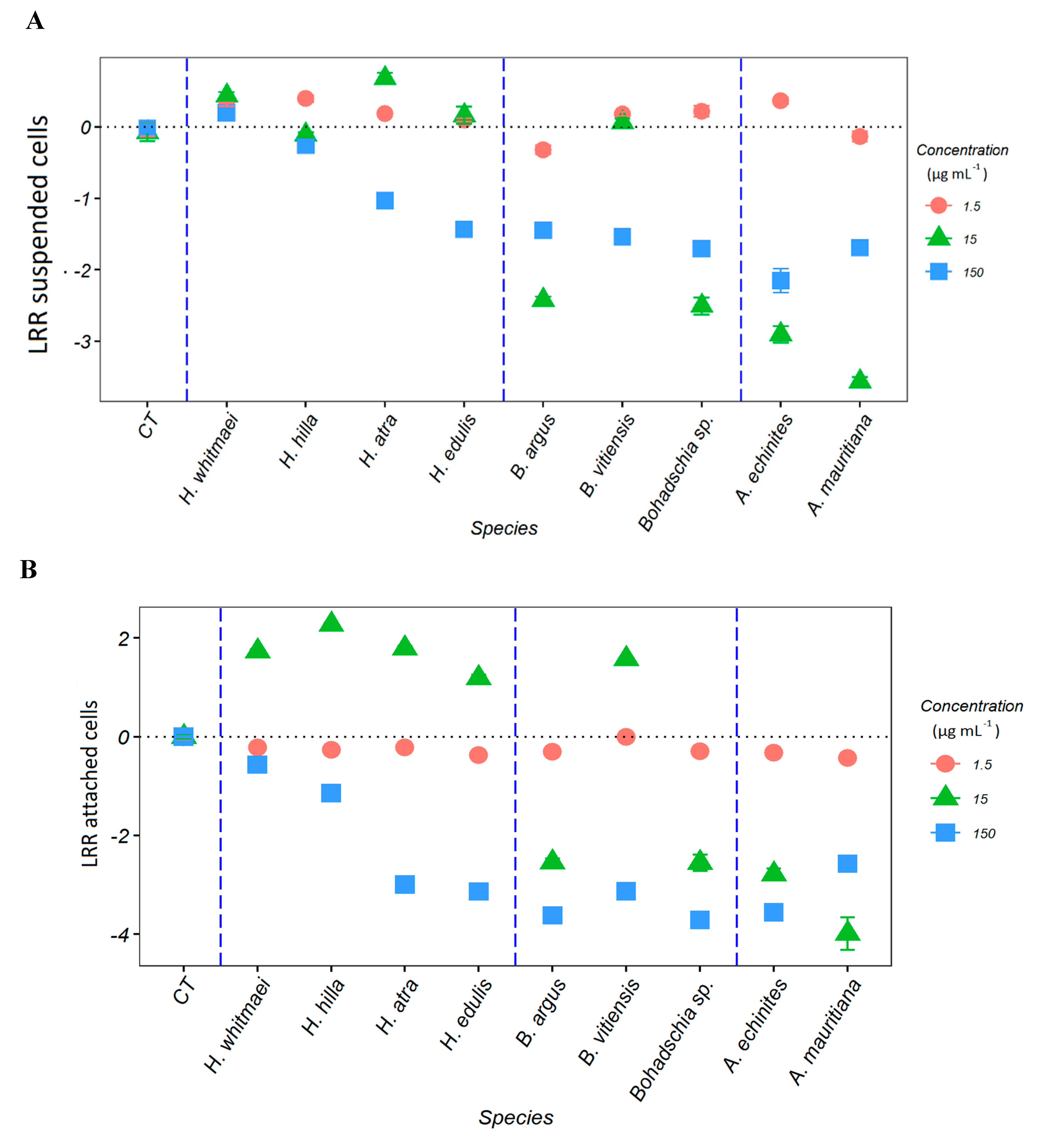
2.1. Anti-Fouling Effects of the Crude Extracts
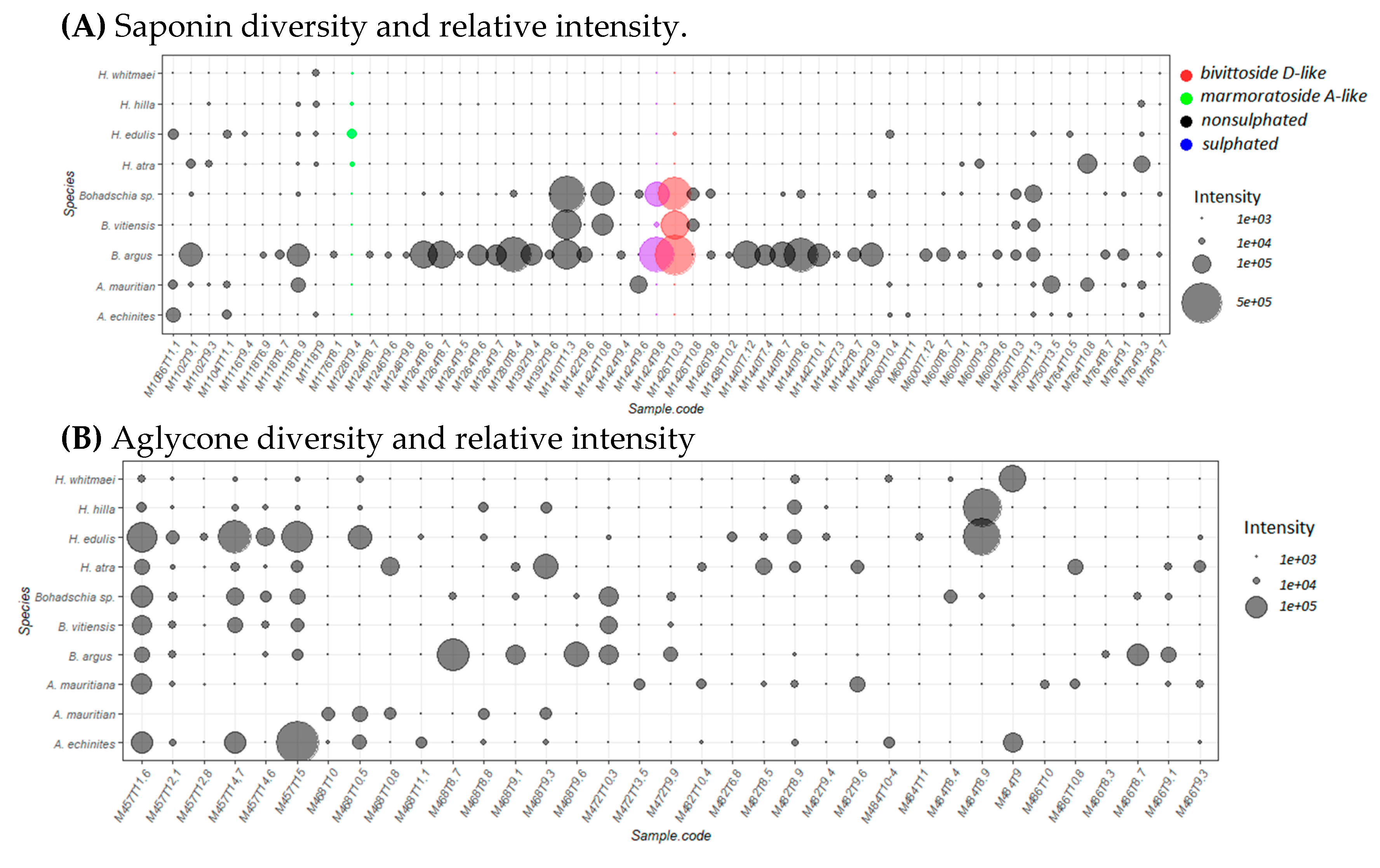
2.2. Saponin Profile of the Crude Extracts
2.2.1. Saponin Composition
2.2.2. Total Saponin Concentration
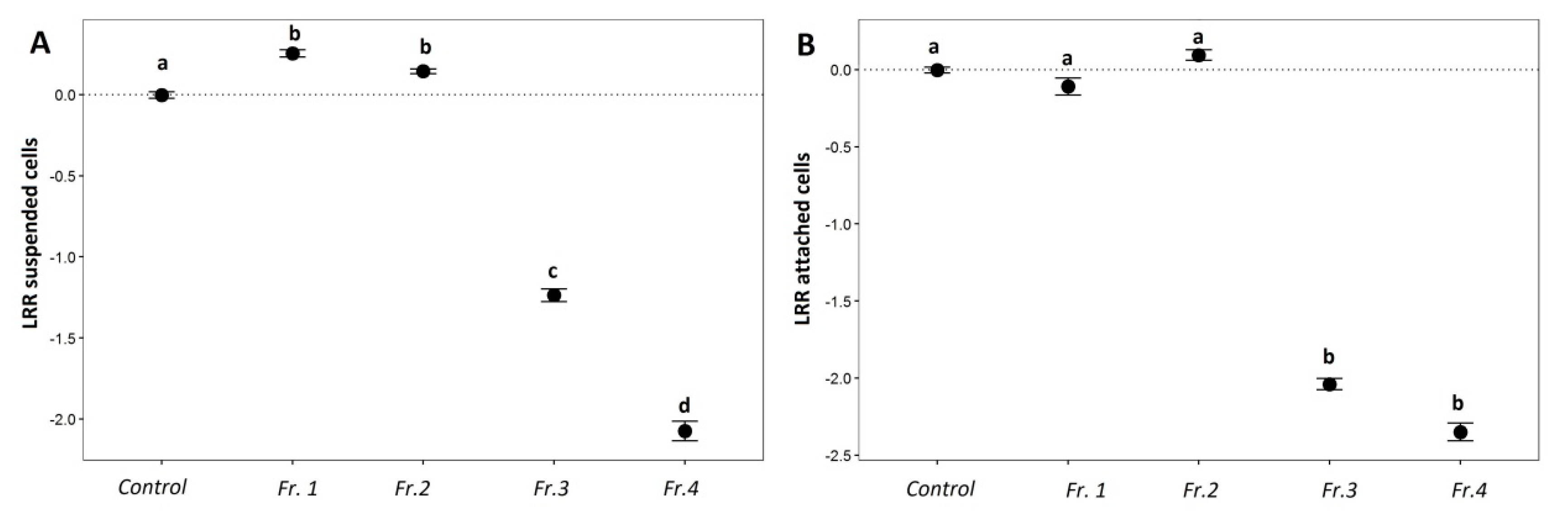
2.3. Anti-Fouling Effects of Purified Saponin Fractions and Pure Compounds
2.3.1. AF Assay with an Emphasis on Saponins
2.3.2. Saponin Profile of the Fractions
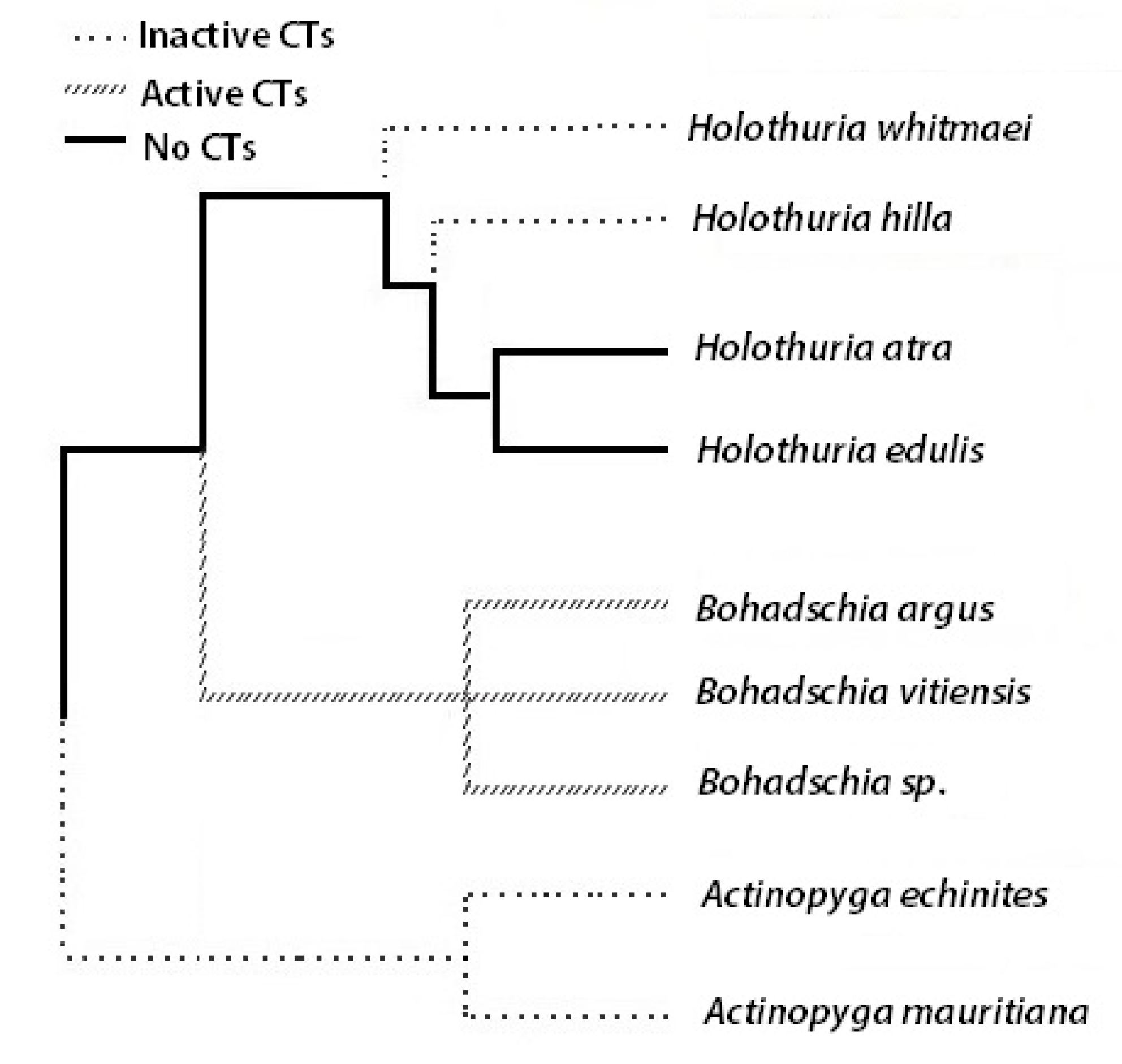
3. Discussion
4. Materials and Methods
4.1. Experimental Setup
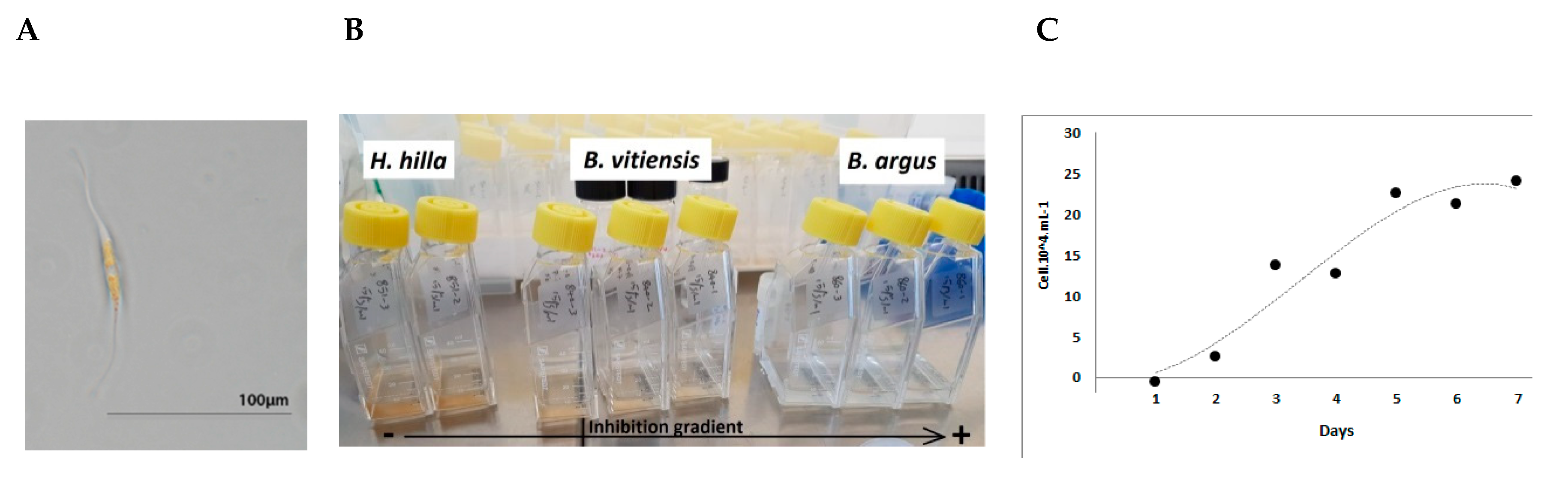
4.1.1. Cylindrotheca Closterium Culture
4.1.2. Preparation of Sea Cucumber Crude Extracts
4.1.3. Anti-Fouling Assay: Experimental Design
4.1.4. Diatom Growth and Settlement Analyses
4.1.5. Anti-Fouling Effects: Data and Statistical Analyses
4.2. Saponins as Potential Bioactive Compounds Affecting the Fouling Organism C. closterium
4.2.1. Dereplication of Saponins
4.2.2. Saponin Compounds Composition: Data and Statistical Analyses
4.2.3. Total Saponin Concentration within the Examined Sea Cucumber Species
4.3. Anti-Fouling Effects of Purified Saponin Fractions
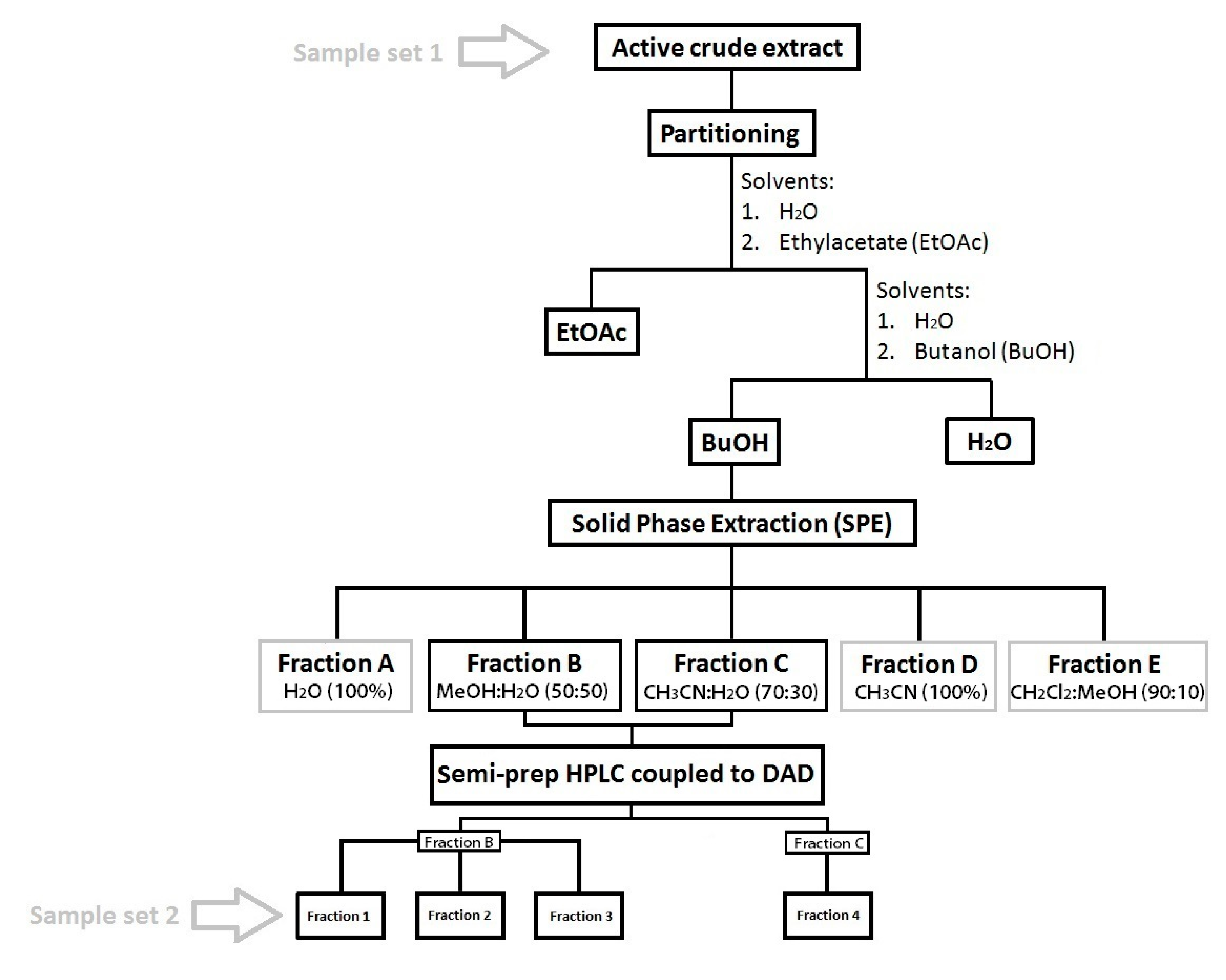
4.4. Sample Fractionation and Purification
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Briand, J.F. Marine antifouling laboratory bioassays: An overview of their diversity. Biofouling 2009, 25, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Railkin, A. Marine biofouling: Colonization processes and defenses; CSC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- McKenzie, J.D.; Grigolava, I.V. The echinoderm surface and its role in preventing microfouling. Biofouling 1996, 10, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Wahl, M. Marine epibiosis.I. fouling and antifouling: Some basice aspects. Mar. Ecol. Prog. Ser. 1989, 58, 175–189. [Google Scholar] [CrossRef]
- Davis, A.R.; Targett, N.M.; McConnell, O.J.; Young, C.M. Epibiosis of Marine Algae and Benthic Invertebrates: Natural Products Chemistry and Other Mechanisms Inhibiting Settlement and Overgrowth; Scheuer, P.J., Ed.; Bioorganic; Springer: Berlin/Heidelberg, Germany, 1989; Vol. 3. [Google Scholar]
- Rohde, S.; Hiebenthal, C.; Wahl, M.; Karez, R.; Bischof, K. Decreased depth distribution of Fucus vesiculosus (Phaeophyceae) in the Western Baltic: Effects of light deficiency and epibionts on growth and photosynthesis. Eur. J. Phycol. 2008, 43, 143–150. [Google Scholar] [CrossRef]
- Yebra, D.M.; Kiil, S.; Dam-Johansen, K. Antifouling technology—Past, present and future steps towards efficient and environmentally friendly antifouling coatings. Prog. Org. Coatings 2004, 50, 75–104. [Google Scholar] [CrossRef]
- Amara, I.; Miled, W.; Slama, R.B.; Ladhari, N. Antifouling processes and toxicity effects of antifouling paints on marine environment. A review. Environ. Toxicol. Pharmacol. 2018, 57, 115–130. [Google Scholar] [CrossRef]
- Puentes, C.; Carreño, K.; Santos-Acevedo, M.; Gómez-León, J.; García, M.; Pérez, M.; Stupak, M.; Blustein, G. Anti-fouling paints based on extracts of marine organisms from the colombian varibbean. Sh. Sci. Technol. 2014, 8, 75–90. [Google Scholar]
- Alzieu, C.; Sanjuan, J.; Michel, P.; Borel, M.; Dreno, J.P. Monitoring and assessement of butyltins in Atlantic coastal waters. Mar. Pollut. Bull. 1989, 20, 22–26. [Google Scholar] [CrossRef]
- Ellis, D.V. New dangerous chemicals in the environment: Lessons from TBT. Mar. Pollut. Bull. 1991, 22, 8–10. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Kato, H.; Hirota, H.; Fusetani, N. Antifouling terpenes and steroids against barnacle larvae from marine sponges. Biofouling 1997, 11, 283–291. [Google Scholar] [CrossRef]
- Champ, M.A. The status of the treaty to ban TBT in marine antifouling paints and alternatives. In Proceedings of the 24th UJNR (US/Japan) Marine Facilities Panel Meeting, Honolulu, HI, USA, 7–8 November 2001; pp. 1–7. [Google Scholar]
- Singh, P.; Cameotra, S.S. Potential applications of microbial surfactants in biomedical sciences. Trends Biotechnol. 2004, 22, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.H.; Zhang, S.; Yang, L.H.; Qian, P.Y. Antifouling and antibacterial compounds from the gorgonians Subergorgia suberosa and Scripearia gracillis. Nat. Prod. Res. 2008, 22, 154–166. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, E.; Boyd, K.G.; Pisacane, A.; Peppiatt, C.J.; Burgess, J.G. Marine microbial natural products in antifouling coatings. Biofouling 2000, 16, 215–224. [Google Scholar] [CrossRef]
- Dusane, D.H.; Pawar, V.S.; Nancharaiah, Y.V.; Venugopalan, V.P.; Kumar, A.R.; Zinjarde, S.S. Anti-biofilm potential of a glycolipid surfactant produced by a tropical marine strain of Serratia marcescens. Biofouling 2011, 27, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Tello, E.; Castellanos, L.; Arevalo-Ferro, C.; Rodríguez, J.; Jiménez, C.; Duque, C. Absolute stereochemistry of antifouling cembranoid epimers at C-8 from the Caribbean octocoral Pseudoplexaura flagellosa. Revised structures of plexaurolones. Tetrahedron 2011, 67, 9112–9121. [Google Scholar] [CrossRef]
- Salta, M.; Wharton, J.A.; Stoodley, P.; Dennington, S.P.; Goodes, L.R.; Werwinski, S.; Mart, U.; Wood, R.J.K.; Stokes, K.R. Designing biomimetic antifouling surfaces. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 4729–4754. [Google Scholar] [CrossRef]
- Jadhav, S.; Shah, R.; Bhave, M.; Palombo, E.A. Inhibitory activity of yarrow essential oil on Listeria planktonic cells and biofilms. Food Control 2013, 29, 125–130. [Google Scholar] [CrossRef]
- Majik, M.; Parvatkar, P. Next Generation Biofilm Inhibitors for Pseudomonas aeruginosa: Synthesis and Rational Design Approaches. Curr. Top. Med. Chem. 2014, 14, 81–109. [Google Scholar] [CrossRef]
- Okino, T.; Yoshimura, E.; Hirota, H.; Fusetani, N. New antifouling kalihipyrans from the marine sponge Acanthella cavernosa. J. Nat. Prod. 1996, 59, 1081–1083. [Google Scholar] [CrossRef]
- Bryan, P.J.; Mcclintock, J.B.; Hopkins, T.S. Structural and chemical defenses of echinoderms from the northern Gulf of Mexico. Exp. Mar. Biol. Ecol. 1997, 210, 173–186. [Google Scholar] [CrossRef]
- Wright, A.D.; McCluskey, A.; Robertson, M.J.; MacGregor, K.A.; Gordon, C.P.; Guenther, J. Anti-malarial, anti-algal, anti-tubercular, anti-bacterial, anti-photosynthetic, and anti-fouling activity of diterpene and diterpene isonitriles from the tropical marine sponge Cymbastela hooperi. Org. Biomol. Chem. 2011, 9, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Canicattí, C.; D’Ancona, G. Biological protective substances in Marthasterias glacialis (Asteroidea) epidermal secretion. J. Zool. 1990, 222, 445–454. [Google Scholar] [CrossRef]
- Pawlik, J.R. Marine Invertebrate Chemical Defenses. Chem. Rev. 1993, 93, 1911–1922. [Google Scholar] [CrossRef]
- Key, M.M.; Jeffries, W.B.; Voris, H.K. Epizoic bryozoans, sea snakes, and other nektonic substrates. Bull. Mar. Sci. 1995, 56, 462–474. [Google Scholar]
- Krupp, D.A. An immunochemical study of the mucus from the solitary coral Fungia scutaria (Scleractinia, Fungiidae). Bull. Mar. Sci. 1985, 36, 163–176. [Google Scholar]
- Wahl, M.; Kröger, K.; Lenz, M. Non-toxic protection against epibiosis. Biofouling 1998, 12, 205–226. [Google Scholar] [CrossRef]
- Fusetani, N. Biofouling and antifouling. Nat. Prod. Rep. 2004, 21, 94–104. [Google Scholar] [CrossRef]
- Qian, P.Y.; Xu, Y.; Fusetani, N. Natural products as antifouling compounds: Recent progress and future perspectives. Biofouling 2010, 26, 223–234. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine Natural Products. Nat. Prod. Chem. Biol. 1994, 11, 355–394. [Google Scholar] [CrossRef]
- Ortlepp, S.; Sjögren, M.; Dahlström, M.; Weber, H.; Ebel, R.; Edrada, R.A.; Thoms, C.; Schupp, P.; Bohlin, L.; Proksch, P. Antifouling activity of bromotyrosine-derived sponge metabolites and synthetic analogues. Mar. Biotechnol. 2007, 9, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Kjelleberg, S.; Steinberg, P.; Givskov, M.; Gram, L.; Manefield, M.; De Nys, R. Do marine natural products interfere with prokaryotic AHL regulatory systems? Aquat. Microb. Ecol. 1997, 13, 85–93. [Google Scholar] [CrossRef]
- Qi, S.H.; Ma, X. Antifouling compounds from marine invertebrates. Mar. Drugs 2017, 15, 263. [Google Scholar]
- De Marino, S.; Iorizzi, M.; Zollo, F.; Amsler, C.D.; Greer, S.P.; McClintock, J.B. Three new asterosaponins from the starfish Goniopecten demonstrans. European J. Org. Chem. 2000, 2000, 4093–4098. [Google Scholar] [CrossRef]
- Dobretsov, S.; Al-Mammari, I.M.; Soussi, B. Bioactive Compounds from Omani Sea Cucumbers. Agric. Mar. Sci. 2009, 14, 49–53. [Google Scholar] [CrossRef][Green Version]
- Soliman, Y.A.; Ibrahim, A.M.; Tadros, H.R.Z.; Abou-Taleb, E.A.; Moustafa, A.H.; Hamed, M.A. Antifouling and antibacterial activities of marine bioactive compounds extracted from some Red Sea sea cucumber. Int. J. Contemp. Appl. Sci. 2016, 3, 83–103. [Google Scholar]
- Dobretsov, S.; Teplitski, M.; Paul, V. Mini-review: Quorum sensing in the marine environment and its relationship to biofouling. Biofouling 2009, 25, 413–427. [Google Scholar] [CrossRef]
- Acevedo, M.S.; Puentes, C.; Carreño, K.; León, J.G.; Stupak, M.; García, M.; Pérez, M.; Blustein, G. Antifouling paints based on marine natural products from Colombian Caribbean. Int. Biodeterior. Biodegrad. 2013, 83, 97–104. [Google Scholar] [CrossRef]
- Ozupek, N.M.; Cavas, L. Triterpene glycosides associated antifouling activity from Holothuria tubulosa and Holothuria polii. Reg. Stud. Mar. Sci. 2017, 13, 32–41. [Google Scholar] [CrossRef]
- Kamyab, E.; Kellermann, M.Y.; Kunzmann, A.; Schupp, P.J. Chemical Biodiversity and Bioactivities of Saponins in Echinodermata with an Emphasis on Sea Cucumbers (Holothuroidea). In YOUMARES 9-The Oceans: Our Research, Our Future; Jungblut, S., Liebich, V., Bode-Dalby, M., Eds.; Springer: Cham, Switzerland, 2020; pp. 121–157. [Google Scholar]
- Van Dyck, S.; Flammang, P.; Meriaux, C.; Bonnel, D.; Salzet, M.; Fournier, I.; Wisztorski, M. Localization of secondary metabolites in marine invertebrates: Contribution of MALDI MSI for the study of saponins in Cuvierian tubules of H. forskali. PLoS ONE 2010, 5, e13923. [Google Scholar] [CrossRef]
- Popov, A.M. A comparative study of the hemolytic and cytotoxic activities of triterpenoids isolated from ginseng and sea cucumbers. Biol. Bull. 2002, 29, 120–128. [Google Scholar] [CrossRef]
- Van Dyck, S.; Gerbaux, P.; Flammang, P. Qualitative and quantitative saponin contents in five sea cucumbers from the Indian ocean. Mar. Drugs 2010, 8, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Kingston, M.B. Growth and motility of the diatom Cylindrotheca closterium: Implications for commercial applications. J. North Carolina Acad. Sci. 2009, 125, 138–142. [Google Scholar]
- Tanaka, N. The cell division rates of ten species of attaching diatoms in natural seawater. Bull. Japanese Soc. Sci. Fish. 1984, 50, 969–972. [Google Scholar] [CrossRef][Green Version]
- Aslam, S.N.; Strauss, J.; Thomas, D.N.; Mock, T.; Underwood, G.J.C. Identifying metabolic pathways for production of extracellular polymeric substances by the diatom Fragilariopsis cylindrus inhabiting sea ice. ISME J. 2018, 12, 1237–1251. [Google Scholar] [CrossRef]
- Decho, A.W. Microbial exopolymer secretions in ocean environments: Their role(s) in food webs and marine processes. Ocean. Mar. Biol. Annu. Rev. 1990, 28, 73–153. [Google Scholar]
- Stal, L.J.; Défarge, C. Structure and dynamics of exopolymers in an intertidal diatom biofilm. Geomicrobiol. J. 2005, 22, 341–352. [Google Scholar] [CrossRef][Green Version]
- Wimpenny, J.; Manz, W.; Szewzyk, U. Heterogeneity in biofilms. FEMS Microbiol. Rev. 2000, 24, 661–671. [Google Scholar] [CrossRef]
- Apoya-Horton, M.D.; Yin, L.; Underwood, G.J.C.; Gretz, M.R. Movement modalities and responses to environmental changes of the mudflat diatom Cylindrotheca closterium (Bacillariophyceae). J. Phycol. 2006, 42, 379–390. [Google Scholar] [CrossRef]
- Roberts, R.D.; Kawamura, T.; Handley, C.M. Factors affecting settlement of abalone (Haliotis Iris) larvae on benthic diatom films. J. Shellfish Res. 2007, 26, 323–334. [Google Scholar] [CrossRef]
- Gallardo, W.G.; Buen, S.M.A. Evaluation of mucus, Navicula, and mixed diatoms as larval settlement inducers for the tropical abalone Haliotis asinina. Aquaculture 2003, 221, 357–364. [Google Scholar] [CrossRef]
- Hellio, C.; Berge, J.P.; Beaupoil, C.; Le Gal, Y.; Bourgougnon, N. Screening of marine algal extracts for anti-settlement activities against microalgae and macroalgae. Biofouling 2002, 18, 205–215. [Google Scholar] [CrossRef]
- Kitagawa, I.; Kobayashi, M.; Hori, M.; Kyogoku, Y. Structures of four new triterpenoidal oligoglycosides, Bivittoside A, B, C, and D, from the sea cucumber Bohadschia bivittata MITSUKURI. Chem. Pharm. Bull 1981, 29, 282–285. [Google Scholar] [CrossRef]
- Kerr, A.M.; Kim, J. Phylogeny of Holothuroidea (Echinodermata) inferred from morphology. Zool. J. Linn. Soc. 2001, 133, 63–81. [Google Scholar] [CrossRef]
- Kamarudin, K.R.; Ridzwan, H.; Usup, G. Phylogeny of sea cucumber (Echinodermata: Holothuroidea) as inferred from 16s mitochondrial rRNA gene sequences. Sains Malaysiana 2010, 39, 209–218. [Google Scholar]
- Wen, J.; Hu, C.; Zhang, L.; Fan, S. Genetic identification of global commercial sea cucumber species on the basis of mitochondrial DNA sequences. Food Control 2011, 21, 72–77. [Google Scholar] [CrossRef]
- Miller, A.K.; Kerr, A.M.; Paulay, G.; Reich, M.; Wilson, N.G.; Carvajal, J.I.; Rouse, G.W. Molecular phylogeny of extant Holothuroidea (Echinodermata). Mol. Phylogenet. Evol. 2017, 111, 110–131. [Google Scholar] [CrossRef]
- Antoine, D.; Andre, J.; Morel, A. Oceanic primary production 2. Estimation at global scale from satellite (coastal zone color scanner) chlorophyll. Global Biogeochem. Cycles 1996, 10, 57–69. [Google Scholar] [CrossRef]
- Thrane, J.E.; Kyle, M.; Striebel, M.; Haande, S.; Grung, M.; Rohrlack, T.; Andersen, T. Spectrophotometric analysis of pigments: A critical assessment of a high-throughput method for analysis of algal pigment mixtures by spectral deconvolution. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Gerhard, M.; Koussoroplis, A.M.; Hillebrand, H.; Striebel, M. Phytoplankton community responses to temperature fluctuations under different nutrient concentrations and stoichiometry. Ecology 2019, 100, e02834. [Google Scholar] [CrossRef]
- Iorizzi, M.; Bryan, P.; McClintock, J.; Minale, L.; Palagiano, E.; Maurelli, S.; Riccio, R.; Zollo, F. Chemical and biological investigation of the polar constituents of the starfish Luidia clathrata, collected in the gulf of mexico. J. Nat. Prod. 1995, 58, 653–671. [Google Scholar] [CrossRef] [PubMed]
- Kubanek, J.; Whalen, K.E.; Engel, S.; Kelly, S.R.; Henkel, T.P.; Fenical, W.; Pawlik, J.R. Multiple defensive roles for triterpene glycosides from two Caribbean sponges. Oecologia 2002, 131, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Bers, A.V.; D’Souza, F.; Klijnstra, J.W.; Willemsen, P.R.; Wahl, M. Chemical defence in mussels: Antifouling effect of crude extracts of the periostracum of the blue mussel Mytilus edulis. Biofouling 2006, 22, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, N.; Dobretsov, S.; Rohde, S.; Schupp, P.J. Comparison of antifouling properties of native and invasive Sargassum (Fucales, Phaeophyceae) species. Eur. J. Phycol. 2017, 52, 116–131. [Google Scholar] [CrossRef]
- Hellio, C.; De La Broise, D.; Dufossé, L.; Le Gal, Y.; Bourgougnon, N. Inhibition of marine bacteria by extracts of macroalgae: Potential use for environmentally friendly antifouling paints. Mar. Environ. Res. 2001, 52, 231–247. [Google Scholar] [CrossRef]
- Gonsalves, C.O.L. Effect of holothurian and zoanthid extracts on growth of some bacterial and diatom species. Indian J. Mar. Sci. 1997, 26, 377–379. [Google Scholar]
- Stebbing, A.R.D. Hormesis-The stimulation of growth by low levels of inhibitors. Sci. Total Environ. 1982, 22, 213–234. [Google Scholar] [CrossRef]
- Stebbing, A.R.D. A theory for growth hormesis. Mutat. Res. Mol. Mech. Mutagen. 1998, 403, 249–258. [Google Scholar] [CrossRef]
- Riccio, R.; Iorizzi, M.; Minale, L.; Oshima, Y.; Yasumoto, T. Starfish saponins. Part 34. Novel steroidal glycoside sulphates from the starfish Asterias amurensis. J. Chem. Soc. Perkin Trans. 1 1988, 6, 1337–1347. [Google Scholar] [CrossRef]
- Haug, T.; Kjuul, A.K.; Styrvold, O.B.; Sandsdalen, E.; Olsen, Ø.M.; Stensvag, K. Antibacterial activity in Strongylocentrotus droebachiensis (Echinoidea), Cucumaria frondosa (Holothuroidea), and Asterias rubens (Asteroidea). J. Invertebr. Pathol. 2002, 81, 94–102. [Google Scholar] [CrossRef]
- Lorent, J.H.; Quetin-Leclercq, J.; Mingeot-Leclercq, M.P. The amphiphilic nature of saponins and their effects on artificial and biological membranes and potential consequences for red blood and cancer cells. Org. Biomol. Chem. 2014, 12, 8803–8822. [Google Scholar] [CrossRef]
- Selvin, J.; Lipton, A.P. Antifouling activity of bioactive substances extracted from Holothuria scabra. Hydrobiologia 2004, 513, 251–253. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Avilov, S.A.; Silchenko, A.S.; Stonik, V.A. Triterpene glycosides of sea cucumbers (Holothuroidea, Echinodermata) as Taxonomic Markers. Nat. Prod. Commun. 2015, 10, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Caulier, G.; Van Dyck, S.; Gerbaux, P.; Eeckhaut, I.; Flammang, P. Review of saponin diversity in sea cucumbers belonging to the family Holothuriidae. SPC Beche-de-mer Inf. Bull. 2011, 31, 48–54. [Google Scholar]
- Kitagawa, I.; Kobayashi, M.; Hori, M.; Kyogoku, Y. Marine Natural Producs. XVIII. Four lanostane- type triterpene oligoglycosides, bivittosides A,B,C, and D from the Okinawan sea cucumber Bohadschia bivittata (Mitsukuri). Chem. Pharm. Bull. 1989, 37, 61–67. [Google Scholar] [CrossRef]
- Grauso, L.; Yegdaneh, A.; Sharifi, M.; Mangoni, A.; Zolfaghari, B.; Lanzotti, V. Molecular networking-based analysis of cytotoxic saponins from sea cucumber Holothuria atra. Mar. Drugs 2019, 17, 86. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Inamoto, T.; Fuchida, M.; Okada, S.; Kobayashi, M.; Nishino, T.; Kyoboku, Y. Structures of Echinoside A and B, two antifungal oligoglycosides from the sea cucumber Actinopyga echinites (JAEGER). Chem. Pharm. Bull. 1980, 28, 1651–1653. [Google Scholar] [CrossRef]
- Van Dyck, S.; Caulier, G.; Todesco, M.; Gerbaux, P.; Fournier, I.; Wisztorski, M.; Flammang, P. The triterpene glycosides of Holothuria forskali: Usefulness and efficiency as a chemical defense mechanism against predatory fish. J. Exp. Biol. 2011, 214, 1347–1356. [Google Scholar] [CrossRef]
- Honey-Escandón, M.; Arreguín-Espinosa, R.; Solís-Marín, F.A.; Samyn, Y. Biological and taxonomic perspective of triterpenoid glycosides of sea cucumbers of the family Holothuriidae (Echinodermata, Holothuroidea). Comp. Biochem. Physiol. Part-B Biochem. Mol. Biol. 2015, 180, 16–39. [Google Scholar] [CrossRef]
- Olsen, S.M. Controlled release of environmentally friendly antifouling agents from marine coatings. Kgs. Lyngby, Denmark: Technical University of Denmark. 2009. Available online: https://backend.orbit.dtu.dk/ws/portalfiles/portal/5008364/Stefan+M%C3%B8ller+Olsen.pdf (accessed on 20 October 2019).
- Lorent, J.; Le Duff, C.S.; Quetin-Leclercq, J.; Mingeot-Leclercq, M.P. Induction of highly curved structures in relation to membrane permeabilization and budding by the triterpenoid saponins, α- And δ-hederin. J. Biol. Chem. 2013, 288, 14000–14017. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hori, M.; Kan, K.; Yasuzawa, T.; Matsu, M.; Suzuki, S.; Kitagawa, I. Marine Natural Products. XXVII Distribution of Lanostane-type triterpene oligoglycosides in ten kind of Okinawan sea cucumbers. Chem. Pharm. Bull. 1991, 39, 2282–2287. [Google Scholar] [CrossRef]
- Guillard, R.R.L. Culture of Phytoplankton for Feeding Marine Invertebrates. In Culture of Marine Invertebrate Animals: Proceedings-1st Conference on Culture of Marine Invertebrate Animals Greenport; Smith, W.L., Chanley, M.H., Eds.; Springer: Boston, MA, USA, 1975; pp. 29–60. ISBN 978-1-4615-8714-9. [Google Scholar]
- Affan, A.; Heo, S.J.; Jeon, Y.J.; Lee, J.B. Optimal growth conditions and antioxidative activities of Cylindrotheca closterium (bacillariophyceae). J. Phycol. 2009, 45, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, Y.; Zhang, W.; Franco, C. Discovery of novel saponins from the viscera of the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 2633–2667. [Google Scholar] [CrossRef] [PubMed]
- Clastres, A.; Ahond, A.; Poupat, C.; Potier, P.; Intes, A. Marine invertebrates from New-Caledonian Lagoon. I. Structural study of a new sapogenin isolated from a sea-cucumber: Bohadschia vitiensis Semper. Experientia 1978, 34, 973–974. [Google Scholar] [CrossRef]
- Mellado, G.G.; Zubía, E.; Ortega, M.J.; López-González, P.J. Steroids from the antarctic octocoral Anthomastus bathyproctus. J. Nat. Prod. 2005, 68, 1111–1115. [Google Scholar] [CrossRef]
- Ioannou, E.; Abdel-Razik, A.F.; Alexi, X.; Vagias, C.; Alexis, M.N.; Roussis, V. 9,11-Secosterols with antiproliferative activity from the gorgonian Eunicella cavolini. Bioorganic Med. Chem. 2009, 17, 4537–4541. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Malyutin, A.N.; Stonik, V.A. Caudinoside A—A novel triterpene glycoside from the holothurian Paracaudina ransonetii. CA 105: 169209x. Khim. Prir. Soedin 1986, 378–379. [Google Scholar]
- Elyakov, G.B.; Kuznetsova, T.A.; Dzizenko, A.K.; Elkin, Y.N. A chemical investigation of the trepang (Stichopus Japonicus Selenka): The structure of triterpenoid aglycones obtained from trepang glycosides. Tetrahedron Lett. 1969, 10, 1151–1154. [Google Scholar] [CrossRef]
- Bhatnagar, S.; Dudouet, B.; Ahond, A.; Poupat, C.; Thoison, O.; Clastres, A.; Laurent, D.; Potier, P. Invertebres marins du lagon Neocaledonien IV. Saponines et sapogenines d’une holothurie, Actinopyga flammea. Bull. Soc. Chim. Fr. 1985, 124–129. [Google Scholar]
- Radhika, P.; Anjaneyulu, V.; Subba Rao, P.V.; Makarieva, T.N.; Kalinovosky, A.I. Chemical examination of the echinoderms of Indian Ocean: The triterpene glycosides of the sea cucumbers: Holothuria nobilis, Bohadschia aff. tenuissima and Actinopyga mauritana from Lakshadweep, Andaman and Nicobar Islands. Indian J. Chem.-Sect. B Org. Med. Chem. 2002, 41, 1276–1282. [Google Scholar]
- Bahrami, Y.; Zhang, W.; Chataway, T.; Franco, C. Structural elucidation of novel saponins in the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 4439–4473. [Google Scholar] [CrossRef]
- Van Dyck, S.; Gerbaux, P.; Flammang, P. Elucidation of molecular diversity and body distribution of saponins in the sea cucumber Holothuria forskali (Echinodermata) by mass spectrometry. Comp. Biochem. Physiol.-B 2009, 152, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Hiai, S.; Oura, H.; T, N. Color Reaction of Some Sapogenins. Planta Med. 1976, 29, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Cutignano, A.; Nuzzo, G.; Ianora, A.; Luongo, E.; Romano, G.; Gallo, C.; Sansone, C.; Aprea, S.; Mancini, F.; D’Oro, U.; et al. Development and application of a novel SPE-method for bioassay-guided fractionation of marine extracts. Mar. Drugs 2015, 13, 5736–5749. [Google Scholar] [CrossRef] [PubMed]
- Ebada, S.; Edrada, R.A.; Lin, W.; Proksch, P. Methods for isolation, purification and structural elucidation of bioactive secondary metabolites from marine invertebrates. Nature Protocols 2008, 3, 1820–1831. [Google Scholar] [CrossRef] [PubMed]
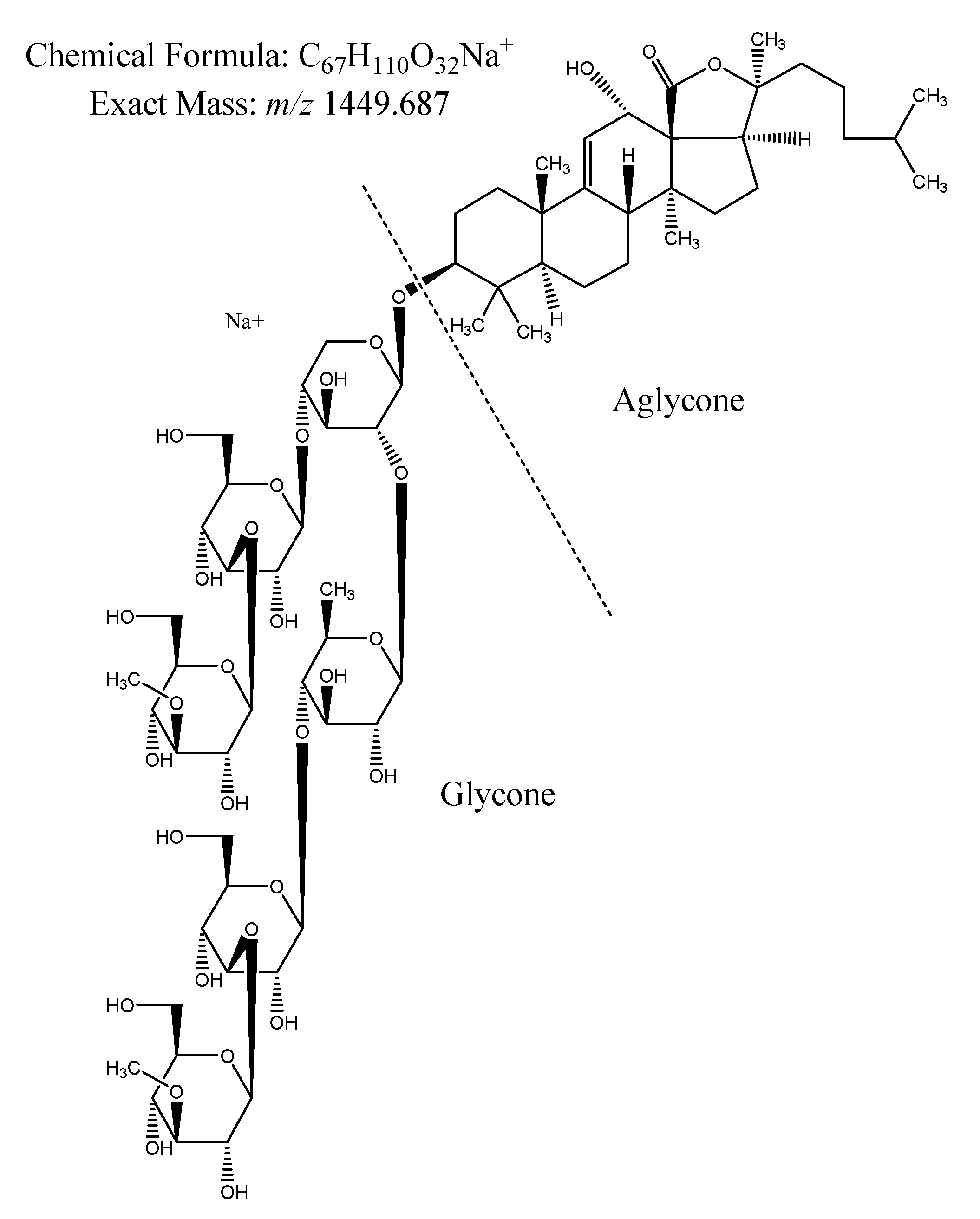


| Diagnostic Ions | Reported Exact Mass (m/z) | Molecular Formula | Organism | References |
|---|---|---|---|---|
| Sapogenin | 472.3552 | C30H48O4 | B. vitiensis | [89] |
| Sapogenin 1 | 482.3032 | C30H42O5 | Octacoral (Anthomastus bathyproctus) | [90] |
| Sapogenin 3 | 457.3318 | C29H45O4 | Gorgonian (Eunicella cavolini) | [91] |
| Caudinoside A | 468.3239 | C30H44O4 | Paracaudina ransonetii | [92] |
| Stichopogenin A4 | 486.3345 | C30H46O5 | Stichopus japonicus | [93] |
| 16 Keto holothurinogenin | 484.3189 | C30H44O5 | A. mauritiana | [94,95] |
| MeGlc-Glc-Qui + Na+ | 507.164 | C19H32O14Na+ | H. lesson, H. forskali | [96,97] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamyab, E.; Goebeler, N.; Kellermann, M.Y.; Rohde, S.; Reverter, M.; Striebel, M.; Schupp, P.J. Anti-Fouling Effects of Saponin-Containing Crude Extracts from Tropical Indo-Pacific Sea Cucumbers. Mar. Drugs 2020, 18, 181. https://doi.org/10.3390/md18040181
Kamyab E, Goebeler N, Kellermann MY, Rohde S, Reverter M, Striebel M, Schupp PJ. Anti-Fouling Effects of Saponin-Containing Crude Extracts from Tropical Indo-Pacific Sea Cucumbers. Marine Drugs. 2020; 18(4):181. https://doi.org/10.3390/md18040181
Chicago/Turabian StyleKamyab, Elham, Norman Goebeler, Matthias Y. Kellermann, Sven Rohde, Miriam Reverter, Maren Striebel, and Peter J. Schupp. 2020. "Anti-Fouling Effects of Saponin-Containing Crude Extracts from Tropical Indo-Pacific Sea Cucumbers" Marine Drugs 18, no. 4: 181. https://doi.org/10.3390/md18040181
APA StyleKamyab, E., Goebeler, N., Kellermann, M. Y., Rohde, S., Reverter, M., Striebel, M., & Schupp, P. J. (2020). Anti-Fouling Effects of Saponin-Containing Crude Extracts from Tropical Indo-Pacific Sea Cucumbers. Marine Drugs, 18(4), 181. https://doi.org/10.3390/md18040181






