A Truncated Galectin-3 Isolated from Skin Mucus of Atlantic Salmon Salmo salar Binds to and Modulates the Proteome of the Gram-Negative Bacteria Moritella viscosa
Abstract
1. Introduction
2. Results
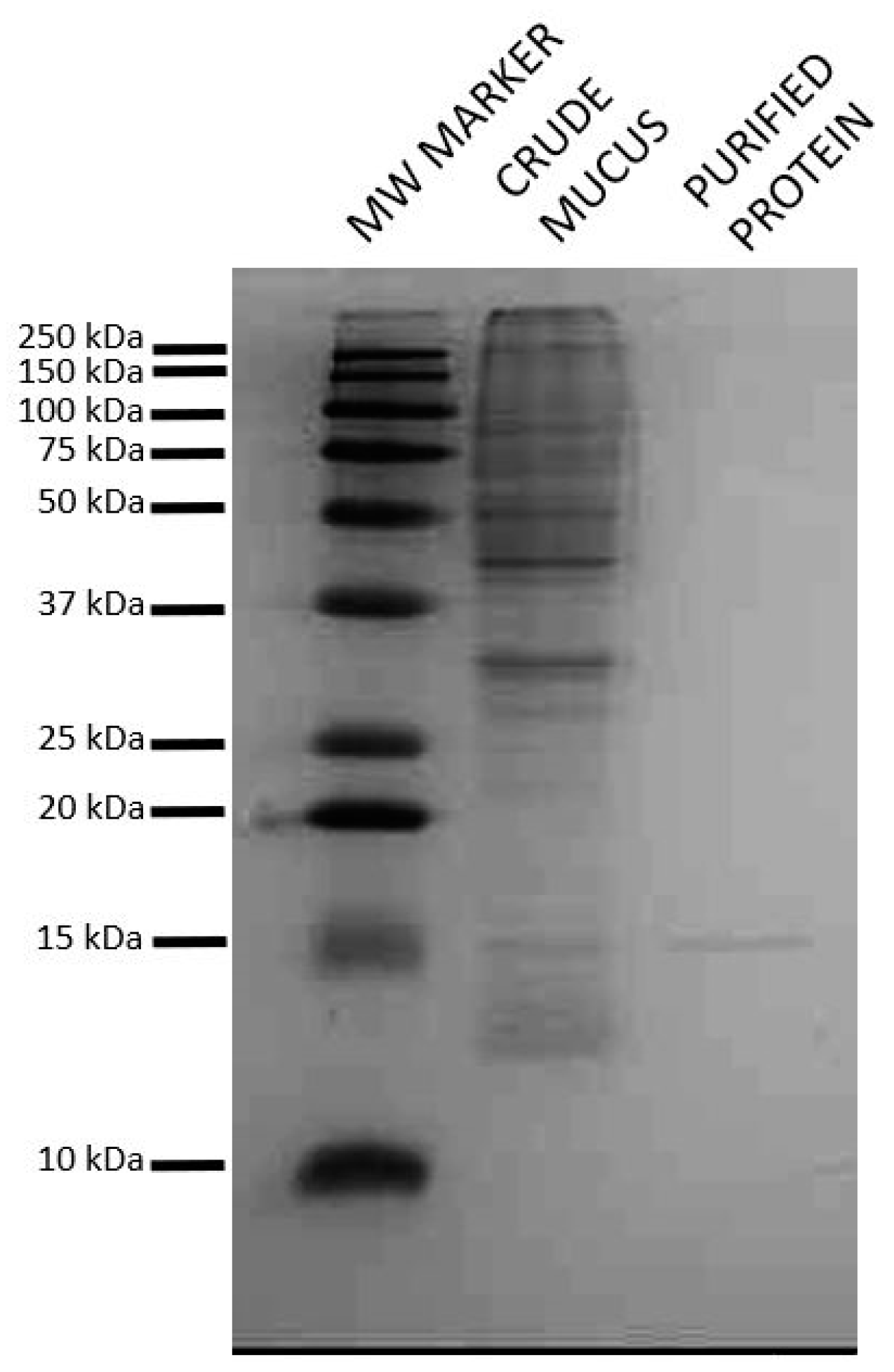
2.1. Isolation of Lactose Binding Protein from Atlantic Salmon Skin Mucus
2.2. Identification of the Isolated Protein as a Short Form of Galectin-3
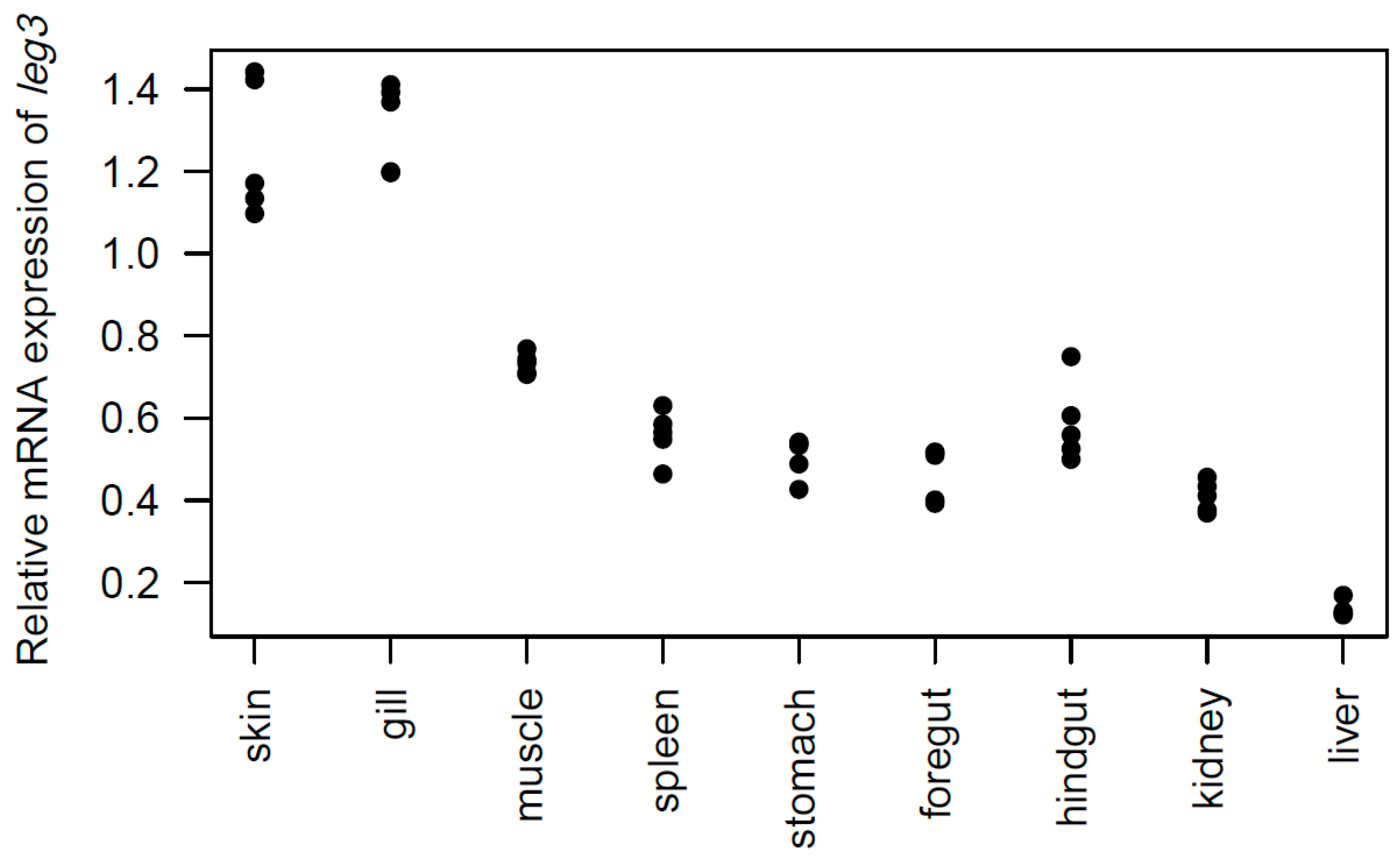
2.3. Tissue Distribution of the leg3 Gene
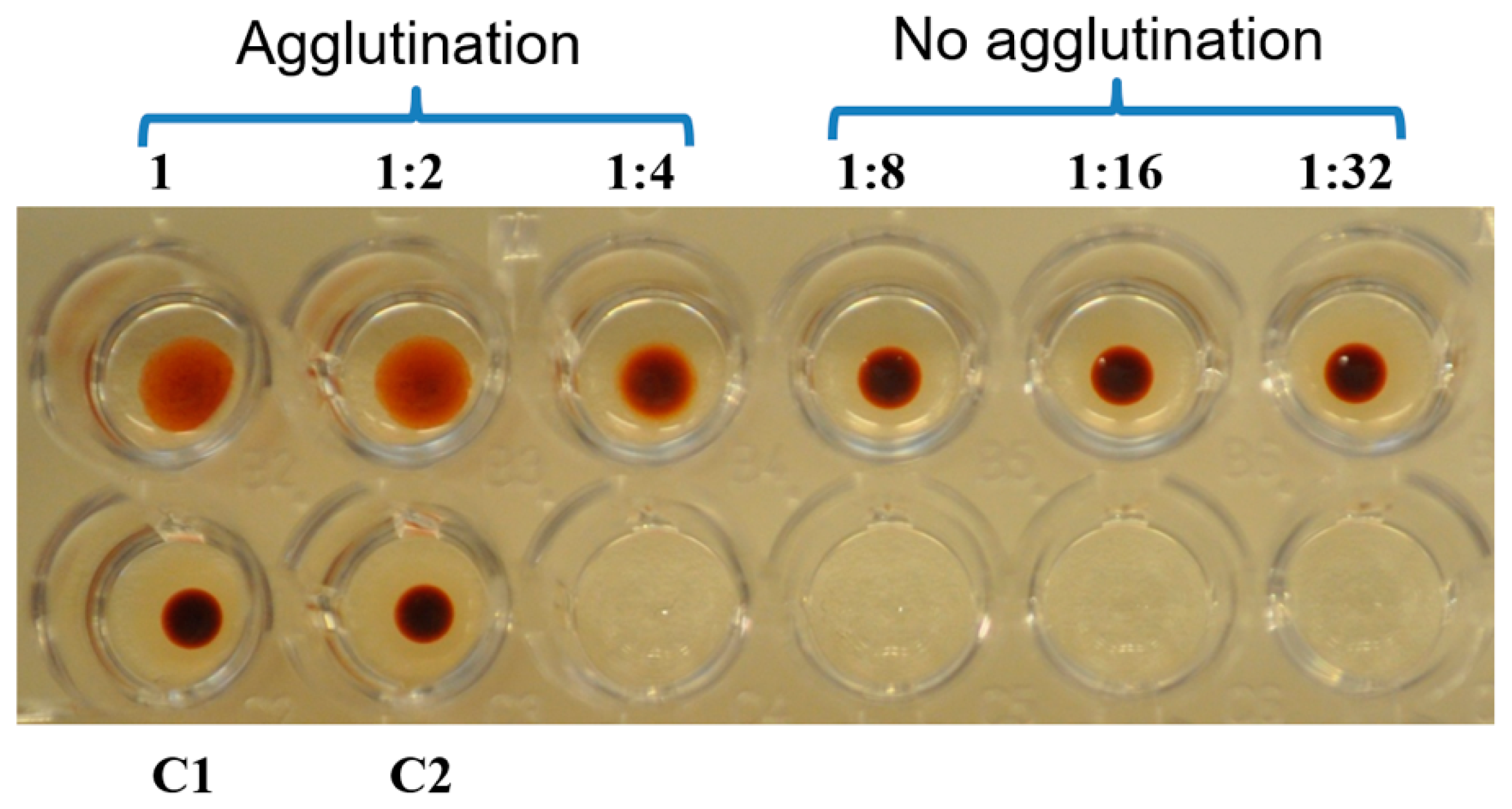
2.4. Hemaglutination Activity of Galectin-3C
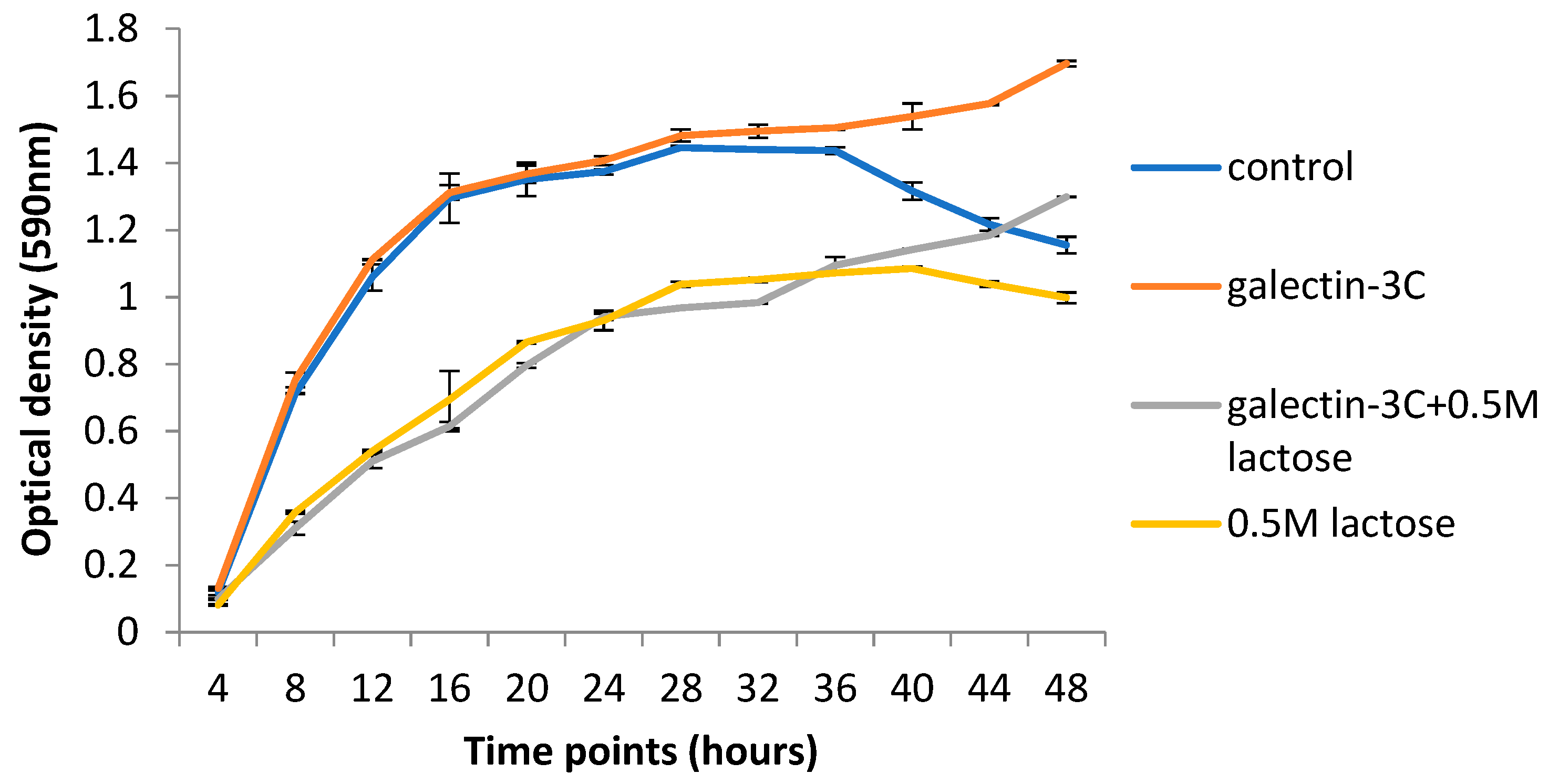
2.5. Galectin-3C and Lactose Change the Growth Curves of Moritella viscosa
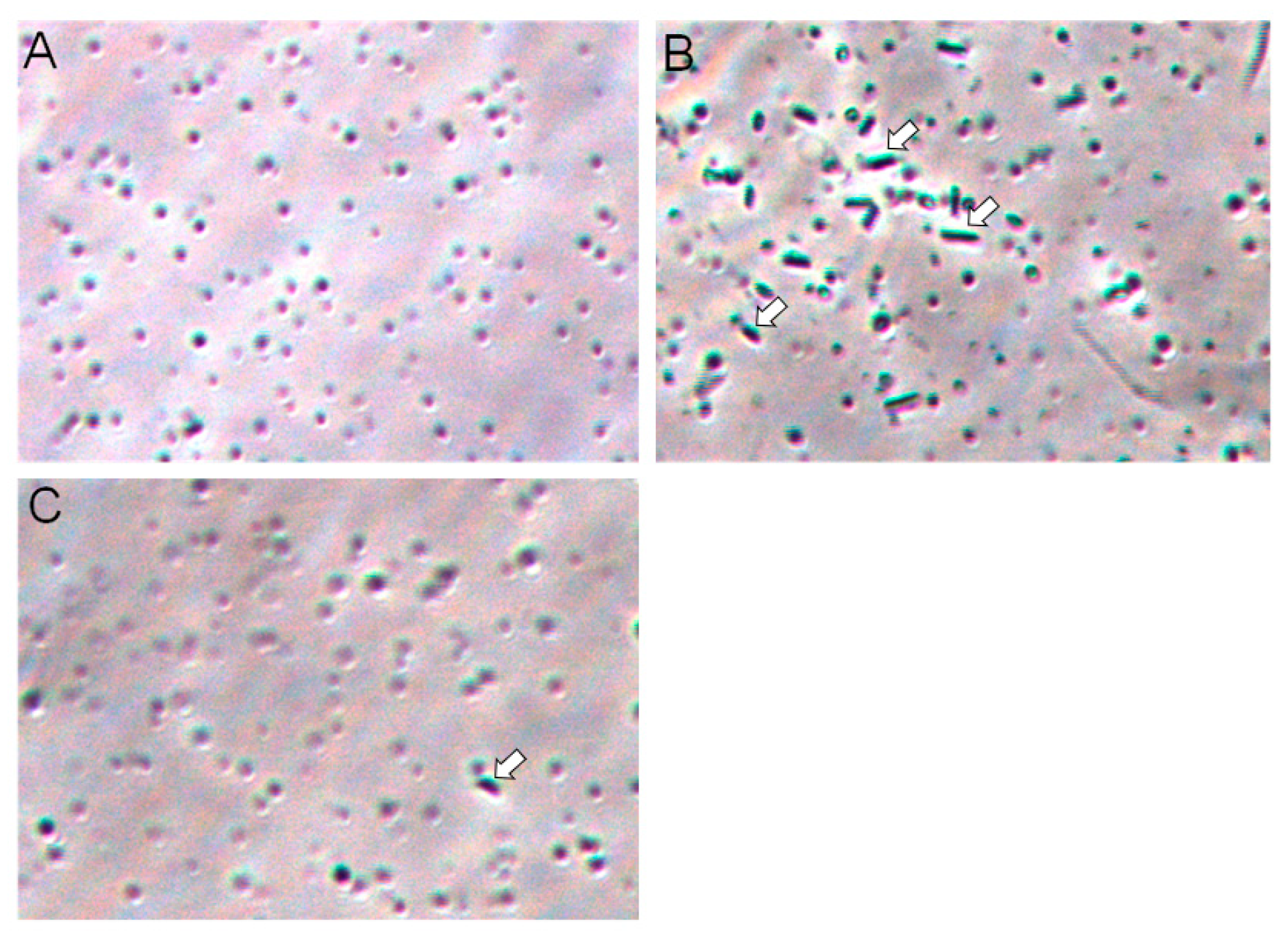
2.6. Galectin-3C agglutinate Moritella viscosa
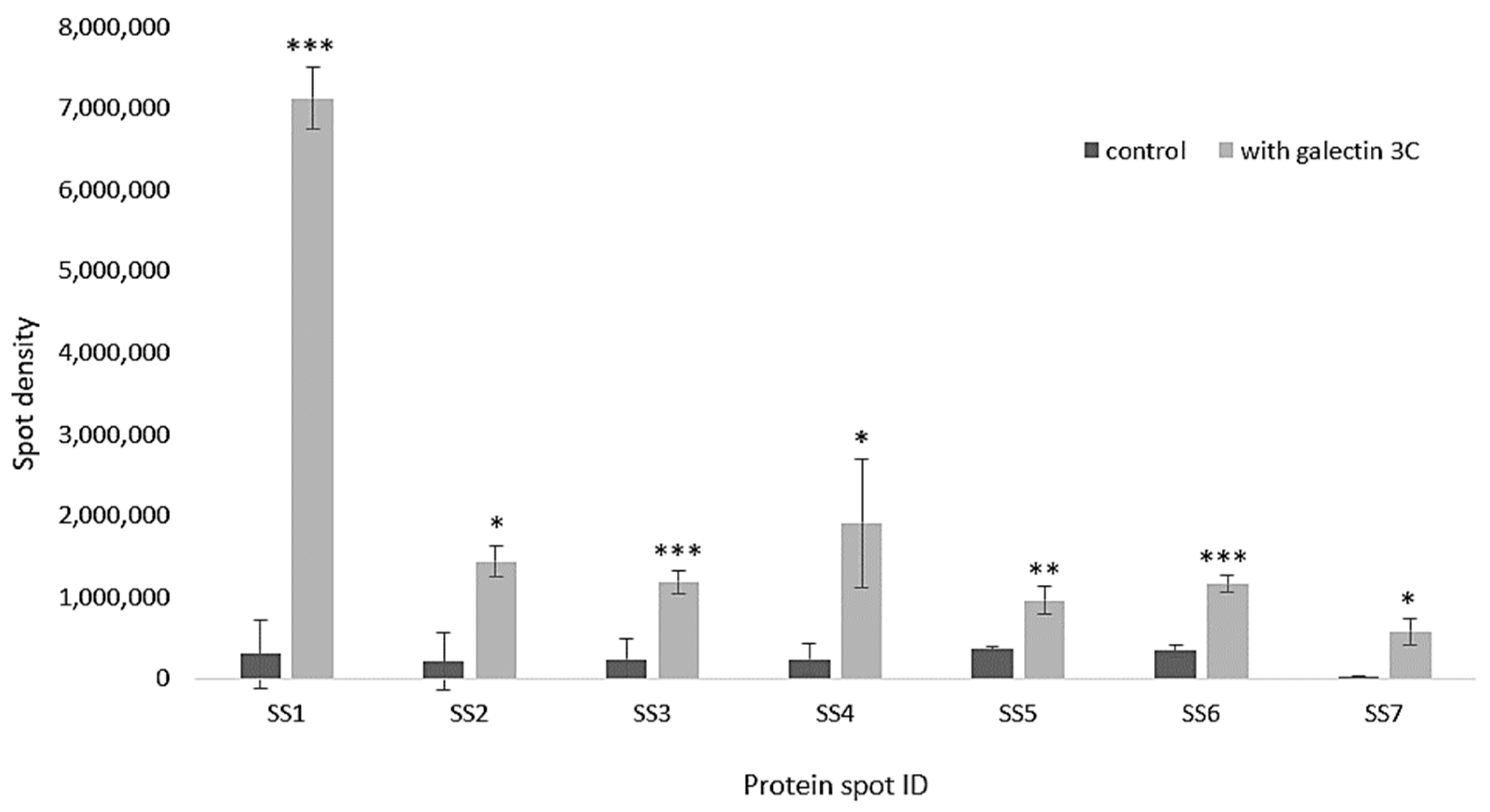
2.7. Galectin-3C Modulate the Proteome of Moritella viscosa
3. Discussion
4. Materials and Methods
4.1. Fish and Their Maintenance
4.2. Sampling of Skin Mucus and Tissues
4.3. Purification of Salmo salar Galectin-3 from Skin Mucus
4.3.1. Preparation of Skin Mucus Sample for Purification
4.3.2. Binding of Skin Mucus Protein to α-Lactose Agarose
4.3.3. Desalting of Purified Protein Using Sephadex G-15
4.4. SDS-PAGE
4.5. LC/MS-MS Analysis
4.6. Bioinformatic Analysis of Mass Spectrometry Files
4.7. Hemagglutination and Inhibition Assays
4.8. Tissue Distribution of Galectin-3 Gene Expression
4.8.1. RNA Extraction and cDNA Synthesis
4.8.2. Primer Design
4.8.3. Reverse Transcription-qPCR for Tissue Distribution Analysis
4.9. Effect of Salmo salar Galectin-3C on the Proteome of Moritella Viscosa and Agglutination of the Bacteria
4.9.1. Bacterial Strain and Experimental Set Up
4.9.2. Sample Preparation for 2D Analysis
4.9.3. Bacterial Agglutination
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ángeles Esteban, M. An overview of the immunological defenses in fish skin. ISRN Immunol. 2012, 2012, 29. [Google Scholar] [CrossRef]
- Brinchmann, M.F. Immune relevant molecules identified in the skin mucus of fish using -omics technologies. Mol. BioSyst. 2016, 12, 2056–2063. [Google Scholar] [CrossRef] [PubMed]
- Wells, V.; Mallucci, L. Identification of an autocrine negative growth factor: Mouse β-galactoside-binding protein is a cytostatic factor and cell growth regulator. Cell 1991, 64, 91–97. [Google Scholar] [CrossRef]
- Shetty, K.N.; Latha, V.L.; Rao, R.N.; Nadimpalli, S.K.; Suguna, K. Affinity of a galactose-specific legume lectin from Dolichos lablab to adenine revealed by X-ray cystallography. IUBMB Life 2013, 65, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Brinchmann, M.; Patel, D.; Pinto, N.; Iversen, M. Functional aspects of fish mucosal lectins—Interaction with non-self. Molecules 2018, 23, 1119. [Google Scholar] [CrossRef]
- Beck, B.H.; Farmer, B.D.; Straus, D.L.; Li, C.; Peatman, E. Putative roles for a rhamnose binding lectin in Flavobacterium columnare pathogenesis in channel catfish Ictalurus punctatus. Fish Shellfish Immunol. 2012, 33, 1008–1015. [Google Scholar] [CrossRef]
- Mandal, P.; Molla, A.R. Solvent perturbation of protein structures―A review study with lectins. Protein Pept. Lett. 2019. [Google Scholar] [CrossRef]
- Thurston, T.L.; Wandel, M.P.; von Muhlinen, N.; Foeglein, Á.; Randow, F. Galectin 8 targets damaged vesicles for autophagy to defend cells against bacterial invasion. Nature 2012, 482, 414–418. [Google Scholar] [CrossRef]
- Patterson, R.J.; Haudek, K.C.; Voss, P.G.; Wang, J.L. Examination of the role of galectins in pre-mRNA splicing. Methods Mol. Biol. 2015, 1207, 431–449. [Google Scholar] [CrossRef]
- Suzuki, Y.; Tasumi, S.; Tsutsui, S.; Okamoto, M.; Suetake, H. Molecular diversity of skin mucus lectins in fish. Comp. Biochem. Physiol. 2003, 136, 723–730. [Google Scholar] [CrossRef]
- Ng, T.B.; Chi Fai Cheung, R.; Cheuk Wing Ng, C.; Fei Fang, E.; Ho Wong, J. A review of fish lectins. Curr. Protein Pept. Sci. 2015, 16, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-W.; Wang, J.-X. Diversity and multiple functions of lectins in shrimp immunity. Dev. Comp. Immunol. 2013, 39, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Rajan, B.; Kiron, V.; Fernandes, J.M.O.; Brinchmann, M.F. Localization and functional properties of two galectin-1 proteins in Atlantic cod (Gadus morhua) mucosal tissues. Dev. Comp. Immunol. 2013, 40, 83–93. [Google Scholar] [CrossRef]
- Tasumi, S.; Yang, W.-J.; Usami, T.; Tsutsui, S.; Ohira, T.; Kawazoe, I.; Wilder, M.N.; Aida, K.; Suzuki, Y. Characteristics and primary structure of a galectin in the skin mucus of the Japanese eel, Anguilla japonica. Dev. Comp. Immunol. 2004, 28, 325–335. [Google Scholar] [CrossRef]
- Kamiya, H.; Muramoto, K.; Goto, R. Purification and properties of agglutinins from conger eel, Conger Myriaster (Brevoort), skin mucus. Dev. Comp. Immunol. 1988, 12, 309–318. [Google Scholar] [CrossRef]
- Lunder, T.; Evensen, Ø.; Holstad, G.; Håstein, T. ‘Winter ulcer’ in the Atlantic salmon Salmo salar. Pathological and bacteriological investigations and transmission experiments. Dis. Aquat. Organ. 1995, 23, 39–49. [Google Scholar] [CrossRef]
- Banfer, S.; Schneider, D.; Dewes, J.; Strauss, M.T.; Freibert, S.A.; Heimerl, T.; Maier, U.G.; Elsasser, H.P.; Jungmann, R.; Jacob, R. Molecular mechanism to recruit galectin-3 into multivesicular bodies for polarized exosomal secretion. Proc. Natl. Acad. Sci. USA 2018, 115, E4396–E4405. [Google Scholar] [CrossRef]
- Brinchmann, M.F.; Patel, D.M.; Iversen, M.H. The role of galectins as modulators of metabolism and inflammation. Mediat. Inflamm. 2018, 2018. [Google Scholar] [CrossRef]
- Díaz-Alvarez, L.; Ortega, E. The many roles of galectin-3, a multifaceted molecule, in innate immune responses against pathogens. Mediat. Inflamm. 2017, 2017. [Google Scholar] [CrossRef]
- Ochieng, J.; Fridman, R.; Nangia-Makker, P.; Kleiner, D.E.; Liotta, L.A.; Stetler-Stevenson, W.G.; Raz, A. Galectin-3 is a novel substrate for human matrix metalloproteinases-2 and -9. Biochemistry 1994, 33, 14109–14114. [Google Scholar] [CrossRef]
- John, C.M.; Leffler, H.; Kahl-Knutsson, B.; Svensson, I.; Jarvis, G.A. Truncated galectin-3 inhibits tumor growth and metastasis in orthotopic nude mouse model of human breast cancer. Clin. Cancer Res. 2003, 9, 2374–2383. [Google Scholar]
- Sato, S.; Burdett, I.; Hughes, R.C. Secretion of the baby hamster kidney 30-kDa galactose-binding lectin from polarized and nonpolarized cells: a pathway independent of the endoplasmic reticulum-Golgi complex. Exp. Cell Res. 1993, 207, 8–18. [Google Scholar] [CrossRef]
- Menon, S.; Kang, C.M.; Beningo, K.A. Galectin-3 secretion and tyrosine phosphorylation is dependent on the calpain small subunit, Calpain 4. Biochem. Biophys. Res. Commun. 2011, 410, 91–96. [Google Scholar] [CrossRef]
- Menon, R.P.; Hughes, R.C. Determinants in the N-terminal domains of galectin-3 for secretion by a novel pathway circumventing the endoplasmic reticulum-Golgi complex. Eur. J. Biochem. 1999, 264, 569–576. [Google Scholar] [CrossRef]
- Salles, C.M.C.; Gagliano, P.; Leitão, S.A.T.; Salles, J.B.; Guedes, H.L.M.; Cassano, V.P.F.; De-Simone, S.G. Identification and characterization of proteases from skin mucus of tambacu, a Neotropical hybrid fish. Fish Physiol. Biochem. 2007, 33, 173. [Google Scholar] [CrossRef]
- Firth, K.J.; Johnson, S.C.; Ross, N.W. Characterization of proteases in the skin mucus of Atlantic salmon (Salmo salar) infected with the salmon louse (Lepeophtheirus salmonis) and in whole-body louse homogenate. J. Parasitol. 2000, 86, 1199–1205. [Google Scholar] [CrossRef]
- Ahmad, N.; Gabius, H.J.; Andre, S.; Kaltner, H.; Sabesan, S.; Roy, R.; Liu, B.; Macaluso, F.; Brewer, C.F. Galectin-3 precipitates as a pentamer with synthetic multivalent carbohydrates and forms heterogeneous cross-linked complexes. J. Biol. Chem. 2004, 279, 10841–10847. [Google Scholar] [CrossRef]
- Yang, R.Y.; Hill, P.N.; Hsu, D.K.; Liu, F.T. Role of the carboxyl-terminal lectin domain in self-association of galectin-3. Biochemistry 1998, 37, 4086–4092. [Google Scholar] [CrossRef]
- Roche, A.M.; Richard, A.L.; Rahkola, J.T.; Janoff, E.N.; Weiser, J.N. Antibody blocks acquisition of bacterial colonization through agglutination. Mucosal Immunol. 2015, 8, 176–185. [Google Scholar] [CrossRef]
- Stowell, S.R.; Arthur, C.M.; Dias-Baruffi, M.; Rodrigues, L.C.; Gourdine, J.P.; Heimburg-Molinaro, J.; Ju, T.Z.; Molinaro, R.J.; Rivera-Marrero, C.; Xia, B.Y.; et al. Innate immune lectins kill bacteria expressing blood group antigen. Nat. Med. 2010, 16, 295–301. [Google Scholar] [CrossRef]
- Biemer, J.J. Antimicrobial susceptibility testing by the Kirby-Bauer disc diffusion method. Ann. Clin. Lab. Sci. 1973, 3, 135–140. [Google Scholar]
- Qin, X.; Emich, J.; Goycoolea, F.M. Assessment of the quorum sensing inhibition activity of a non-toxic chitosan in an N-acyl homoserine lactone (AHL)-based Escherichia coli biosensor. Biomolecules 2018, 8, 87. [Google Scholar] [CrossRef]
- Lunder, T.; Sorum, H.; Holstad, G.; Steigerwalt, A.G.; Mowinckel, P.; Brenner, D.J. Phenotypic and genotypic characterization of Vibrio viscosus sp. nov. and Vibrio wodanis sp. nov. isolated from Atlantic salmon (Salmo salar) with ‘winter ulcer’. Int. J. Syst. Evol. Microbiol. 2000, 50, 427–450. [Google Scholar] [CrossRef]
- Moor, K.; Diard, M.; Sellin, M.E.; Felmy, B.; Wotzka, S.Y.; Toska, A.; Bakkeren, E.; Arnoldini, M.; Bansept, F.; Co, A.D.; et al. High-avidity IgA protects the intestine by enchaining growing bacteria. Nature 2017, 544, 498–502. [Google Scholar] [CrossRef]
- Alav, I.; Sutton, J.M.; Rahman, K.M. Role of bacterial efflux pumps in biofilm formation. J. Antimicrob. Chemother. 2018, 73, 2003–2020. [Google Scholar] [CrossRef]
- Dunon, V.; Bers, K.; Lavigne, R.; Top, E.M.; Springael, D. Targeted metagenomics demonstrates the ecological role of IS1071 in bacterial community adaptation to pesticide degradation. Environ. Microbiol. 2018, 20, 4091–4111. [Google Scholar] [CrossRef]
- Nagel, A.; Michalik, S.; Debarbouille, M.; Hertlein, T.; Gesell Salazar, M.; Rath, H.; Msadek, T.; Ohlsen, K.; van Dijl, J.M.; Völker, U.; et al. Inhibition of Rho activity increases expression of SaeRS-dependent virulence factor genes in Staphylococcus aureus, showing a link between transcription termination, antibiotic action, and virulence. mBio 2018, 9, e01332-18. [Google Scholar] [CrossRef]
- Brodersen, D.E.; Clemons, W.M., Jr.; Carter, A.P.; Morgan-Warren, R.J.; Wimberly, B.T.; Ramakrishnan, V. The structural basis for the action of the antibiotics tetracycline, pactamycin, and hygromycin B on the 30S ribosomal subunit. Cell 2000, 103, 1143–1154. [Google Scholar] [CrossRef]
- Vazquez-Laslop, N.; Mankin, A.S. How macrolide antibiotics work. Trends Biochem. Sci. 2018, 43, 668–684. [Google Scholar] [CrossRef]
- Bandow, J.E.; Brötz, H.; Leichert, L.I.O.; Labischinski, H.; Hecker, M. Proteomic approach to understanding antibiotic action. Antimicrob. Agents Chemother. 2003, 47, 948–955. [Google Scholar] [CrossRef]
- Takayama, S.; Saitoh, E.; Kimizuka, R.; Yamada, S.; Kato, T. Effect of eel galectin AJL-1 on periodontopathic bacterial biofilm formation and their lipopolysaccharide-mediated inflammatory cytokine induction. Int. J. Antimicrob. Agents 2009, 34, 355–359. [Google Scholar] [CrossRef]
- Magnusson, S.; Baldursson, B.T.; Kjartansson, H.; Rolfsson, O.; Sigurjonsson, G.F. Regenerative and antibacterial properties of acellular fish skin grafts and human amnion/chorion membrane: Implications for tissue preservation in combat casualty care. Milit. Med. 2017, 182, 383–388. [Google Scholar] [CrossRef]
- Panjwani, N. Role of galectins in re-epithelialization of wounds. Ann. Transl. Med. 2014. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Øverbye, A.; Fengsrud, M.; Seglen, P.O. Proteomic analysis of membrane-associated proteins from rat liver autophagosomes. Autophagy 2007, 3, 300–322. [Google Scholar] [CrossRef]
- Nowak, T.P.; Kobiler, D.; Roel, L.E.; Barondes, S.H. Developmentally regulated lectin from embryonic chick pectoral muscle: Purification by affinity chromatography. J. Biol. Chem. 1977, 252, 6026–6030. [Google Scholar] [CrossRef]
- Olsvik, P.A.; Lie, K.K.; Jordal, A.E.; Nilsen, T.O.; Hordvik, I. Evaluation of potential reference genes in real-time RT-PCR studies of Atlantic salmon. BMC Mol. Biol. 2005, 6. [Google Scholar] [CrossRef]
- Ingerslev, H.C.; Pettersen, E.F.; Jakobsen, R.A.; Petersen, C.B.; Wergeland, H.I. Expression profiling and validation of reference gene candidates in immune relevant tissues and cells from Atlantic salmon (Salmo salar L.). Mol. Immunol. 2006, 43, 1194–1201. [Google Scholar] [CrossRef]
- Raeder, I.L.U.; Paulsen, S.M.; Smalas, A.O.; Willassen, N.P. Effect of fish skin mucus on the soluble proteome of Vibrio salmonicida analyzed by 2-D gel electrophoresis and tandem mass spectrometry. Microb. Pathog. 2007, 42, 36–45. [Google Scholar] [CrossRef]
- Rajan, B.; Fernandes, J.M.O.; Caipang, C.M.A.; Kiron, V.; Rombout, J.H.W.M.; Brinchmann, M.F. Proteome reference map of the skin mucus of Atlantic cod (Gadus morhua) revealing immune competent molecules. Fish Shellfish Immunol. 2011, 31, 224–231. [Google Scholar] [CrossRef]


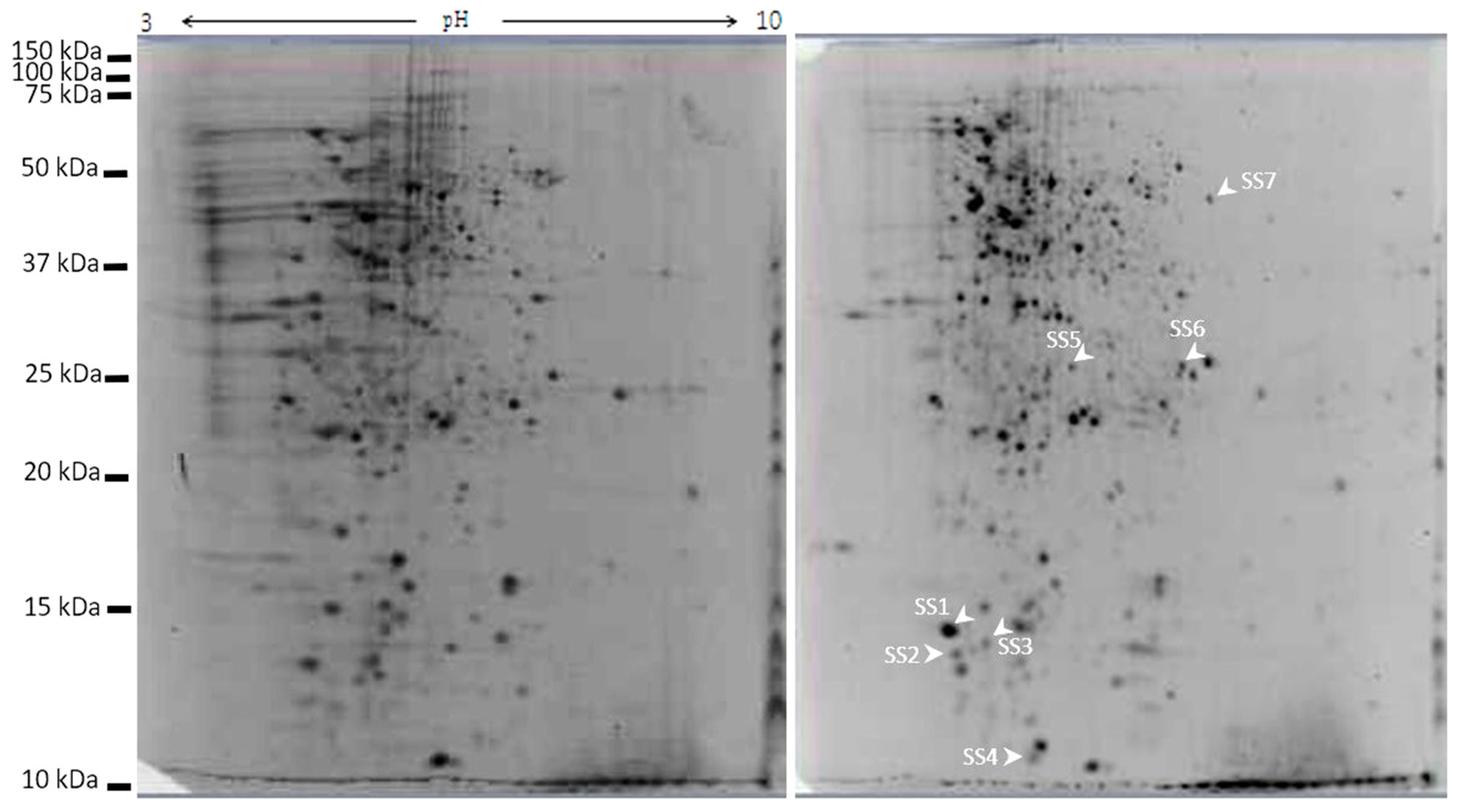
| Spot ID | Accession ID/Database | Protein Name/Organism/ Major Protein Domain | Calculated pI/Nominal Mass (Mr) | Mascot ion Score/ p-Value | SC (%)/ UP Score | ST | Peptide Sequences |
|---|---|---|---|---|---|---|---|
| SS1 | WP_002958882 NCBIprot | 50S ribosomal protein L7/L12 Pseudoalteromonas Ribosomal_L7_L12 superfamily | 4.45/12194 | 407/ p < 0.01 | 50/406 | 69 | KDLTEAGAEVEVK KEGVSKEEAEALAKD KTEFDVILTGAGANKV KFGVTAAAAMVAGPA-AEAAEEKT |
| SS2 | Q3ILQ0 NCBIprot | 50S ribosomal protein L7/L12/Pseudoalteromonas haloplanktis Ribosomal_L19 superfamily | 4.45/12222 | 161/ p < 0.008 | 19/161 | 71 | KALVEAAPTPVKE KDLTEAGAEVEVK |
| SS3 | Q3IJK6 Swissprot | 30S ribosomal protein S13/Pseudoalteromonas haloplanktis Ribosomal_S13 superfamily | 10.95/13162 | 123/ p < 0.0005 | 11/123 | 72 | KIGELSDETLDVLRD |
| SS4 | P21219 SwissProt | High-molecular weight cobalt-containing nitrile hydratase subunit alpha/Rhodococcus rhodochrous | 4.85/22991 | 58/ p < 0.05 | 6/58 | 37 | RDFGFDIPDEVEVRV |
| SS5 | WP_016707503 NCBIpro | multidrug transporter/Pseudoalteromonas SapC superfamily | 5.38/26438 | 178/ p < 0.01 | 15/178 | 70 | KEALVSFIEFSHVTEAFTKY KYLADKELLVAQTLTVDIKG |
| SS6 | WP_045111824 NCBIprot | 30S ribosomal protein S2/Moritella viscosa RPS2 superfamily | 6.60/26834 | 169/ p < 0.01 | 12/169 | 66 | KLYAGAVAASVNEGRN RNQDIAAQAESDFIEEAK |
| SS7 | CCQ10658 NCBIprot | Transcription termination factor Rho/Pseudoalteromonas luteoviolacea Rho superfamily | 6.55/47155 | 149/ p < 0.01 | 6/77 | 66 | KGEVIASTFDEPASRH KILFENLTPIHANERL |
| Gene Name | Gene Symbol | Sequence (5′>3′) |
|---|---|---|
| Galectin-3 | leg3 | Forward: GAGTTCAAACACCGCATCCG Reverse: GGCTGAAACCAACCCTGCTA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, D.M.; Kitani, Y.; Korsnes, K.; Iversen, M.H.; Brinchmann, M.F. A Truncated Galectin-3 Isolated from Skin Mucus of Atlantic Salmon Salmo salar Binds to and Modulates the Proteome of the Gram-Negative Bacteria Moritella viscosa. Mar. Drugs 2020, 18, 102. https://doi.org/10.3390/md18020102
Patel DM, Kitani Y, Korsnes K, Iversen MH, Brinchmann MF. A Truncated Galectin-3 Isolated from Skin Mucus of Atlantic Salmon Salmo salar Binds to and Modulates the Proteome of the Gram-Negative Bacteria Moritella viscosa. Marine Drugs. 2020; 18(2):102. https://doi.org/10.3390/md18020102
Chicago/Turabian StylePatel, Deepti Manjari, Yoichiro Kitani, Kjetil Korsnes, Martin Haugmo Iversen, and Monica Fengsrud Brinchmann. 2020. "A Truncated Galectin-3 Isolated from Skin Mucus of Atlantic Salmon Salmo salar Binds to and Modulates the Proteome of the Gram-Negative Bacteria Moritella viscosa" Marine Drugs 18, no. 2: 102. https://doi.org/10.3390/md18020102
APA StylePatel, D. M., Kitani, Y., Korsnes, K., Iversen, M. H., & Brinchmann, M. F. (2020). A Truncated Galectin-3 Isolated from Skin Mucus of Atlantic Salmon Salmo salar Binds to and Modulates the Proteome of the Gram-Negative Bacteria Moritella viscosa. Marine Drugs, 18(2), 102. https://doi.org/10.3390/md18020102







