Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia
Abstract
1. Introduction
2. Results
2.1. Sample Characterization
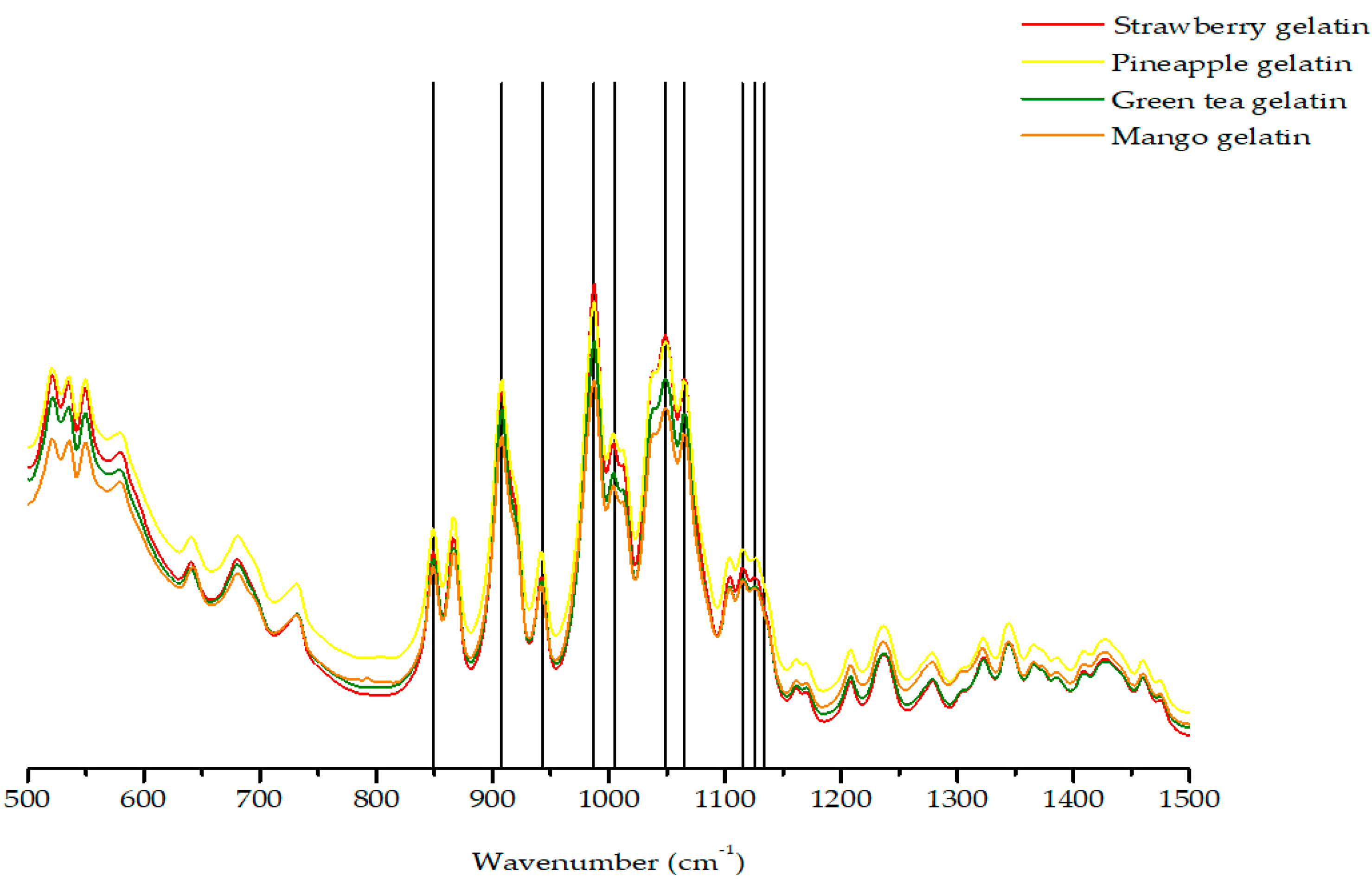
2.2. FTIR-ATR Spectroscopy
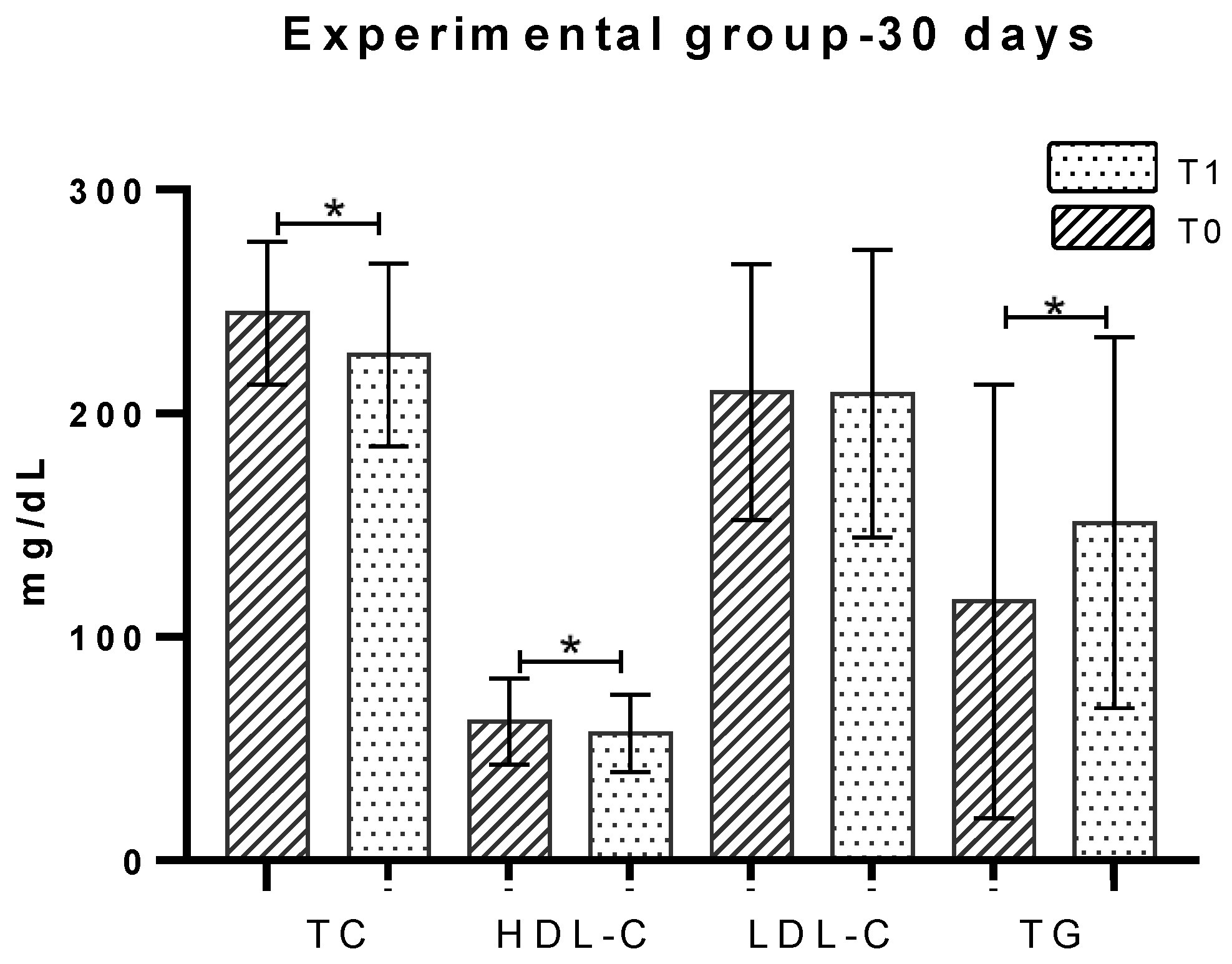
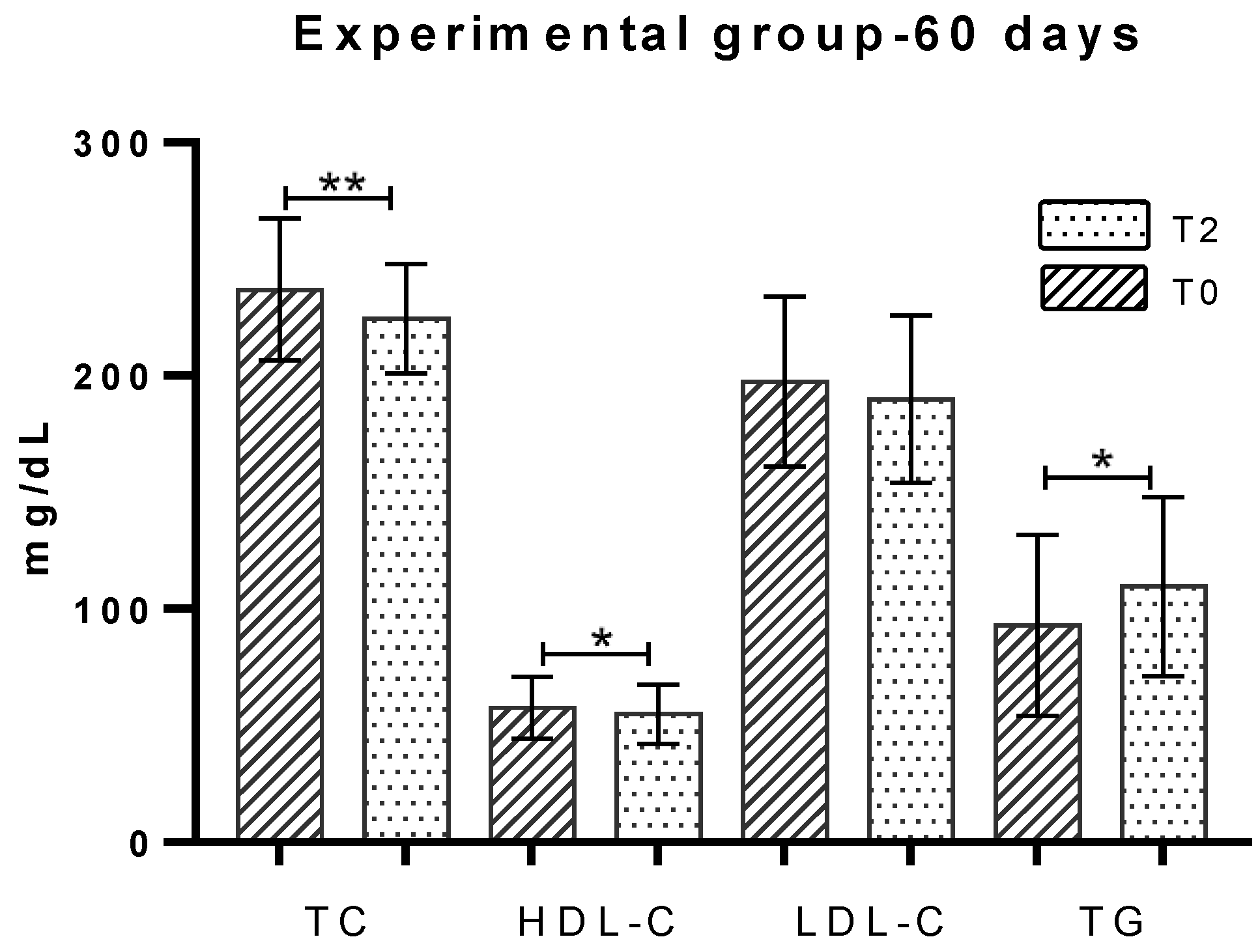
2.3. Total Cholesterol
2.4. HDL-Cholesterol
2.5. LDL-Cholesterol
2.6. Triglycerides
2.7. MeDiet-Adherence to Mediterranean Diet
3. Discussion
4. Materials and Methods
4.1. Sample Characterization
4.2. Sample Collection and Preparation
4.3. Statistical Analysis
4.4. Confidentiality and Data Protection
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klop, B.; Elte, J.W.; Cabezas, M.C. Dyslipidemia in obesity: Mechanisms and potential targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef] [PubMed]
- WHO. Cardiovascular Diseases CVDs 2018. Available online: https://www.who.int/cardiovascular_diseases/en/ (accessed on 19 September 2019).
- INE. Anuário Estatistico, 109rd ed.; Instituto Nacional de Estatística: Lisboa, Portugal, 2017; p. 254.
- Shimazu, T.; Kuriyama, S.; Hozawa, A.; Ohmori, K.; Sato, Y.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Dietary patterns and cardiovascular disease mortality in Japan: A prospective cohort study. Int. J. Epidemiol. 2007, 36, 600–609. [Google Scholar] [CrossRef] [PubMed]
- Burtin, P. Nutritional value of seaweeds. Electron. J. Environ. Agric. Food Chem. 2003, 2, 498–503. [Google Scholar]
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar. Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Filipic, M.; Jose Frutos, M.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Kuhnle, G.G.; et al. Re-evaluation of carrageenan (E 407) and processed Eucheuma seaweed (E 407a) as food additives. EFSA J. 2018, 16, e05238. [Google Scholar]
- Kenn, H. Genu Carrageenan Book; CP Kelco Inc.: Lille Skensved, Denmark, 2002; p. 28. [Google Scholar]
- Pereira, L.; Amado, A.M.; Critchley, A.T.; van de Velde, F.; Ribeiro-Claro, P.J.A. Identification of selected seaweed polysaccharides (phycocolloids) by vibrational spectroscopy (FTIR-ATR and FT-Raman). Food Hydrocoll. 2009, 23, 1903–1909. [Google Scholar] [CrossRef]
- Pereira, L. Algae, Uses in Agriculture, Gastronomy and Food Industry; Câmara Municipal de Viana do Castelo: Viana do Castelo, Portugal, 2010; p. 67. [Google Scholar]
- García, I.A.F.; Castrovielo, R.A.; Neira, C.D. Las Algas en Galicia—Alimentacion y Otros Usos. Xunta de Galicia; Consellería de Pesca; Marisqueo e Acuicultura: Galicia, Spain, 1993; p. 229. [Google Scholar]
- Soares, P.A.; de Seixas, J.R.; Albuquerque, P.B.; Santos, G.R.; Mourão, P.A.; Barros, W., Jr.; Correia, M.T.; Carneiro-da-Cunha, M.G. Development and characterization of a new hydrogel based on galactomannan and κ-carrageenan. Carbohydr. Polym. 2015, 134, 673–679. [Google Scholar] [CrossRef]
- van de Velde, F.; de Ruiter, G.A. Carrageenan. In Biopolymers Polysaccharides II, Polysaccharides from Eukaryotes; Vandame, S.D., Baets, S.D., Steinbèuchej, A., Eds.; Wiley-VCH: Weinheim, Germany, 2002; Volume 6, pp. 245–274. [Google Scholar]
- Pereira, L. Biological and therapeutic properties of the seaweed polysaccharides. Int. Biol. Rev. 2018, 2, 1–50. [Google Scholar]
- Silva, F.R.F.; Dore, C.M.P.G.; Marques, C.T.; Nascimento, M.S.; Benevides, N.M.B.; Rocha, H.A.O.; Chavante, S.F.; Leite, E.L. Anticoagulant activity, paw edema and pleurisy induced carrageenan: Action of major types of commercial carrageenans. Carbohydr. Polym. 2010, 79, 26–33. [Google Scholar] [CrossRef]
- Blackwood, A.; Salter, J.; Dettmar, P.; Chaplin, M. Dietary fibre, physicochemical properties and their relationship to health. J. R. Soc. Promot. Health 2000, 120, 242–247. [Google Scholar] [CrossRef]
- Chen, F.; Deng, Z.; Zhang, Z.; Zhang, R.; Xu, Q.; Fan, G.; Luo, T.; Julian, D.; Clements, Mc. Controlling lipid digestion profiles using mixtures of different types of microgel: Alginate beads and carrageenan beads. J. Food Eng. 2018, 238, 156–163. [Google Scholar] [CrossRef]
- Amano, H.; Kakinuma, M.; Coury, D.; Ohno, H.; Hara, T. Effect of a seaweed mixture on serum lipid level and platelet aggregation in rats. Fish. Sci. 2005, 71, 1160–1166. [Google Scholar] [CrossRef]
- Gomez, E.; Jimenez, A.; Ruperez, P. Effect of the red seaweed Mastocarpus stellatus intake on lipid metabolism and antioxidant status in healthy Wistar rats. Food Chem. 2012, 135, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Sokolova, E.; Bogdanovich, L.; Ivanova, T.; Byankina, A.; Kryzhanovskiy, S.; Yermak, I. Effect of carrageenan food supplement on patients with cardiovascular disease results in normalization of lipid profile and moderate modulation of immunity system markers. PharmaNutrition 2014, 2, 33–37. [Google Scholar] [CrossRef]
- Panlasigui, L.; Baello, O.; Dimatangal, J.; Dumelod, B. Blood cholesterol and lipid-lowering effects of carrageenan on human volunteers. Asia Pac. J. Clin. Nutr. 2003, 12, 209–214. [Google Scholar]
- Cardoso, S.; Pereira, O.; Seca, A.; Pinto, D.; Silva, A. Seaweeds as preventive agents for cardiovascular diseases: From nutrients to functional foods. Mar. Drugs 2015, 13, 6838–6865. [Google Scholar] [CrossRef]
- Williams, M.; Ragasa, C. Functional foods: Opportunities and challenges for developing countries. World Bank 2006, 19, 37683. [Google Scholar]
- Siro, I.; Kápolna, E.; Kapolna, B.; Lugasi, A. Functional food. Product development, marketing and consumer acceptance—A review. Appetite 2008, 51, 456–467. [Google Scholar] [CrossRef]
- Dickson-Spillman, M.; Siegrist, M.; Keller, C. Attitudes toward chemicals are associated with preference for natural food. Food Qual. Pref. 2011, 22, 149–156. [Google Scholar] [CrossRef]
- Campo, V.; Kawano, D.; Silva, D.; Carvalho, I. Carrageenans: Biological properties, chemical modifications and structural analysis—A review. Carbohydr. Polym. 2009, 77, 167–180. [Google Scholar] [CrossRef]
- Bui, V.; Nguyen, B.; Renou, F.; Nicolai, T. Rheology and microstructure of mixtures of iota and kappa-carrageenan. Food Hydrocoll. 2019, 89, 180–187. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Ros, E.; Covas, M.I.; Fiol, M.; Wärnberg, J.; Arós, F.; Ruíz-Gutiérrez, V.; Lamuela-Raventós, R.M.; et al. PREDIMED Study Investigators. Cohort profile: Design and methods of the PREDIMED study. Int. J. Epidemiol. 2012, 41, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Schroder, H.; Fito, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A short screener is valid for assessing Mediterranean diet adherence among older Spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Brenner, T.; Xie, J.; Liu, Z.; Wang, S.; Matsukawa, S. Gelation of iota/kappa Carrageenan Mixtures. In Gums and Stabilisers for the Food Industry 18: Hydrocolloid Functionality for Affordable and Sustainable Global Food Solutions, 1st ed.; Williams, P., Philips, G.R.S.C., Eds.; Royal Society of Chemistry: Cambridge, UK, 2016; pp. 47–55. [Google Scholar]
- Pereira, L.; Gheda, S.; Ribeiro-Claro, P. Analysis by vibrational spectroscopy of seaweed polysaccharides with potential use in food, pharmaceutical, and cosmetic industries. Int. J. Carbohydr. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Anderson, J.; Baird, P.; Davis, R.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Matthan, N.; Zhu, L.; Pencina, M.; D’Agostino, R.; Schaefer, E.; Lichtenstein, A. Sex-specific differences in the predictive value of cholesterol homeostasis markers and 10-year cardiovascular disease event rate in Framingham Offspring Study participants. Am. Heart J. 2013, 2, e005066. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. ESC Scientific Document Group. European guidelines on cardiovascular disease prevention in clinical practice: The sixth joint task force of the European society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of 10 societies and by invited experts) developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Elsevier 2016, 252, 207–274. [Google Scholar]
- McKim, J. Food additive carrageenan: Part I: A critical review of carrageenan in vitro studies, potential pitfalls, and implications for human health and safety. Crit. Rev. Toxicol. 2014, 44, 211–243. [Google Scholar] [CrossRef]
- Weiner, M.; Nuber, D.; Blakemore, W.; Harriman, J.; Cohen, S. A 90-day dietary study on kappa carrageenan with emphasis on the gastrointestinal tract. Food Chem. Toxicol. 2007, 45, 98–106. [Google Scholar] [CrossRef]
- Awang, A.; Ng, J.; Matanjun, P.; Sulaiman, M.; Tan, T.; Ooi, Y. Anti-obesity property of the brown seaweed, Sargassum polycystum using an in vivo animal model. J. Appl. Phycol. 2014, 26, 1043–1048. [Google Scholar] [CrossRef]
- Weiner, M. Food additive carrageenan: Part II: A critical review of carrageenan in vivo safety studies. Crit. Rev. Toxicol. 2014, 44, 244–269. [Google Scholar] [CrossRef] [PubMed]
- Chater, P.I.; Wilcox, M.; Cherry, P.; Herford, A.; Mustar, S.; Wheater, H.; Wheater, H.; Brownlee, I.; Seal, C.; Pearson, J. Inhibitory activity of extracts of Hebridean brown seaweeds on lipase activity. J. Appl. Phycol. 2016, 28, 1303–1313. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Onnagawa, M.; Yoshie, Y.; Suzuki, T. Binding of bile salts to soluble and insoluble dietary fibers of seaweeds. Fish. Sci. 2001, 67, 1169–1173. [Google Scholar] [CrossRef]
- Sokolova, E.; Kravchenko, A.; Sergeeva, N.; Davydova, V.; Bogdanovich, L.; Yermak, I. Effect of carrageenans on some lipid metabolism components in vitro. Carbohydr. Polym. 2019. [Google Scholar] [CrossRef]
- McIntosh, M.; Miller, C. A diet containing food rich in soluble and insoluble fiber improves glycemic control and reduces hyperlipidemia among patients with type 2 diabetes mellitus. Nutr. Rev. 2001, 59, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Andrade, I.; Santos, L.; Ramos, F. Cholesterol absorption and synthesis markers in Portuguese hypercholesterolemic adults: A cross-sectional study. Eur. J. Int. Med. 2016, 28, 85–90. [Google Scholar] [CrossRef] [PubMed]
- De Marinis, E.; Matini, C.; Trentalance, A.; Pallottini, V. Sex differences in hepatic regulation of cholesterol homeostasis. J. Endocrinol. 2008, 198, 635–643. [Google Scholar] [CrossRef]
- Mehta, R.; Reyes-Rodriguez, E.; Yaxmehen, B.-C.O.; Guerrero-Díaz, A.C.; Vargas-Vázquez, A.; Cruz-Bautista, I.; AAguilar-Salinas, C. Performance of LDL-C calculated with Martin’s formula compared to the Friedewald equation in familial combined hyperlipidemia. Atherosclerosis 2018, 277, 204–210. [Google Scholar] [CrossRef]
- Schulze, M.B.; Hoffmann, K. Methodological approaches to study dietary patterns in relation to risk of coronary heart disease and stroke. Br. J. Nutr. 2006, 95, 860–869. [Google Scholar] [CrossRef]
| Ingestion Period | 30 Days | 60 Days | |
|---|---|---|---|
| n = 12 | n = 30 | ||
| Age (years) | - | 44.55 ± 11.61 | 48.68 ± 10.63 |
| (min–max) | (23–64) | (20–64) | |
| Sex | F | 8 | 24 |
| M | 4 | 6 | |
| MeDiet | ≥10 | 4 | 15 |
| <10 | 8 | 15 | |
| Sex | ||
|---|---|---|
| Female | Male | |
| TC (T0) | 234.51 ± 31.06 | 244.95 ± 29.62 |
| TC (T2) | 220.43 ± 21.16 ** | 237.76 ± 27.82 |
| HDL-C (T0) | 60.85 ± 13.15 | 46.78 ± 6.65 |
| HDL-C (T2) | 58.41 ± 12.10 | 43.46 ± 7.28 |
| LDL-C (T0) | 190.58 ± 33.85 | 220.12 ± 38.77 |
| LDL-C (T2) | 180.35 ± 29.07 * | 221.48 ± 40.11 |
| TG (T0) | 87.88 ± 38.06 | 109.75 ± 40.18 |
| TG (T2) | 101.65 ± 31.16 | 135.87 ± 50.83 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valado, A.; Pereira, M.; Caseiro, A.; Figueiredo, J.P.; Loureiro, H.; Almeida, C.; Cotas, J.; Pereira, L. Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia. Mar. Drugs 2020, 18, 19. https://doi.org/10.3390/md18010019
Valado A, Pereira M, Caseiro A, Figueiredo JP, Loureiro H, Almeida C, Cotas J, Pereira L. Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia. Marine Drugs. 2020; 18(1):19. https://doi.org/10.3390/md18010019
Chicago/Turabian StyleValado, Ana, Maria Pereira, Armando Caseiro, João P. Figueiredo, Helena Loureiro, Carla Almeida, João Cotas, and Leonel Pereira. 2020. "Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia" Marine Drugs 18, no. 1: 19. https://doi.org/10.3390/md18010019
APA StyleValado, A., Pereira, M., Caseiro, A., Figueiredo, J. P., Loureiro, H., Almeida, C., Cotas, J., & Pereira, L. (2020). Effect of Carrageenans on Vegetable Jelly in Humans with Hypercholesterolemia. Marine Drugs, 18(1), 19. https://doi.org/10.3390/md18010019









