Abalone Collagen Extracts Potentiate Stem Cell Properties of Human Epidermal Keratinocytes
Abstract
:1. Introduction
2. Results and Discussion
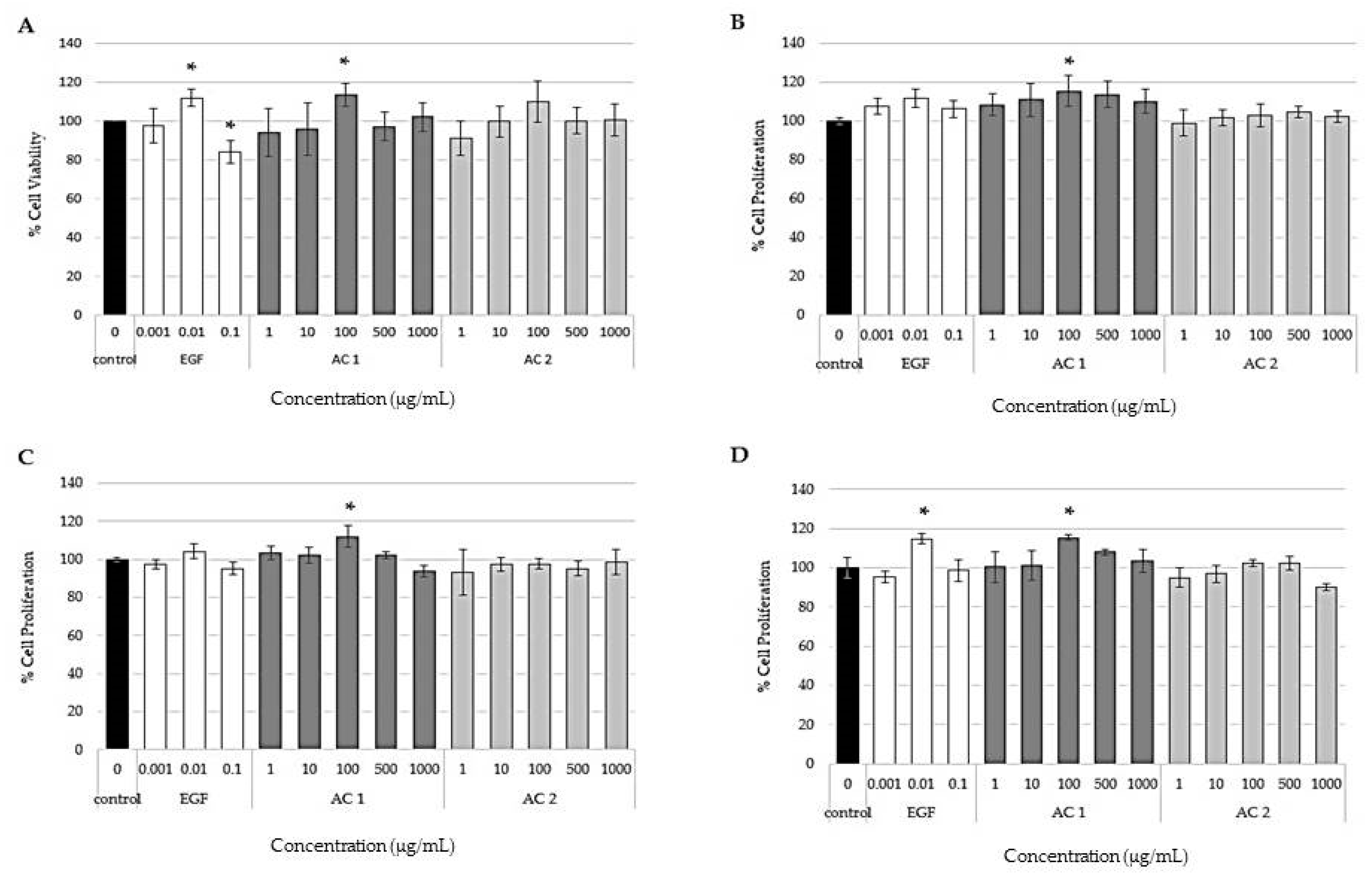
2.1. Effects of Abalone Collagen Extracts on Cell Viability and Cell Proliferation
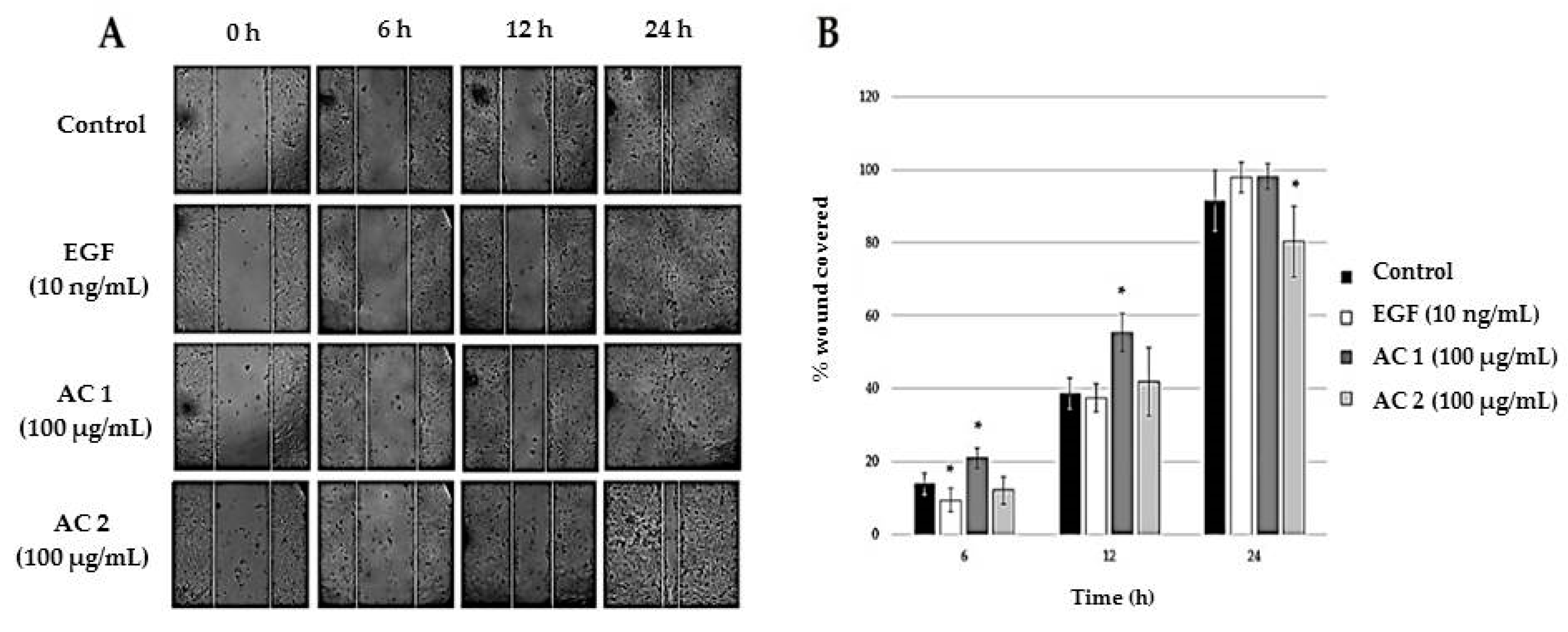
2.2. Abalone Collagen Extracts Induces Epithelization
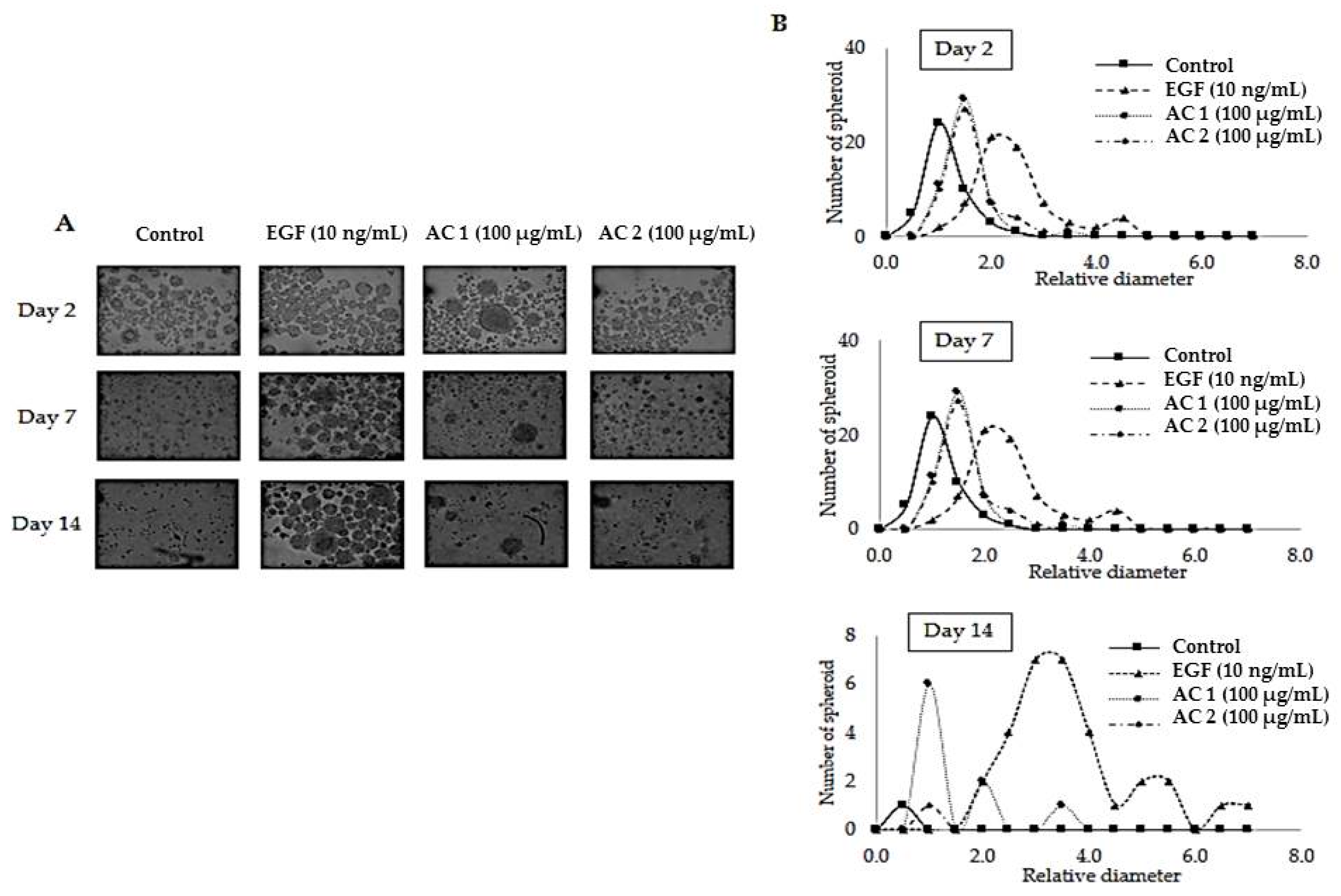
2.3. Abalone Collagen Extracts Potentiates 3D Spheroid Forming Activity
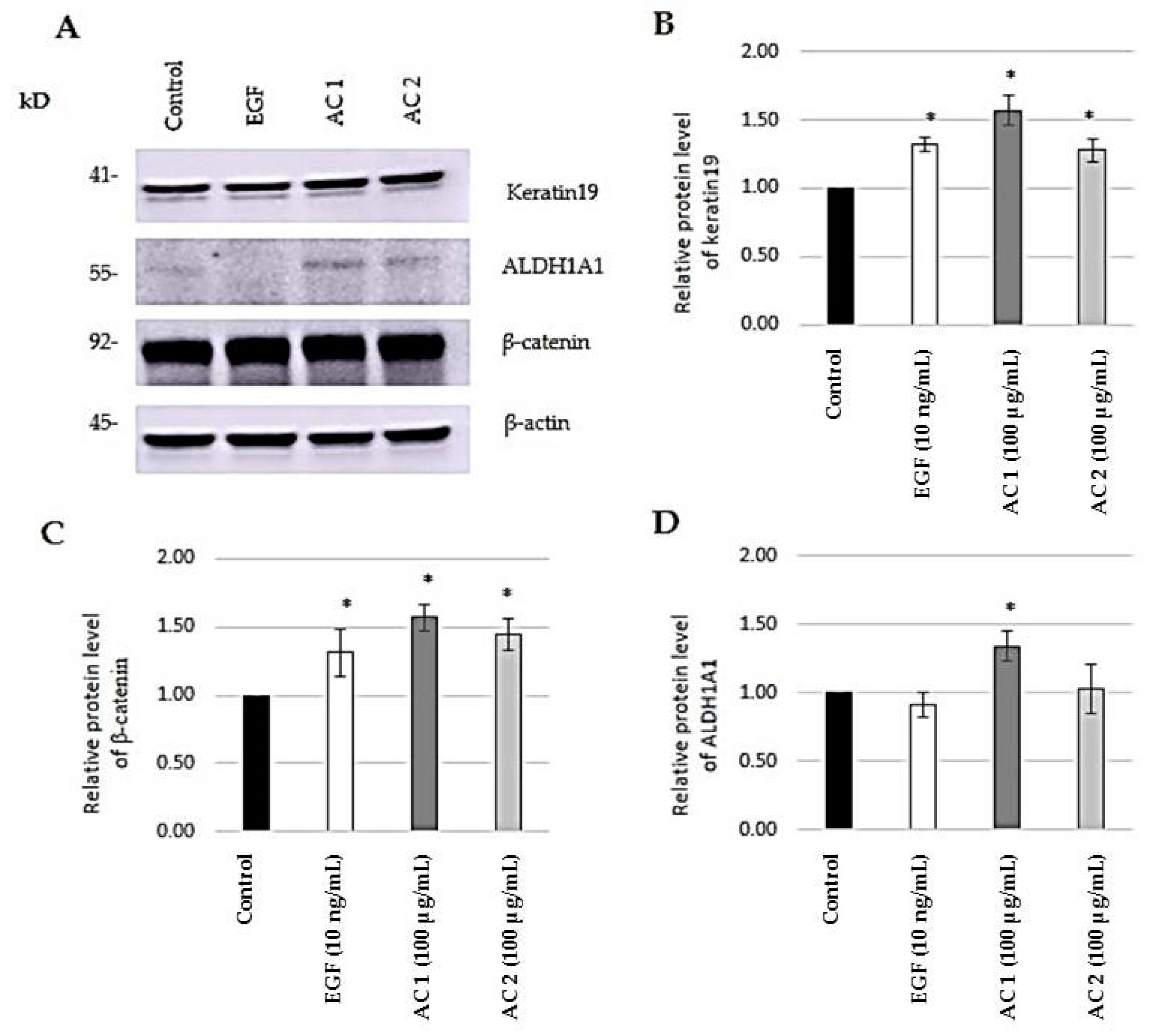
2.4. Evaluation of Stem Cell Markers
3. Materials and Methods
3.1. Materials
3.2. Cell Culture
3.3. Cell Viability by MTT Assay
3.4. Cell Proliferation by ATP content Analysis
3.5. Cell Proliferation by DNA Assay
3.6. Cell Proliferation by Total Cellular Protein Content Assay (SRB Assay)
3.7. Cell Migration (Wound Healing) by Scratch Test
3.8. Spheroid Formation
3.9. Stem Cell Marker Quantification by Western Blot Analysis.
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geerligs, M. Skin Layer Mechanics; Doctor of Philosophy; Department of Biomedical Engineering: Eindhoven, The Netherlands, 2010. [Google Scholar]
- Chandra, P.; Willi, P. The Anatomy and Functions of Skin. In Advances in Wound Healing Materials: Science and Skin Engineering, 1st ed.; Smithers Rapra Technology: Shropshire, UK, 2015; pp. 25–33. [Google Scholar]
- Wickett, R.R.; Visscher, M.O. Structure and function of the epidermal barrier. Am. J. Infect. Control 2006, 34, S98–S110. [Google Scholar] [CrossRef]
- Cueva, A. Epidermal Differentiation & the Common Ichthyoses. Master’s Thesis, Cardiff University, Cardiff, UK, 2013. [Google Scholar]
- Alberts, B.; Johnson, A.; Lewis, J. Epidermis and Its Renewal by Stem Cells. In Molecular Biology of the Cell, 4th ed.; H. Freeman & Company: New York, NY, USA, 2002. [Google Scholar]
- Wikramanayake, T.C.; Stojadinovic, O.; Tomic-Canic, M. Epidermal Differentiation in Barrier Maintenance and Wound Healing. Adv. Wound Care 2014, 3, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulou, P.A.; Blanpain, C. Development and Homeostasis of the Skin Epidermis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008383. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, E. Skin stem cells: Rising to the surface. J. Cell Biol. 2008, 180, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Marconi, A.; Pincelli, C. Keratinocyte Stem Cells: Friends and Foes. J. Cell. Physiol. 2010, 225, 310–315. [Google Scholar]
- Doles, J.; Storer, M.; Cozzuto, L.; Roma, G.; Keyes, W.M. Age-associated inflammation inhibits epidermal stem cell function. Genes Dev. 2012, 26, 2144–2153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michel, M.; Torok, N.; Godbout, M.J.; Lussier, M.; Gaudreau, P.; Royal, A.; Germain, L. Keratin 19 as a biochemical marker of skin stem cells in vivo and in vitro: Keratin 19 expressing cells are differentially localized in function of anatomic sites, and their number varies with donor age and culture stage. J. Cell Sci. 1996, 109, 1017–1028. [Google Scholar]
- Agache, A.; Humbert, P. Measuring the Skin, 1st ed.; Springer-Verlag: Berlin/Heidelberg, Germany; New York, NY, USA, 2004; pp. 165–180. [Google Scholar]
- Cadigan, K.M.; Nusse, R. Wnt signaling: A common theme in animal development. Genes Dev. 1997, 11, 3286–3305. [Google Scholar] [CrossRef]
- Nelson1, W.J.; Nusse, R. Convergence of Wnt, ß-catenin, and cadherin pathways. Science 2004, 303, 1483–1487. [Google Scholar] [CrossRef]
- Augustin, I. Wnt signaling in skin homeostasis and pathology. JDDG J. Dtsch. Dermatol. Ges. 2015, 13, 302–306. [Google Scholar] [CrossRef] [Green Version]
- Kretzschmar, K.; Clevers, H. Wnt/β-catenin signaling in adult mammalian epithelial stem cells. Dev. Biol. 2017, 428, 273–282. [Google Scholar] [CrossRef]
- Tomita, H.; Tanaka, K.; Tanaka, T.; Hara, A. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget 2016, 7, 11018–11032. [Google Scholar] [CrossRef]
- Levi, B.P.; Yilmaz, Ö.H.; Duester, G.; Morrison, S.J. Aldehyde dehydrogenase 1a1 is dispensable for stem cell function in the mouse hematopoietic and nervous systems. Hematop. Stem Cells 2009, 13, 1670–1680. [Google Scholar] [CrossRef]
- Taboonpong, S.; Kiratipaiboon, C.; Phiboonchaiyanan, P.P.; Junthongjin, P.; Trithossadech, P.; Chanvorachote, P. Vanillin increases stem cell signal and cell adhesion in keratinocytes. Thai J. Pharm. Sci. 2017, 41, 47–51. [Google Scholar]
- Chowjarean, V.; Nimmannit, U.; Chaotham, C.; Eaknai, W.; Sucontphunt, A.; Plaimee, P.P.; Tengamnuay, P.; Chanvorachote, P. Grammatophyllum speciosum Extract Potentiates Stemness in Keratinocyte Cells. Chiang Mai J. Sci. 2017, 44, 1–12. [Google Scholar]
- Fratzl, P. Collagen: Structure and Mechanics; Springer: New York, NY, USA, 2008; pp. 1–496. [Google Scholar]
- Gelse, K.; Poschl, E.; Aigner, T. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1532–1546. [Google Scholar] [CrossRef]
- Sandhu, S.V.; Gupta, S.; Bansal, H.; Singla, K. Collagen in Health and Disease. J. Orofac. Res. 2012, 2, 153–159. [Google Scholar] [CrossRef]
- Ricard-Blum, S. The collagen family. Cold Spring Harb. Perspect. Biol. 2011, 3, a004978. [Google Scholar] [CrossRef]
- Berardesca, E.; Serio, A.M.; Cameli, N. Effects of topical glucooligosaccharide and collagen tripeptide F in the treatment of sensitive atopic skin. Int. J. Cosmet. Sci. 2009, 31, 271–277. [Google Scholar] [CrossRef]
- Avila Rodríguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar] [CrossRef]
- Cen, L.; Liu, W.; Cui, L.; Zhang, W.; Cao, Y. Collagen tissue engineering: Development of novel biomaterials and applications. Pediatric Res. 2008, 63, 492–496. [Google Scholar] [CrossRef]
- Hoyer, B.; Bernhardt, A.; Lode, A.; Heinemann, S.; Sewing, J.; Klinger, M.; Notbohm, H.; Gelinsky, M. Jellyfish collagen scaffolds for cartilage tissue engineering. Acta Biomater. 2014, 10, 883–892. [Google Scholar] [CrossRef]
- Martínez, A.; Blanco, M.D.; Davidenko, N.; Cameron, R.E. Tailoring chitosan/collagen scaffolds for tissue engineering: Effect of composition and different crosslinking agents on scaffold properties. Carbohydr. Polym. 2015, 132, 606–619. [Google Scholar] [CrossRef] [Green Version]
- Sun, W.; Inayathullah, M.; Manoukian, M.A.; Malkovskiy, A.V.; Manickam, S.; Marinkovich, M.P.; Lane, A.T.; Tayebi, L.; Seifalian, A.M.; Rajadas, J. Transdermal Delivery of Functional Collagen Via Polyvinylpyrrolidone Microneedles. Ann. Biomed. Eng. 2015, 43, 2978–2990. [Google Scholar] [CrossRef] [Green Version]
- Agban, Y.; Lian, J.; Prabakar, S.; Seyfoddin, A.; Rupenthala, I.D. Nanoparticle cross-linked collagen shields for sustained delivery of pilocarpine hydrochloride. Int. J. Pharm. 2016, 501, 96–101. [Google Scholar] [CrossRef]
- Tihan, G.T.; Rau, I.; Zgarian, R.G.; Ghica, M.V. Collagen-based biomaterials for ibuprofen delivery. C. R. Chim. 2016, 19, 390–394. [Google Scholar] [CrossRef]
- Silvipriya, K.S.; Kumar, K.K.; Kumar, A.R.D.; John, A.; Lakshmanan, P. Collagen: Animal Sources and Biomedical Application. J. Appl. Pharm. Sci. 2015, 5, 123–127. [Google Scholar] [CrossRef]
- Pati, F.; Adhikar, B.; Dhara, S. Isolation and characterization of fish scale collagen of higher thermal stability. Bioresour. Technol. 2010, 101, 3737–3742. [Google Scholar] [CrossRef]
- Leonga, L.M.; Sahalana, A.Z.; Tan, L.H.; Mustafaa, N.H.; Rajaba, N.F. Clarias batrachus collagen extract increases fibroblast cell adhesion, migration and proliferation. J. Appl. Pharm. Sci. 2015, 5, 19–23. [Google Scholar] [CrossRef]
- Hu, Z.; Yang, P.; Zhou, C.; Li, S.; Hong, P. Marine Collagen Peptides from the Skin of NileTilapia (Oreochromis niloticus): Characterization and Wound Healing Evaluation. Mar. Drugs 2017, 15, 102. [Google Scholar] [CrossRef]
- Pozzolini, M.; Millo, E.; Oliveri, C.; Mirata, S.; Salis, A.; Damonte, G.; Arkel, M.; Scarfì, S. Elicited ROS Scavenging Activity, Photoprotective, and Wound-Healing Properties of Collagen-Derived Peptides from the Marine Sponge Chondrosia reniformis. Mar. Drugs 2018, 16, 465. [Google Scholar] [CrossRef] [PubMed]
- Coelho, R.C.; Marques, A.L.; Oliveira, S.M.; Diogo, G.S.; Pirraco, R.P.; Moreira-Silva, J.; Xavier, J.C.; Reis, R.L.; Silva, T.H.; Mano, J.F. Extraction and characterization of collagen from antarctic and sub-antarctic squid and its potential application in hybrid scaffolds for tissue engineering. Mater. Sci. Eng. C 2017, 78, 787–795. [Google Scholar] [CrossRef]
- Cheng, X.C.; Shao, Z.Y.; Li, C.B.; Yu, L.J.; Raja, M.A.; Liu, C.G. Isolation, characterization and evaluation of collagen from jellyfish rhopilema esculentum kishinouye for use in hemostatic applications. PLoS ONE 2017, 12, e0169731. [Google Scholar] [CrossRef] [PubMed]
- Jae-Young, J.; Soo, Y.P.; Hwang, J.-Y.; Chang-Bum, A. Amino acid composition and in vitro antioxidant and cytoprotective activity of abalone viscera hydrolysate. J. Funct. Foods 2015, 16, 94–103. [Google Scholar]
- Suleria, H.A.R.; Masci, P.P.; Gobe, G.C.; Osborne, S.A. Therapeutic potential of abalone and status of bioactive molecules: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2017, 57, 1742–1748. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Lim, H.K.; Lee, S.; Hwang, H.C.; Cho, S.K.; Cho, M. Pepsin-solubilised collagen (PSC) from Red Sea cucumber (Stichopus japonicus) regulates cell cycle and the fibronectin synthesis in HaCaT cell migration. Food Chem. 2012, 132, 487–492. [Google Scholar] [CrossRef]
- Lin, X.; Chen, Y.; Jin, H.; Zhao, Q.; Liu, C.; Li, R.; Yu, F.; Chen, Y.; Huang, F.; Yang, Z.; et al. Collagen Extracted from Bigeye Tuna (Thunnus obesus) Skin by Isoelectric Precipitation: Physicochemical Properties, Proliferation, and Migration Activities. Mar. Drugs 2019, 17, 261. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Yue, J.; Gou, X.; Wu, X. Epidermal Stem Cells in Skin Wound Healing. Adv. Wound Care 2017, 6, 297–307. [Google Scholar] [CrossRef] [Green Version]
- Strong, A.L.; Neumeister, M.W.; Levi, B. Stem cells and tissue engineering: Regeneration of the skin and its contents. Clin. Plast. Surg. 2017, 44, 635–650. [Google Scholar] [CrossRef]
- Wang, P.; Henning, S.M.; Heber, D. Limitations of MTT and MTS-Based Assays for Measurement of Antiproliferative Activity of Green Tea Polyphenols. PLoS ONE 2010, 5, e10202. [Google Scholar] [CrossRef]
- Van Tonder, A.; Joubert, A.M.; Cromarty, A.D. Limitations of the 3-(4,5-dimethylthiazol-2-yl)- 2,5-diphenyl-2H-tetrazolium bromide (MTT) assay when compared to three commonly used cell enumeration assays. BMC Res. Notes 2015, 8, 47. [Google Scholar] [CrossRef]
- Jost, M.; Kari, C.; Rodeck, U. The EGF receptor-an essential regular of multiple epidermal functions. Eur. J. Dermatol. 2000, 10, 505–510. [Google Scholar]
- Biteau, B.; Heinrich, J. EGF signaling regulates the proliferation of intestinal stem cells in Drosophila. Development 2011, 138, 1045–1055. [Google Scholar] [CrossRef]
- Knight, C.; James, S.; Kuntin, D.; Fox, J.; Newling, K.; Hollings, S.; Pennock, R.; Genever, P. Epidermal growth factor can signal via β-catenin to control proliferation of mesenchymal stem cells independently of canonical Wnt signaling. Cell. Signal. 2019, 53, 256–268. [Google Scholar] [CrossRef]
- Kanji, S.; Das, H. Advances of Stem Cell Therapeutics in Cutaneous Wound Healing and Regeneration. Mediat. Inflamm. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Sart, S.; Tsai, A.; Li, Y.; Ma, T. Three-Dimensional Aggregates of Mesenchymal Stem Cells: Cellular Mechanisms, Biological Properties, and Applications. Tissue Eng. Part B 2014, 20, 365–380. [Google Scholar] [CrossRef] [Green Version]
- Cesarz, Z.; Tamama, K. Spheroid Culture of Mesenchymal Stem Cells. Stem Cells Int. 2015, 2016, 9176357. [Google Scholar] [CrossRef]
- Saleh, F.A.; Whyte, M.; Genever, P.G. Effects of endothelial cells on human mesenchymal stem cell activity in a three-dimensional in vitro model. Eur. Cells Mater. 2011, 22, 242–257. [Google Scholar] [CrossRef]
- Fennema, E.; Rivron, N.; Rouwkema, J.; van Blitterswijk, C.; de Boer, J. Spheroid culture as a tool for creating 3D complex tissues. Trends Biotechnol. 2013, 31, 108–115. [Google Scholar] [CrossRef]
- Lyle, S.; Christofidou-Solomidou, M.; Liu, Y.; Elder, D.E.; Albelda, S.; Cotsarelis, G. The C8/144B monoclonal antibody recognizes cytokeratin 15 and defines the location of human hair follicle stem cells. J. Cell Sci. 1998, 111, 3179–3188. [Google Scholar]
- Watt, F.M.; Collins, C.A. Role of β-catenin in Epidermal Stem Cell Expansion, Lineage Selection, and Cancer. Cold Spring Harb. Symp. Quant. Biol. 2008, 73, 503–512. [Google Scholar] [CrossRef]
- Li, X.; Wan, L.; Geng, J.; Wu, C.L.; Bai, X. Aldehyde dehydrogenase 1A1 possesses stem-like properties and predicts lung cancer patient outcome. J. Thorac. Oncol. 2012, 7, 1235–1245. [Google Scholar] [CrossRef]
- Farwick, M.; Lersch, P.; Strutz, G. Low Molecular Weight Hyaluronic Acid: Its Effects on Epidermal Gene Expression and Skin Ageing. SOFW J. 2008, 11, 134. [Google Scholar]
- Benadiba, M.; Serruya, R.; Maor, Y. Bioaccessibility of Shore Magic® collagen, a low-molecular-weight collagen supplement, in different in vitro barrier models. Heliyon. 2018, 4, e00821. [Google Scholar] [CrossRef]
- Kim, S.K.; Kim, Y.T.; Byun, H.G. Isolation and Characterization of antioxidative peptides from gelatin hydrolysate of Alaska Pollack skin. J. Agric. Food Chem. 2001, 49, 1984–1989. [Google Scholar] [CrossRef]
- Je, J.-Y.; Park, P.-J.; Kwon, J.Y.; Kim, S.-K. A novel angiotensin I converting enzyme inhibitory peptide from Alaska Pollack (Theragra chalcogramma) frame protein hydrolysate. J. Agric. Food Chem. 2004, 52, 7842–7845. [Google Scholar] [CrossRef]
- Kirkland, S.C. Type I collagen inhibits differentiation and promotes a stem cell-like phenotype in human colorectal carcinoma cells. Br. J. Cancer 2009, 101, 320–326. [Google Scholar] [CrossRef] [Green Version]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Crampton, S.P.; Wu, B.; Park, E.J.; Kim, J.H.; Solomon, C.; Waterman, M.L.; Hughes, C.C. Integration of the b-Catenin-Dependent Wnt Pathway with Integrin Signaling through the Adaptor Molecule Grb2. PLoS ONE 2009, 4, e7841. [Google Scholar] [CrossRef]
- Panatta, E.; Lena, A.M.; Mancini, M.; Affinati, M.; Smirnov, A.; Annicchiarico-Petruzzelli, M.; Piro, M.C.; Campione, E.; Bianchi, L.; Mazzanti, C.; et al. Kruppel-like factor 4 regulates keratinocyte senescence. Biochem. Biophys. Res. Commun. 2018, 499, 389–395. [Google Scholar] [CrossRef]
- Segrelles, C.; Santos-de-Frutos, K.; Paramio, J.M.; Lorz, C. Competitive Repopulation Assay of Long-Term Epidermal Stem Cell Regeneration Potential. In Methods in Molecular Biology; Springer: New York, NY, USA, 2019; pp. 1–9. [Google Scholar]
- Pagano, S.; Chieruzzi, M.; Balloni, S.; Lombardo, G.; Torre, L.; Bodo, M.; Cianetti, S.; MArinucci, L. Biological, thermal and mechanical characterization of modified glass ionomer cements: The role of nanohydroxyapatite, ciprofloxacin and zinc lcarnosine. Mater. Sci. Eng. C 2019, 94, 76–85. [Google Scholar] [CrossRef]
- Pagano, S.; Lombardo, G.; Balloni, S.; Bodo, M.; Cianetti, S.; Barbati, A.; Montaseri, A.; Marinucci, L. Cytotoxicity of Universal dental adhesive systems: Assessment in vitro assays on human gingival fibroblasts. Toxicol Vitr. 2019, 60, 252–260. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thaweekitphathanaphakdee, S.; Chanvorachote, P.; Prateepchinda, S.; Khongkow, M.; Sucontphunt, A. Abalone Collagen Extracts Potentiate Stem Cell Properties of Human Epidermal Keratinocytes. Mar. Drugs 2019, 17, 424. https://doi.org/10.3390/md17070424
Thaweekitphathanaphakdee S, Chanvorachote P, Prateepchinda S, Khongkow M, Sucontphunt A. Abalone Collagen Extracts Potentiate Stem Cell Properties of Human Epidermal Keratinocytes. Marine Drugs. 2019; 17(7):424. https://doi.org/10.3390/md17070424
Chicago/Turabian StyleThaweekitphathanaphakdee, Sajee, Pithi Chanvorachote, Sagaw Prateepchinda, Mattaka Khongkow, and Apirada Sucontphunt. 2019. "Abalone Collagen Extracts Potentiate Stem Cell Properties of Human Epidermal Keratinocytes" Marine Drugs 17, no. 7: 424. https://doi.org/10.3390/md17070424
APA StyleThaweekitphathanaphakdee, S., Chanvorachote, P., Prateepchinda, S., Khongkow, M., & Sucontphunt, A. (2019). Abalone Collagen Extracts Potentiate Stem Cell Properties of Human Epidermal Keratinocytes. Marine Drugs, 17(7), 424. https://doi.org/10.3390/md17070424




